Abstract
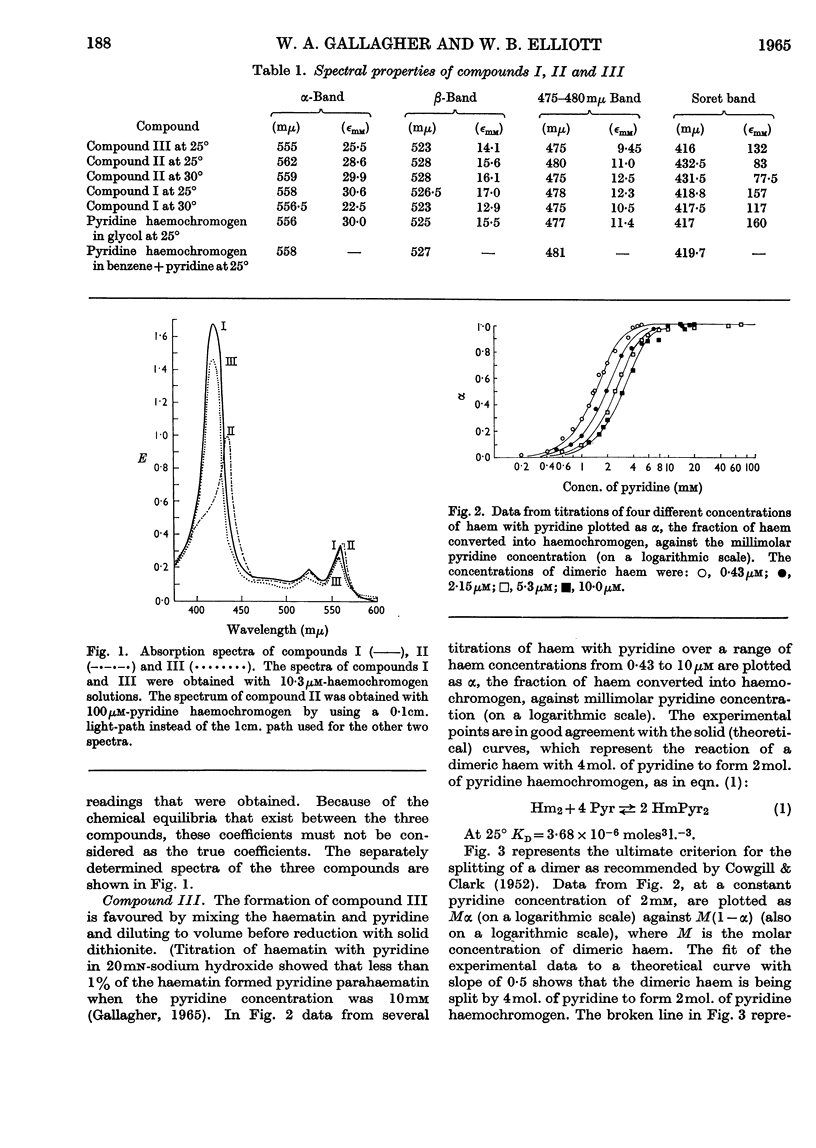
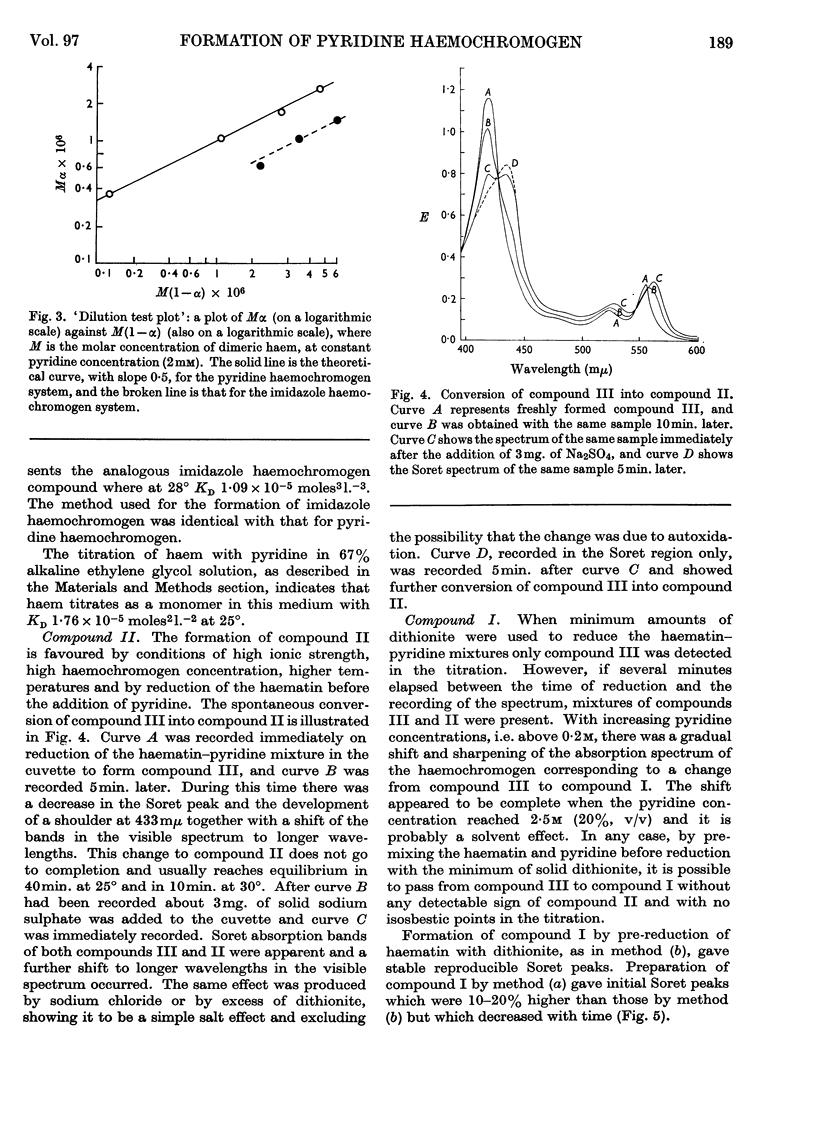
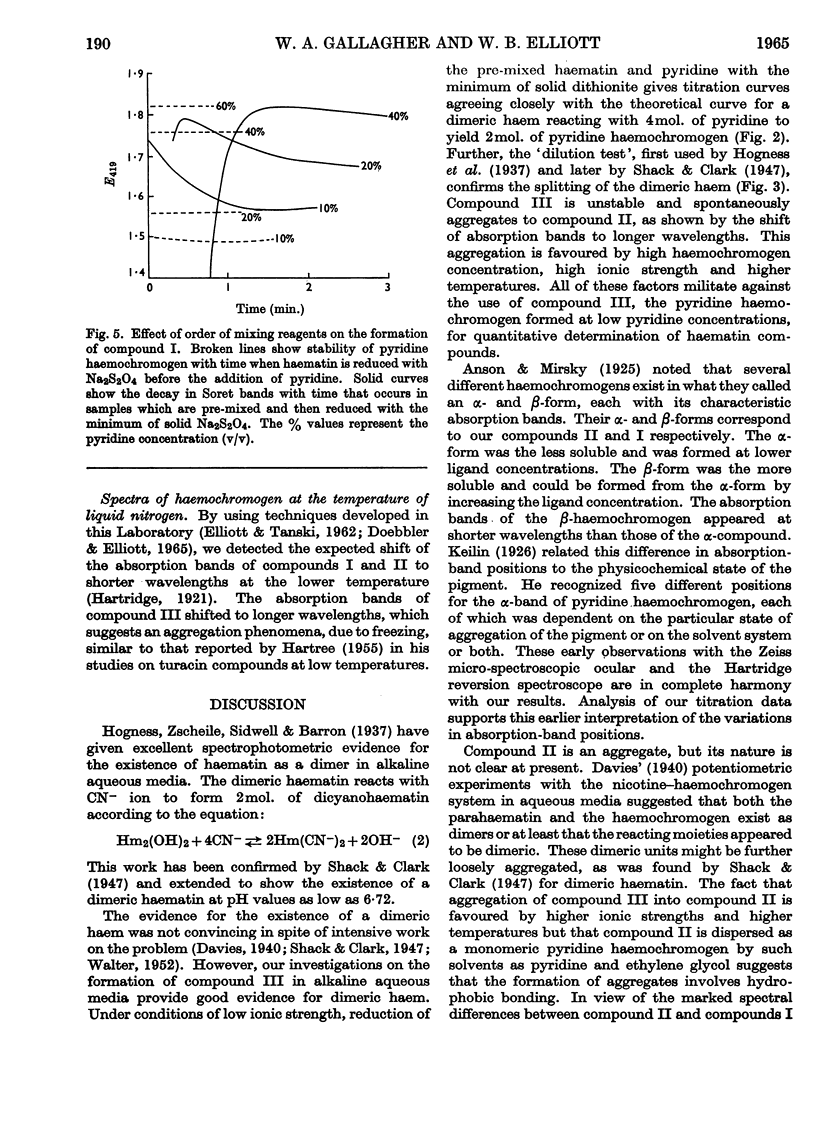
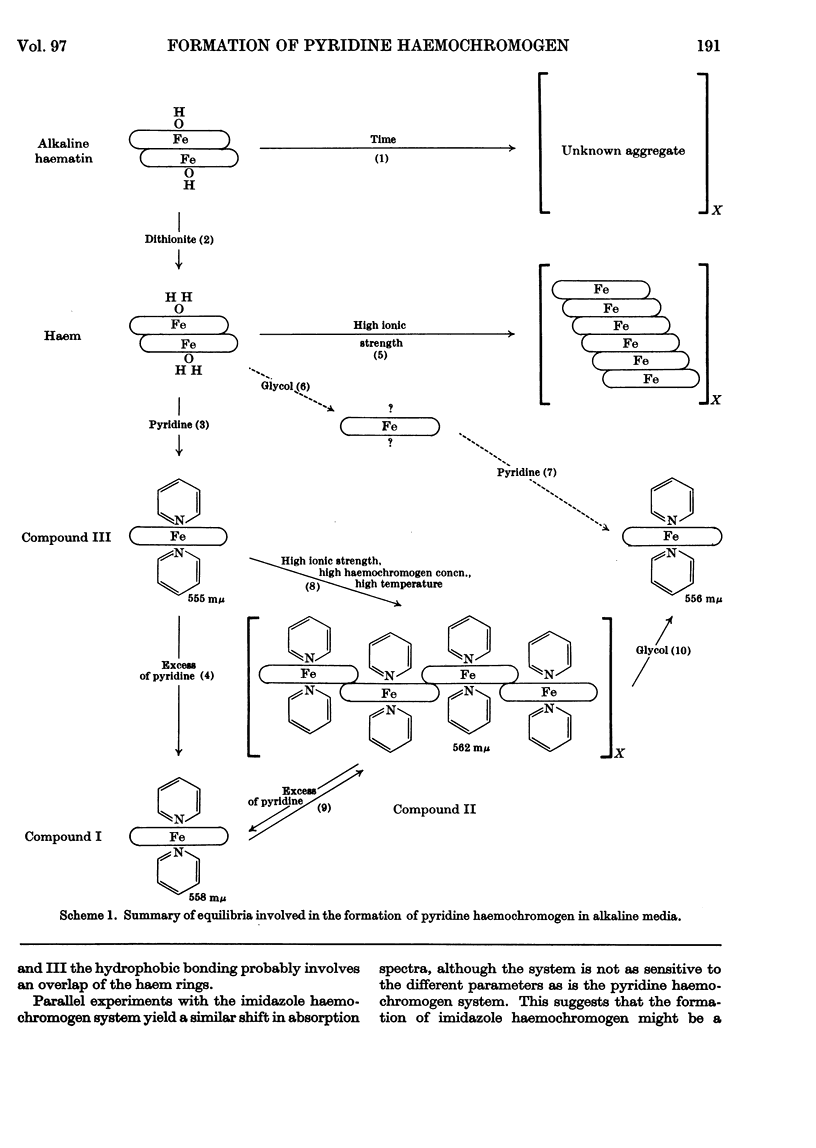
1. Titration of haem with pyridine in alkaline media of low ionic strength yields a true pyridine haemochromogen, compound III, at very low concentrations of pyridine. 2. Graphical analysis of this titration gives the first spectrophotometric evidence for a dimeric haem. 3. Compound III is unstable and tends to aggregate to a second compound, compound II, whose formation is enhanced under those conditions favourable to hydrophobic bonding. 4. At higher concentrations of pyridine, compound II is dispersed to yield the classical pyridine haemochromogen, compound I, whose spectral properties are essentially those of pyridine haemochromogen in a non-aqueous medium.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- COWGILL R. W., CLARK W. M. Metalloporphyrins. VII. Coordination of imidazoles with ferrimesoporphyrin. J Biol Chem. 1952 Sep;198(1):33–61. [PubMed] [Google Scholar]
- DOEBBLER G. F., ELLIOTT W. B. A SIMPLE METHOD FOR ABSORPTION SPECTROPHOTOMETRY OF BIOLOGICAL MATERIALS AT LOW TEMPERATURE. Biochim Biophys Acta. 1965 Mar 29;94:317–323. doi: 10.1016/0926-6585(65)90040-3. [DOI] [PubMed] [Google Scholar]
- SMITH M. H. Kinetics and equilibria in systems containing haem, carbon monoxide and pyridine. Biochem J. 1959 Sep;73:90–101. doi: 10.1042/bj0730090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WALTER R. I. Potentiometric studies of a sulfonated iron porphyrin. J Biol Chem. 1952 May;196(1):151–174. [PubMed] [Google Scholar]


