Abstract
During maize infection, the fungal pathogen Ustilago maydis undergoes a dimorphic transition from budding, yeast-like cells to a filamentous dikaryon that proliferates in the host. This transition is regulated by mating and environmental signals. Septation is likely to be important in the growth of the infectious dikaryon because of the need to maintain specific cellular compartments during dikaryotic growth. Recently, we found that the transcript level for a septin gene was influenced by the conserved cyclic AMP (cAMP)/protein kinase A signaling pathway that participates in regulating dimorphism in U. maydis. In this study, we describe the detailed analysis of the function of this septin gene, designated sep3, in the growth, development, and pathogenesis of U. maydis. We show that sep3 is required for normal cellular morphology and the division of budding haploid cells. The gene is also required for lipid-induced filamentous growth in culture but not during the formation of mating filaments on agar medium or in planta. Strains with a deletion in sep3 have a reduction in symptom development in maize, with filamentous cells in planta displaying morphological defects. In addition, sep3 influences the differentiation of hyphae into teliospores and the germination of these teliospores to produce the meiotic haploid progeny that complete the disease life cycle. Finally, the deletion of sep3 was found to influence the multiple budding phenotype of a mutant with a defect in the regulatory subunit of protein kinase A. This result is consistent with a link between sep3 and the control of morphogenesis by cAMP signaling. Overall, this study highlights the importance of regulating septation and changes in morphology during phytopathogenesis.
Fungal pathogenesis is dependent upon changes in morphology to generate specific cell types capable of host penetration, host colonization, and dispersal to reinitiate infection. The infection of maize by the basidiomycete fungal pathogen Ustilago maydis requires haploid, budding yeast cells of compatible mating types to fuse and grow as dikaryotic, filamentous cells which penetrate and colonize plant tissue. Infection results in anthocyanin pigment production and the formation of plant tumors at sites of infection. Within these tumors, the dikaryon proliferates and morphologically differentiates into melanized spores (teliospores) that eventually rupture plant cells and the tumors to disperse. Teliospores germinate to produce meiotic haploid progeny capable of reinitiating the life cycle (2, 8, 33, 34).
Changes in morphology in U. maydis result from the activation of two conserved signaling pathways: a mitogen-activated protein kinase (MAPK) signaling cascade and a cyclic AMP (cAMP)/protein kinase A (PKA)-dependent pathway (12). Both pathways are thought to transduce environmental signals, such as nutrient availability, presence of lipids, air exposure, acidic pH, and pheromones from cells of opposite mating type, to regulate the transition from budding to filamentous growth (6, 22, 27).
Mating is essential for the formation of the filamentous dikaryon, and the process is regulated by two unlinked mating loci designated a and b (6, 7). The a locus encodes a pheromone (mfa1/2) and a pheromone receptor (pra1/2) that are required for cell recognition and that allow successful mating between cells of opposite a mating types (7). The b locus encodes two homeodomain proteins (bE and bW) required for dikaryotic growth and completion of the sexual phase of the life cycle (21, 23). The MAPK cascade encoded by ubc4 (MAPK kinase kinase [MAPKKK]), fuz7 (MAPKK), and ubc3/kpp2 (MAPK) leads to the activation of pheromone response factor Prf1 (1, 3, 17, 28).
The cAMP/PKA pathway is also required for dimorphism and pathogenesis in U. maydis (15, 25). Mutations in gpa3, the alpha subunit of a heterotrimeric G protein, uac1 (encoding adenylyl cyclase), or adr1 (encoding the catalytic subunit of PKA) result in a constitutively filamentous phenotype and reduced virulence (4, 13, 14, 30). Mutation of the ubc1 gene, encoding the regulatory subunit of PKA, results in a multibudding phenotype; these mutants are capable of proliferating in the plant, but they are unable to induce tumor symptoms (16). These phenotypes indicate that high PKA activity leads to a budding phenotype but that low PKA activity results in filamentous growth. Recently, the presence of lipids was shown to induce a switch from budding to filamentous growth (22). The lipid signal was shown to require both the cAMP and MAPK signaling pathways because mutants in components of either of these pathways could no longer respond morphologically to the presence of lipids (22).
In a related study to identify genes up- or down-regulated during budding or filamentous growth, serial analysis of gene expression (SAGE) was performed to compare the transcriptomes of wild-type mutants with those of mutants with a defect in ubc1 (regulatory subunit of PKA; multibudded phenotype) or adr1 (catalytic subunit of PKA; constitutively filamentous phenotype) (24). Among other results, SAGE revealed that expression of a U. maydis septin gene (ortholog of the Saccharomyces cerevisiae CDC11 gene and designated sep3) was down-regulated in the ubc1 mutant (24). Septins are a conserved family of filament-forming, GTP-binding proteins that have a conserved role during cellular division and morphology (26). Therefore, sep3 represents a possible link between the cAMP signaling pathway and changes in U. maydis morphology.
In this study, we investigated the role of sep3 in the development and pathogenesis of U. maydis. We found that sep3 is required for normal morphology and the division of haploid cells. sep3 is also required for the formation of hyphae during the filamentous growth response to lipids but is not required to produce normal mating filaments. This finding is of significant interest because it may indicate that filaments induced by lipids require different morphogenetic machinery than those induced by mating. In addition, we investigated the role of sep3 in the pathogenicity of U. maydis and found that the deletion of sep3 resulted in a minor decrease in the production of tumor symptoms in maize. Our results show that sep3 was required for correct cellular morphology during infection and during the production and germination of teliospores, an important aspect in the ability of the fungus to disseminate during the completion of the disease cycle. This study also further investigated the connection between PKA signaling and sep3 by generating Δubc1 Δsep3 double mutants that showed the phenotype of the Δsep3 strain and by analyzing the growth of Δsep3 strains in exogenous cAMP.
MATERIALS AND METHODS
Molecular techniques.
Plasmid DNA was isolated using the Eppendorf fast plasmid mini kit. Genomic DNA was isolated as previously described (19). Southern transfers were performed using Amersham Hybond N+ membranes as per the manufacturer's instructions. Southern blots were hybridized using standard methods and [α-32P]dCTP (31).
The genomic sequence of sep3 was obtained from the U. maydis genome sequence at http://mips.gsf.de/genre/proj/ustilago/ (Um03449). The Δsep3::hygBr construct was generated by overlap PCR (9). Primers KO1 (5′GTTTTTGAGGCCGCATAG3′) and KO3 (5′TAGCACACGACTCACATCTGATGACGGACGAAATGG3′) and primers KO4 (5′ACTGTGCTTCAATCGCTGTGCAGGCTCAGAACATCA3′) and KO6 (5′ATTGGACACGGAAGACTT3′) were used to amplify the 945-bp left arm and 905-bp right arm, respectively, from genomic DNA. Primers KO2 (5′CCATTTCGTCCGTCATCAGATGTGACTCGTGTGCTA3′) and KO5 (5′TGATGTTCTGAGCCTGCACAGCGATTGAAGCACAGT3′) amplified a 2.7-kb hygromycin resistance marker region from pIC19RHL. Overlap PCR was performed using the three PCR fragments as templates and primers KO1 and KO6. The 4.5-kb overlap PCR product was cloned into TOPO-TA (Invitrogen), generating the Δsep3::hygBr construct (pKB001). This construct deletes from −8 to +1,216, relative to the ATG translation start codon (leaves 8 bp of coding region at the 3′ end), and has 945 bp and 905 bp of flanking region on either side of the selectable marker for homologous recombination.
Strains.
Δsep3::hygBr a2b2 and Δsep3::hygBr a1b1 strains were generated by the transformation of a2b2 (strain 518) and a1b1 (strain 521) protoplasts with pKB001. Δubc1::phleor and Δsep3::hygBr a2b2 strains were generated by the transformation of strain 0505 (Δubc1-4::phleor a2b2 [15]) with pKB001. Transformants were selected on double complete medium (DCM) with 1 M sorbitol and 250 μg/ml hygromycin B and purified on complete medium plates containing 150 μg/ml hygromycin B. Transformants were screened by colony PCR using a primer outside the construct (KB053, 5′TTGACCACCTGGAGCCGC3′) and a primer within hygB (KB062, 5′ACTGTGCTTCAATCGCTG3′). Gene deletion and the absence of ectopic copies were confirmed by Southern blot analysis. A high frequency (80%) of targeted integration was noted, and four independent mutants in each strain background were analyzed.
Media and growth conditions.
Analysis of haploid morphology was performed by growing strain 518 (a2b2), strain 521 (a1b1), and the Δsep3::hygBr a1b1, Δsep3::hygBr a2b2, and Δubc1::phelor Δsep3::hygBr a2b2 strains in potato dextrose broth (PDB) medium overnight in a shaking 30°C incubator. Characterization of the response of strains to lipids was performed by growing 518 (a2b2), 521 (a1b1), the Δsep3::hygBr a1b1 mutant, and the Δsep3::hygBr a2b2 mutant in a 30°C shaking incubator for 5 days in minimal medium supplemented with 1% Tween 40 (polyethylene sorbitans of the fatty acids palmitic, stearic, and oleic acid) or 1% corn oil. Mating assays were performed on plates with complete medium and activated charcoal as previously described (20).
Microscopic analysis.
For cell wall staining, 1 μl of the fluorescent brightener 28 calcofluor white (calcofluor [CAL]; Sigma) was added directly to 3 μl cell culture on a slide. For nuclear staining, 1 ml of cell culture (approximately 1 × 107 cells) was centrifuged at 14,000 rpm for 1 minute and resuspended in 100 μl of 1-μg/ml 4′,6′-diamidino-2-phenylindole (DAPI; Sigma). The cell suspension was incubated for 10 min at room temperature and washed three times in sterile distilled water. To observe the formation of mating filaments, filaments were scraped off a mating medium and resuspended in single-distillation H2O plus 1 μl calcofluor. For analysis of the effect of exogenous cAMP, 1 × 106 cells were inoculated into minimal medium plus 1% glucose and with 1, 5, or 10 mM or no cAMP (adenosine 3′,5′-cyclic monophosphate sodium salt; Sigma) and grown overnight at 30°C. Cells were viewed on a Zeiss Axioplan 2 fluorescence microscope under differential-interference-contrast (DIC) or UV fluorescence to observe CAL-stained walls and DAPI-stained nuclei. Images were captured with a DVC camera and processed with Northern Eclipse imaging software and Adobe Photoshop 7.
Pathogenicity assays.
Mating cultures were generated by growing the a2b2 (strain 518), a1b1 (strain 521), Δsep3::hygBr a1b1, and Δsep3::hygBr a2b2 strains in PDB medium overnight in a shaking 30°C incubator. Cultures were diluted to 1 × 106 or 1 × 105 cells ml−1 and combined in the following combinations: the a2b2 strain (518) × the a1b1 strain (521), the a2b2 strain (518) × the Δsep3::hygBr a1b1strain, the Δsep3::hygBr a2b2 strain × the a1b1 strain (521), and the Δsep3::hygBr a2b2 strain × the Δsep3::hygBr a1b1 strain. One-week-old Zea mays (golden bantam variety) plants grown in the greenhouse (temperature ranged from 25 to 30°C during the day) were inoculated by stem injection with approximately 100 μl of mating cultures. Plants were scored for disease after 2 weeks. Disease ratings used are as follows: 0, no disease; 1, pigment production; 2, small leaf tumors; 3, small stem tumors; 4, large stem tumors/teliospore production; and 5, plant death. Pathogenicity assays for both 1 × 106 and 1 × 105 cells ml−1 cultures were performed on three separate occasions, and a minimum of 100 plants for each cross were scored for disease. Three-month-old maize plants were inoculated by injecting approximately 2 ml of mating culture (1 × 106 cells ml−1) into the silk channels of developing cobs. Teliospore production was observed 3 weeks after inoculation.
In planta phenotypic analysis.
Epidermal peels were performed on leaves 5 days postinfection, and these were placed on a 20-μl drop of water containing 3 μl CAL (infection described above). Horizontal and vertical dissections of tumors isolated from infected plants 12 days postinfection were performed with a razor blade, and these were also placed on a 20-μl drop of water containing 3 μl CAL.
Teliospore isolation and germination.
Tumors were isolated from infected corn plants 14 days postinfection (described above). Whole tumors were dipped in 10% bleach for 30 seconds, washed twice in sterile double-distilled H2O (ddH2O), and ground in 20 ml 1.5% CuSO4 5H2O. Teliospore preparations were filtered through cheesecloth and left overnight. Teliospore suspensions were centrifuged and washed twice in sterile ddH2O. Teliospore preparation (200 μl) was spread over a petri dish containing a microscopic slide covered in ∼3 mm of 1.5% water agar. After approximately 14 h of incubation at 30°C, the slides were cut out of the petri dish. One microliter of CAL in 5 μl sterile ddH2O was placed onto the coverslip prior to inversion. Teliospores were isolated from three independent experiments, and germination was observed in three separate experiments.
RESULTS
Identification of the U. maydis CDC11 septin homolog, sep3.
Larraya et al. identified a SAGE tag with lower abundance in a library from a ubc1 mutant than in the wild-type and adr1 libraries and whose sequence corresponded to the U. maydis homolog of the yeast CDC11 septin gene (24). The SAGE result was confirmed by Northern blot analysis (data not shown), and the gene was subsequently named sep3 (named in the same order as the S. cerevisiae septins to facilitate future cross-species comparisons). Similar to what occurs in septin homologs in other organisms, the predicted sequence of Sep3 from U. maydis contains a nucleotide binding site motif (P-loop) and has divergent C and N termini with a highly conserved central region. Sep3 shows high sequence conservation with other Cdc11 homologs, with 43.2% identity to the yeast Cdc11 protein (over 95% of the sequence) and 72.3% identity with the closest homolog (found in Coprinus cinereus). The U. maydis genome contains four septin genes that correspond to four S. cerevisiae septins: Cdc3p (Ustilago gene identifier Um10503), Cdc10p (Um10644), Cdc11p (Um03449), and Cdc12p (Um03599). BLASTP searches against the U. maydis database with three other septin proteins from S. cerevisiae (Spr28p and Spr3p, involved in sporulation, and Sep7p, involved in division) detected the U. maydis orthologs of only Cdc3, Cdc10, Cdc11, and Cdc12.
Deletion of sep3 results in cells with aberrant morphology and division.
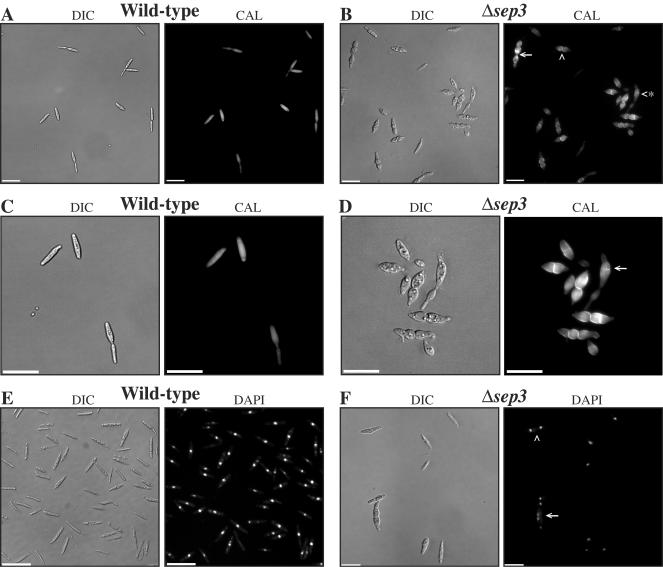
The coding region of the sep3 gene was deleted in strains of the a1b1 and a2b2 mating types, and two deletion strains from each background were used in phenotypic analyses (see Materials and Methods). Initially, we noted that the colonial morphology of the Δsep3 mutants was indistinguishable from that of wild-type strains (not shown). However, haploid cells showed a distinctive morphological phenotype (Fig. 1). Wild-type cells of U. maydis are cylindrical and uninucleate and display budding from the distal end of the cell (Fig. 1A and C). Cells of the Δsep3 strains were often observed in clumps and frequently appeared swollen and misshapen (Fig. 1B and D). Calcofluor staining of cell walls showed that some of the mutant cells had an uneven distribution or an increased accumulation of chitin (Fig. 1B). A common phenotype was the presence of a single septum located in the center of the cell that resulted in a distinctive lemon drop shape (Fig. 1B). Some cells possessed multiple septa. These inappropriately located septa could possibly be the result of an unsuccessful attempt of the cell to divide (Fig. 1B). This is also suggested by nuclear staining, which shows that in contrast to the uninucleate wild-type cells, Δsep3 cells often have multiple nuclei and the number of nuclei correlate with the number of septa (Fig. 1F).
FIG. 1.
Morphology of Δsep3 mutants. Wild-type 521 (a1b1) (A, C, and E) and Δsep3 (in a strain 521 a1b1 background) (B, D, and F) haploid strains were grown overnight in PDB medium and stained to visualize cell walls (CAL) or nuclei (DAPI). (A) Cell wall staining of wild-type cells showing septation at the mother-daughter bud junction at the distal end of the mother cell. The cells display even chitin distribution. (B) Cells of the Δsep3 mutant have an abnormal morphology resulting from aberrant division (white arrowhead) or multiple aberrant division sites (white arrow). Cells also often appear swollen and misshapen (white arrowhead with asterisk). Some cells also display an increase in chitin staining and uneven chitin distribution (white arrow). (C) Higher magnification of wild-type cells in panel A. (D) Higher magnification of Δsep3 cells in panel B. (E) Uninucleate cells of the wild-type strain 521 (a1b1). (F) Cells of the Δsep3 mutant are often multinucleate (white arrowhead and arrow). Cells with two nuclei appear morphologically similar to those in panel B, which result from one aberrant division (white arrowhead), while those with four nuclei appear morphologically similar to those in panel B, which result from three aberrant division sites (white arrow). Images were captured using DIC or epifluorescence to observe calcofluor-stained cell walls (CAL) or nuclei (DAPI). Scale bars = 50 μm.
Δsep3 mutants fail to respond morphologically to lipids.
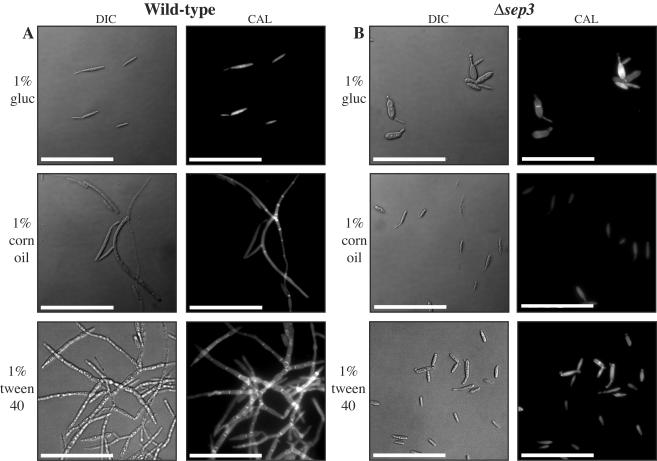
Corn oil and fatty acids have previously been shown to induce filamentous growth in U. maydis, and this morphological response suggests that the lipid content of plant cells could potentially be a host signal that influences pathogenicity (22). To investigate whether the deletion of sep3 affected the ability of Δsep3 mutants to morphologically respond to lipids, Δsep3 mutants were grown in liquid minimal medium supplemented with 1% corn oil (a mixture of triacylglycerides of various fatty acids esterified to glycerol) or 1% Tween 40 (polyethylene sorbitans of the fatty acids palmitic, stearic, and oleic acid) (Fig. 2). In minimal medium supplemented with 1% glucose as the sole carbon source, wild-type cells of U. maydis displayed yeast-like, budding growth (Fig. 2A). In contrast, wild-type cells grown in minimal medium supplemented with either 1% corn oil or 1% Tween 40 as the sole carbon source underwent a dimorphic transition to grow as septate, branched hyphae (Fig. 2A). In 1% glucose, the Δsep3 strains grew as budding yeast cells with the same morphological features described in the previous section (Fig. 2B). In contrast to wild-type cells, the Δsep3 mutants in 1% corn oil or 1% Tween 40 also grew as budding yeast. Intriguingly, the morphological defects observed in 1% glucose are not as severe as those observed in 1% corn oil or 1% Tween 40 (Fig. 2B). Overall, these results indicate that the sep3 gene is required for the formation of filaments that arise from growth on lipids as the sole carbon source.
FIG. 2.
The Δsep3 mutants fail to respond morphologically to lipids. Wild-type 521 (a1b1) (A) and Δsep3 (in a strain 521 a1b1 background) (B) haploid strains were grown for 5 days in minimal medium plus 1% glucose (gluc), 1% corn oil, or 1% Tween 40 and stained to visualize cell walls (CAL). (A) Wild-type haploid cells grow with a budding yeast cellular morphology in minimal medium plus 1% glucose. In minimal medium plus either 1% corn oil or 1% Tween 40, wild-type cells switch morphology to grow as septate, branched hyphae. (B) In minimal medium plus 1% glucose, the Δsep3 mutants appear swollen and have abnormal septation sites and morphology. In contrast to wild-type cells, Δsep3 cells have a budding yeast morphology in minimal medium plus 1% corn oil or 1% Tween 40. In addition, cells of the Δsep3 strains grown in lipids do not display morphology defects seen with mutant cells grown in 1% glucose. Images were captured using DIC or epifluorescence to observe calcofluor-stained cell walls (CAL). Scale bars = 50 μm.
Mating is unaffected in Δsep3 mutants.
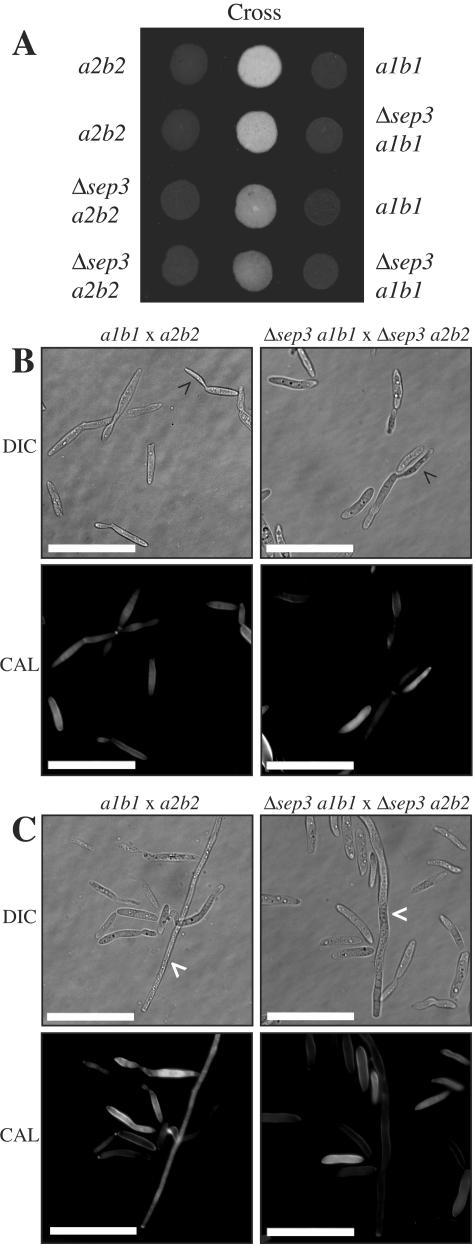
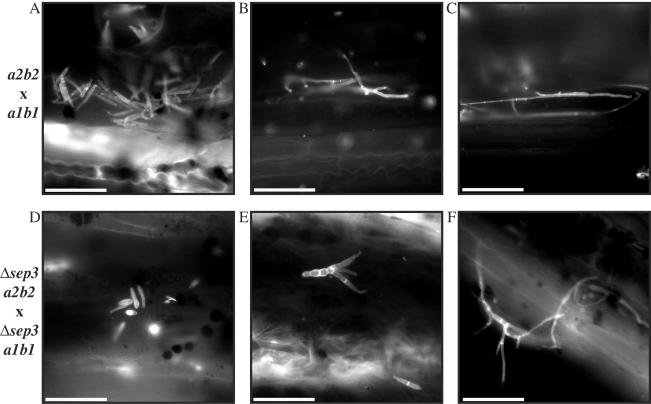
To investigate whether sep3 is required for mating, strains of opposite mating types were combined on charcoal plates and evaluated for the filamentous growth and fuzzy colony appearance indicative of successful mating. By this assay, the deletion of sep3 had no effect on the ability of the mutant cells to mate with either wild-type mating partner 521 (a1b1) or 518 (a2b2) (Fig. 3A). In addition, no effect was observed when sep3 deletion strains of opposite mating types were combined on mating medium (Fig. 3A). To look more closely at the formation of mating filaments, filaments were scraped off a mating plate and observed microscopically. Wild-type cells were present in a number of cellular morphologies: yeast-like cells (Fig. 3B and C), yeast-like cells extending conjugation tubes (Fig. 3B), and hyphal filaments (Fig. 3C). Similar to wild-type cells, Δsep3 cells from a mating plate were observed as yeast-like cells (Fig. 3B and C), yeast-like cells extending conjugation tubes (Fig. 3B), and hyphal filaments (Fig. 3C). Interestingly, in contrast to haploid cells in culture, the Δsep3 cells from mating plates showed very few morphological defects. This could be due to a reduction in cellular division events during mating or the nutritional conditions of the mating medium. The mutant cells did show reduced numbers of calcofluor-stained septa, and other minor morphological effects were still observed in the Δsep3 strains, with cells appearing swollen with various amounts of chitin deposition (Fig. 3B and C). These results indicate that sep3 is not required for the establishment of filaments during mating but does contribute to establishing the normal morphology of the filaments.
FIG. 3.
Mating is unaffected in Δsep3 mutants. (A) Approximately 1 × 105 cells were dropped onto DCM-plus-charcoal plates and incubated for 2 days at 30°C. Drops of single wild-type and mutant strains were included as controls along with combinations of wild-type and mutant strains to test for mating. Deletion of sep3 does not reduce the ability to mate. (B and C) To examine mating filaments, cells were scraped from DCM-plus-charcoal plates, resuspended in single-distillation H2O, and stained with CAL. Wild-type cells are present in a number of different morphological stages: yeast cells (B and C), yeast cells that have extended a conjugation tube (white arrowhead) (B), and mating filaments (black arrowhead) (C). Likewise, cells of the Δsep3 strains also are visible as yeast cells (B and C), yeast cells extending conjugation tubes (white arrowhead) (B), or hyphal filaments (black arrowhead) (C). Images were captured using DIC or epifluorescence to observe calcofluor-stained cell walls (CAL). Scale bar = 20 μm.
The sep3 gene is required for full symptom development during maize infection.
U. maydis infection of maize results in anthocyanin pigment production by the plant and the formation of tumors that are filled with proliferating fungal cells that eventually differentiate into black teliospores. To assess the ability of Δsep3 strains to infect and cause disease symptoms, 1-week-old plants were inoculated by stem injection with mating cultures of either 1 × 105 cells or 1 × 106 cells. Plants were infected with mixtures of wild-type strains, mixtures of wild-type and mutant strains, or mixtures of mating-compatible mutants. Plants were scored for disease after 2 weeks, and a disease index was calculated (Table 1). The results indicated that the Δsep3 mutants were capable of inducing pigmentation and tumors on the stems and leaves of infected plants (not shown). As indicated in Table 1, there was a small decrease in the disease index produced in plants inoculated with the higher inoculum level of the compatible mixture of the mutants. A more dramatic difference in the disease indices for the wild-type and mutant infections was observed when plants were inoculated with the lower concentration of cells (Table 1). Overall, these results indicated that the deletion of the sep3 gene attenuated symptom formation but that the mutants were still capable of forming tumors and teliospores (see below).
TABLE 1.
Disease indices for wild-type and mutant infections in maize plantsa
| Cross | Size of culture inoculated (cells) | No. of plants with disease rating: |
Total no. of plants | Disease index | |||||
|---|---|---|---|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | 4 | 5 | ||||
| 518 × 521 | 106 | 0 | 14 | 22 | 9 | 11 | 73 | 129 | 3.8 |
| 518 × Δsep3 a1b1 | 106 | 0 | 12 | 19 | 4 | 22 | 67 | 124 | 3.9 |
| Δsep3 a2b2 × 521 | 106 | 0 | 12 | 17 | 7 | 17 | 57 | 110 | 3.8 |
| Δsep3 a2b2 × Δsep3 a1b1 | 106 | 0 | 19 | 17 | 11 | 32 | 37 | 116 | 3.4 |
| 518 × 521 | 105 | 6 | 41 | 15 | 8 | 11 | 29 | 110 | 2.6 |
| 518 × Δsep3 a1b1 | 105 | 5 | 26 | 14 | 11 | 5 | 37 | 98 | 2.9 |
| Δsep3 a2b2 × 521 | 105 | 5 | 32 | 8 | 7 | 9 | 31 | 92 | 2.8 |
| Δsep3 a2b2 × Δsep3 a1b1 | 105 | 57 | 17 | 15 | 0 | 16 | 13 | 118 | 1.5 |
Disease ratings are as follows: 0, no disease; 1, anthocyanin production; 2, leaf tumors; 3, small stem tumors; 4, large stem tumors; and 5, death. The numbers of plants from three individual experiments are pooled for each cross. Infection assays were performed on three separate occasions using both 1 × 106 cells and 1 × 105 cells, and approximately 100 plants for each cross were scored for disease.
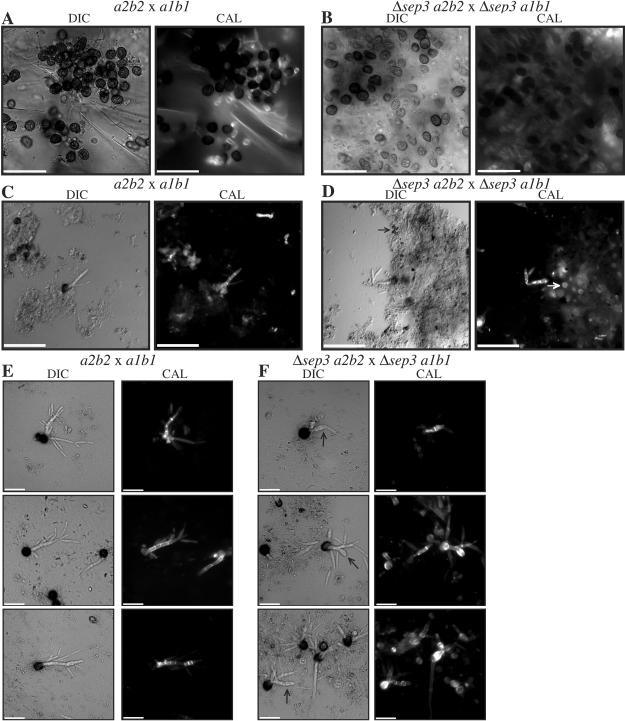
Mating of Δsep3 mutants in planta resulted in cells with abnormal morphology.
Septin proteins are proposed to form scaffolds at sites of division or new growth and have been shown to be required during polarized growth of hyphae and branching in filamentous fungi (29, 35, 36, 37). Therefore, we examined the role of sep3 during the morphological changes from budding yeast to hyphae by observing the fungal cells in planta during maize infection. One-week-old maize plants were inoculated with mating mixtures of the wild-type strains 518 (a2b2) and 521 (a1b1) or the Δsep3 a2b2 and Δsep3 a1b1 mutants. Epidermal peels were performed on infected plants at 5 days postinoculation; at this time, cells can be observed in a variety of different morphological stages within a single infected leaf (Fig. 4A to C). In maize infected with a wild-type cross, numerous yeast cells could be observed in areas surrounding plant epidermal cells (Fig. 4A), along with elongated yeast cells that appeared to be beginning to grow as hyphae (Fig. 4B). In addition, some regions of the plant epidermis were completely colonized with hyphal filaments, which form a network of hyphae along edges and connections of epidermal cells (Fig. 4C). In maize infected with the mixture of sep3 mutants (Δsep3 a2b2 with Δsep3 a1b1 mutants), yeast cells were also observed in areas surrounding plant epidermal cells. These in planta cells showed morphological defects similar to those grown in liquid culture, that is, yeast cells that were swollen, misshapen, and that had abnormal septa and chitin distribution were observed (Fig. 4D). Abnormal yeast cells that were beginning to extend and grow as hyphae were also visible in plant tissue, and these appeared to extend out of the swollen, misshapen cells (Fig. 4E). In some sections of plant epidermis, the Δsep3 hyphal cells had begun to colonize the plant tissue but to a much lesser extent than was seen in the maize infected with wild-type cells. The Δsep3 hyphal cells were evident but did not form an extensive network along edges and connections of epidermal cells. In addition, although the hyphal cells of the mutants branch and have septa, the hyphae had altered morphology in that they appeared slightly curved or curled (Fig. 4F). These results suggest that Sep3 plays a role in determining the morphology of the infectious, filamentous dikaryon that proliferates in host tissue; the altered morphology may account for the reduction in disease symptoms (Table 1).
FIG. 4.
Cellular morphology in planta. Epidermal peels of leaves of infected maize plants were examined 5 days postinoculation. Maize seedlings were inoculated with crosses of wild-type strains 518 (a2b2) and 521 (a1b1) (A, B, and C) or Δsep3 (a2b2) and Δsep3 (a1b1) (D, E, and F). Epidermal peels were stained with calcofluor to reveal cell wall chitin and observed under epifluorescence. (A) Yeast cells of the wild-type strain 521 in infected leaves. (B) Filamentous growth of the wild-type cells, possibly towards mating partners. (C) Wild-type cells in a hyphal network around surrounding plant epidermal cells. Hyphal cells branch and have visible septa. (D) The Δsep3 yeast cells in infected leaves display morphological defects, including aberrant cell shape and swelling. (E) Yeast cells of the Δsep3 strain grow filamentously, possibly towards cells of the opposite mating partner. The cells display morphological defects, such as aberrant septation sites and abnormal cell shape. (F) Δsep3 hyphal cells on plant epidermal cells. Hyphal cells of the Δsep3 cells branch and have visible septa but have altered morphology, with hyphae appearing slightly curved. Images were captured using epifluorescence to observe calcofluor-stained cell walls. Scale bar = 50 μm.
Teliospore maturation is decreased in seedlings infected with Δsep3 mutants.
To investigate whether the deletion of sep3 affected the differentiation of hyphae into teliospores during infection, sections of tumors were isolated either from maize plants inoculated with a cross of wild-type strains 518 (a2b2) and 521 (a1b1) or from plants infected with a cross of the Δsep3 a2b2 and Δsep3 a1b1 strains. Tumor sections from plants infected with wild-type cells contained masses of mature teliospores that display an echinulated surface coat. Immature teliospores could also be visualized in tumor sections when stained with the calcofluor cell wall stain (Fig. 5A). Both mature and immature teliospores were visible in tumor sections from plants infected with a cross of the Δsep3 a2b2 and Δsep3 a1b1 strains. However, the number of mature teliospores appeared to be reduced in these plants, and many of the teliospores seen in tumor sections appeared to have cell walls that were not as thick as those seen in mature spores (Fig. 5B). It was difficult to quantify relative numbers of mature and immature spores because of variability in the stages of infection between tumors and even within different parts of the same tumor. However, observations of several independent infections indicated that the number of immature teliospores that stain with calcofluor was increased in the infections with the mutants (Fig. 5B). This decrease in the number of mature teliospores was not sufficiently dramatic to be observed in infected corn ears of 3-month-old maize plants (Fig. 6). That is, we extended the examination of whether the deletion of sep3 affected teliospore production by inoculating developing embryos (ears) of plants with mixtures of compatible wild-type or mutant strains. Both types of inoculations produced tumors filled with black teliospores, and differences in teliospore maturity were not evident in this assay (Fig. 6 and unpublished observations).
FIG. 5.
Teliospore production and germination. Sections of tumors isolated from maize plants 14 days postinoculation (A and B), isolated teliospores harvested from maize plants 14 days postinoculation (C and D), and harvested teliospores germinated on potato dextrose agar slides for 24 h (E and F). Maize plants were inoculated with crosses of wild-type strains 518 (a2b2) and 521 (a1b1) (A, C, and E) or the Δsep3 a2b2 and Δsep3 a1b1 strains (B, D, and F). (A) Numerous mature teliospores (black with a rough surface coat which does not stain with calcofluor) are visible in sections from plants infected with wild-type strains. Immature teliospores could also be visualized in tumor sections when stained with the calcofluor cell wall stain. (B) Mature and immature teliospores are visible in tumor sections from plants infected with a cross of the Δsep3 a2b2 and Δsep3 a1b1 strains. However, the number of mature teliospores is reduced in these plants, and the number of immature teliospores is increased. (C) Teliospore preparations from plants infected with a wild-type cross. Ungerminated and germinating teliospores and some plant material are visible, but there are very few immature teliospores. (D) Teliospore preparations from plants infected with a Δsep3 a2b2 × Δsep3 a1b1 strain cross contain ungerminated and germinating teliospores and some plant material in addition to a large number of immature teliospores (white arrow) and many small, abnormal teliospores (black arrow). (E) Germinating teliospores from a wild-type cross. The teliospores extend a promycelium, from which haploid progeny are generated. (F) Germinating teliospores from a Δsep3 a2b2 × Δsep3 a1b1 strain cross show a variety of morphological defects. The promycelium and haploid progeny can be misshapen and swollen (black arrows). Images were captured under DIC or epifluorescence to reveal calcofluor-stained cell walls (CAL). Scale bars = 25 μm (A and B) and 50 μm (C, D, E, and F).
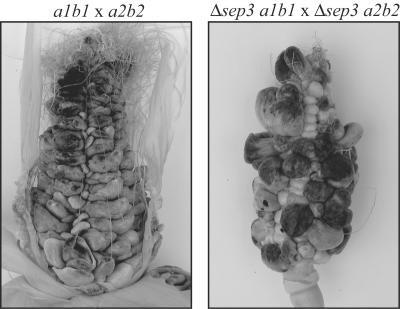
FIG. 6.
Pathogenicity of sep3 strains. Three-month-old maize plants were inoculated via injection into the silk channels of developing cobs. Plants were inoculated with crosses of wild-type strains 518 (a2b2) and 521 (a1b1) or the Δsep3 a1b1 and Δsep3 a2b2 mutant strains. Tumor formation and teliospore production were assessed 3 weeks postinoculation. Representative cobs of a 518 (a2b2) × 521 (a1b1) cross and a Δsep3 a1b1 × Δsep3 a2b2 strain cross are shown. A wild-type U. maydis infection results in the formation of large tumors filled with black teliospores. The deletion of sep3 does not inhibit the production of tumors or teliospores in infected plants.
The morphology of germinating teliospores is affected in Δsep3 strains.
Tumors from infected seedlings were ground and filtered to isolate teliospores so that the influence of the Δsep3 mutation on germination could be assessed (Materials and Methods). In addition to mature teliospores, a small amount of plant material is present in the teliospore preparations. Teliospores harvested from plants infected with a wild-type cross did not contain many immature spores (Fig. 5C). In contrast, teliospores harvested from plants infected with the cross of Δsep3 a2b2 and Δsep3 a1b1 strains contained many immature teliospores (Fig. 5D). This correlates with the increased numbers of immature spores seen in tumor sections as described above. In addition, many small, abnormal teliospores were observed in the Δsep3 a2b2 × Δsep3 a1b1 strain cross teliospore preparation (Fig. 5D).
Teliospore preparations were plated onto potato dextrose agar-coated slides and incubated for 24 h to observe teliospore germination. Wild-type teliospores germinate by extending a promycelium, with subsequent meiosis and the formation of haploid progeny as buds from the promycelium (Fig. 5E). These haploid progeny then continue to reproduce by budding to establish a new colony. Germinating teliospores from a Δsep3 a2b2 × Δsep3 a1b1 strain cross were capable of producing a promycelium and haploid progeny; however, both the promycelium and haploid progeny were observed to have abnormal morphology for many germinating spores (Fig. 5F). Morphological defects for the promycelium included swelling and aberrant shape. Haploid progeny were also observed to be swollen and misshapen, but in addition, these cells also possessed apparent septation defects (Fig. 5F).
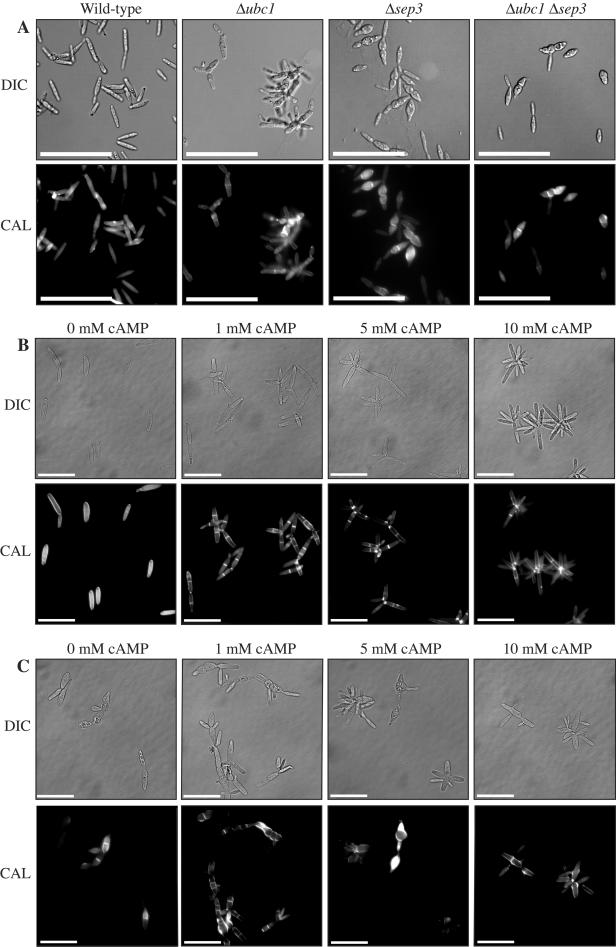
Deletion of sep3 suppressed the multibudded phenotype of the ubc1 mutant, but cAMP restored the phenotype in a Δsep3 strain.
Initially, our attention was drawn to sep3 because of its reduced transcript level in a ubc1 mutant defective in the regulatory subunit of PKA. To further investigate the relationship between sep3 and ubc1, Δubc1 sep3 double mutants were generated and phenotypically analyzed (Materials and Methods). Wild-type strains of U. maydis grow as apically budding yeast cells, and in contrast, cells of the Δubc1 mutant have a multibudded phenotype (Fig. 7A). As described earlier, the Δsep3 cells have a distinctive lemon drop shape, are swollen and misshapen, have multiple septa, and are multinucleate (Fig. 1 and 7A). The Δubc1 sep3 double mutants appear phenotypically similar to the single Δsep3 mutants in that the cells are swollen, misshapen, have multiple septa, and are multinucleate (Fig. 7A). In particular, the majority of the Δubc1 sep3 double mutant cells do not display the multibudded phenotype observed in the single ubc1 strains. This suggests that the Δsep3 mutation can suppress the multibudded phenotype of the Δubc1 mutant and that Sep3 may act downstream of PKA (Ubc1).
FIG.7.
Morphology of Δubc1 sep3 mutants and growth of Δsep3 strains in cAMP. (A) Wild-type 518 (a2b2) and Δubc1 a2b2, Δsep3 a2b2, and Δubc1 Δsep3 a2b2 haploid strains were grown overnight in PDB media and stained to visualize cell walls (CAL). The Δubc1 mutant exhibits a multibudded phenotype. The Δsep3 mutant cells are swollen, with abnormal morphology and septation sites. The Δubc1 sep3 double mutant strains have a cellular morphology similar to that of the Δsep3 single mutant strains. The cells are not multibudded like those of the Δubc1 mutant and have a swollen morphology with inappropriate septation sites. (B and C) Wild-type 518 (a2b2) (B) and Δsep3 a2b2 (C) haploid strains were grown overnight in minimal medium plus 1% glucose and either 0, 1, 5, or 10 mM cAMP and stained to visualize cell walls (CAL). (B) In 1 mM cAMP, wild-type cells exhibit an increase in budding and a greatly increased level of septation. Cells grown in 5 mM cAMP display less septation and more budding than cells grown in 1 mM cAMP. Wild-type cells grown in 10 mM cAMP exhibit little septation and greatly increased budding, such that cells appear as rosettes. (C) In 0 mM cAMP, cells of the Δsep3 strains appear misshapen and swollen and exhibit inappropriately localized septa. This phenotype is increased in severity in 1 mM cAMP, with swollen cells forming misshapen chains of septate cells. An increase in budding is evident in 5 mM cAMP and 10 mM cAMP. Like wild-type cells, Δsep3 cells grown in 10 mM cAMP form rosettes. The morphological defects of these strains at no or low levels of cAMP are reduced at high levels of cAMP. Images were captured under DIC or epifluorescence to reveal calcofluor stained cell walls (CAL). Scale bars = 50 μm (A) and 20 μm (B and C).
The ability of the Δsep3 mutation to suppress the multibudded phenotype of cells grown in exogenous cAMP was also tested. Similar to wild-type cells in high levels of cAMP, the Δsep3 strains could mimic the Δubc1 phenotype and become multibudded, with buds extending from the swollen and misshapen Δsep3 cells (Fig. 7B and C). However, the mutant response to a high exogenous level of cAMP differed from the wild-type response in that the mutant cells showed lateral as well as apical budding (Fig. 7C). In general, the multiple budding phenotype resulting from exposure to high levels of exogenous cAMP was not entirely suppressed by the Δsep3 mutation, in contrast to the phenotype of the Δubc1 sep3 double mutants. It is possible that the lack of multiple-bud cells in the Δubc1 sep3 double mutants might result from decreased budding (due to the Δsep3 mutation), which could be overcome with high levels of cAMP. In addition, the activation of PKA by loss of the Ubc1 regulatory subunit may be physiologically different than activation by exogenous cAMP, and this treatment may also have other unforeseen influences besides the influence on PKA activity.
We also varied the levels of exogenous cAMP to examine the response in more detail. Interestingly, the growth of wild-type cells in various concentrations of cAMP revealed that in addition to regulating budding frequency, cAMP levels influence septation frequency and position. In 1 mM cAMP, wild-type cells became highly septate, with septa forming in the cell centers as well as at bud sites (Fig. 7B). The number of septa decreased with increasing amounts of cAMP, suggesting that low levels of cAMP promote septation (Fig. 7B). This was further supported by the morphological phenotype of the Δsep3 strains, which was the most severe in low levels of cAMP (where septation is promoted and the loss of Sep3 function would therefore have the most effect) (Fig. 7C). Overall, these results suggest that PKA regulates septation events during budding at least in part via Sep3.
DISCUSSION
sep3 encodes a conserved U. maydis homolog of the yeast Cdc11 septin.
Septins are important components for cell growth and are proposed to form heteromeric complexes that establish filament scaffolds at sites of division or new growth. In S. cerevisiae, the septins Cdc3p, Cdc10p, Cdc11p, Cdc12p, and Sep7p are required during primary septum formation to localize the myosin Myo1p (which is part of the actomyosin ring) and the chitin synthase II complex at the mother bud neck and to localize chitin synthase III early in the cell cycle (5, 10, 26, 32). Septins are also involved in mitotic spindle orientation, cell cycle checkpoints, and targeted secretion (reviewed in reference 26). There are also two septins in S. cerevisiae, Spr3p and Spr28p, which are specifically required during sporulation (11). Septin homologs in Candida albicans and Aspergillus nidulans have also been shown to be required during polarized growth of hyphae, branching, and the formation of complex cell types (conidiophores during asexual development in A. nidulans) (35, 36, 37).
U. maydis appears to have four septin genes based on analysis of the genome sequence, and we have focused our attention on the sep3 gene (accession no. Um03449) that appears to be an ortholog of the yeast CDC11 septin gene. This gene drew our attention because SAGE analysis (24) indicated that this gene may be important for the control of morphology exerted by the cAMP/PKA pathway. SAGE tags were not found for the three other genes with similarity to the yeast septins Cdc3 (Um10503; 41.5% identity), Cdc10 (Um10644; 39.3% identity), and Cdc12 (Um03599; 38.9% identity). Searches of the Sep3 amino acid sequence against the annotated genomes of Fusarium graminearum, Neurospora crassa, Magnaporthe grisea, Coprinus cinereus, Cryptococcus neoformans, and Aspergillus nidulans revealed that these fungi have at least five putative septin homologs, four of which fall into each of the conserved Cdc3, Cdc10, Cdc11, and Cdc12 subclasses. The additional gene with homology to septins is often named after sepE in A. nidulans. In addition, N. crassa, F. graminearum, M. grisea and C. cinereus also possess an additional gene with weak homology (http://www.broad.mit.edu/annotation).
The sep3 gene is required for normal budding and hyphal morphology in U. maydis.
Haploid cells of Δsep3 strains often grew in clusters, with many cells exhibiting multiple nuclei, inappropriately positioned and/or multiple septa, and an uneven distribution of chitin. These phenotypes suggest a failure of separation during budding and possible defects in cell wall deposition. Likewise, S. cerevisiae and C. albicans CDC11 mutants also fail to complete cellular division, and cell clustering is observed in budding cells of C. albicans CDC11 mutants (18, 36). However, unlike the U. maydis Δsep3 mutants, the C. albicans CDC11 mutant lacks chitin staining between cellular compartments (36).
The dimorphic transition from the haploid budding yeast form to the dikaryotic filamentous growth stage in U. maydis is crucial for pathogenic development in maize. Specifically, filamentous growth during the life cycle generally occurs during the biotrophic phase, when hyphal cells colonize maize epidermal tissue, penetrate plant cells, and proliferate within tumors induced in the host. The ability to grow filamentously is therefore crucial to life cycle progression and the pathogenesis of U. maydis. The dimorphic transition is initiated by the mating of two haploid strains of opposite mating types and is also induced by the presence of lipids (6, 7, 22). It is tempting to speculate that lipids may provide a nutritional signal within the plant host that contributes to hyphal proliferation. The haploid Δsep3 strains were unable to form filaments in response to lipids, but the mutants were able to form dikaryotic mating filaments. However, the morphology of the mating filaments was abnormal. These findings suggest that sep3 is required for the formation of lipid-induced hyphae but not dikaryotic filaments, and this is intriguing because it suggests that the protein machinery required to initiate hyphal growth differs between the two types of filaments. During mating, the cells of opposite mating types extend conjugation tubes towards each other and fuse to form a dikaryon. It is possible that the fusion of the two cells, which consequently grow as a hypha, bypasses the requirement of the septins to initiate hyphal growth by establishing a scaffold at the site of new growth. In contrast, the lipid signal might direct haploid cells to extend from both ends of the cell, and initiation of this polarized cell growth may require septin scaffolds. It is also possible that the cAMP and MAPK pathways play differential roles during different types of filamentous growth.
The dikaryotic hyphal filaments from a Δsep3 a1b1 × Δsep3 a2b2 strain cross in planta did show morphological defects consistent with Sep3 playing a role during hyphal growth in U. maydis. Hyphal filamentation was reduced, and filaments often appeared curved. The CDC11 mutant in C. albicans also displays curved hyphae and a reduction of filamentation in agar (36).
The sep3 gene influences symptom formation, the differentiation of hyphae into teliospores, and teliospore germination.
Maize infected with U. maydis produces disease symptoms, including anthocyanin pigment production and the formation of tumors at sites of infection. Within these tumors, hyphae differentiate into masses of black teliospores in a process that involves a morphological change from cylindrical hyphal cells to rounded teliospores. Hyphae become bloated, produce bulbous, rounded tips, and twist around each other to form what is known as the “worm-like stage.” Hyphae then undergo fragmentation to produce immature teliospores that eventually mature into black melanized spores with echinulated walls (2). Maize infected with the sep3 deletion mutants (a mix of strains of opposite mating types) showed a reduction in disease symptoms, which was exacerbated at lower inoculation density. This decrease is consistent with the morphological defects of Δsep3 cells that may result in decreased proliferation. Dissections of stem tumors on maize seedlings infected with Δsep3 mutants indicated a reduced number of mature teliospores, suggesting that Sep3 plays a role during the differentiation of hyphae into teliospores. This influence was reproducible in multiple independent inoculations of seedlings, but it was not sufficiently dramatic to result in a decrease in visible mature teliospores in infected maize ears. This result could reflect differences in physiological aspects of fungal growth in the host plants that might arise due to the maturity of the plants and/or the difference in infection between floral and vegetative tissue. It is also possible that the septins in U. maydis have some overlapping roles and that the deletion of more than one gene (or possibly all four) would be required to have a dramatic effect on teliospore development. A role of Sep3 during teliospore differentiation would be consistent with the requirement for Sep3 during cellular division. The differentiation of teliospores requires hyphal fragmentation, a process that likely requires multiple rounds of septation. In a similar process, the CDC3 septin homolog in A. nidulans, aspB, is required for the formation of asexual spores (conidia) from a specialized structure termed the conidiophore (37). AspB localizes to the tips of sporogenic cells during the formation of conidia and is likely to be involved in septation events partitioning spores from the progenitor cells. An aspB mutant produces only immature conidiophores that fail to differentiate into spores (37).
Teliospores isolated from maize plants infected with the Δsep3 mutants displayed morphological defects during germination. The germination of teliospores requires the teliospore to break dormancy, extend a promycelium, and undergo meiosis. Following meiosis, the haploid progeny bud off the promycelium, and this process is likely to involve tight regulation of septation. The defects seen in germinating teliospores from cells mutated in sep3 indicate that Sep3 functions during this septation process. Again, one or more of the other septins may also function at this stage of the life cycle.
PKA signaling and septin regulation.
This study was initiated because we observed that the SAGE tag for the sep3 gene had lower abundance in the multibudded ubc1 mutant (16, 24). We initially postulated that the failure of buds to separate from the mother cell in the ubc1 strain could be due in part to a reduction in the amount of Sep3. Consistent with this idea, the Δubc1 sep3 double mutants did not exhibit the multibudded phenotype of the ubc1 single mutant, suggesting that sep3 is essential to generate the multibudded phenotype. Thus, PKA signaling to promote budding may occur partly through the regulation of sep3 expression. However, multibudding was observed in the Δsep3 mutant when a high level of exogenous cAMP was added to the cells, indicating that cAMP/PKA-dependent signaling to promote budding occurs through additional morphogenetic machinery. Also, the Δubc1 sep3 double mutants display a decrease in budding that can be overcome with additional cAMP. Therefore, there are likely to be numerous factors leading to the phenotype of the ubc1 mutant and, consistent with this idea, the role of PKA during U. maydis development is highly pleiotrophic (15). It is also possible that the effect of sep3 expression on the ubc1 mutant is indirect such that Ubc1 might affect the expression of a gene that regulates sep3 at a specific time in development, such as during the differentiation of hyphae into teliospores.
Our observations of wild-type cells growing in various levels of cAMP led to the discovery that cAMP promotes septation at lower concentrations. This influence of cAMP levels on septation fits with the model of PKA regulation of budding versus filamentous growth. Mutation in one of the genes encoding a catalytic subunit of PKA, adr1, causes a constitutively filamentous phenotype, indicating that reduced PKA activity results in filamentous growth. Consistent with this observation, a relatively low level of exogenous cAMP promotes septation at the cell center, a phenomenon that occurs during filamentous growth but not during yeast-like growth. In contrast, when PKA activity (e.g., in a ubc1 mutant) and exogenous cAMP levels are high, budding is promoted and the location of septa is restricted to the mother-bud junction. The ubc1 regulatory PKA mutant often displays septa that are mislocalized in the cell center (Fig. 7A), a phenotype that is consistent with a reduction in sep3 expression. The increase in severity of the Δsep3 morphological phenotype at low cAMP levels also supports the idea that cAMP-dependent regulation of septation requires Sep3. Cultural or nutritional conditions that remediated the phenotype of the Δsep3 haploid mutants were also observed (e.g., on mating medium or in the presence of lipids), and these could potentially impinge on the cAMP/PKA pathway.
In summary, we have shown that the sep3 gene has important roles in the morphogenesis of budding and filamentous cells of U. maydis and that sep3 influences several aspects of the infection process, including symptom formation. The results suggest that further investigation of the influence of PKA on septation, as well as the determination of the roles of the other septins in U. maydis, is warranted.
Acknowledgments
This work was supported by the Canadian Institutes of Health Research and by the Natural Sciences and Engineering Research Council of Canada. J.W.K. is a Burroughs Wellcome Fund Scholar in Molecular Pathogenic Mycology.
REFERENCES
- 1.Andrews, D. L., J. D. Egan, M. E. Mayorga, and S. E. Gold. 2000. The Ustilago maydis ubc4 and ubc5 genes encode members of a MAP kinase cascade required for filamentous growth. Mol. Plant-Microbe Interact. 13:781-786. [DOI] [PubMed] [Google Scholar]
- 2.Banuett, F., and I. Herskowitz. 1996. Discrete developmental stages during teliospore formation in the corn smut fungus, Ustilago maydis. Development 122:2965-2976. [DOI] [PubMed] [Google Scholar]
- 3.Banuett, F., and I. Herskowitz. 1994. Identification of fuz7, a Ustilago maydis MEK/MAPKK homolog required for a-locus-dependent and -independent steps in the fungal life cycle. Genes Dev. 8:1367-1378. [DOI] [PubMed] [Google Scholar]
- 4.Barrett, K. J., S. E. Gold, and J. W. Kronstad. 1993. Identification and complementation of a mutation to constitutive filamentous growth in Ustilago maydis. Mol. Plant-Microbe Interact. 6:274-283. [DOI] [PubMed] [Google Scholar]
- 5.Bi, E., P. Maddox, D. J. Lew, E. D. Salmon, J. N. McMillan, E. Yeh, and J. R. Pringle. 1998. Involvement of an actomyosin contractile ring in Saccharomyces cerevisiae cytokinesis. J. Cell Biol. 142:1301-1312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bölker, M., S. Genin, C. Lehmler, and R. Kahmann. 1995. Genetic regulation of mating and dimorphism in Ustilago maydis. Can. J. Bot. 73:320-325. [Google Scholar]
- 7.Bölker, M., M. Urban, and R. Kahmann. 1992. The a mating type locus of U. maydis specifies cell signaling components. Cell 68:441-450. [DOI] [PubMed] [Google Scholar]
- 8.Christensen, J. J. 1963. Corn smut induced by Ustilago maydis. Am. Phytopathol. Soc. Monogr. 2:1-41. [Google Scholar]
- 9.Davidson, R. C., J. R. Blankenship, P. R. Kraus, M. DeJesus Berrios, C. M. Hull, C. D'Souza, P. Wang, and J. Heitman. 2002. A PCR-based strategy to generate integrative targeting alleles with large regions of homology. Microbiology 148:2607-2615. [DOI] [PubMed] [Google Scholar]
- 10.DeMarini, D. J., A. E. Adams, H. Fares, C. De Virgilio, G. Valle, J. S. Chuang, and J. R. Pringle. 1997. A septin-based hierarchy of proteins required for localized deposition of chitin in the Saccharomyces cerevisiae cell wall. J. Cell Biol. 139:75-93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.De Virgilio, C., D. J. DeMarini, and J. R. Pringle. 1996. SPR28, a sixth member of the septin gene family in Saccharomyces cerevisiae that is expressed specifically in sporulating cells. Microbiology 142:2897-2905. [DOI] [PubMed] [Google Scholar]
- 12.D'Souza, C. A., and J. Heitman. 2001. Conserved cAMP signaling cascades regulate fungal development and virulence. FEMS Microbiol. Rev. 25:349-364. [DOI] [PubMed] [Google Scholar]
- 13.Dürrenberger, F., R. D. Laidlaw, and J. W. Kronstad. 2001. The hgl1 gene is required for dimorphism and teliospore formation in the fungal pathogen Ustilago maydis. Mol. Microbiol. 41:337-348. [DOI] [PubMed] [Google Scholar]
- 14.Dürrenberger, F., K. Wong, and J. W. Kronstad. 1998. Identification of a cAMP-dependent protein kinase catalytic subunit required for virulence and morphogenesis in Ustilago maydis. Proc. Natl. Acad. Sci. USA 95:5684-5689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gold, S., G. Duncan, K. Barrett, and J. W. Kronstad. 1994. cAMP regulates morphogenesis in the fungal pathogen Ustilago maydis. Genes Dev. 8:2805-2816. [DOI] [PubMed] [Google Scholar]
- 16.Gold, S. E., S. M. Brogdon, M. E. Mayorga, and J. W. Kronstad. 1997. The Ustilago maydis regulatory subunit of a cAMP-dependent protein kinase is required for gall formation in maize. Plant Cell 9:1585-1594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hartmann, H. A., R. Kahmann, and M. Bolker. 1996. The pheromone response factor coordinates filamentous growth and pathogenicity in Ustilago maydis. EMBO J. 15:1632-1641. [PMC free article] [PubMed] [Google Scholar]
- 18.Hartwell, L. H. 1971. Genetic control of the cell division cycle in yeast. IV. Genes controlling bud emergence and cytokinesis. Exp. Cell Res. 69:265-276. [DOI] [PubMed] [Google Scholar]
- 19.Holliday, R. 1961. The genetics of Ustilago maydis. Genet. Res. 2:204-230. [Google Scholar]
- 20.Holliday, R. 1974. Ustilago maydis, p. 575-595. In R. C. King (ed.), Handbook of genetics, vol. 1. Plenum, New York, N.Y. [Google Scholar]
- 21.Kahmann, R., T. Romeis, M. Bolker, and J. Kamper. 1995. Control of mating and development in Ustilago maydis. Curr. Opin. Genet. Dev. 5:559-564. [DOI] [PubMed] [Google Scholar]
- 22.Klose, J., M. M. de Sá, and J. W. Kronstad. 2004. Lipid-induced filamentous growth in Ustilago maydis. Mol. Microbiol. 52:823-835. [DOI] [PubMed] [Google Scholar]
- 23.Kronstad, J. W., and C. Staben. 1997. Mating type in filamentous fungi. Annu. Rev. Genet. 31:245-276. [DOI] [PubMed] [Google Scholar]
- 24.Larraya, L. M., K. J. Boyce, A. So, B. R. Steen, S. Jones, M. Marra, and J. W. Kronstad. 2005. Serial analysis of gene expression reveals conserved links between protein kinase A, ribosome biogenesis, and phosphate metabolism in Ustilago maydis. Eukaryot. Cell 4:2029-2043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lee, N., C. A. D'Souza, and J. W. Kronstad. 2003. Of smuts, blasts, mildews, and blights: cAMP signaling in phytopathogenic fungi. Annu. Rev. Phytopathol. 41:399-427. [DOI] [PubMed] [Google Scholar]
- 26.Longtine, M. S., and E. Bi. 2003. Regulation of septin organization and function in yeast. Trends Cell Biol. 13:403-409. [DOI] [PubMed] [Google Scholar]
- 27.Martinez-Espinoza, A. D, J. Ruiz-Herrera, C. G. Leon-Ramirez, and S. E. Gold. 2004. MAP kinase and cAMP signaling pathways modulate the pH-induced yeast-to-mycelium dimorphic transition in the corn smut fungus Ustilago maydis. Curr. Microbiol. 49:274-281. [DOI] [PubMed] [Google Scholar]
- 28.Mayorga, M. E., and S. E. Gold. 1999. A MAP kinase encoded by the ubc3 gene of Ustilago maydis is required for filamentous growth and full virulence. Mol. Microbiol. 34:485-497. [DOI] [PubMed] [Google Scholar]
- 29.Momany, M., and J. E. Hamer. 1997. The Aspergillus nidulans septin encoding gene, aspB, is essential for growth. Fungal Genet. Biol. 21:92-100. [DOI] [PubMed] [Google Scholar]
- 30.Regenfelder, E., T. Spellig, A. Hartmann, S. Lauenstein, M. Bolker, and R. Kahmann. 1997. G proteins in Ustilago maydis: transmission of multiple signals? EMBO J. 16:1934-1942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sambrook, J., E. F. Fritsch, and T. Maniatis. 1989. Molecular cloning: a laboratory manual, 2nd ed. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.
- 32.Schmidt, M., B. Bowers, A. Varma, D. H. Roh, and E. Cabib. 2002. In budding yeast, contraction of the actomyosin ring and formation of the primary septum at cytokinesis depend on each other. J. Cell Sci. 115:293-302. [DOI] [PubMed] [Google Scholar]
- 33.Snetselaar, K. M., and C. W. Mims. 1992. Sporidial fusion and infection of maize seedlings by the smut fungus Ustilago maydis. Mycologia 84:193-203. [Google Scholar]
- 34.Snetselaar, K. M., and C. W. Mims. 1994. Light and electron microscopy of Ustilago maydis hyphae in maize. Mycol. Res. 98:347-365. [Google Scholar]
- 35.Warenda, A. J., S. Kauffman, T. P. Sherrill, J. M. Becker, and J. B. Konopka. 2003. Candida albicans septin mutants are defective for invasive growth and virulence. Infect. Immun. 71:4045-4051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Warenda, A. J., and J. B. Konopka. 2002. Septin function in Candida albicans morphogenesis. Mol. Biol. Cell 13:2732-2746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Westfall, P. J., and M. Momany. 2002. Aspergillus nidulans septin AspB plays pre- and post-mitotic roles in septum, branch, and conidiophore development. Mol. Biol. Cell 13:110-118. [DOI] [PMC free article] [PubMed] [Google Scholar]