Abstract
The ability of Streptococcus pyogenes (group A streptococcus [GAS]) to respond to changes in environmental conditions is essential for this gram-positive organism to successfully cause disease in its human host. The two-component system CovRS controls expression of about 15% of the GAS genome either directly or indirectly. In most operons studied, CovR acts as a repressor. We previously linked CovRS to the GAS stress response by showing that the sensor kinase CovS is required to inactivate the response regulator CovR so that GAS can grow under conditions of heat, acid, and salt stress. Here, we sought to identify CovR-repressed genes that are required for growth under stress. To do this, global transcription profiles were analyzed by microarrays following exposure to increased temperature (40°C) and decreased pH (pH 6.0). The CovR regulon in an M type 6 strain of GAS was also examined by global transcriptional analysis. We identified a gene, rscA (regulated by stress and Cov), whose transcription was confirmed to be repressed by CovR and activated by heat and acid. RscA is a member of the MDR1 family of ABC transporters, and we found that it is required for growth of GAS at 40°C but not at pH 6.0. Thus, for GAS to grow at 40°C, CovR repression must be alleviated so that rscA can be transcribed to allow the production of this potential exporter. Possible explanations for the thermoprotective role of RscA in this pathogen are discussed.
Strains of Streptococcus pyogenes (group A streptococcus [GAS]) are capable of causing very diverse types of infection in the human host. Colonization by this gram-positive organism may lead to mild, self-limiting infections of the throat or skin (pharyngitis and pyoderma, respectively) in one host, whereas the same strain may cause a more serious, invasive disease in another patient. The more severe diseases are caused by the invasion of GAS into deeper tissues and include necrotizing fasciitis, septicemia, and streptococcal toxic shock syndrome, all of which can be life threatening (9).
For GAS to cause disease in such diverse host tissues, this organism must be able to withstand a variety of environmental factors and grow in different microenvironments. GAS can face many stress conditions within the human host, such as the increased salt concentration on the skin, the low pH of the vagina, and the pH fluctuations within the nasopharynx. Also, the bacteria must be able to overcome the stress conditions that arise as a consequence of the GAS infection itself, such as high temperature resulting from fever and decreased pH as a result of abscess formation.
Most bacteria use two-component regulatory systems to sense the various environments they encounter and to alter their gene expression in response to these signals (25, 42). In general, these systems consist of a membrane-bound histidine kinase and a cytoplasmic response regulator. In response to a specific environmental signal, the histidine kinase autophosphorylates and transfers the newly acquired phosphoryl group to an aspartate residue on the response regulator protein. The phosphorylated response regulator undergoes a conformational rearrangement that allows it to bind to the promoter region of target genes, resulting in a change in the level of transcription. Many histidine kinases also have phosphatase activity that catalyzes the reverse reaction; i.e., the phosphate is removed from the response regulator, causing it to release the DNA. The CovRS two-component system has been shown by microarray analysis to regulate ∼15% of the M1 GAS genome, including genes encoding several virulence factors (21). Unlike most response regulators that activate gene expression, CovR functions predominately as a repressor of transcription (16, 21). We have shown previously that this system plays a role in the GAS stress response (10). Inactivation of CovR by its cognate sensor kinase, CovS, is required for growth of GAS under heat, acid, and salt stress. A CovS mutant, in either M6 or M3, does not grow under these stress conditions unless a second site mutation occurs that inactivates CovR, suggesting that CovR represses genes whose products are needed for growth under stress (10). The goal of this work was to identify the CovR-repressed gene or genes responsible for this phenotype.
The best-studied stress response genes in GAS and other gram-positive organisms are those required for growth at elevated temperatures. Heat shock proteins primarily function either as chaperones that promote the refolding of denatured proteins or as proteases that degrade misfolded proteins. Heat shock genes are commonly divided into five classes. The class I heat shock genes, which include the chaperones GroEL and DnaK, are under the control of the repressor HrcA. The class II heat shock genes are transcriptionally regulated by an alternative sigma factor, σB. There are no σB homologs encoded in the GAS genome sequences, so this class of heat shock gene either is not present in GAS or is under different regulatory control (4, 18, 45). Class III heat shock genes, which include the clp ATPases, are repressed by CtsR, which is found in most low-G+C gram-positive organisms (8, 50). Class IV genes are those that are not under the control of HrcA, CtsR, or σB (52). Class V genes are heat stress genes that are under the control of the two-component regulatory system CssRS (11).
Many heat shock genes are also induced in response to acid in several gram-positive bacteria. For example, in addition to being induced by high temperature, acid shock also induces groEL and dnaK in Streptococcus mutans, while in Lactococcus lactis, the levels of both class I- and class III-regulated gene products are increased at low pH (19, 26, 31). In addition to genes regulated by both heat and acid, some genes are specifically upregulated only in acidic conditions. For example, in many species of streptococci, the activities of the F1Fo ATPase and arginine deiminase are induced in response to acid (1, 46, 50). The F1Fo ATPase seems to be critical for the protection of bacteria in acidic environments by exporting excess protons from the cell (40). Arginine deiminase activity leads to the generation of ammonia under acidic conditions to help maintain pH homeostasis (22).
In this work, we have used microarray analysis of transcription to gain insight into the heat and acid stress regulons of GAS. Because we used a different strain of GAS and different growth conditions from those previously used by Graham et al. (21), we also performed microarray analysis to identify the CovR regulon of our M type 6 strain of GAS. The global gene expression data we obtained were used to identify CovR-repressed genes that are activated in response to heat and acid. We demonstrate here that one such gene, which we name rscA (regulated by stress and Cov), is required for optimal growth of GAS at 40°C.
MATERIALS AND METHODS
Bacterial growth conditions and media.
GAS strain JRS4 is a streptomycin-resistant derivative of D471, an M type 6 strain (44). Unless otherwise noted, GAS strains were grown at 37°C without agitation in Todd-Hewitt broth supplemented with 0.2% yeast extract (THY). THY broth buffered with HEPES (pH 7.5) and THY broth buffered with morpholineethanesulfonic acid (MES) (pH 6.0) were made as previously described (10). Antibiotics were used at the following concentrations: chloramphenicol, 5 μg/ml for GAS and 20 μg/ml for Escherichia coli; erythromycin, 0.5 μg/ml for GAS and 250 μg/ml for E. coli; and kanamycin, 200 μg/ml for GAS and 50 μg/ml for E. coli.
Microarray construction.
Our microarrays consist of 2,328 unique 70-mer oligonucleotide probes designed and synthesized by Operon Technologies (Valencia, CA) using a proprietary algorithm to target unique nonrepetitive open reading frames (TIGR annotation as of 2 December 2002) present in the sequenced genomes of a strain of serotype M1 (SF370; 1,932 probes), M3 (MGAS315; 165 probes), and M18 (MGAS8232; 231 probes). Oligonucleotides were spotted in triplicate onto UltraGAPS2 microarray slides (catalog number 40017; Corning) by Microarrays Inc. (Nashville, TN). Slides were rehydrated, cross-linked, and blocked in a prehybridization solution (50% formamide, 5× SSC [1× SSC is 0.15 M NaCl plus 0.015 M sodium citrate], 0.1% sodium dodecyl sulfate [SDS], 0.1 mg/ml bovine serum albumin) according to the manufacturer's instructions (Corning).
cDNA labeling and hybridization conditions.
Total RNA was isolated as described previously (35). Labeled cDNA was generated from RNA samples by incorporation of aminoalloyl-dUTP (Fairplay microarray labeling kit, catalog number 252003; Stratagene) using 3 μg random hexamers (catalog number 48190-011; Invitrogen) in each reaction mixture. The cDNA was then directly labeled with Cy3 or Cy5 monoreactive dyes (catalog numbers PA23001 and PA25001; Amersham) according to the manufacturer's instructions. Labeled cDNA was purified using QiaQuick PCR purification columns (catalog number 28104; QIAGEN) and concentrated using Microcon YM30 columns (catalog number 42410; Amicon). The amount of incorporated label was determined by measuring the absorbance at 550 nm (Cy3) or 650 nm (Cy5).
For each set of microarray experiments, a reference cDNA sample consisted of pooled Cy5-labeled cDNA generated from all RNA samples to be examined. The test samples consisted of cDNA generated from RNA from each condition that was labeled with Cy3 in triplicate and pooled before hybridization. Therefore, the 37°C arrays for the heat shock experiments consisted of Cy5-labeled reference cDNA (pooled cDNA samples from 37°C, 40°C/5 min, 40°C/10 min, and 40°C/20 min) and Cy3-labeled test cDNA (37°C cDNA labeled in triplicate and pooled). For each array, an aliquot of reference cDNA and test cDNA containing 60 pmol each Cy dye was combined in 60 μl hybridization buffer 1 (catalog number 8861; Ambion), heated at 95°C for 5 min, and immediately centrifuged at 12,000 × g for 2 min at room temperature before being applied to the array. The probe was allowed to hybridize to the array at 42°C for at least 16 h. Following hybridization, the arrays were washed once with 2× SSC-0.1% SDS, once with 0.1× SSC-0.1% SDS, and twice with 0.1× SSC.
Microarray data analysis.
The washed arrays were scanned on a GenePix 4000A scanner (Axon instruments). The resulting images were inspected to remove values from bad spots (for example, those affected by scratches) and analyzed using GenePix Pro 3.0 software. For each of the hybridizations, signals below a threshold level (the mean background level of the whole experiment plus 1 standard deviation) were set to the threshold level to avoid spurious expression ratios. The data for each gene from each of three individual spots on the microarray were averaged and treated as a single value for the experiment. The data were then filtered to remove values for genes that were not present in strain JRS4 (the strain examined) as identified independently by hybridization of Cy3-labeled genomic DNA to the array.
The expression value for each gene was then expressed as a fraction of the total amount of expression on each array (global normalization) in order to make comparisons between arrays valid. Normalized data were then log2 transformed, and statistical analysis was performed using Significance Analysis of Microarrays software (49).
Microarray data have been deposited in the Gene Expression Omnibus database (http://www.ncbi.nlm.nih.gov/geo) (accession no. GSE 3367).
Probe construction and RPAs.
Probes (∼250 bp in length) for regions within the coding sequence of indicated genes were constructed as described previously (10). The sequences of primer pairs used to amplify specific probes from the JRS4 chromosome will be provided upon request. Probes were labeled with [α-32P]UTP as previously described (10). T7 polymerase was used in the in vitro transcription reactions for the ska, groEL, ctsR, and atpB probes. SP6 polymerase was used in the in vitro transcription reactions for the clpC, purC, and rscA probes. RNase protection assays (RPAs) were performed as described previously (10). Transcript levels were measured by densitometry using ImageQuant software.
Insertional inactivation of rscA.
An internal region of rscA was PCR amplified from the JRS4 chromosome using primers YdaG-S1 (gccgcgcgaattcTCATGGGTTTGGTGATGCGTCAA) and YdaG-A1 (gccgcgcgaattcTTGGCTCATCATCATTAGGATAT) (lowercase letters indicate bases added; underlined letters are introduced restriction sites). The PCR product was then digested with EcoRI and ligated into EcoRI-digested pSK-Erm, a vector that cannot replicate in GAS (6). The resulting plasmid, pEU7248, was then transformed into electrocompetent JRS4 cells, and erythromycin-resistant transformants were selected. The resulting strain is JRS1819. The insertional inactivation of rscA was confirmed by PCR amplification of the junctions between the chromosome and the plasmid using the following primer pairs: 5′ junction, ggccgggatccATATGCTAACTTATAAAGG (ydaG-F) and GTAAAACGACGGCCAG (M13F); 3′ junction, ggccgctgcagTTTTCATCACTACTCCTC (ydaG-R) and CAGGAAACAGCTATGA (M13R). The same primer sets were used to confirm the rscA insertion mutation following growth of JRS1819 at 40°C.
Construction of the rscA-complementing plasmid pJRS334.
To clone rscA and rscB under the constitutively expressed lactococcal promoter P23 (39), a PCR fragment containing the coding regions of rscA and rscB and their ribosome-binding sites was amplified from JRS4 using primers ydaG-F-EcoRI (ggccgggaattcATATGCTAACTTATAAAGG) and ydaG-R-BamHI (ggccggggatccTTATTCAAAGACAAATTG). To construct pJRS334, the 3.5-kb PCR product was digested with EcoRI and BamHI and ligated into EcoRI/BamHI-digested pJRS7759. To construct pJRS7759, P23 was digested out of pORI23 (39) with PstI/EcoRI and ligated into PstI/EcoRI-digested pLZ12 (A. Benitez and S. Chancey, unpublished data). pLZ12 is a chloramphenicol-resistant shuttle vector that is able to replicate in GAS (13).
Temperature shift growth curves.
Strains were grown at 37°C until they reached the early exponential phase, at which time the cultures were diluted 1:20 in THY that had been prewarmed to 40°C. Growth was monitored with a colorimeter (Klett-Summerson).
RESULTS
Identification of the CovR-regulated stress response genes. (i) Experimental design.
Because we had previously shown that growth of GAS under stress conditions requires CovS to inactivate CovR, we sought to identify the genes regulated by CovR that are responsible for this phenotype (10). We expected that there might be both CovR-repressed genes needed for growth under stress and CovR-activated genes detrimental to growth under stress. To identify these genes, we employed a multistep strategy based on global transcription patterns in GAS. We first used microarrays to define the CovR regulon in GAS. Second, microarrays were used to identify genes differentially expressed in response to exposure to high temperatures or low pH in wild-type strain JRS4. The stress conditions we chose were 40°C and pH 6.0, because GAS is expected to encounter these conditions during an infection and because we had used these conditions previously to characterize the covS mutant (10). A CovS mutant could not be used for these experiments because it does not grow under stress; therefore, we performed our arrays using the wild-type strain. Data acquired from the microarray experiments were used to identify genes that are regulated by both CovR and one or both stress conditions.
(ii) The CovR regulon of strain JRS4.
To identify stress response genes that are repressed by CovR, we first used microarrays to determine the global transcriptional profiles of a wild-type M6 strain (JRS4) and an isogenic strain with a deletion of covR (JRS948) (16). These strains were grown at 37°C in THY broth until late in the exponential growth phase, when covR expression is maximal (16), and total RNA was harvested. To minimize variation that may occur due to differences in culture conditions and RNA isolation procedures, we harvested RNA individually from four independent cultures of both JRS4 and JRS948. All RNA samples were used to make cDNA that was then labeled with the appropriate monoreactive dye. Independent cDNA preparations (test) were hybridized individually to at least two array slides in combination with the reference RNA standard (total pooled RNA). Data were then normalized as described in Materials and Methods and analyzed using Significance Analysis of Microarrays software (49). The genes regulated by CovR in JRS4 are listed in the supplemental material (see Tables S1 and S2 in the supplemental material).
Previously identified, well-studied CovR-repressed genes and operons were shown to be upregulated in the absence of CovR in our arrays, thus validating our results. These genes include ska, sda, and grab as well as the has, sag, and covR operons. However, there are significant differences in the genes identified as being CovR regulated between our results and those previously reported by Graham et al. for an M1 strain (21). Possible explanations for the differences between these two sets of data include differences in growth conditions and the time at which the RNA was harvested (Graham et al. grew their strains in 5% CO2, while we grew ours in ambient air), variations in stringency and/or standardization used for analysis, and strain-specific differences. For example, in our arrays, we found that many phage-associated genes, which are not present in the M1 strain, are activated (either directly or indirectly) by CovR.
(iii) Global effects of high temperature and low pH on gene expression in GAS.
The above-described microarray results indicated that, although expressed at a detectable level, the well-studied stress response genes such as groEL, clpC, and dnaK showed little difference in transcript level in the CovR− strain compared to that in the CovR+ strain (1.1-, 1.2-, and 0.7-fold differences, respectively). Consequently, we felt that it was unlikely that these genes are responsible for the growth defect of covS mutant strains under stress conditions. Therefore, we looked for new genes that were differentially expressed in a wild-type strain following either an increase in temperature or a decrease in pH. For the heat response experiment, wild-type strain JRS4 was grown to early logarithmic phase at 37°C in THY broth, and RNA was isolated from a portion of the culture. The remaining culture was centrifuged at 37°C, and the cell pellet was resuspended in prewarmed THY broth and incubated at 40°C. RNA was isolated from the cells 5, 10, and 20 min after the temperature shift.
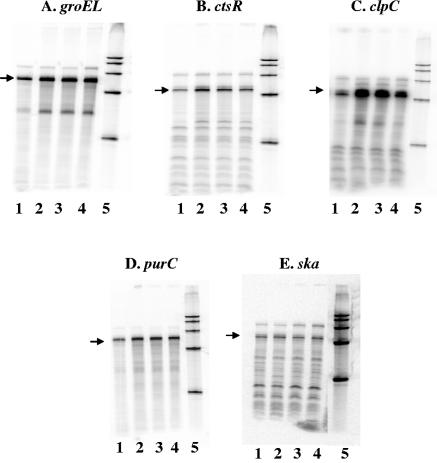
Both heat-activated and heat-repressed genes were identified, and they included those genes predicted to encode proteins involved in such functions as fatty acid biosynthesis, metabolism, regulation, transport, and protein synthesis (data not shown). Because RNA was harvested from only one culture at each time point, these data should not be considered conclusive. However, the array analysis indicated that the well-characterized heat shock genes groEL, ctsR, clpC, and purC were activated at 40°C, and we confirmed this activation in response to heat using RPAs (Fig. 1A to D). The transcript level of groEL increased approximately threefold at 5, 10, and 20 min following the shift to 40°C. The RPAs also showed that ctsR was activated ∼5-fold after 5 min, ∼3-fold after 10 min, and ∼2-fold after 20 min at 40°C compared to that at 37°C. Likewise, transcript levels of clpC increased ∼3-fold at 5 and 10 min, and these levels fell back to ∼2-fold above the 37°C value 20 min following the temperature shift. Under these same conditions, there was a ∼2-fold increase in purC transcript at all 40°C time points. As a control, we also assayed the ska transcript levels by RPA. As expected, we found by both microarray analysis and RPA that ska was not regulated by heat (Fig. 1E).
FIG. 1.
RNase protection assays confirm temperature stress microarray analysis. Fifty micrograms of RNA from strain JRS4 that was grown to early log phase at 37°C (lane 1) and then shifted to 40°C for 5 min (lane 2), 10 min (lane 3), or 20 min (lane 4) was hybridized to [α-32P]UTP-labeled groEL (A), ctsR (B), clpC (C), purC (D), and ska (E) probes. Lane 5 contains a size marker. Arrows indicate the band of the size expected for the hybridization product with each probe.
Microarray experiments were also used to identify the acid stress regulon. To do this, strain JRS4 was grown at 37°C to early logarithmic phase in THY broth buffered with HEPES (pH 7.5), and RNA was harvested. The remaining culture was centrifuged and resuspended in THY broth buffered with MES (pH 6.0). RNA was harvested 5, 10, and 20 min after the shift to pH 6.0 and was used to generate cDNA that was hybridized to two array slides. Genes differentially regulated in response to acid included those predicted to encode proteins involved in protein synthesis, regulation, intermediary metabolism, DNA metabolism, and transport (data not shown).
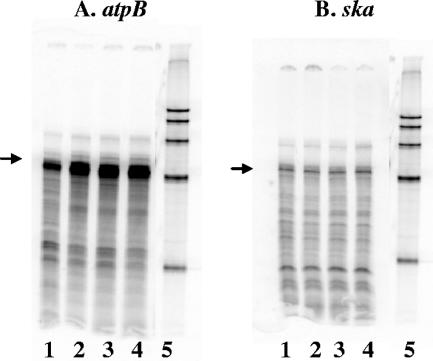
Acid-activated genes included those that encode the F1Fo ATPase, which is a major component of the acid shock response in many organisms (50). The microarray results for one member of this ATPase operon, atpB, were confirmed by RPA using newly isolated RNA (Fig. 2A). Quantitation of the transcript levels indicated in the RPA showed a ∼3-fold increase in atpB transcription levels at 5, 10, and 20 min following the shift to pH 6.0. ska was used again as a control, and expression was unaffected by shifting the culture to low pH as determined both by microarray analysis and RPA (Fig. 2B).
FIG. 2.
RNase protection assays confirm acid stress microarrays. Fifty micrograms of RNA from strain JRS4 that was grown to early log phase at pH 7.5 (lane 1) and then shifted to pH 6.0 for 5 min (lane 2), 10 min (lane 3), or 20 min (lane 4) was hybridized to [α-32P]UTP-labeled atpB (A) and ska (B) probes. Lane 5 contains a size marker. Arrows indicate the band of the size expected for the hybridization product with each probe.
In the microarray analyses, 15 genes, 9 of which appear to be part of operons, were found to be activated by both heat and acid stress. These genes encode proteins homologous to those involved in such functions as transport, DNA metabolism, fatty acid and phospholipid metabolism, purine biosynthesis, and oxidative stress (Table 1).
TABLE 1.
Genes activated by both heat and acid in JRS4
| Locusa | Description (gene)b | Time after shift (min)c
|
|
|---|---|---|---|
| Acid | Heat | ||
| SPy0024 | Phosphoribosylaminoimidazole-succinocarboxamide synthase (purC) | 5, 10 | 5 |
| SPy0025 | Phosphoribosylformylglycinamidine synthase | 5 | 5 |
| SPy0033 | Phosphoribosylaminoimidazole carboxylase (purE) | 10 | 5 |
| SPy0229* | ABC transporter, ATP-binding protein, putative | 5, 10 | 5 |
| SPy0230* | Transport ATP-binding protein, putative | 5, 10 | 5 |
| SPy0570* | Drug transporter, putative | 5, 10, 20 | 5 |
| SPy1135 | GMP reductase (guaC) | 5, 10 | 5 |
| SPy1136 | Xanthine phosphoribosyltransferase (xpt) | 5, 10, 20 | 5, 10 |
| SPy1159 | Predicted membrane protein, hemolysin, putative (hly) | 5 | 5 |
| SPy1216 | Domain of unknown function, putative | 5 | 5 |
| SPy1284 | DNA polymerase III, alpha subunit (dnaE) | 5 | 5 |
| SPy1404* | Hypothetical protein | 10 | 5 |
| SPy1406 | Superoxide dismutase (sodA) | 5, 10, 20 | 5 |
| SPy1746 | (3R)-hydroxymyristoyl-(acyl carrier protein) dehydratase (fabZ) | 20 | 10 |
| SPy2080 | Putative NADH oxidase (nox1) | 10, 20 | 10 |
Locus is listed for GAS M1 strain SF370. Genes with an * represent the genes found to be CovR repressed in JRS4 (this study). Loci in boldface type represent genes previously found by Graham et al. to be CovR repressed in MGAS5005 (21).
Description is the proposed function as annotated by TIGR or by sequence homology identified by BLASTP analysis.
Times after the shift to the stress condition at which the gene is activated.
Classes of CovR-regulated stress response genes.
We examined which of the genes regulated in response to the individual stress conditions were also CovR regulated. A total of 83 CovR-regulated genes were also regulated by one or both of the environmental stresses. The majority of these genes, 66 out of 83, were regulated by only a single stress condition, either heat or acid, but not both. Since our goal was to identify CovR-regulated genes necessary for growth under both stress conditions, we analyzed the remaining 17 genes, some of which appear to be part of operons. These fall into five distinct classes: (i) repressed by CovR and activated under heat and acid stress (SPy0229, SPy0230, SPy0570, and Spy1404), (ii) activated by CovR and repressed by heat and acid stress (SPy0145, SPy0706, SPy0967, and SPyM3_1347), (iii) activated by CovR and activated by heat and acid stress (SPy0033), (iv) repressed by CovR and repressed under heat and acid stress (SPy0165, SPy0166, SPy0167, and SPy0170), and (v) repressed by CovR and activated by heat but repressed by acid (SPy2200, SPy2201, SPy2202, and NT01SP0037).
Since a CovS mutant is unable to grow under either acid or heat stress in the presence of CovR, we reasoned that genes that are activated under both stress conditions might be involved in a response that is repressed by CovR in the absence of stress (10). Thus, we further investigated the members of class 1, the CovR-repressed genes that are activated by both heat and acid stress (Table 1). We identified four genes in this class: SPy0229, SPy0230, SPy0570, and SPy1404. SPy1404 encodes a protein of unknown function with no known homologs. SPy0570 is predicted to encode an exporter because of its homology to members of the major facilitator superfamily, including LmrB of Bacillus subtilis (28% identity and 45% similarity). Transcriptional regulation of SPy0570 by heat, acid, and CovR was confirmed by RPAs (data not shown).
SPy0229 and SPy0230 form an operon and encode products homologous to the MDR1 family of ATP-binding cassette (ABC) transporters. The closest relatives of SPy0229 and SPy0230 appear to be YdaG and YdaB of L. lactis (53% identity and 69% similarity and 55% identity and 72% similarity, respectively), which are also encoded by adjacent genes. In L. lactis, YdaG and YdbA function to export toxic substances such as ethidium bromide and daunomycin (33).
CovR is a repressor of SPy0229, a gene activated in response to temperature and pH.
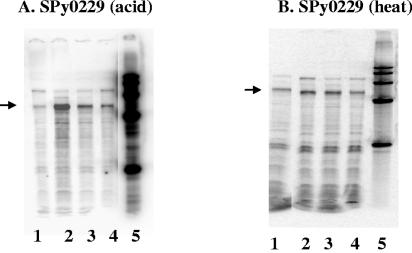
To confirm our microarray results, we performed RPAs with newly isolated RNA from strain JRS4 to look at SPy0229 transcript levels following exposure to increased temperature and decreased pH (Fig. 3). There was a ∼4-fold increase in the amount of SPy0229 transcript following 5 min at pH 6.0, and this level decreased to ∼2-fold after 10 and 20 min at a low pH (Fig. 3A). Similarly, when cells were subjected to 40°C for 5 or 10 min, SPy0229 transcript levels increased (∼1.5-fold) and then began to decrease by 20 min (Fig. 3B). These results confirm that transcription of SPy0229 increases transiently under conditions of heat and acid stress. Transient activation of this type is characteristic of many heat shock genes in other bacteria (43).
FIG. 3.
Transcription of SPy0229 is increased at pH 6.0 and at 40°C. RNase protection assays in which 50 μg of RNA from strain JRS4 was hybridized to the [α-32P]UTP-labeled SPy0229 probe are depicted. (A) RNA from JRS4 was grown to early log phase at pH 7.5 (lane 1) and then shifted to pH 6.0 for 5 min (lane 2), 10 min (lane 3), or 20 min (lane 4). (B) RNA was harvested from JRS4 grown to early log phase at 37°C (lane 1) and then shifted to 40°C for 5 min (lane 2), 10 min (lane 3), or 20 min (lane 4). Lane 5 contains a size marker in both panels. Arrows indicate the band of the size expected for the hybridization product with the SPy0229 probe.
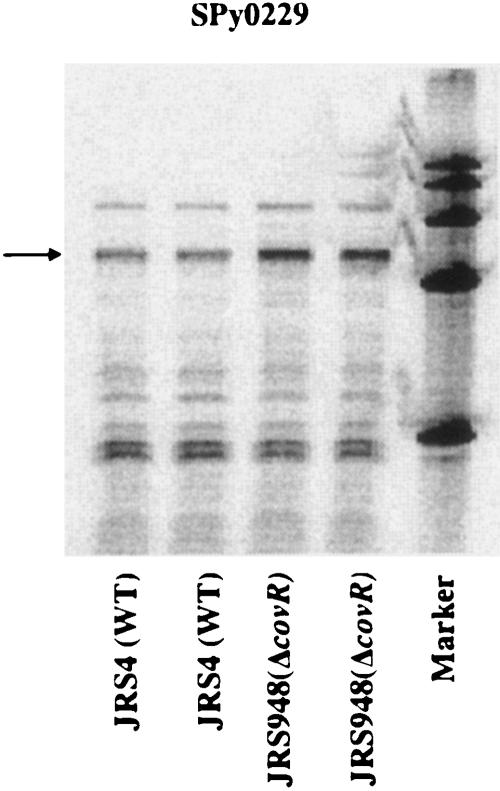
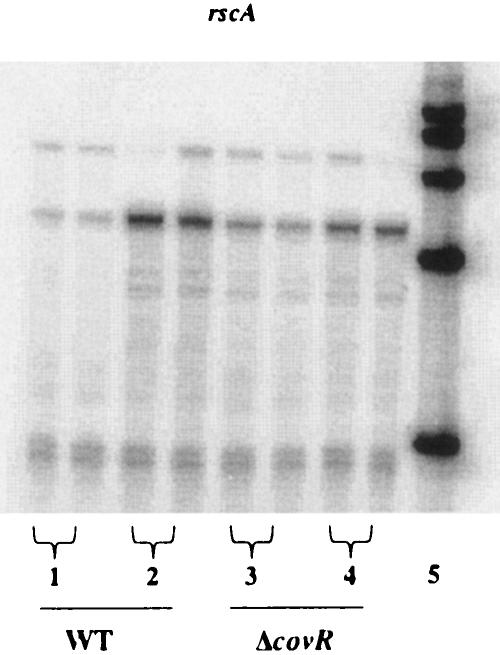
Next, the CovR microarray results were confirmed using an independent RNA preparation. RNA was harvested during the late exponential phase of growth from JRS4, the wild-type strain, and from JRS948, the covR deletion strain, and analyzed by RPA. Figure 4 shows that CovR represses SPy0229 twofold. Therefore, transcription of SPy0229 is activated in response to heat and acid stress and repressed in the presence of the GAS global regulator CovR. For these reasons and based on homologies (see above), we named SPy0229/SPy0230 “rscA/rscB” (regulated by stress and Cov).
FIG. 4.
CovR represses transcription of SPy0229. For the RNase protection assay, 50 μg of RNA from strain JRS4 or JRS948 harvested at the late exponential phase was hybridized in duplicate to an [α-32P]UTP-labeled SPy0229 probe. Arrows indicate the band of the size expected for the hybridization product. WT, wild type.
To determine whether the stress activation of rscA transcription results from the inactivation of CovR, stress induction of this gene in a covR deletion strain (JRS948) was compared to that of its wild-type parent. To do this, RNA was harvested from both JRS4 and JRS948 at pH 7.5 as well as following a 5-min exposure to pH 6.0, and transcript levels of rscA were analyzed by RPA. In the wild-type strain, rscA was induced sevenfold by acid stress, while in the covR deletion strain, rscA was only induced twofold (Fig. 5), which indicates that at low pH, induction of rscA is due mostly to derepression by CovR.
FIG. 5.
Acid-dependent increase in rscA transcript levels is largely CovR dependent. For the RNase protection assay, 50 μg of RNA from strain JRS4 or JRS948 was hybridized in duplicate to an [α-32P]UTP-labeled rscA (SPy0229) probe. Each panel contains RNA from JRS4 (wild type [WT]) grown to early log phase at pH 7.5 (lanes labeled 1) and shifted to pH 6.0 for 5 min (lanes labeled 2) and RNA from JRS948 (ΔcovR) grown to early log phase at pH 7.5 (lanes labeled 3) and shifted to pH 6.0 for 5 min (lanes labeled 4). Lane 5 contains a size marker in both panels. Arrows indicate the band of the size expected for the hybridization product.
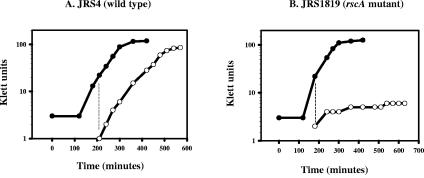
A rscA mutant does not grow at 40°C.
To test whether the rscA product was necessary for growth under stress conditions, we constructed a rscA mutant in JRS4, strain JRS1819, and examined its ability to grow following a shift to 40°C in THY broth. The wild-type strain grew with the same doubling time following the temperature shift, whereas the rscA mutant ceased growing when shifted to 40°C (Fig. 6A and B). No further growth of JRS1819 was detected at 40°C, even when incubation was continued overnight (data not shown). This is similar to the phenotype of a CovS mutant (10).
FIG. 6.
Growth of JRS4 and JRS1819 cultures shifted from 37°C to 40°C. Cultures grown in THY broth at 37°C (closed circles) were diluted and shifted to 40°C at early log phase (open circles). (A) JRS4 (wild type); (B) JRS1819 (rscA mutant). Growth curves were repeated at least twice for each strain with similar results.
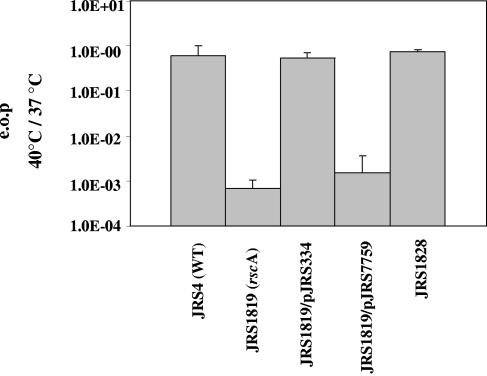
We also examined the ability of the rscA mutant strain to form colonies under stress conditions. Unexpectedly, we found no difference in the efficiency of plating (EOP) between JRS1819 and JRS4 at pH 6.0 (data not shown). The ability of the rscA mutant to grow at pH 6.0 may be due to the presence of another transporter in the cell that provides this function. However, as expected, JRS1819 had a much lower EOP at 40°C than wild-type strain JRS4, approximately 7 × 10−4 (Fig. 7). In addition, a strain with a deletion of the SPy0570 gene had an efficiency of plating similar to that of JRS4 and was thus no longer investigated (data not shown).
FIG. 7.
Efficiency of plating (e.o.p) (rscA). Cultures grown overnight at 37°C in THY broth were plated at 37°C and 40°C and incubated for up to 48 h. Numbers are reported as CFU/milliliter at 40°C divided by CFU/milliliter at 37°C and are the means of at least two experiments, with error bars indicating the standard deviations.
We isolated and reassayed four colonies that formed when JRS1819, the rscA mutant, was grown at 40°C. Three of the four colonies had an EOP at 40°C similar to that of JRS4 (Fig. 7 shows results for one representative survivor mutant, called JRS1828). In these strains, the presence of the rscA insertion mutation was confirmed by PCR. Therefore, most JRS1819-derived colonies that grow at 40°C contain suppressor mutations that allow them to grow.
To be sure that the phenotype attributed to rscA resulted from the insertion mutation, a complementation experiment was performed. The gene directly downstream of rscA, SPy0230 (which we named rscB), has homology with ydbA of L. lactis and is thus likely to form a protein that complexes with RscA to form a functional transporter. The rscA insertion mutation in JRS1819 is likely to be polar. Therefore, we complemented the rscA insertion mutation in JRS1819 with pJRS334, a plasmid that contains both rscA and rscB under the lactococcal P23 promoter (39). We also constructed a control strain by transforming JRS1819 with the same vector without the rscA and rscB genes, pJRS7759. The EOP at 40°C of JRS1819 carrying the vector alone was similar to that of the rscA mutant (Fig. 7). However, the EOP of the complemented strain at 40°C was restored to that of the wild-type strain, confirming that the mutation in rscAB is responsible for the inability of JRS1819 to grow at high temperatures (Fig. 7).
DISCUSSION
In this work, we have identified genes that are regulated by the global regulator CovR and that are likely to belong to the heat and acid stress regulons of GAS. These genes were identified when strain JRS4, an M6 strain, was grown in THY medium in ambient air. This analysis led to the identification of the rscAB operon as both CovR repressed and necessary for growth of this pathogen at 40°C. It seems likely that CovR represses rscAB directly because the promoter for this operon, which we mapped by primer extension (data not shown), contains three consensus CovR binding sites (17), one of which overlaps the −35 sequence.
Stress specificity of CovR-regulated GAS stress response genes.
The purpose of this work was to identify stress genes that are CovR regulated, so we focused our analyses on those genes. Although we anticipated that many CovR-regulated stress response genes would be activated under both acid and heat stress conditions, we found that transcription of most of these genes was affected by only one of the two stress conditions studied. This suggests that the ability of CovR-regulated genes to respond differentially to different stresses may play an important role in the pathogenic potential of the GAS, and the identification of these differentially regulated genes may help identify factors needed at different stages of GAS disease, when the organism is exposed to a variety of conditions in the human host.
The specificity of the stress response of CovR-regulated genes may be mediated by several mechanisms. For instance, an additional stress-activated “pathway” may function to regulate CovR-specific promoters independently of CovR. This was observed to be the case for rscAB, since even in the absence of CovR, transcription is partially activated by lowering the pH (Fig. 5). This second pathway may be mediated by a regulatory protein whose synthesis is stress specific or by the degree of local DNA supercoiling at the promoter. In some bacteria, the degree of local superhelicity can be altered in response to low pH, high temperature, and increased osmolarity, which in turn may alter the level of transcription from certain promoters (2, 3, 5, 15, 24, 27, 30, 48).
In addition, the degree of CovR phosphorylation may be stress dependent. Stress conditions appear to result in the conversion of CovS to a phosphatase that dephosphorylates CovR, leading to the derepression of some CovR-regulated promoters (10). It seems likely that different stresses may affect the phosphatase/kinase ratio of CovS. Thus, the amount of phosphorylated CovR (CovR-P) may depend on the environmental condition in which the GAS is grown. Some promoters appear to have a greater differential affinity for CovR and CovR-P than others (20, 23); thus, the CovR/CovR-P ratio may lead to differential responses of some CovR-regulated promoters to specific stress conditions.
CovR-repressed proteins activated by heat and acid stress have homology to exporters.
To identify genes responsible for the inability of a CovS mutant to grow at 40°C and at pH 6.0, we investigated the four genes that we found to be repressed by CovR and activated in response to both heat and acid. The product of one of these genes, SPy1404, has no homology to known proteins. However, the other two operons identified, rscAB and SPy0570, encode proteins homologous to members of each of the two major exporter protein families: the MDR1 class of the ABC transporter family and the major facilitator superfamily, respectively. Proteins in these two families are usually responsible for multidrug resistance in many different organisms, including gram-positive and gram-negative bacteria and humans (reviewed in reference 38). Furthermore, members of these protein families export unrelated small molecules besides antibiotics and other drugs (38). We found that SPy0570 is not required for growth at high temperature or low pH (data not shown), although it is possible that this may be due to the presence of a functionally redundant protein in GAS. In contrast, the MDR1 homolog RscA, while not required for growth at pH 6.0, is essential for GAS growth at 40°C.
MDR1 exporters.
The members of the MDR1 class of the ABC transporter family mediate export using the energy of ATP hydrolysis. Proteins in this group, including RscA and RscB, contain at least two ATP-binding domains and two transmembrane domains (38). These proteins localize to the cytoplasmic membrane and function as either homodimers or heterodimers to move amphipathic substances including drugs and lipids across this membrane (reviewed in reference 12). The protein encoded by MDR1 itself, P glycoprotein, which has 28% identity and 47% similarity to both RscA and RscB, functions as a homodimer in humans. It exports a diverse assortment of amphipathic drugs, including anthracyclines, actinomycin D, valinomycin, and gramicidin, as well as lipids and steroids (12, 53). This ABC transporter also limits the success of cancer treatment due to its ability to export many drugs used for chemotherapy (32).
Many MDR1 family members are found in bacterial species where they are responsible for inherent resistance to multiple drugs that are not chemically related (37). In gram-negative bacteria, the MDR1 homolog msbA is an essential gene because, in addition to conferring drug resistance, it is responsible for the transport of lipid A, a major outer membrane constituent, across the cytoplasmic membrane (14, 41, 54). It seems likely that all MDR1 family members of different organisms are able to perform the same functions because of their very close structural relationship. Furthermore, the similarity of the MDR1 proteins of bacteria and humans is so great that LmrA from L. lactis can functionally be substituted not only for MsbA of E. coli but also for the human P glycoprotein (41, 51).
Like RscA, some other MDR1 class members have a demonstrated role in the bacterial response to heat stress. For example, MsbA of E. coli was first identified as a multicopy suppressor of a mutant that lacks the HtrB heat shock protein (28, 29). A deletion in htrB leads to the accumulation of lethal amounts of such precursor lipids as tetra-acylated lipid A and glycerophospholipids in the inner membrane at 42°C. The rescue of growth of an htrB mutant by the addition of msbA on a multicopy plasmid at high temperatures suggests that the MsbA exporter facilitates the removal of these precursor lipids from the inner membrane (36, 54). Since the MDR1 family members can export lipids, these transporters may protect the cell from heat stress by transporting lipids with specific fatty acid compositions to yield the appropriate membrane composition and fluidity for growth at high temperatures (7, 34, 47, 54).
In addition to their role in increasing thermotolerance, some MDR1 ABC transporters are involved in resistance to other stresses. For example, in the bacterium Oenococcus oeni, which is responsible for producing the flavors of wine, OmrA, a RscA homolog, is required for resistance of this organism to the low pH, ethanol, tannins, and free fatty acids found in wine. Production of OmrA is induced at high temperatures and by osmotic stress, consistent with its role in the stress response (7).
Potential roles for RscAB in GAS.
Based on their homology to the MDR1 class of ABC transporters, along with the functional similarities between the members of these classes of proteins, we can propose several possibilities for the requirement of RscAB for GAS growth only under stress conditions. For example, like MsbA, RscAB may affect the movement of specific membrane lipids and therefore alter the fatty acid composition of the cytoplasmic membrane. Membrane structure and fluidity appear to be important for cell growth at high temperatures (7, 34, 47, 54).
Alternatively, the need for RscAB only when GAS is subjected to stress conditions might result from a functional requirement for these proteins to export toxic products. Such toxins might either accumulate under specific stress conditions or be produced constitutively during GAS growth but exert toxicity only under stress conditions. If RscAB is needed to export toxic products that are always produced in GAS, a homolog with a redundant function is expected to remove this toxin under nonstress conditions but should be nonfunctional under stress. A candidate operon for this function is SPy1148/SPy1149, which encodes predicted proteins that are 29% identical and 49% (SPy1148) or 50% (SPy1149) similar to RscAB. It is possible that these proteins are inactive at 40°C or are not synthesized at that temperature, making the export of a specific toxin, and thus growth of GAS, dependent on RscAB at 40°C. Likewise, proteins encoded by SPy1148/SPy1149 may be functional at a low pH, thus alleviating the need for RscAB under this condition. Further investigation of the survivors that we isolated when the rscA strain was plated at 40°C should help us understand the role of this exporter in the growth of GAS at high temperatures.
Supplementary Material
Acknowledgments
The oligonucleotide design and syntheses were performed in collaboration with Kevin McIver. We thank Virginia Stringer for her help with various aspects of the RNase protection assays and the construction of strains.
This work was supported by Public Health Service grant R01-AI20723 from the National Institutes of Health. T.L.D. and J.T.C. were supported in part by NIH training grant T32-AI07470.
Footnotes
Supplemental material for this article may be found at http://jb.asm.org/.
REFERENCES
- 1.Abrams, A., and C. Jensen. 1984. Altered expression of the H+ ATPase in Streptococcus faecalis membranes. Biochem. Biophys. Res. Commun. 122:151-157. [DOI] [PubMed] [Google Scholar]
- 2.Adamcik, J., V. Viglasky, F. Valle, M. Antalik, D. Podhradsky, and G. Dietler. 2002. Effect of bacteria growth temperature on the distribution of supercoiled DNA and its thermal stability. Electrophoresis 23:3300-3309. [DOI] [PubMed] [Google Scholar]
- 3.Bang, I. S., J. P. Audia, Y. K. Park, and J. W. Foster. 2002. Autoinduction of the OmpR response regulator by acid shock and control of the Salmonella enterica acid tolerance response. Mol. Microbiol. 44:1235-1250. [DOI] [PubMed] [Google Scholar]
- 4.Beres, S. B., G. L. Sylva, K. D. Barbian, B. Lei, J. S. Hoff, N. D. Mammarella, M. Y. Liu, J. C. Smoot, S. F. Porcella, L. D. Parkins, D. S. Campbell, T. M. Smith, J. K. McCormick, D. Y. Leung, P. M. Schlievert, and J. M. Musser. 2002. Genome sequence of a serotype M3 strain of group A streptococcus: phage-encoded toxins, the high-virulence phenotype, and clone emergence. Proc. Natl. Acad. Sci. USA 99:10078-10083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bhriain, N. N., C. J. Dorman, and C. F. Higgins. 1989. An overlap between osmotic and anaerobic stress responses: a potential role for DNA supercoiling in the coordinate regulation of gene expression. Mol. Microbiol. 3:933-942. [DOI] [PubMed] [Google Scholar]
- 6.Biswas, I., and J. R. Scott. 2003. Identification of rocA, a positive regulator of covR expression in the group A streptococcus. J. Bacteriol. 185:3081-3090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bourdineaud, J. P., B. Nehme, S. Tesse, and A. Lonvaud-Funel. 2004. A bacterial gene homologous to ABC transporters protect Oenococcus oeni from ethanol and other stress factors in wine. Int. J. Food Microbiol. 92:1-14. [DOI] [PubMed] [Google Scholar]
- 8.Chastanet, A., J. Fert, and T. Msadek. 2003. Comparative genomics reveal novel heat shock regulatory mechanisms in Staphylococcus aureus and other gram-positive bacteria. Mol. Microbiol. 47:1061-1073. [DOI] [PubMed] [Google Scholar]
- 9.Cunningham, M. W. 2000. Pathogenesis of group A streptococcal infections. Clin. Microbiol. Rev. 13:470-511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dalton, T. L., and J. R. Scott. 2004. CovS inactivates CovR and is required for growth under conditions of general stress in Streptococcus pyogenes. J. Bacteriol. 186:3928-3937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Darmon, E., D. Noone, A. Masson, S. Bron, O. P. Kuipers, K. M. Devine, and J. M. van Dijl. 2002. A novel class of heat and secretion stress-responsive genes is controlled by the autoregulated CssRS two-component system of Bacillus subtilis. J. Bacteriol. 184:5661-5671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Dean, M., Y. Hamon, and G. Chimini. 2001. The human ATP-binding cassette (ABC) transporter superfamily. J. Lipid Res. 42:1007-1017. [PubMed] [Google Scholar]
- 13.de Vos, W. M. 1986. Genetic improvement of starter streptococci by the cloning and expression of the gene coding for a non-bitter proteinase, p. 465-472. In E. Magnien (ed.), Biomolecular engineering in the European community: achievements of the research programme (1982-1986)—final report. Martinus Nijhoff, Lancaster, England.
- 14.Doerrler, W. T., H. S. Gibbons, and C. R. Raetz. 2004. MsbA-dependent translocation of lipids across the inner membrane of Escherichia coli. J. Biol. Chem. 279:45102-45109. [DOI] [PubMed] [Google Scholar]
- 15.Dorman, C. J. 1996. Flexible response: DNA supercoiling, transcription and bacterial adaptation to environmental stress. Trends Microbiol. 4:214-216. [DOI] [PubMed] [Google Scholar]
- 16.Federle, M. J., K. S. McIver, and J. R. Scott. 1999. A response regulator that represses transcription of several virulence operons in the group A streptococcus. J. Bacteriol. 181:3649-3657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Federle, M. J., and J. R. Scott. 2002. Identification of binding sites for the group A streptococcal global regulator CovR. Mol. Microbiol. 43:1161-1172. [DOI] [PubMed] [Google Scholar]
- 18.Ferretti, J. J., W. M. McShan, D. Ajdic, D. J. Savic, G. Savic, K. Lyon, C. Primeaux, S. Sezate, A. N. Suvorov, S. Kenton, H. S. Lai, S. P. Lin, Y. Qian, H. G. Jia, F. Z. Najar, Q. Ren, H. Zhu, L. Song, J. White, X. Yuan, S. W. Clifton, B. A. Roe, and R. McLaughlin. 2001. Complete genome sequence of an M1 strain of Streptococcus pyogenes. Proc. Natl. Acad. Sci. USA 98:4658-4663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Frees, D., F. K. Vogensen, and H. Ingmer. 2003. Identification of proteins induced at low pH in Lactococcus lactis. Int. J. Food Microbiol. 87:293-300. [DOI] [PubMed] [Google Scholar]
- 20.Gao, J., A. A. Gusa, J. R. Scott, and G. Churchward. 20 September 2005, posting date. Binding of the global response regulator protein CovR to the sag promoter of Streptococcus pyogenes reveals a new mode of CovR-DNA interaction. J. Biol. Chem. [Online.] 10.1074/bc.M506121200. [DOI] [PubMed]
- 21.Graham, M. R., L. M. Smoot, C. A. Migliaccio, K. Virtaneva, D. E. Sturdevant, S. F. Porcella, M. J. Federle, G. J. Adams, J. R. Scott, and J. M. Musser. 2002. Virulence control in group A streptococcus by a two-component gene regulatory system: global expression profiling and in vivo infection modeling. Proc. Natl. Acad. Sci. USA 99:13855-13860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Griswold, A., Y. Y. Chen, J. A. Snyder, and R. A. Burne. 2004. Characterization of the arginine deiminase operon of Streptococcus rattus FA-1. Appl. Environ. Microbiol. 70:1321-1327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gusa, A. A., and J. R. Scott. 2005. The CovR response regulator of group A streptococcus (GAS) acts directly to repress its own promoter. Mol. Microbiol. 56:1195-1207. [DOI] [PubMed] [Google Scholar]
- 24.Higgins, C. F., C. J. Dorman, D. A. Stirling, L. Waddell, I. R. Booth, G. May, and E. Bremer. 1988. A physiological role for DNA supercoiling in the osmotic regulation of gene expression in S. typhimurium and E. coli. Cell 52:569-584. [DOI] [PubMed] [Google Scholar]
- 25.Hoch, J. A. 2000. Two-component and phosphorelay signal transduction. Curr. Opin. Microbiol. 3:165-170. [DOI] [PubMed] [Google Scholar]
- 26.Jayaraman, G. C., J. E. Penders, and R. A. Burne. 1997. Transcriptional analysis of the Streptococcus mutans hrcA, grpE and dnaK genes and regulation of expression in response to heat shock and environmental acidification. Mol. Microbiol. 25:329-341. [DOI] [PubMed] [Google Scholar]
- 27.Karem, K., and J. W. Foster. 1993. The influence of DNA topology on the environmental regulation of a pH-regulated locus in Salmonella typhimurium. Mol. Microbiol. 10:75-86. [DOI] [PubMed] [Google Scholar]
- 28.Karow, M., O. Fayet, A. Cegielska, T. Ziegelhoffer, and C. Georgopoulos. 1991. Isolation and characterization of the Escherichia coli htrB gene, whose product is essential for bacterial viability above 33°C in rich media. J. Bacteriol. 173:741-750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Karow, M., and C. Georgopoulos. 1993. The essential Escherichia coli msbA gene, a multicopy suppressor of null mutations in the htrB gene, is related to the universally conserved family of ATP-dependent translocators. Mol. Microbiol. 7:69-79. [DOI] [PubMed] [Google Scholar]
- 30.Krispin, O., and R. Allmansberger. 1995. Changes in DNA supertwist as a response of Bacillus subtilis towards different kinds of stress. FEMS Microbiol. Lett. 134:129-135. [DOI] [PubMed] [Google Scholar]
- 31.Lemos, J. A., Y. Y. Chen, and R. A. Burne. 2001. Genetic and physiologic analysis of the groE operon and role of the HrcA repressor in stress gene regulation and acid tolerance in Streptococcus mutans. J. Bacteriol. 183:6074-6084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Leonard, G. D., T. Fojo, and S. E. Bates. 2003. The role of ABC transporters in clinical practice. Oncologist 8:411-424. [DOI] [PubMed] [Google Scholar]
- 33.Lubelski, J., P. Mazurkiewicz, R. van Merkerk, W. N. Konings, and A. J. Driessen. 2004. ydaG and ydbA of Lactococcus lactis encode a heterodimeric ATP-binding cassette-type multidrug transporter. J. Biol. Chem. 279:34449-34455. [DOI] [PubMed] [Google Scholar]
- 34.Mansilla, M. C., L. E. Cybulski, D. Albanesi, and D. de Mendoza. 2004. Control of membrane lipid fluidity by molecular thermosensors. J. Bacteriol. 186:6681-6688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.McIver, K. S., and J. R. Scott. 1997. Role of Mga in growth phase regulation of virulence genes of the group A streptococcus. J. Bacteriol. 179:5178-5187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Polissi, A., and C. Georgopoulos. 1996. Mutational analysis and properties of the msbA gene of Escherichia coli, coding for an essential ABC family transporter. Mol. Microbiol. 20:1221-1233. [DOI] [PubMed] [Google Scholar]
- 37.Putman, M., H. W. Van Veen, J. E. Degener, and W. N. Konings. 2000. Antibiotic resistance: era of the multidrug pump. Mol. Microbiol. 36:772-773. [DOI] [PubMed] [Google Scholar]
- 38.Putman, M., H. W. van Veen, and W. N. Konings. 2000. Molecular properties of bacterial multidrug transporters. Microbiol. Mol. Biol. Rev. 64:672-693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Que, Y. A., J. A. Haefliger, P. Francioli, and P. Moreillon. 2000. Expression of Staphylococcus aureus clumping factor A in Lactococcus lactis subsp. cremoris using a new shuttle vector. Infect. Immun. 68:3516-3522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Quivey, R. G., Jr., W. L. Kuhnert, and K. Hahn. 2000. Adaptation of oral streptococci to low pH. Adv. Microb. Physiol. 42:239-274. [DOI] [PubMed] [Google Scholar]
- 41.Reuter, G., T. Janvilisri, H. Venter, S. Shahi, L. Balakrishnan, and H. W. van Veen. 2003. The ATP binding cassette multidrug transporter LmrA and lipid transporter MsbA have overlapping substrate specificities. J. Biol. Chem. 278:35193-35198. [DOI] [PubMed] [Google Scholar]
- 42.Russo, F. D., and T. J. Silhavy. 1993. The essential tension: opposed reactions in bacterial two-component regulatory systems. Trends Microbiol. 1:306-310. [DOI] [PubMed] [Google Scholar]
- 43.Schumann, W. 2003. The Bacillus subtilis heat shock stimulon. Cell Stress Chaperones 8:207-217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Scott, J. R., P. C. Guenthner, L. M. Malone, and V. A. Fischetti. 1986. Conversion of an M− group A streptococcus to M+ by transfer of a plasmid containing an M6 gene. J. Exp. Med. 164:1641-1651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Smoot, J. C., K. D. Barbian, J. J. Van Gompel, L. M. Smoot, M. S. Chaussee, G. L. Sylva, D. E. Sturdevant, S. M. Ricklefs, S. F. Porcella, L. D. Parkins, S. B. Beres, D. S. Campbell, T. M. Smith, Q. Zhang, V. Kapur, J. A. Daly, L. G. Veasy, and J. M. Musser. 2002. Genome sequence and comparative microarray analysis of serotype M18 group A streptococcus strains associated with acute rheumatic fever outbreaks. Proc. Natl. Acad. Sci. USA 99:4668-4673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Takahashi, N., and T. Yamada. 1999. Acid-induced acid tolerance and acidogenicity of non-mutans streptococci. Oral Microbiol. Immunol. 14:43-48. [DOI] [PubMed] [Google Scholar]
- 47.Tolner, B., B. Poolman, and W. N. Konings. 1997. Adaptation of microorganisms and their transport systems to high temperatures. Comp. Biochem. Physiol. A Physiol. 118:423-428. [DOI] [PubMed] [Google Scholar]
- 48.Tse-Dinh, Y. C., H. Qi, and R. Menzel. 1997. DNA supercoiling and bacterial adaptation: thermotolerance and thermoresistance. Trends Microbiol. 5:323-326. [DOI] [PubMed] [Google Scholar]
- 49.Tusher, V. G., R. Tibshirani, and G. Chu. 2001. Significance analysis of microarrays applied to the ionizing radiation response. Proc. Natl. Acad. Sci. USA 98:5116-5121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.van de Guchte, M., P. Serror, C. Chervaux, T. Smokvina, S. D. Ehrlich, and E. Maguin. 2002. Stress responses in lactic acid bacteria. Antonie Leeuwenhoek 82:187-216. [PubMed] [Google Scholar]
- 51.van Veen, H. W., R. Callaghan, L. Soceneantu, A. Sardini, W. N. Konings, and C. F. Higgins. 1998. A bacterial antibiotic-resistance gene that complements the human multidrug-resistance P-glycoprotein gene. Nature 391:291-295. [DOI] [PubMed] [Google Scholar]
- 52.Versteeg, S., A. Escher, A. Wende, T. Wiegert, and W. Schumann. 2003. Regulation of the Bacillus subtilis heat shock gene htpG is under positive control. J. Bacteriol. 185:466-474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Zhou, Y., M. M. Gottesman, and I. Pastan. 1999. Studies of human MDR1-MDR2 chimeras demonstrate the functional exchangeability of a major transmembrane segment of the multidrug transporter and phosphatidylcholine flippase. Mol. Cell. Biol. 19:1450-1459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Zhou, Z., K. A. White, A. Polissi, C. Georgopoulos, and C. R. Raetz. 1998. Function of Escherichia coli MsbA, an essential ABC family transporter, in lipid A and phospholipid biosynthesis. J. Biol. Chem. 273:12466-12475. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.