Abstract
The development of transmission-blocking vaccines is one approach to malaria control. To identify novel Plasmodium zygote- and ookinete-secreted proteins as targets of blocking malaria transmission, monoclonal antibodies (MAbs) were produced against parasite-secreted proteins found in Plasmodium gallinaceum ookinete culture supernatants. Four MAbs—1A6, 2A5, 2B5, and 4B6—were identified that bound to P. gallinaceum zygotes and ookinetes in diverse patterns in terms of spatial localization on parasites, time course of antigen expression, and Western immunoblot patterns. MAbs 2A5 and 4B6 recognized more than one protein band as detected by Western immunoblot of P. gallinaceum ookinete supernatants. Beginning at 0 h postfertilization, MAb 2A5 recognized a diverse set of antigens; at 10 h postfertilization, MAb 4B6 recognized several antigens as well. MAb 1A6 recognized a single ∼17-kDa protein, and 2B5 recognized a single ∼32-kDa protein at 15 h postfertilization. In membrane feeding assays to assess the effect of these MAbs on P. gallinaceum infectivity for Aedes aegypti mosquitoes, the addition of MAbs 1A6 and 2B5 to infectious blood meals significantly inhibited oocyst development in the mosquito midgut. In contrast, MAb 2A5 seemed to enhance infectivity. These results demonstrate that Plasmodium ookinetes secrete proteins (in addition to previously characterized chitinases) that may be targets for blocking malaria transmission. Future investigation of ookinete-secreted neutralization-sensitive molecules should provide valuable insight into mechanisms by which ookinetes exit the blood meal, penetrate and transverse the peritrophic matrix, and invade the mosquito midgut epithelium.
One strategy to achieve global malaria control includes the development of transmission blocking vaccines. This approach targets antigens expressed by Plasmodium parasites either on gametocytes within the human host or within the mosquito (3). Antigens expressed by Plasmodium stages within the mosquito midgut are logical to consider for transmission blocking vaccines since the mosquito midgut stages of the malaria parasite represent a genetic bottleneck: many asexual-stage parasites in the vertebrate host are winnowed down by orders of magnitude to a few ookinetes ending up as oocysts on the mosquito midgut epithelium. Studies over the past two decades have begun to elucidate Plasmodium ookinete molecules that are necessary for invasion of the mosquito midgut. Secreted ookinete antigens may be useful as synergistic components of transmission blocking vaccines since molecules sequentially secreted from secretory organelles (micronemes, rhoptries, and dense granules) in apicomplexan parasites play a critical role in target cell invasion (2, 4, 6, 13, 17, 18). Few Plasmodium ookinete-secreted antigens have been characterized, despite numerous molecules being secreted at this stage (as determined by Coomassie blue staining of ookinete culture supernatants; R. C. Langer and J. M. Vinetz, unpublished data). Plasmodium ookinete-secreted chitinases have been shown to be necessary for the ookinete to exit the blood meal and transverse the peritrophic matrix (5, 20, 21, 23, 24). Characterization of Plasmodium chitinases has demonstrated that ookinete-secreted molecules are targets of blocking parasite invasion of the mosquito midgut. Inhibition of chitinase activity with either chitinase antagonists, such as allosamidin (20), or Plasmodium chitinase-specific antibodies (R. C. Langer, F. Li, and J. M. Vinetz, unpublished data) significantly reduces the number of oocysts developing in the mosquito midguts. A successful Plasmodium transmission blocking vaccine will likely target numerous zygote and ookinete molecules, both surface-associated and soluble secreted proteins, such as the 25- and 28-kDa families of sexual-stage surface proteins (7, 8, 10, 22). To dissect further molecular mechanisms by which Plasmodium ookinetes invade the mosquito midgut and simultaneously identify potential transmission-blocking vaccine candidates, we produced a panel of monoclonal antibodies (MAbs) against soluble proteins found in supernatants of Plasmodium gallinaceum ookinetes. Because large numbers of ookinetes can be cultured in vitro in chemically defined, axenic medium for the avian malaria parasite, P. gallinaceum (11), our studies have focused on that species. P. gallinaceum is closely related to the human malaria parasite, Plasmodium falciparum (15, 16, 25). Delineation of antigens secreted by the avian parasite will likely lead to the identification of homologs in the P. falciparum genome database, which would be an attempt to define one component of the proteome of the P. falciparum ookinete which, to date, has not been cultivatable in vitro.
MATERIALS AND METHODS
Production of P. gallinaceum ookinete culture supernatants.
The 8A strain of P. gallinaceum was used to infect 4- to 6-week-old White Leghorn chickens. A gametocyte-producing line was maintained by subpassage in chickens and periodic passage through mosquitoes. Ookinetes were cultured from purified zygotes in serum-free and protease-free M199 culture medium as described previously (11). Twenty-four- to thirty-hour cultures of ookinetes were centrifuged (4,000 × g, 10 min), parasites were pelleted, and culture supernatants were collected and stored at −20°C until use. Pooled supernatants were concentrated 60-fold by centrifugal ultrafiltration (Centriprep 10; Amicon).
Production of MAbs against P. gallinaceum ookinete culture supernatant antigens.
P. gallinaceum ookinete culture supernatants were prepared as described above and protein concentration determined by the bicinchoninic acid protein assay (Pierce, Rockford, Ill.). Chitinase activity present in ookinete culture supernatants was confirmed by a microfluorimeter assay using the substrate 4-methylumbelliferyl-N,N′N′-β -d-triacetylchitotrioside (Calbiochem, La Jolla, Calif.) as previously described (24), confirming the presence of known ookinete-secreted proteins (chitinases) at biologically detectable concentrations.
Eight-week-old BALB/c mice (Charles River Laboratories) were immunized intraperitoneally with P. gallinaceum ookinete culture supernatant emulsified in complete Freund adjuvant (Difco, Detroit, Mich.) and boosted (50 μ g/mouse in incomplete Freund adjuvant) by the same routes 2, 6, and 10 weeks later. At 4 weeks after the last boost, a final injection was administered (50 μ g/mouse intraperitoneally and 50 μ g/mouse intravenously). Spleen cells were fused 3 days later with SP2/0 myeloma cells as previously described (9). Hybridomas were screened by enzyme-linked immunosorbent assay for immunoreactivity against concentrated P. gallinaceum supernatants. Positive hybridomas were cloned by single cell limiting dilution in Dulbecco modified Eagle medium containing 1% penicillin-streptomycin and 10% fetal bovine serum (Omega Scientific, Tarzana, Calif.), and the isotype was determined by using a mouse antibody isotyping kit (Gibco, Grand Island, N.Y.). Hybridoma culture supernatants were collected for each clone (1A6, 2A5, 2B5, and 4B6), and the MAb concentration was determined by radial immunodiffusion (Binding Site, San Diego, Calif.).
Characterization of anti-P. gallinaceum ookinete supernatant MAbs by immunofluorescence microscopy and Western immunoblotting.
Aliquots were taken from in vitro ookinete cultures at 0, 10, 15, and 20 h postexflagellation and fertilization (hpf), centrifuged (10 min, 3,000 × g, 22°C) and parasite pellets suspended in phosphate-buffered saline (PBS) containing 3% (wt/vol) bovine serum albumin. The parasite preparations were divided into aliquots (20 μ l/well) on multichamber glass slides (PGC Scientific, Frederick, Md.). Slides were allowed to air dry and were then heat fixed. Ookinete slides were incubated (1 h, 22°C) with PBS-3% bovine serum albumin-3% (vol/vol) Triton X-100 to block nonspecific binding sites and permeabilize parasites. Slides were incubated (30 min, 22°C) in a humidified chamber with primary MAbs (including isotype-matched negative controls), washed extensively with PBS, incubated with affinity-purified fluorescein-conjugated goat anti-mouse immunoglobulin G (IgG)/M/A (Kirkegaard & Perry, Gaithersburg, N.J.), and washed with PBS, followed by a final H2O wash. Slides were mounted with Permafluor (Immunon Shandon, Pittsburgh, Pa.) and observed and photographed on a Zeiss Axiophot 2 immunofluorescence microscope.
For Western immunoblotting (nonreducing, denaturing), P. gallinaceum ookinete culture supernatants collected at 0, 10, 15, and 20 hpf (50 μ g) were heated (5 min, 90°C) in sample buffer (25 mM Tris, pH 6.8; 2.2% [wt/vol] sodium dodecyl sulfate; 15% [vol/vol] glycerol; 0.001% [wt/vol] bromophenol blue), centrifuged (10,000 × g, 5 min) to remove insoluble debris and resolved in 4 to 20% Tris-glycine gradient gels (Invitrogen, Carlsbad, Calif.). Resolved proteins were electroblotted to nitrocellulose membranes by using the Novex Xcell Blot II module. After blocking with Buffer A (0.17 M NaCl, 0.1 M Tris [pH 7.5]) containing 5% (wt/vol) nonfat dry milk (NDM), blots were incubated (22°C, 1 h) with primary antibodies in Buffer B (Buffer A containing 0.1% [wt/vol] sodium dodecyl sulfate and 0.1% [vol/vol] Triton X-100) containing 5% (wt/vol) NDM. Primary MAbs included IgG1 isotype control of irrelevant specificity, IgM isotype control of irrelevant specificity, and the MAbs 1A6, 2A5, 2B5, and 4B6 (∼25 μ g of each/ml). After six washes (5 min each) with Buffer B containing NDM, blots were incubated (22°C, 1 h) with alkaline phosphatase-conjugated goat anti-mouse IgG/M/A (Kirkegaard & Perry) diluted 1:2,000 in Buffer B containing NDM. Blots were washed three times with Buffer B containing NDM, three times with Buffer B only, and one time with 0.1 M Tris (pH 9.0) and then developed with BCIP/NBT alkaline phosphatase substrate (Kirkegaard & Perry).
Membrane feeding assay.
Mosquitoes were fed blood meals through a membrane as previously described (19), with the exception that chicken skin was used in place of parafilm. Allosamidin (10 μ g/ml), a potent chitinase inhibitor used as a positive control in these experiments (20), or anti-P. gallinaceum supernatant or isotype control MAbs (10 μ g/ml) were mixed with freshly drawn P. gallinaceum-infected (∼10% parasitemia) chicken blood (200 μ l) containing heparin and fed (20 min, 37°C) to overnight-starved Aedes aegypti mosquitoes. To assess the transmission blocking activity of the MAbs, mosquitoes were dissected at 7 days post-blood meal, midguts stained with 1% (vol/vol) mercurochrome in water, and the number of oocysts per midgut was enumerated under light microscopy. Oocyst numbers in experimental and control groups were compared statistically by the Mann-Whitney U test as previously described (8).
RESULTS AND DISCUSSION
Development of MAbs that recognize distinct, novel P. gallinaceum zygote-ookinete, time-dependent epitopes.
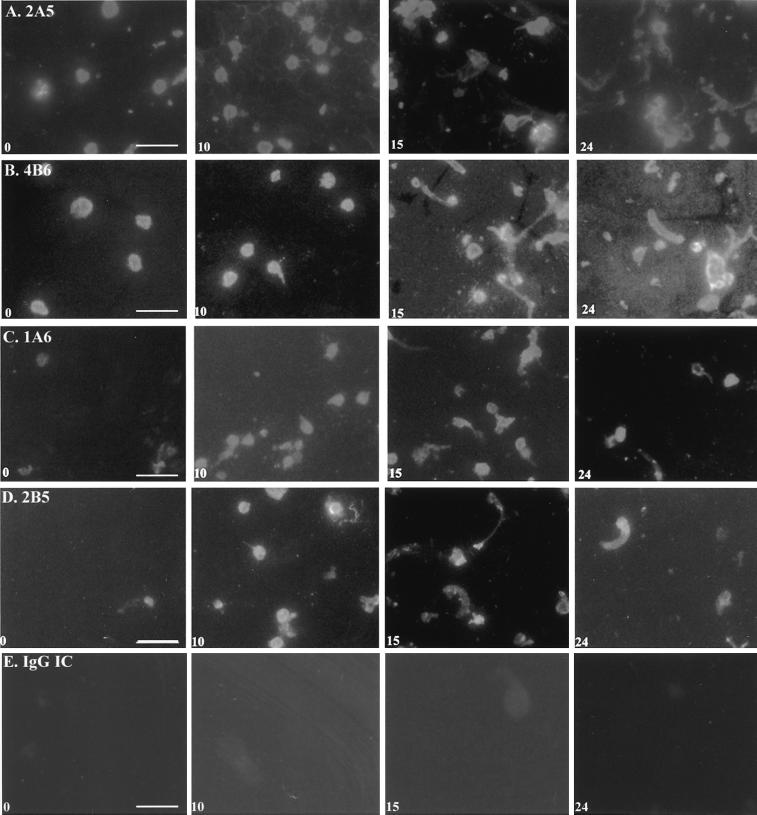
Four stable hybridomas (1A6-IgG1, 2A5-IgG1, 2B5-IgG1, and 4B6-IgM) were identified to secrete MAbs that bound in distinct patterns to the surface, internal apical, and middle regions of ookinetes, zygotes, retorts, and extracellular (exogenous) antigen secreted by the parasites (Fig. 1), as determined by immunofluoresence microscopy. MAb 2A5 reacted with 100% of early zygotes (2.5 hpf and continued to react with 100% of the parasites through 24 hpf); this immunofluorescence pattern continued until full ookinete maturation, at which point the parasites appeared in clumps (Fig. 1A, 24 h). MAb 2A5 also bound in a pattern resembling cobwebs that seem to connect various parasites. MAb 4B6 reacted with ∼50% of the early zygotes but 100% of the later stages. The majority of parasite-specific reactivity began 10 hpf on the surface of retorts, which progressed to the surface and apical tip of ookinetes at 15 and 24 hpf (Fig. 1B). MAb 4B6 recognized a remarkable amount of secreted antigen deposited on the slide at 15 and 24 hpf; this has been seen previously with ookinete-secreted chitinase (12). At time zero, few parasites were reactive with MAb 1A6, but at 10 and 15 hpf 100% of the parasites were strongly reactive. At 24 hpf, only ∼60% of the parasites were immunoreactive with 1A6 (Fig. 1C). MAb 1A6 recognized structures resembling multiple pinpoint dots in the 24-hpf cultures; simultaneous light microscopy (not shown) showed that the 1A6-recognized antigen was localized on the extreme apical end of the ookinete. MAb 2B5 reacted with <5% of the parasites at time zero, with 100% at 10 and 15 hpf, and with ∼60% of parasites 24 hpf. MAb 2B5 usually bound diffusely to zygotes and ookinetes but occasionally produced a granular pattern suggestive of subcellular organellar localization. Immunofluorescence microscopy with an IgG isotype-matched control showed insignificant background immunofluorescence (Fig. 1E).
FIG. 1.
Immunofluorescence microscopy of P. gallinaceum ookinete cultures collected at 0, 10, 15, and 24 h postfertilization. Immunofluorescence was performed with MAbs 2A5 (A), 4B6 (B), 1A6 (C), 2B5 (D), and an antigen-irrelevant IgG isotype control (E). Bar, 5 μ m.
Western immunoblot characterization of zygote or ookinete proteins recognized by MAbs.
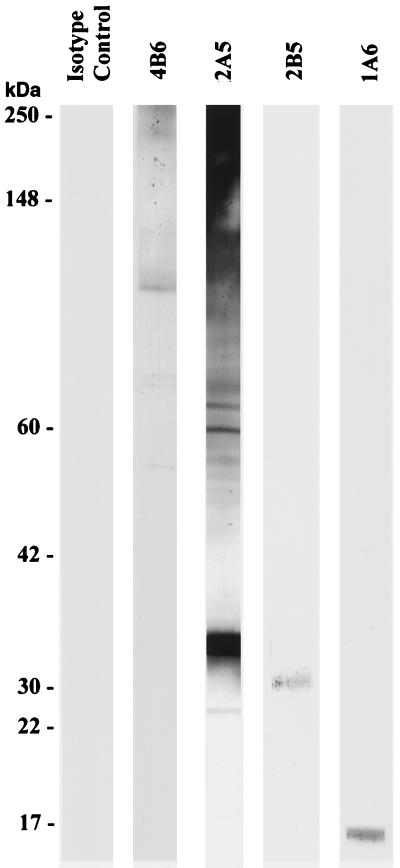
MAbs also reacted in distinct binding patterns with ookinete antigens derived from ookinete culture supernatants as determined by Western immunoblot (Fig. 2, antigen from 24 hpf culture supernatant shown). Both 2A5 and 4B6 recognized a range of nonreduced proteins. 1A6 recognized a single ∼17-kDa protein, and 2B5 recognized an ∼32-kDa protein; both of these proteins appeared most prominently 15 and 24 hpf but were also faintly visible at 10 hpf, as determined by the immunofluorescence microscopy approach described above.
FIG. 2.
Western immunoblot analysis of P. gallinaceum ookinete culture supernatants collected 24 h postfertilization. Molecular masses are indicated in kilodaltons at the left.
Concentrations of proteins in ookinete culture supernatants may have been too low to detect minimal amounts in 10 hpf cultures by Western immunoblotting with the MAbs (data not shown), suggesting that these antigens had not been secreted in sufficient quantities for detection but remained in the apical organelles of the parasite detectable by immunofluorescence assay. The detection of immunoreactive antigens in the 24-hpf culture supernatants by Western immunoblot, despite diminishing reactivity by microscopic immunofluorescence assay, could indicate a higher concentration of the antigen in a secreted form as opposed to nonsecreted form.
Effect of MAbs on infectivity of P. gallinaceun for mosquitoes.
Anti-P. gallinaceum ookinete supernatant MAbs were added to infectious blood meals at a concentration of 10 μ g/ml and fed to Aedes aegypti mosquitoes, and the development of oocysts in the mosquito midgut was enumerated. These membrane feeding experiments were performed four times. MAbs 1A6 and 2B5 significantly reduced oocyst counts, with a geometric mean number of oocysts/gut somewhat less than the maximal effect produced by allosamidin (Table 1). The addition of similar quantities of irrelevant, isotype-matched control MAb had no effect on oocyst counts. Further molecular characterization and subcellular location of the two neutralization-sensitive antigens defined by MAbs 1A6 and 2B5 will be of substantial interest.
TABLE 1.
Effect of MAbs raised against P. gallinaceum ookinete-secreted proteins on oocyst development in A. aegypti mosquitoes
| Treatment | No. of oocysts/guta | Infectivity (%)b | No. infected/totalc | Pd |
|---|---|---|---|---|
| Expt 1 | ||||
| 2A5 | 7.17 | 208 | 34/40 | 0.001 |
| 4B6 | 4.63 | 135 | 31/37 | NS |
| 1A6 | 0.74 | 21.6 | 20/37 | <0.001 |
| 2B5 | 0.49 | 14.2 | 12/40 | <0.001 |
| IgG-IgM ICe | 3.44 | 100 | 34/40 | |
| Allosamidinf | 0.12 | 3.6 | 31/37 | <0.001 |
| Expt 2 | ||||
| 2A5 | NDg | ND | ND | |
| 4B6 | 2.11 | 57.4 | 29/40 | NS |
| 1A6 | 0.40 | 10.8 | 17/40 | <0.001 |
| 2B5 | 1.30 | 35.4 | 21/40 | 0.002 |
| IgG-IgM IC | 3.69 | 100 | 29/36 | |
| Allosamidin | 0.05 | 1.2 | 2/40 | <0.001 |
| Expt 3 | ||||
| 2A5 | 52.0 | 137.4 | 37/40 | 0.009 |
| 4B6 | 33.65 | 88.9 | 38/40 | NS |
| 1A6 | 3.50 | 9.2 | 27/40 | <0.001 |
| 2B5 | 7.64 | 20.2 | 24/40 | 0.001 |
| IgG-IgM IC | 37.83 | 100 | 39/40 | |
| Allosamidin | 0.07 | 0.2 | 3/40 | <0.001 |
| Expt 4 | ||||
| 2A5 | 57.88 | 155.4 | 39/39 | 0.011 |
| 4B6 | 36.54 | 98.1 | 40/40 | NS |
| 1A6 | 3.41 | 9.1 | 28/40 | <0.001 |
| 2B5 | 12.37 | 33.2 | 36/40 | <0.001 |
| IgG-IgM IC | 37.28 | 100 | 40/40 | |
| Allosamidin | 0.15 | 0.4 | 4/40 | <0.001 |
Oocyst counts are presented as geometric means.
Infectivity is expressed as the percentage of geometric mean oocyst counts compared to the control group.
Number of mosquitoes infected/total number of mosquitoes dissected. Only mosquitoes with eggs, indicating blood meal ingestion, were dissected.
P values were determined by Mann-Whitney U test. NS, not significant.
IC, Isotype control MAb of irrelevant specificity.
Allosamidin, a chitinase inhibitor, was used as a positive control for transmission-blocking activity.
ND, not determined.
MAb 4B6 and isotype-matched control MAbs of irrelevant specificity had no significant effect on parasite development. MAb 2A5 seemed to enhance oocyst development; the presence of this MAb in an infectious blood meal was associated with oocyst counts significantly above the negative control. It is possible that 2A5 recognizes an antigen targeted by a mosquito defense molecule or other similar enzymes and is efficiently serving as an inhibitor to a naturally occurring process. Additionally, 2A5 bound prominently to an ∼36-kDa band often seen comigrating with an antigen that was immunoreactive with different IgG isotype control MAbs of irrelevant specificities (data not shown). These results suggest the possibility of an IgG receptor expressed on Plasmodium ookinetes. If the parasites are expressing such a receptor that is tightly bound by a MAb specifically recognizing the receptor, the parasite may be protected from such events as host-mediated lysis. While these results do not indicate that 2A5 is a viable target of blocking transmission of malaria parasites, further characterization of the defined protein may provide detailed insight into ookinete interactions with the midgut and the response of the midgut to this process.
This study demonstrates that some of the numerous proteins secreted by P. gallinaceum ookinetes contain epitopes that may be new targets for blocking malaria transmission. None of the MAbs described above produced Western immunoblot patterns resembling those produced by other known zygote or ookinete proteins. The proteins recognized by the transmission-blocking MAbs—1A6 and 2B5—were primarily expressed 10 h or later, suggesting that these MAbs are present and biologically active relatively late after blood meal ingestion by the mosquito. In contrast, the leading transmission-blocking vaccine, the 25-kDa protein Pfs25, is expressed early in the mosquito midgut. Previous studies have also shown that MAbs against Pfs25 and against mosquito salivary gland proteins persist long enough to be present in the oocyst, hemolymph, and even salivary gland (1, 14). Therefore, these data provide additional rationale for pursuing transmission-blocking vaccine candidates by targeting ookinete-specific proteins.
By creating a panel of MAbs and screening for the ability to block transmission of the parasite to natural hosts, we have been able to begin the preliminary characterization of new zygote and ookinete proteins. More detailed analysis of ookinete-secreted antigens should provide valuable insight into mechanisms by which ookinetes invade the mosquito midgut. Future efforts will include focusing on molecular characterization of the transmission-blocking epitopes identified in this study and identification of their homologues in the P. falciparum genome database.
Acknowledgments
We thank Abdel Desoukey for excellent technical assistance in preparing ookinetes.
This work was supported by a U.S. Public Health Service postdoctoral training grant in Emerging and Reemerging Infectious Diseases from the National Institute of Allergy and Infectious Diseases (T32-AI07536) (R.C.L), NIH grants RO1 AI45999 and KO2 AI50049 (J.M.V.), and United Nations Development Program/World Bank/World Health Organization Special Programme for Research and Training in Tropical Diseases (A00407). J.M.V. is a Culpeper Medical Sciences Scholar supported by the Rockefeller Brothers Fund.
Editor: W. A. Petri, Jr.
REFERENCES
- 1.Brennan, J. D., M. Kent, R. Dhar, H. Fujioka, and N. Kumar. 2000. Anopheles gambiae salivary gland proteins as putative targets for blocking transmission of malaria parasites. Proc. Natl. Acad. Sci. USA 97: 13859–13864 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Carruthers, V. B. 1999. Armed and dangerous: Toxoplasma gondii uses an arsenal of secretory proteins to infect host cells. Parasite Int. 48: 1–10 [DOI] [PubMed] [Google Scholar]
- 3.Carter, R. 2001. Transmission blocking malaria vaccines. Vaccine 19: 2309–2314 [DOI] [PubMed] [Google Scholar]
- 4.Dessens, J. T., A. L. Beetsma, G. Dimopoulos, K. Wengelnik, A. Crisanti, F. C. Kafatos, and R. E. Sinden. 1999. CTRP is essential for mosquito infection by malaria ookinetes. EMBO J. 18: 6221–6227 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Dessens, J. T., J. Mendoza, C. Claudianos, J. M. Vinetz, E. Khater, S. Hassard, G. R. Ranawaka, and R. E. Sinden. 2001. Knockout of the rodent malaria parasite chitinase pbcht1 reduces infectivity to mosquitoes. Infect. Immun. 69: 4041–4047 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dubremetz, J. F., N. Garcia-Reguet, V. Conseil, and M. N. Fourmaux. 1998. Apical organelles and host-cell invasion by Apicomplexa. Int. J. Parasitol. 28: 1007–1013 [DOI] [PubMed] [Google Scholar]
- 7.Duffy, P., P. Pimenta, and D. Kaslow. 1993. Pgs28 belongs to a family of epidermal growth factor-like antigens that are targets of malaria transmission-blocking antibodies. J. Exp. Med. 177: 505–510 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gozar, M. M., V. L. Price, and D. C. Kaslow. 1998. Saccharomyces cerevisiae-secreted fusion proteins Pfs25 and Pfs28 elicit potent Plasmodium falciparum transmission-blocking antibodies in mice. Infect. Immun. 66: 59–64 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Harlow, E., and D. Lane. 1988. Antibodies: a laboratory manual. Cold Spring Harbor Laboratory, Cold Spring Harbor, N.Y
- 10.Kaslow, D. 1997. Transmission-blocking vaccines: uses and current status of development. Int. J. Parasitol. 27: 183–189 [DOI] [PubMed] [Google Scholar]
- 11.Kaushal, D. C., and R. Carter. 1984. Characterization of antigens on mosquito midgut stages of Plasmodium gallinaceum. II. Comparison of surface antigens of male and female gametes and zygotes. Mol. Biochem. Parasitol. 11: 145–156 [DOI] [PubMed] [Google Scholar]
- 12.Langer, R. C., R. Hayward, T. Tsuboi, M. Tachibana, M. Torii, and J. M. Vinetz. 2000. Micronemal transport of Plasmodium ookinete chitinases to the electron-dense area of the apical complex for extracellular secretion. Infect. Immun. 68: 6461–6465 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Langer, R. C., and M. W. Riggs. 1999. Cryptosporidium parvum apical complex glycoprotein CSL contains a sporozoite ligand for intestinal epithelial cells. Infect. Immun. 67: 5282–5291 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lensen, A., G. Van Gemert, M. Bolmer, J. Meis, D. Kaslow, J. Meuwissen, and T. Ponnudurai. 1992. Transmission blocking antibody of the Plasmodium falciparum zygote/ookinete surface protein Pfs25 also influences sporozoite development. Parasite Immunol. 14: 471–479 [DOI] [PubMed] [Google Scholar]
- 15.McCutchan, T., J. Kissinger, M. Touray, M. J. Rogers, J. Li, M. Sullivan, E. Braga, A. Krettli, and L. Miller. 1996. Comparison of circumsporozoite proteins from avian and mammalian malarias: biological and phylogenetic implications. Proc. Natl. Acad. Sci. USA 93: 11889–11894 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.McCutchan, T. F., J. B. Dame, L. H. Miller, and J. Barnwell. 1984. Evolutionary relatedness of Plasmodium species as determined by the structure of DNA. Science 225: 808–811 [DOI] [PubMed] [Google Scholar]
- 17.Menard, R., A. A. Sultan, C. Cortes, R. Altszuler, M. P. Van Dijk, C. J. Janse, A. P. Waters, R. S. Nussenzweig, and V. Nussenzweig. 1997. Circumsporozoite protein is required for development of malaria sporozoites in mosquitoes. Nature 385: 336–340 [DOI] [PubMed] [Google Scholar]
- 18.Ngo, H. M., H. C. Hoppe, and K. A. Joiner. 2000. Differential sorting and post-secretory targeting of proteins in parasitic invasion. Trends Cell Biol. 10: 67–72 [DOI] [PubMed] [Google Scholar]
- 19.Shahabuddin, M., M. Criscio, and D. C. Kaslow. 1995. Unique specificity of in vitro inhibition of mosquito midgut trypsin-like activity correlates with in vivo inhibition of malaria parasite infectivity. Exp. Parasitol. 80: 212–219 [DOI] [PubMed] [Google Scholar]
- 20.Shahabuddin, M., T. Toyoshima, M. Aikawa, and D. C. Kaslow. 1993. Transmission-blocking activity of a chitinase inhibitor and activation of malarial parasite chitinase by mosquito protease. Proc. Natl. Acad. Sci. USA 90: 4266–4270 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tsai, Y. L., R. E. Hayward, R. C. Langer, D. A. Fidock, and J. M. Vinetz. 2001. Disruption of Plasmodium falciparum chitinase markedly impairs parasite invasion of mosquito midgut. Infect. Immun. 69: 4048–4054 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Tsuboi, T., D. C. Kaslow, M. M. Gozar, M. Tachibana, Y. M. Cao, and M. Torii. 1998. Sequence polymorphism in two novel Plasmodium vivax ookinete surface proteins, Pvs25 and Pvs28, that are malaria transmission-blocking vaccine candidates. Mol. Med. 4: 772–782 [PMC free article] [PubMed] [Google Scholar]
- 23.Vinetz, J., S. Dave, C. Specht, K. Brameld, R. Hayward, and D. Fidock. 1999. The chitinase PfCHT1 from the human malaria parasite Plasmodium falciparum lacks proenzyme and chitin-binding domains and displays unique substrate preferences. Proc. Natl. Acad. Sci. USA 96: 14061–14066 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Vinetz, J. M., J. G. Valenzuela, C. A. Specht, L. Aravind, R. C. Langer, J. M. Ribeiro, and D. C. Kaslow. 2000. Chitinases of the avian malaria parasite Plasmodium gallinaceum, a class of enzymes necessary for parasite invasion of the mosquito midgut. J. Biol. Chem. 275: 10331–10341 [DOI] [PubMed] [Google Scholar]
- 25.Waters, A. P., D. G. Higgins, and T. F. McCutchan. 1991. Plasmodium falciparum appears to have arisen as a result of lateral transfer between avian and human hosts. Proc. Natl. Acad. Sci. USA 88: 3140–3144. [DOI] [PMC free article] [PubMed] [Google Scholar]