Abstract
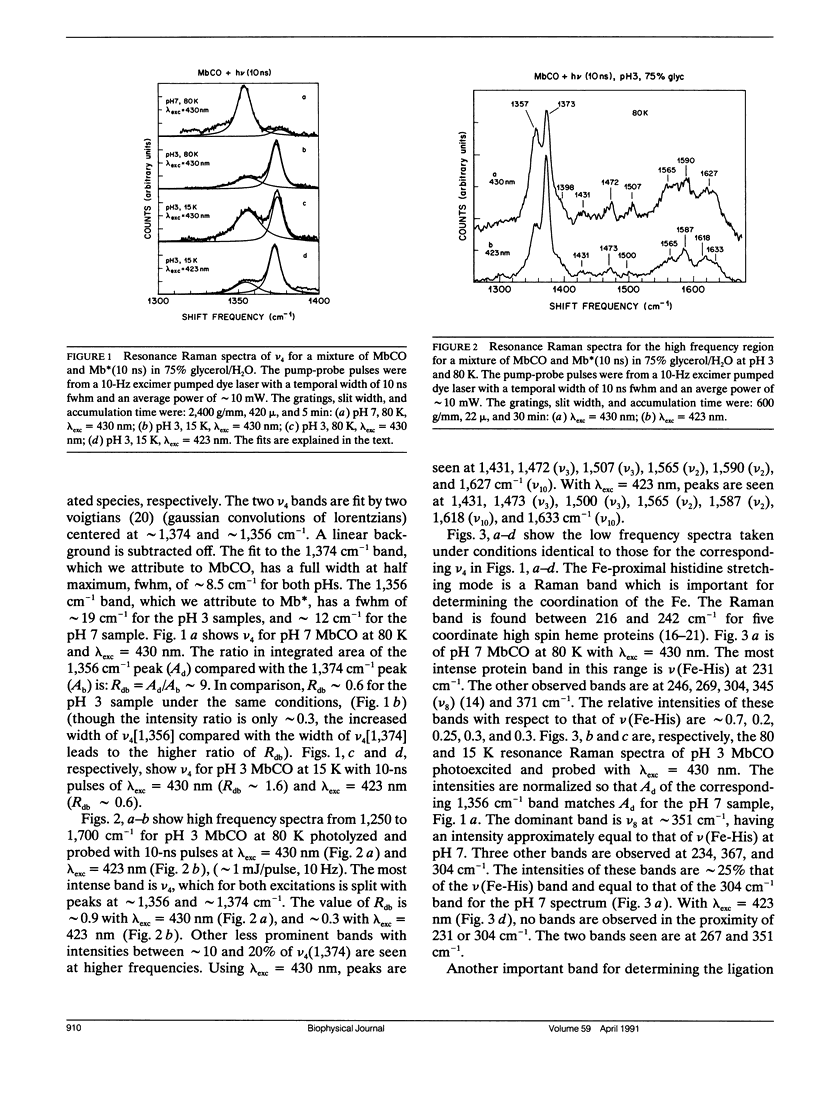
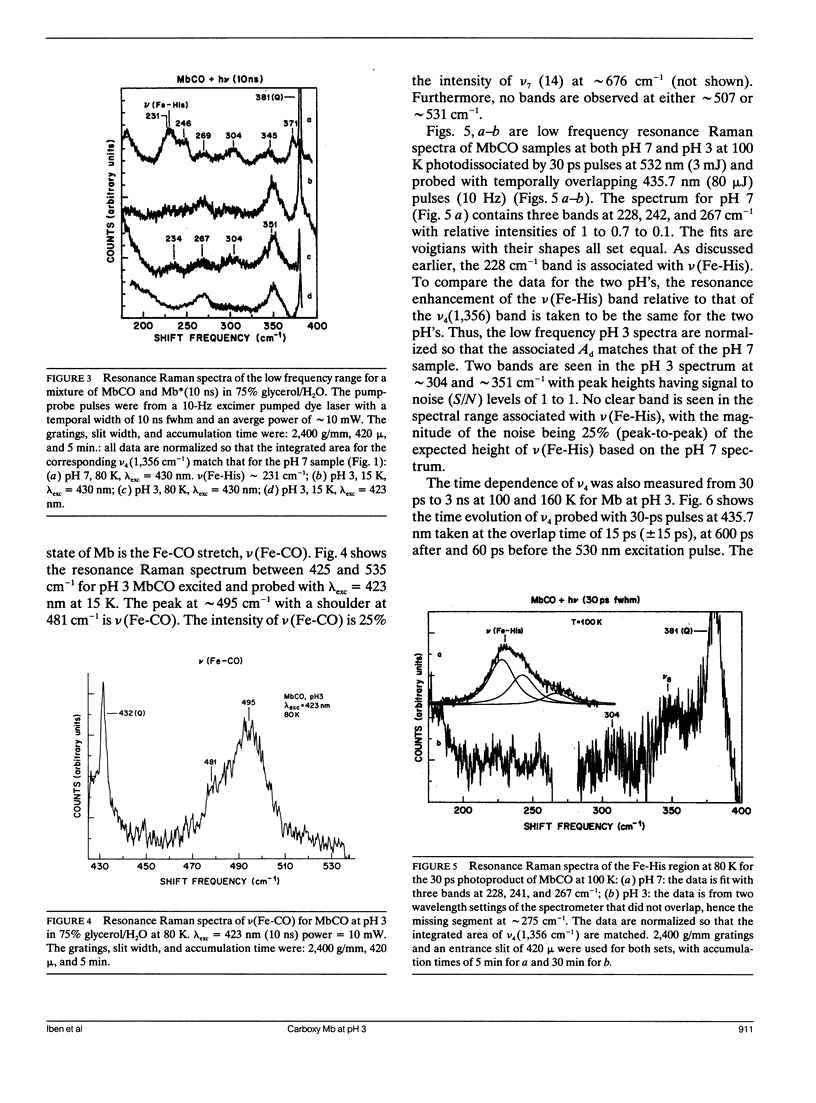
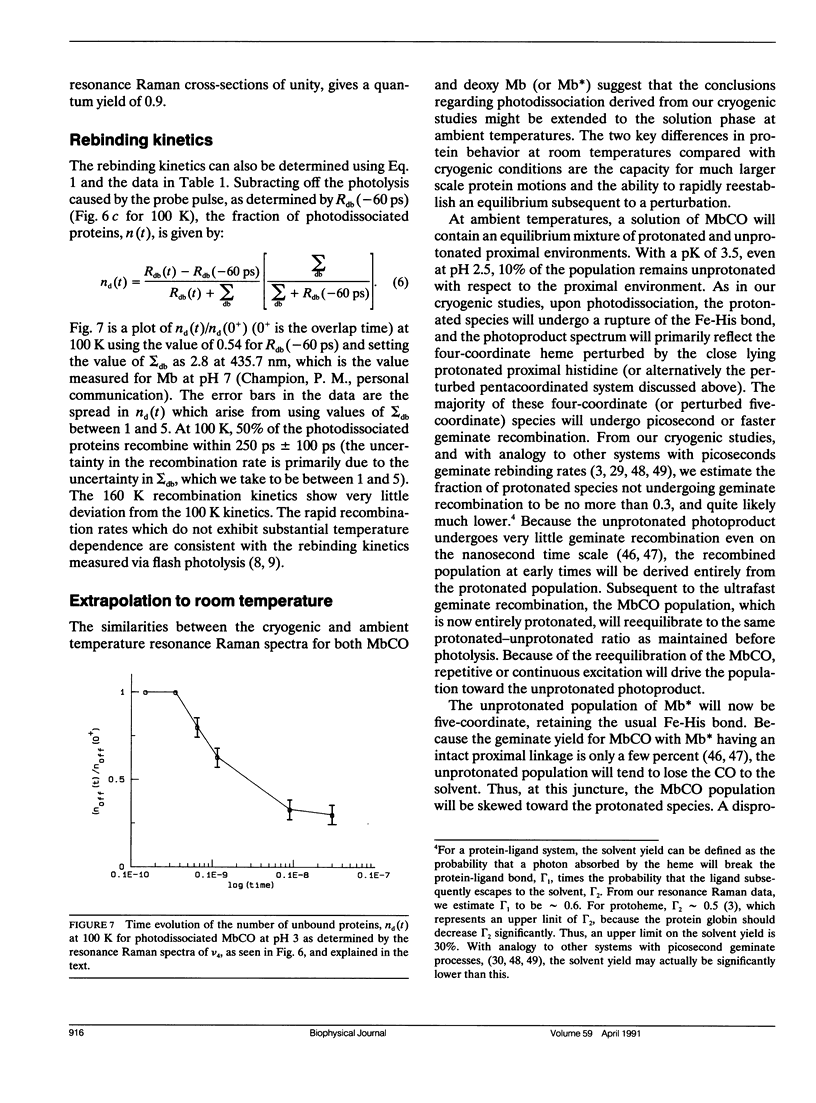
Cryogenic samples of MbCO at pH3 are studied using nanosecond and picosecond time-resolved resonance Raman spectroscopy. It is observed that under excitation conditions sufficient to completely photodissociate MbCO at pH7, the pH3 sample at 10 ns remains substantially unphotolyzed even at 15 K. The similarity in the optical and resonance Raman spectra of MbCO at pH3 with that of pH7 indicates that at pH3 the iron remains six-coordinate and low-spin. The Fe-CO stretch frequency is consistent with a more upright CO orientation. The absence of the v(Fe-His) band in the 30 ps photoproduct Raman spectrum suggests that the Fe-His(F8) bond is broken within 30 ps of photodissociation. Other Raman bands, though, are not consistent with a normal four-coordinate heme for the photoproduct, Mb*. Suggested possible interpretations include a four-coordinate heme highly perturbed by the close lying protonated proximal histidine or a five-coordinate heme with the Fe-His bond significantly weakened. The partial photolysis monitored at 30 ps and 100 K indicates either a significant amount of geminate recombination within 30 ps or low quantum yield or photolysis. The time course for CO recombination is monitored via the Raman spectra from 30 ps to 3 ns at 100 K and 160 K. Of the fraction of protein-ligand pairs that remain photodissociated at 30 ps, 50% recombine by approximately 250 ps at 100 K and 160 K, supporting the flash photolysis rebinding data of Cowen et al. (Cowen, B. R. 1990. Ph. D. thesis. University of Illinois at Urbana-Champaign; Cowen, B. R., D. Braunstein, H. Frauenfelder, P. J. Steinbach, and R. D. Young. 1989. Biophys. J. 55:55a. [Abstr.].) The conclusions from these resonance Raman studies are extended to solution phase studies at ambient temperatures.
Full text
PDF








Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Amiconi G., Antonini E., Brunori M., Formaneck H., Huber R. Functional properties of native and reconstituted hemoglobins from Chironomus thummi thummi. Eur J Biochem. 1972 Nov 21;31(1):52–58. doi: 10.1111/j.1432-1033.1972.tb02499.x. [DOI] [PubMed] [Google Scholar]
- Ansari A., Berendzen J., Braunstein D., Cowen B. R., Frauenfelder H., Hong M. K., Iben I. E., Johnson J. B., Ormos P., Sauke T. B. Rebinding and relaxation in the myoglobin pocket. Biophys Chem. 1987 May 9;26(2-3):337–355. doi: 10.1016/0301-4622(87)80034-0. [DOI] [PubMed] [Google Scholar]
- Austin R. H., Beeson K. W., Eisenstein L., Frauenfelder H., Gunsalus I. C. Dynamics of ligand binding to myoglobin. Biochemistry. 1975 Dec 2;14(24):5355–5373. doi: 10.1021/bi00695a021. [DOI] [PubMed] [Google Scholar]
- Coletta M., Ascenzi P., Traylor T. G., Brunori M. Kinetics of carbon monoxide binding to monomeric hemoproteins. Role of the proximal histidine. J Biol Chem. 1985 Apr 10;260(7):4151–4155. [PubMed] [Google Scholar]
- Friedman J. M., Campbell B. F., Noble R. W. A possible new control mechanism suggested by resonance Raman spectra from a deep ocean fish hemoglobin. Biophys Chem. 1990 Aug 31;37(1-3):43–59. doi: 10.1016/0301-4622(90)88006-e. [DOI] [PubMed] [Google Scholar]
- Friedman J. M., Lyons K. B. Transient Raman study of CO-haemoprotein photolysis: origin of the quantum yield. Nature. 1980 Apr 10;284(5756):570–572. doi: 10.1038/284570a0. [DOI] [PubMed] [Google Scholar]
- Friedman J. M. Structure, dynamics, and reactivity in hemoglobin. Science. 1985 Jun 14;228(4705):1273–1280. doi: 10.1126/science.4001941. [DOI] [PubMed] [Google Scholar]
- Giacometti G. M., Traylor T. G., Ascenzi P., Brunori M., Antonini E. Reactivity of ferrous myoglobin at low pH. J Biol Chem. 1977 Nov 10;252(21):7447–7448. [PubMed] [Google Scholar]
- Gilson M. K., Honig B. H. The dielectric constant of a folded protein. Biopolymers. 1986 Nov;25(11):2097–2119. doi: 10.1002/bip.360251106. [DOI] [PubMed] [Google Scholar]
- Han S., Rousseau D. L., Giacometti G., Brunori M. Metastable intermediates in myoglobin at low pH. Proc Natl Acad Sci U S A. 1990 Jan;87(1):205–209. doi: 10.1073/pnas.87.1.205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henry E. R., Sommer J. H., Hofrichter J., Eaton W. A. Geminate recombination of carbon monoxide to myoglobin. J Mol Biol. 1983 May 25;166(3):443–451. doi: 10.1016/s0022-2836(83)80094-1. [DOI] [PubMed] [Google Scholar]
- Martin J. L., Migus A., Poyart C., Lecarpentier Y., Antonetti A., Orszag A. Femtosecond photodissociation and picosecond recombination of O2 in myoglobin: a plausible explanation for the low quantum yield in MbO2. Biochem Biophys Res Commun. 1982 Aug;107(3):803–810. doi: 10.1016/0006-291x(82)90594-0. [DOI] [PubMed] [Google Scholar]
- Maxwell J. C., Caughey W. S. An infrared study of NO bonding to heme B and hemoglobin A. Evidence for inositol hexaphosphate induced cleavage of proximal histidine to iron bonds. Biochemistry. 1976 Jan 27;15(2):388–396. doi: 10.1021/bi00647a023. [DOI] [PubMed] [Google Scholar]
- Morikis D., Champion P. M., Springer B. A., Sligar S. G. Resonance raman investigations of site-directed mutants of myoglobin: effects of distal histidine replacement. Biochemistry. 1989 May 30;28(11):4791–4800. doi: 10.1021/bi00437a041. [DOI] [PubMed] [Google Scholar]
- Norvell J. C., Nunes A. C., Schoenborn B. P. Neutron diffraction analysis of myoglobin: structure of the carbon monoxide derivative. Science. 1975 Nov 7;190(4214):568–570. doi: 10.1126/science.1188354. [DOI] [PubMed] [Google Scholar]
- Perutz M. F., Kilmartin J. V., Nagai K., Szabo A., Simon S. R. Influence of globin structures on the state of the heme. Ferrous low spin derivatives. Biochemistry. 1976 Jan 27;15(2):378–387. doi: 10.1021/bi00647a022. [DOI] [PubMed] [Google Scholar]
- Petrich J. W., Poyart C., Martin J. L. Photophysics and reactivity of heme proteins: a femtosecond absorption study of hemoglobin, myoglobin, and protoheme. Biochemistry. 1988 May 31;27(11):4049–4060. doi: 10.1021/bi00411a022. [DOI] [PubMed] [Google Scholar]
- Phillips S. E. Structure and refinement of oxymyoglobin at 1.6 A resolution. J Mol Biol. 1980 Oct 5;142(4):531–554. doi: 10.1016/0022-2836(80)90262-4. [DOI] [PubMed] [Google Scholar]
- Phillips S. E. Structure of oxymyoglobin. Nature. 1978 May 18;273(5659):247–248. doi: 10.1038/273247a0. [DOI] [PubMed] [Google Scholar]
- Ramsden J., Spiro T. G. Resonance Raman evidence that distal histidine protonation removes the steric hindrance to upright binding of carbon monoxide by myoglobin. Biochemistry. 1989 Apr 18;28(8):3125–3128. doi: 10.1021/bi00434a001. [DOI] [PubMed] [Google Scholar]
- Smulevich G., Mauro J. M., Fishel L. A., English A. M., Kraut J., Spiro T. G. Heme pocket interactions in cytochrome c peroxidase studied by site-directed mutagenesis and resonance Raman spectroscopy. Biochemistry. 1988 Jul 26;27(15):5477–5485. doi: 10.1021/bi00415a014. [DOI] [PubMed] [Google Scholar]
- Spiro T. G., Burke J. M. Protein control of porphyrin conformation. Comparison of resonance Raman spectra of heme proteins with mesoporphyrin IX analogues. J Am Chem Soc. 1976 Sep 1;98(18):5482–5489. doi: 10.1021/ja00434a013. [DOI] [PubMed] [Google Scholar]
- Spiro T. G., Smulevich G., Su C. Probing protein structure and dynamics with resonance Raman spectroscopy: cytochrome c peroxidase and hemoglobin. Biochemistry. 1990 May 15;29(19):4497–4508. doi: 10.1021/bi00471a001. [DOI] [PubMed] [Google Scholar]
- Spiro T. G., Strekas T. C. Resonance Raman spectra of heme proteins. Effects of oxidation and spin state. J Am Chem Soc. 1974 Jan 23;96(2):338–345. doi: 10.1021/ja00809a004. [DOI] [PubMed] [Google Scholar]
- Steigemann W., Weber E. Structure of erythrocruorin in different ligand states refined at 1.4 A resolution. J Mol Biol. 1979 Jan 25;127(3):309–338. doi: 10.1016/0022-2836(79)90332-2. [DOI] [PubMed] [Google Scholar]
- Takano T. Structure of myoglobin refined at 2-0 A resolution. II. Structure of deoxymyoglobin from sperm whale. J Mol Biol. 1977 Mar 5;110(3):569–584. doi: 10.1016/s0022-2836(77)80112-5. [DOI] [PubMed] [Google Scholar]
- Warshel A. Energy-structure correlation in metalloporphyrins and the control of oxygen binding by hemoglobin. Proc Natl Acad Sci U S A. 1977 May;74(5):1789–1793. doi: 10.1073/pnas.74.5.1789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zipp A., Kauzmann W. Pressure denaturation of metmyoglobin. Biochemistry. 1973 Oct 9;12(21):4217–4228. doi: 10.1021/bi00745a028. [DOI] [PubMed] [Google Scholar]


