Abstract
When the filamentous cyanobacterium Anabaena PCC 7120 is exposed to combined nitrogen starvation, 5 to 10% of the cells along each filament at semiregular intervals differentiate into heterocysts specialized in nitrogen fixation. Heterocysts are terminally differentiated cells in which the major cell division protein FtsZ is undetectable. In this report, we provide molecular evidence indicating that cell division is necessary for heterocyst development. FtsZ, which is translationally fused to the green fluorescent protein (GFP) as a reporter, is found to form a ring structure at the mid-cell position. SulA from Escherichia coli inhibits the GTPase activity of FtsZ in vitro and prevents the formation of FtsZ rings when expressed in Anabaena PCC 7120. The expression of sulA arrests cell division and suppresses heterocyst differentiation completely. The antibiotic aztreonam, which is targeted to the FtsI protein necessary for septum formation, has similar effects on both cell division and heterocyst differentiation, although in this case, the FtsZ ring is still formed. Therefore, heterocyst differentiation is coupled to cell division but independent of the formation of the FtsZ ring. Consistently, once the inhibitory pressure of cell division is removed, cell division should take place first before heterocyst differentiation resumes at a normal frequency. The arrest of cell division does not affect the accumulation of 2-oxoglutarate, which triggers heterocyst differentiation. Consistently, a nonmetabolizable analogue of 2-oxoglutarate does not rescue the failure of heterocyst differentiation when cell division is blocked. These results suggest that the control of heterocyst differentiation by cell division is independent of the 2-oxoglutarate signal.
The filamentous cyanobacterium Anabaena sp. PCC 7120 is a simple model for the study of cell differentiation and pattern formation. In response to nitrogen deprivation, 5 to 10% of the cells terminally differentiate within 24 h into nitrogen-fixing heterocysts regularly spaced along each filament (16, 31). Heterocyst differentiation involves integration and coordination of internal and external signals (54). One of the earliest steps of heterocyst differentiation is the accumulation of 2-oxoglutarate, which constitutes a signal of nitrogen starvation (26). NtcA is a global nitrogen regulator necessary for heterocyst differentiation (14, 17, 45). In vitro, 2-oxoglutarate enhances the DNA-binding activity of NtcA in both the unicellular strain Synechococcus PCC 7942 and Anabaena PCC 7120 (26, 42, 44). Specific molecular actors such as HetR or PatS play a central role in the decision of a nitrogen-starved cell to differentiate. The serine-type protease HetR is a DNA-binding protein required for heterocyst differentiation (7, 8, 19, 23, 55), and PatS is thought to be a diffusible inhibitor produced by differentiating cells to suppress heterocyst formation in neighboring cells, possibly by inhibiting HetR activities (19, 23, 49, 51, 52). The hetN gene, whose product is similar to ketoacyl reductase, is involved in the suppression of heterocyst differentiation under conditions of combined nitrogen sufficiency, as well as the maintenance of heterocyst pattern (5, 10). When both patS and hetN are inactivated, the percentage of heterocysts increases as a function of time and reaches a level of near-complete differentiation within 8 days after the removal of ammonium (6). Interestingly, after the first day of induction, clusters of heterocysts appear nonrandomly in this double mutant. This observation suggests that there is another limiting factor for heterocyst differentiation in the absence of PatS and HetN, so that heterocyst differentiation proceeds nonsynchronously along each filament. In addition, how clusters of differentiating cells arise along each filament upon deprivation of combined nitrogen is an unresolved question. One of the possibilities could be the constraint on the ability of each cell to differentiate by its position in the cell cycle or cell lineage.
Several studies suggest that the cell cycle plays a role in heterocyst development. In Anabaena catenula, normal cell division proceeds asymmetrically, and after division both cells reach the same size before they divide again. In this organism, it is always a small daughter cell that becomes a heterocyst (32), and heterocyst differentiation is suggested to occur within the first 8 h following cell division, with a generation time of about 14 h (33). In Anabaena cylindrica, most cell division occurs asymmetrically, and only about 5% of cell division is symmetrical. Exposure to extremely high light intensity results in an increase in the frequency of symmetrical cell division, correlated to the appearance of double heterocysts (1). No asymmetrical cell division has ever been observed for Anabaena PCC 7120.
Bacterial cell division is initiated by FtsZ, a GTPase well conserved in most bacteria, chloroplasts, and the mitochondria of some organisms (13, 37). The polymerization of FtsZ into a ring structure at the mid-cell position triggers a sequential series of events leading to the assembly of the septum and driving the process of cell constriction. The same could be true for cyanobacteria (30, 34). Heterocysts are believed to be terminally differentiated cells unable to divide (47). It has been shown previously that FtsZ becomes undetectable in mature heterocysts (25); a gene, hetC, has been identified whose function could be involved in the arrest of cell division in developing cells (50). In this study, we investigated if heterocyst differentiation may be coupled to cell division, one of the key steps of the bacterial cell cycle. Using ftsZ-gfp fusion, we show here that once the GTPase activity of FtsZ is inhibited in Anabaena PCC 7120, no heterocysts are formed. These results indicate that heterocyst differentiation is tightly coupled to cell division in Anabaena PCC 7120.
MATERIALS AND METHODS
Strains, culture conditions, and measurement of oxygen evolution, 2-oxoglutarate, and cell size.
Escherichia coli strains were grown at 37°C in LB medium in the presence of appropriate antibiotics. Anabaena sp. PCC 7120 and its derivatives were grown in BG11 or BG110 (which lacks sodium nitrate) at 30°C with a light intensity of 15 to 30 μmol of photons m−2 s−1. Under such conditions, proheterocysts were seen 18 h after nitrogen starvation; mature heterocysts with polar nodules of cyanophycin were seen around 24 h after induction. When strains containing the petE promoter were being constructed, we used a modified BG11 or BG110 medium without copper sulfate, and precautions were taken to remove trace amounts of copper, as previously described (8). Conjugation was carried out as previously described (12, 48). The antibiotics aztreonam and cefsulodin were purchased from ICN Biomedicals, Inc.
The measurement of oxygen evolution was carried out as previously described (22). The average cell size was estimated by measuring 400 cells. Measurement of 2-oxogluratae was carried out as previously described (26). 2,2-Difluoro-pentanedioic acid (DFPA), a nonmetabolizable analogue of 2-oxoglutarate, and the strain KGTP, expressing a permease for 2-oxogluratate and DFPA, were described as in Laurent et al. (26).
Microscopy.
A Nikon DXM 1200 digital camera mounted on a Nikon Eclipse E800 microscope was used to capture images. Filaments were visualized under a 100× objective. Confocal microscopy images were acquired with a Zeiss Axiovert 200 M microscope, and the images were deconvoluted with Huygens Essential software (Scientific Volume Imaging Company) and visualized with Imaris software (Bitplane AG Company).
Plasmid constructions.
To construct the pBS-PetE vector, a 490-bp fragment was amplified from the genomic DNA of Anabaena PCC 7120 using primers PetE-f (5′-CTTTCTAGAGGATCCTAAAGCCTGTGAA-3′) and PetE-r (5′-CTTCTGCAGCATATGCGTTCTCCTAACCTGTAGTTT-3′). The fragment that contained a ribosome-binding site and an NdeI site was cloned into pBluescript after digestion with BamHI and PstI. The sulA gene was amplified by PCR with the genomic DNA of E. coli as template and SulA-f (5′-ATTCCATATGTACACTTCAGGCTATGC-3′) and SulA-r (5′-CCGGAATTCTTAATGATACAAATTAGAGTG-3′) as primers. The amplified fragment was digested by NdeI and EcoRI and ligated into pBS-PetE to generate pSS5a. The BamHI-EcoRI or BamHI-HindIII fragment from pSS5a containing the sulA gene under the control of the petE promoter was subcloned into pRL25c (based on the pDU1 replicon) (48) or pRL1272 (digested by BglII and HindIII, a plasmid based on RSF1010 replicon) (56), generating pSS5b or pSS14, respectively. The ftsZ-gfp fusion was constructed in two steps. First, ftsZ was amplified by PCR with Anabaena PCC 7120 genomic DNA as a template and ftsZ-f (5′-GGAATTCCATATGACACTTGATAATAACCA-3′) and ftsZ-r (5′-AAAACTGCAGATTTTTGGGTGGTCGCCG-3′) as primers. The PCR product was digested with NdeI and PstI and ligated into the same sites of pBS-PetE, yielding pSS10a. Second, gfp encoding green fluorescent protein (GFP) was amplified by PCR using pGFPuv (Clontech) as a template and Gfp-f (5′-AAACTGCAGTCTAGACTTGAAATGAGTAAAGGAGAAGAA-3′) and Gfp-r (5′-CGGAATTCTTATTTGTAGAGCTCATC-3′) as primers. The second PCR fragment was digested by PstI and EcoRI and ligated into the same sites of pSS10a, generating pSS10b. The linker sequence is KNLQSRLEMS, where KN are the last two residues of FtsZ and MS are the first two residues of GFP. Finally, the BamHI-EcoRI fragment from pSS10b was subcloned into pRL25c to form pSS10c.
Purification and interaction of FtsZAna and MBP-SulA.
FtsZ of Anabaena PCC 7120 (FtsZAna) was prepared from E. coli BL21(DE3) carrying pET15b-FtsZAna and purified to homogeneity by the procedure described previously (25). The fusion product between the maltose-binding protein (MBP) and SulA was prepared from E. coli DH5α carrying pMal-c-SulA (kindly provided by A. N. Higashitani) (18) and purified to near homogeneity using an amylose resin (New England Biolabs) according to the supplier's instructions.
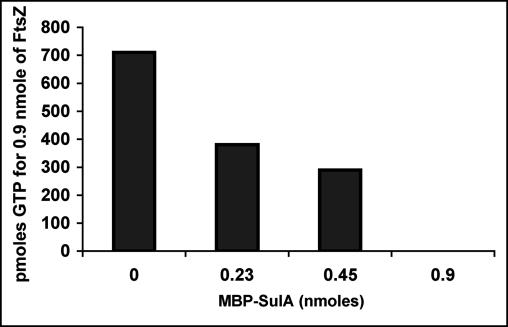
For the GTPase assay, a coupled GTP-regenerating enzyme system was used as described by Margalit et al. (29). A total of 0.9 nmol of FtsZAna was preincubated in the presence of 10 mM GTP without or with MBP-SulA (0.9, 0.45, or 0.225 nmol) for 30 min at 30°C. Preincubated proteins were mixed in a total volume of 360 μl with 3 mM phosphoenolpyruvate, 0.3 mM NADH, 6.5 mM magnesium acetate, 34 mM KCl, 2 mM GTP, 4.17 μg (3 U) lactate dehydrogenase, and 12.5 μg (6 U) pyruvate kinase in 50 mM Tris-HCl (pH 7.5) at 25°C. The decrease in absorbance at 340 nm (oxidation of NADH) was recorded during 30 min. In the regenerating enzyme system, 1 mol of oxidized NADH corresponds to 1 mol of hydrolyzed GTP. Consequently, the amount of hydrolyzed GTP (in picomoles) by FtsZAna was calculated with a molar extinction coefficient for NADH of 6,220 liter−1 mole−1 cm−1.
RESULTS
FtsZAna forms a Z ring at mid-cell position.
FtsZ is conserved in Anabaena PCC 7120 (11, 25, 53). Since little was known about cell division in this organism, we first determined the subcellular localization of FtsZ (thereafter FtsZAna). For this purpose, an ftsZAna-gfp translational fusion under the control of a copper-regulated promoter petE (8) was constructed in the replicative vector pRL25c (48), and the final construct was called pSS10c. The strain expressing ftsZAna-gfp grown in BG11 medium either without copper or with 0.15 μM copper corresponding to that in the normal BG11 medium led to a moderate expression of the fusion that did not affect growth, as measured by following optical density at 700 nm (data not shown). Cell division appeared also normal, since cell size and morphology after continuous subculturing were comparable to those observed for the wild-type strain (Fig. 1A). Visualization of GFP fluorescence in the ftsZAna-gfp-expressing strain under an optical microscope (Fig. 1B) or a confocal microscope (Fig. 1C) showed that FtsZAna-GFP formed a ring structure at mid-cell positions, suggesting that as in other bacteria, FtsZAna drives cytokinesis. Therefore, we tested an approach to impair FtsZ ring formation in Anabaena PCC 7120.
FIG. 1.
Subcellular localization of FtsZAna. The strain containing the plasmid pSS10c bearing an ftsZ-gfp fusion was grown in BG11 for 48 h and visualized under bright light (A) or by epifluorescence excited by light of 465 nm (B). (C) The same results as in panel B are shown but observed with a confocal microscope to show the ring structure of FtsZ in one cell. Bars, 5 μm.
Overexpression of SulA of E. coli prevents FtsZ-ring formation in Anabaena.
In Escherichia coli, SulA is an SOS-inducible protein that inhibits the formation of ring-like structures of FtsZ and, consequently, cell division (4, 18, 20, 21, 36, 43). SulA interacts directly with FtsZ and inhibits the GTPase activity necessary for dynamic polymerization (18, 36, 43). It has been reported recently that proteins similar to SulA were found in cyanobacteria (34). However, the best candidate from Anabaena PCC 7120, All2390, only shares 11% of sequence identity to SulA. In addition, when the sequence of All2390 was used as a probe to search the database, it was a family of proteins of unknown function, rather than SulA, that was found. When all2390 was overexpressed, no clear inhibition of cell division was observed, although alteration in cell morphology was visible (data not shown). Interestingly, as shown below, the expression of sulA of E. coli in Anabaena PCC 7120 inhibited cell division.
The sulA-encoding region from E. coli under the control of the copper-inducible promoter of petE was cloned into pRL1272, a replicative vector in Anabaena PCC 7120 (56). The resulted plasmid was called pSS14. pSS14 and pSS10c bearing a ftsZAna-gfp fusion were based, respectively, on pRL1272 and pRL25c, two compatible replicative plasmids in Anabaena PCC 7120 (48, 56). pSS14 was transferred by conjugation into the strain of Anabaena PCC 7120 containing pSS10c. The culture of this strain in a medium depleted of copper allowed a basal level expression and the formation of a fluorescent FtsZ ring (Fig. 2A and B). Therefore, the basal level expression of sulA did not perturb FtsZ-ring formation. When 0.2 μM copper was added, the FtsZ ring was absent or occasionally very faintly present at mid-cell positions, and a diffuse fluorescence of FtsZAna-GFP was found for most of the observed cells (Fig. 2C and D). Under the same conditions, only background fluorescence due to photosynthetic pigments was found in the wild type (Fig. 2E and F). As a control, FtsZ localization and cell division in a strain containing only pSS10c were not affected by the presence of 0.2 μM copper. These results indicate that the synthesis of E. coli SulA prevents FtsZ-ring formation in vivo.
FIG. 2.
Effect of sulA expression on the formation of FtsZ rings. The strain carrying two compatible plasmids pSS10c (with an ftsZ-gfp fusion) and pSS14 (with the sulA gene) were grown in BG11 without (A and B) or with (C and D) 0.2 μM CuCl2. The same filaments were pictured under bright light (A and C) or under epifluorescence excited by light of 465 nm (B and D). (E and F) Controls representing the wild-type strain grown in BG11 with 0.2 μM CuCl2, pictured under the microscope under the same conditions. The background fluorescence is due to the presence of photosynthetic pigments. Scale bars, 5 μm.
The effects of MBP-SulA (SulA was fused to the carboxyl terminus of the MBP) (18) on the GTPase activity of FtsZAna were examined in vitro (Fig. 3). The GTPase activity of FtsZAna was inhibited when increasing amounts of MBP-SulA were preincubated with FtsZAna (Fig. 3). When the molar ratio of the MBP-SulA fusion to FtsZ was 0.5, GTPase activity of FtsZAna was inhibited by 50%. When this molar ratio was increased to 1, the inhibition was raised to almost 100%. These results indicated that SulA of E. coli inhibited the GTPase activity of FtsZAna with a efficiency similar to that of FtsZ of E. coli. These results were also consistent with the absence of the FtsZ ring when both proteins were coexpressed in Anabaena PCC 7120 (Fig. 2).
FIG. 3.
Effect of SulA on the GTPase activity of FtsZAna. The GTPase activity of FtsZAna was assayed by a coupled GTP-regenerating enzyme system (29). A total of 0.9 nmol of FtsZ was preincubated without or with 0.225, 0.45, or 0.9 nmol of MBP-SulA, and the amount of hydrolyzed GTP was determined.
Inhibition of FtsZ ring formation blocks cell division and suppresses heterocyst differentiation.
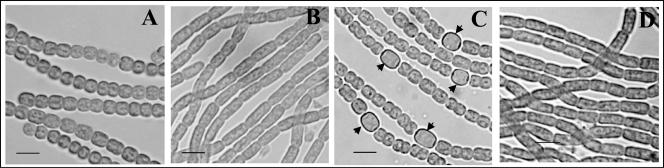
Cell division and heterocyst differentiation of the wild-type filaments were not affected by the presence of 0.2 μM copper (Fig. 4A and C). Heterocyst frequency after 24 h of induction was about 7.8% in the wild type cultured in the normal BG110 medium, whereas it was 7.9% and 7.6% in the wild type cultured in BG110 without copper or with 0.2 μM copper, respectively. Cells overexpressing sulA grown in the presence of 0.2 μM copper were longer in size (Fig. 4B). After induction of sulA expression by copper for 24 h, some cells became longer while others still showed signs of division. After 48 h, all observed cells had lengths longer than normal in the presence of copper (Fig. 4B), indicating that cell division was uniformly blocked. At this stage, the average cell length was 7.1 μm, compared to 3.1 μm for the control. We therefore transferred them into a nitrogen-depleted medium supplemented with copper. No morphologically distinguishable heterocyst was observed after 24 h (Fig. 4D), 48 h, or 4 days of nitrogen step-down (data not shown), showing that differentiation was prevented at least for 4 days. Nitrogen step-down of the sulA-expressing strain in a medium depleted of copper resulted in heterocyst differentiation with a normal frequency and pattern. Thus, when all cells on the filament fail to divide, heterocyst differentiation may not take place normally. These results suggested that the formation of the FtsZ ring at mid-cell positions or cell division in general was necessary for heterocyst development.
FIG. 4.
Inhibition of FtsZ ring formation leads to cell elongation and suppresses heterocyst differentiation. The wild-type strain (A and C) and a strain bearing pSS5b containing the sulA gene (B and D) were grown for 48 h in BG11 containing 0.2 μM CuCl2 (A and B) and then transferred into BG110 with the same amount of CuCl2. At 24 h after the transfer, heterocyst differentiation (indicated by arrows) was visualized (C and D). Bars, 5 μm.
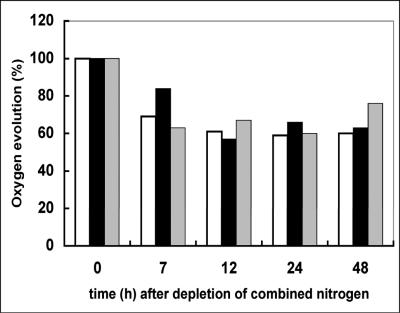
To show that filaments were still metabolically active after failure of cell division, we studied the activity of photosynthesis by measuring the oxygen evolution (Fig. 5). The rate of oxygen evolution in cells overexpressing sulA for 48 h just before the step-down of combined nitrogen was similar to that observed with the wild-type strain (1.24 ± 0.15 nmol oxygen · min−1 · μg chlorophyll−1). During the 2 days following deprivation of combined nitrogen, the changes in the rate of oxygen evolution were also similar to those observed with wild-type cells under similar conditions. These results indicate that the photosynthetic activity of the cells overexpressing sulA is normal in the presence or absence of a source of combined nitrogen.
FIG. 5.
Oxygen evolution of filaments of the wild-type strain, the strain overexpressing sulA, or the wild-type strain treated with the antibiotic aztreonam. The rate of oxygen evolution was measured as previously described (22) using cell suspensions containing to 7 to 10 μg chlorophyll/ml. The rate of oxygen evolution is calculated relative to that obtained just before the depletion of combined nitrogen (time zero). Open bars, wild-type filaments incubated in nitrate-containing BG11 and then transferred into BG110 without nitrate; black bars, wild-type filaments treated with aztreonam for 2 days in BG11 and then transferred into BG110 with aztreonam; dotted bars, wild-type filaments overexpressing sulA cultured in BG11 containing 0.2 μM copper as an inducer of sulA expression and then transferred into BG110 containing the same concentration of copper.
Heterocyst differentiation is independent of the formation of FtsZ rings.
To rule out the possibility that the formation of FtsZ ring itself at mid-cell position was required for heterocyst development, we inhibited septum formation without affecting the formation of the FtsZ ring. In E. coli, a dozen proteins are recruited through a sequential series of events initiated by FtsZ to form the septal ring structure and mediate cytokinesis (13, 46). Among these proteins, a homologue of FtsI (Alr0718) was found in Anabaena PCC 7120 (34). FtsI in E. coli is a penicillin-binding protein required for septal peptidoglycan synthesis, as mutation of its gene abolishes peptidoglycan synthesis at the septum but still supports cell elongation (40, 41). The antibiotic aztreonam specifically inhibits FtsI without impairing other penicillin-binding proteins involved in cell elongation (38).
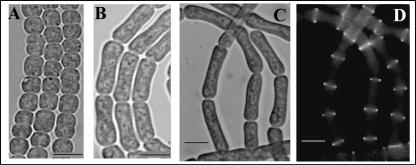
Different concentrations of aztreonam were tested with filaments of Anabaena PCC 7120, and the results showed that the presence of 40 μg · ml−1 aztreonam was efficient to block cell division without cell lysis (data not shown). When wild-type cells were grown for 48 h in the presence of aztreonam, all observed cells were longer (average cell length, 7.4 μm) than normal (average cell length, 3.1 μm) (Fig. 6), indicating that as in E. coli, only septal peptidoglycan synthesis was impaired. These long cells remained viable and metabolically active, since their photosynthetic activity as measured by oxygen evolution was comparable to that of a nontreated culture (Fig. 5), and removal of the antibiotic allowed the cells to divide normally again (see below).
FIG. 6.
Effect of aztreonam on cell division and FtsZ localization. Wild-type Anabaena grown for 48 h in BG11 (A) or BG11 with 40 μg · ml−1 of aztreonam (B) was visualized by bright-field light microscopy. A strain carrying the plasmid pSS10c (with an ftsZ-gfp fusion) was treated with aztreonam for 2 (B) or 4 (C) days and visualized by bright-field light microscopy (B and C) or epifluorescence (D) to show the effect on FtsZ ring formation. Bars, 5 μm.
Owing to the fact that the generation time of the wild-type strain was about 27 h under our growth conditions, 48 h in the presence of aztreonam should block all cells at the same stage, namely, septal ring formation. Nitrogen step-down with cultures previously blocked for 48 h in the presence of aztreonam resulted in the absence of differentiation even after 4 days (data not shown), whereas a normal heterocyst pattern was formed with filaments cultured under similar conditions without aztreonam.
The ftsZAna-gfp-expressing strain grown for 2 or 4 days in the presence of aztreonam clearly showed that the Z-ring formation could still take place under these conditions (Fig. 6C and D) and indicated that the failure of cell differentiation in a strain overexpressing sulA was independent of the formation of FtsZ ring, but rather the result of the blockage of cell division. We have also used the antibiotic cefsulodin, which inhibits penicillin-binding proteins 1a and 1b (9, 27). At a concentration of 30 or 50 μg/ml, the growth of the culture was severely inhibited, but no cell elongation was found. When such a culture was exposed to combined nitrogen step-down, heterocyst differentiation was delayed by 1 or 2 days; afterward, heterocysts were still formed at a frequency comparable to that of the control (Table 1). It appeared that inhibition of cell growth had only a limited impact on heterocyst formation.
TABLE 1.
Effect of cefsulodine on the percentage of heterocyst differentiation
| No. of days after induction | % Heterocysts at:
|
||
|---|---|---|---|
| 0 μg/ml | 50 μg/mla | 75 μg/mla | |
| 1 | 13 | 0 | 0 |
| 2 | 9.5 | 0 | 0 |
| 4 | 6.8 | 6.5 | 1.5 |
| 6 | 5.1 | 5.1 | 4.2 |
Cell growth was measured by spectroscopy at 700 nm; 50 μg of cefsulodine/ml reduced growth by 50%, and 75 μg/ml arrested cell growth almost completely.
Heterocyst differentiation after reinitiation of cell division.
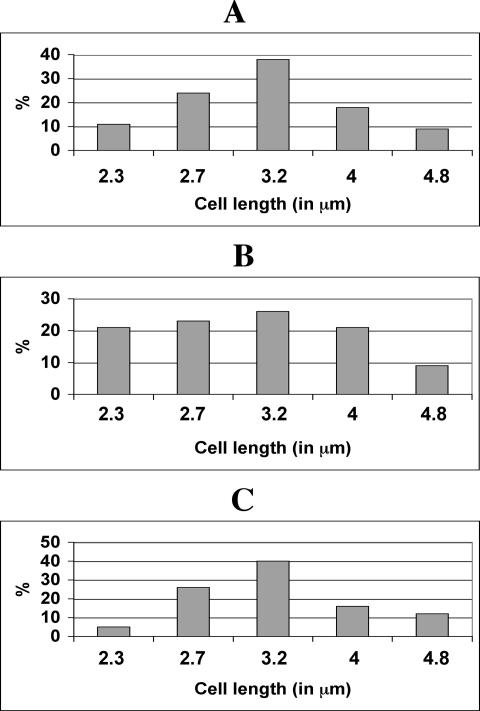
Since sulA overexpression or treatment with aztreonam arrested cell division and suppressed heterocyst differentiation, we studied whether both processes would resume once the inhibitory pressure of cell division was removed. After the filaments were treated with aztreonam for 48 h, the average length of the cells was 7.4 μm, whereas that of untreated cells was 3.1 μm. Filaments were then washed with and resuspended in a medium without antibiotic and observed 24 h later after a period of about one generation. The cell population varied in cell length from 2.3 to 4.8 μm, and cells of small size ranging from 2.3 to 2.7 μm (presumably, those having just completed division) represented 43% of the whole population (Fig. 7B). In comparison, for cell culture at the exponential growth phase in the absence of antibiotic treatment, cells ranging from 2.3 to 2.7 μm represented 35% (Fig. 7A). These results could suggest that once the inhibitor was removed, cell division was not synchronized or was synchronized only in a subpopulation of cells. Some cells formed 24 h after the removal of the antibiotic were much smaller than those normally found, resulting possibly from the localization of FtsZ ring near the pole of some cells treated with the antibiotic (Fig. 6D; Fig. 8B). At 48 h after the removal of aztreonam, cell size analysis gave rise to a Gaussian distribution (Fig. 7C), similar to that of a normal cell population at the exponential growth phase (Fig. 7A).
FIG. 7.
Reinitiation of cell division after removal of aztreonam. (A) A cell population in the exponential phase of growth. (B and C) Cells treated with aztreonam for 48 h and then transferred into a medium without aztreonam for 24 h (B) or 48 h (C) are shown. The lengths of 400 cells for each experiment shown in panels A to C were measured and classified into fractions corresponding to those 2.1 to 2.5 μm in length (labeled as 2.3), those 2.6 to 2.9 μm in length (labeled as 2.7), those 2.9 to 3.5 μm in length (labeled as 3.2), those 3.6 to 4.4 μm in length (labeled as 4), and 4.5 to 5.2 μm in length (labeled as 4.8).
FIG. 8.
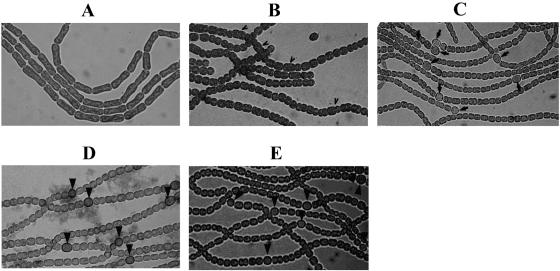
Induction of heterocyst differentiation in the presence and following removal of aztreonam. A culture was treated with aztreonam for 48 h in the presence of nitrate as nitrogen source (A), then aztreonam and nitrate were both removed to induce heterocyst differentiation, and heterocysts were observed 24 h (B) or 48 h (C) after induction. In the control, heterocysts were induced from a culture untreated with aztreonam and observed for 24 h (D) or 48 h (E) after induction. (B and D) Filaments were incubated with Alcian blue, which specifically stains the polysaccharide layer of heterocysts. Some cells, as indicated by arrowheads (B), are still irregular in size; heterocysts are indicated by arrows (C to E).
When aztreonam and the source of combined nitrogen were removed simultaneously, only 0.8% of heterocysts were found after 24 h, compared to 7.8% in the control. To see if proheterocysts could be formed at this stage, filaments were treated with Alcian blue, which specifically stained the polysaccharide layer of heterocysts (15). This treatment did not reveal more cells at the stage of formation of heterocyst envelope layer (Fig. 8). Heterocyst frequency increased to 7.1% at 48 h after the induction, a frequency similar to that found in the wild type (Fig. 8). Since heterocyst development takes about 24 h and generation time is about 24 to 27 h, these results could suggest that the completion of one round of cell division after the removal of aztreonam could be necessary for full heterocyst differentiation to resume, so that a normal heterocyst frequency was found only after 48 h. Similar results were also obtained with the addition and then removal of copper in the sulA overexpression strain (data not shown).
Control of heterocyst differentiation by cell division is independent of 2-oxoglutarate.
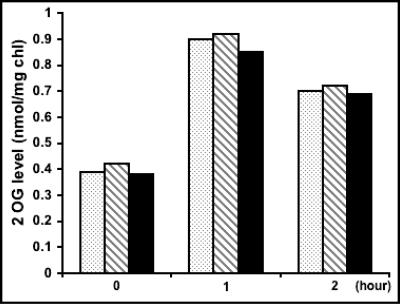
It was shown recently that the levels of 2-oxoglutarate in Anabaena increased by a factor of 2 to 2.5, reaching a maximal level within 1 h following the removal of ammonium as a source of combined nitrogen and then decreasing gradually to the basal level (26). DFPA, a nonmetabolizable analogue of 2-oxoglutarate, added to the KGTP strain expressing a permease of 2-oxoglurate and DFPA could trigger heterocyst differentiation under repressive conditions (26). To determine if the suppression of heterocyst differentiation by the inhibition of cell division was due to the failure of the filaments to accumulate the 2-oxoglutarate signal, the pool of 2-oxoglutarate was measured under different conditions (Fig. 9). In the presence of nitrate, the wild-type strain, the strain bearing the plasmid pSS14 with 0.2 μM copper to induce the expression of sulA for 48 h, and the wild type treated with aztreonam for 48 h to block cell division all showed similar levels of 2-oxoglutarate. During and after the removal of nitrate, copper was always present in the sulA-expressing strain, and aztreonam was still present in the culture previously treated with the same antibiotic. One hour after the removal of nitrate, all three strains displayed an increase in the levels of 2-oxogluratate in a similar manner. Two hours after the removal of nitrate, the levels of 2-oxoglutarate started to decline. These results were consistent with those reported previously (26).
FIG. 9.
The levels of 2-oxoglutarate (2OG) were not affected by inhibition of cell division. The wild-type strain (dotted bars), the strain bearing pSS5b cultured in the presence of copper to induce the expression of sulA for 2 days (striped bars), and the wild-type strain treated with aztreonam for 2 days (black bars) were cultured in the presence of nitrate (time zero) and then shifted to a medium without nitrate for 1 or 2 h. The levels of 2OG were then measured. The results were the means of two experiments.
To know if DFPA could rescue heterocyst formation after inhibition of cell division, the KGTP strain (26) was treated with aztreonam similarly as the wild-type strain, and cell division was also inhibited (data not shown). At 48 h after the addition of the antibiotic, DFPA was added in the presence of ammonium, and the filaments were observed under the microscope. No heterocysts could be seen even after 48 h of incubation with DFPA, while heterocysts could be observed with filaments untreated with aztreonam (data not shown). These results showed that the addition of DFPA did not lead to heterocyst differentiation once cell division was inhibited, and they were consistent with the results obtained with the measurement of the levels of 2-oxoglutarate (Fig. 9).
DISCUSSION
In this study, we show that FtsZ is placed at mid-cell position in Anabaena PCC 7120 as in two unicellular cyanobacteria (Synechocystis sp. PCC 6803 and Synechococcus PCC 7942) (30, 34). Consistent with the arrangement of the division planes in the filamentous strain, FtsZ rings are found in parallel among individual cells along each filament. The ftsZ gene is likely essential in cyanobacteria (30, 34, 53). We were unable to obtain temperature-sensitive ftsZ mutants by replacing conserved residues of FtsZ, which led to conditional mutant alleles in E. coli. We therefore designed an alternative strategy through the controlled expression of sulA, a strong inhibitor of FtsZ. Ectopic expression of All2390, the candidate SulA homologue (34), did not lead to cell elongation, although effects on cell morphology could be observed. These results did not exclude the possibility that All2390 was involved in the regulation of the cell cycle. The level of expression under the control of petE promoter was difficult to control, and the difference between All2390 and SulA of E. coli could be attributed to different amounts of proteins produced with different coding regions. The GTPase activity of FtsZAna could be inhibited almost completely by equimolar concentrations of SulA from E. coli, indicating that SulA inhibited FtsZAna as effectively as it inhibited FtsZ of E. coli. These results were consistent with the effect of sulA expression in vivo, where FtsZAna-GFP was still present but unable to form a ring structure. As a result, cells appeared in elongated form along each filament. The sulA-expressing strain enabled us to assess the effect of an arrest of cell division on heterocyst development. Our results provide evidence that an arrest of cell division suppresses heterocyst development. Similar results were also obtained when FtsI was inhibited by the antibiotic aztreonam, although in this case, the formation of FtsZ rings could still take place. Consistently, once the inhibition of cell division was relieved, cell division resumed before heterocyst differentiation could occur. One possible explanation is that a defect in cell division prevents the cell cycle from proceeding to a stage from which heterocyst differentiation is possible. The completion of cell division could be necessary to relieve the effect of inhibitors of heterocyst development or to enhance the effect of activators of this process. This possibility is consistent with observations of Anabaena catenula and Anabaena cylindrica, in which an early period of the cell cycle could correspond to the competent phase for heterocyst differentiation (1, 33). However, it should be noted that cell division in Anabaena PCC 7120 is symmetrical, in contrast to the asymmetrical division in A. catenula and A. cylindrica.
An alternative explanation is that the arrest of cell division leads to a defect of cell growth, which in turn results in the absence of heterocyst differentiation. There are at least two arguments against this possibility. The first one is that in the strain overexpressing sulA under conditions of nitrogen deprivation, cells continue to grow, as seen by their increased cell length (Fig. 4B) and optical density (data not shown) during the first 24 h of heterocyst induction. This sustained growth may be supported by the degradation of nitrogen reserves such as phycobiliproteins or cyanophycin. Consistently, filaments treated with aztreonam or overexpressing sulA were fully active photosynthetically; once relieved of the inhibition of cell division, they resumed cell division. Secondly, we used the antibiotic cefsulodin, which in E. coli inhibits penicillin-binding proteins 1a and 1b, required for peptidoglycan synthesis during cell elongation (9). The results obtained showed that although cell growth was severely affected under such conditions, heterocyst formation could still occur, although it was delayed by 1 or 2 days. These results are consistent with the possible involvement of two PBP complexes required for cell elongation and septation, respectively (9, 27). In contrast, in a strain overexpressing sulA or inhibited by aztreonam, no heterocyst differentiation was found, even after 4 days. Heterocyst formation is a process of morphogenesis, and these results suggest that it can proceed without much cell growth unless protein or RNA synthesis is inhibited (2).
Two other mutants are reported with impaired cell division in Anabaena PCC 7120, and they were obtained by the inactivation of ftn2 and ftn6 orthologues involved in cell division (24). In these mutants, although some cells elongate and enlarge because of cell division defect, others in the filaments appeared normal. In this case, one would expect that some cells still continue their cell division and thus allow heterocyst development. It is possible that ftn2 and ftn6 have a redundant function in cell division in Anabaena PCC 7120 (24). The level of 2-oxoglutarate was shown to be an early signal of heterocyst development (26). Our data indicated that the inhibition of cell division by the expression of sulA or treatment with aztreonam did not affect the accumulation of 2-oxogluratate, following the deprivation of combined nitrogen. Consistently, the addition of DFPA, a trigger of heterocyst differentiation (26), did not elicit heterocyst differentiation after inhibition of cell division. These results suggest that the control of heterocyst development by the cell cycle is independent of the 2-oxoglutarate signal.
In both prokaryotes and eukaryotes, cell division is tightly regulated during the cell cycle or development (3, 28, 35, 39). In Caulobacter crescentus, where morphogenesis is an integral part of the cell cycle, genes encoding cell division are regulated at multiple levels; the timing of their expression is important for the progression of the cell cycle and development (39). In Bacillus subtilis, cells fail to sporulate without the expression of ftsZ (3), a situation similar to what is observed in the present study. In B. subtilis, FtsZ is necessary for asymmetric cell division in the early phase of sporulation as well as the proper localization of the developmental protein SpoIIE (3, 28). In all these well-studied cases, interaction between components of cell division and those involved in developmental processes constitutes an important mechanism for the coupling between cell division and differentiation. The same could be true for heterocyst development in Anabaena PCC 7120.
Acknowledgments
We thank A. Bernadac for help with confocal microscopy, A. Janicky for technical assistance, and J. W. Golden, C. P. Wolk, and A. N. Higashitani for plasmids and strains.
We acknowledge support from the Lebanese National Council for Scientific Research (S.S., Ph.D. fellowship). This work was financially supported by the CNRS, the ATIP-Microbiologie program and the Environnement et Santé program (AFSSE).
REFERENCES
- 1.Adams, D. G., and N. G. Carr. 1981. Heterocyst differentiation and cell division in the cyanobacterium Anabaena cylindrica: effect of high light intensity. J. Cell Sci. 49:341-352. [DOI] [PubMed] [Google Scholar]
- 2.Adams, D. G., and N. G. Carr. 1989. Control of heterocyst development in the cyanobacterium Anabaena cylindrica. J. Gen. Microbiol. 135:839-849. [Google Scholar]
- 3.Beall, B., and J. Lutkenhaus. 1991. FtsZ in Bacillus subtilis is required for vegetative septation and for asymmetric septation during sporulation. Genes Dev. 5:447-455. [DOI] [PubMed] [Google Scholar]
- 4.Bi, E., and J. Lutkenhaus. 1993. Cell division inhibitor SulA and MinCD prevent formation of the FtsZ ring. J. Bacteriol. 175:1118-1125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Black, T. A., and C. P. Wolk. 1994. Analysis of a Het− mutation in Anabaena sp. strain PCC 7120 implicates a secondary metabolite in the regulation of heterocyst spacing. J. Bacteriol. 176:2282-2292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Borthakur, P. B., C. C. Orozco, S. S. Young-Robbins, R. Haselkorn, and S. M. Callahan. 2005. Inactivation of patS and hetN causes lethal levels of heterocyst differentiation in the filamentous cyanobacterium Anabaena sp. PCC 7120. Mol. Microbiol. 57:111-123. [DOI] [PubMed] [Google Scholar]
- 7.Buikema, W. J., and R. Haselkorn. 1991. Characterization of a gene controlling heterocyst differentiation in the cyanobacterium Anabaena 7120. Genes Dev. 5:321-330. [DOI] [PubMed] [Google Scholar]
- 8.Buikema, W. J., and R. Haselkorn. 2001. Expression of the Anabaena hetR gene from a copper-regulated promoter leads to heterocyst differentiation under repressing conditions. Proc. Natl. Acad. Sci. USA 98:2729-2734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Cabeen, M. T., and C. Jacobs-Wagner. 2005. Bacterial cell shape. Nat. Rev. Microbiol. 3:601-610. [DOI] [PubMed] [Google Scholar]
- 10.Callahan, S. M., and W. J. Buikema. 2001. The role of HetN in maintenance of the heterocyst pattern in Anabaena sp. PCC 7120. Mol. Microbiol. 40:941-950. [DOI] [PubMed] [Google Scholar]
- 11.Doherty, H. M., and D. G. Adams. 1995. Cloning and sequence of ftsZ and flanking regions from the cyanobacterium Anabaena PCC 7120. Gene 163:93-96. [DOI] [PubMed] [Google Scholar]
- 12.Elhai, J., A. Vepritskiy, A. M. Muro-Pastor, E. Flores, and C. P. Wolk. 1997. Reduction of conjugal transfer efficiency by three restriction activities of Anabaena sp. strain PCC 7120. J. Bacteriol. 179:1998-2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Errington, J., R. A. Daniel, and D. J. Scheffers. 2003. Cytokinesis in bacteria. Microbiol. Mol. Biol. Rev. 67:52-65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Frías, J. E., E. Flores, and A. Herrero. 1994. Requirement of the regulatory protein NtcA for the expression of nitrogen assimilation and heterocyst development genes in the cyanobacterium Anabaena sp. PCC 7120. Mol. Microbiol. 14:823-832. [DOI] [PubMed] [Google Scholar]
- 15.Gantar, M., J. Elhai, J. Jia, and M. Ow. 1995. Abstr. 5th Cyanobact. Mol. Biol. Workshop, Pacific Grove, CA, p. 25.
- 16.Golden, J. W., and H. S. Yoon. 2003. Heterocyst development in Anabaena. Curr. Opin. Microbiol. 6:557-563. [DOI] [PubMed] [Google Scholar]
- 17.Herrero, A., A. M. Muro-Pastor, A. Valladares, and E. Flores. 2004. Cellular differentiation and the NtcA transcription factor in filamentous cyanobacteria. FEMS Microbiol. Rev. 28:469-487. [DOI] [PubMed] [Google Scholar]
- 18.Higashitani, A., N. Higashitani, and K. Horiuchi. 1995. A cell division inhibitor SulA of Escherichia coli directly interacts with FtsZ through GTP hydrolysis. Biochem. Biophys. Res. Commun. 209:198-204. [DOI] [PubMed] [Google Scholar]
- 19.Huang, X., Y. Dong, and J. Zhao. 2004. HetR homodimer is a DNA-binding protein required for heterocyst differentiation, and the DNA-binding activity is inhibited by PatS. Proc. Natl. Acad. Sci. USA 101:4848-4853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Huisman, O., and R. D'Ari. 1981. An inducible DNA replication-cell division coupling mechanism in E. coli. Nature 290:797-799. [DOI] [PubMed] [Google Scholar]
- 21.Huisman, O., R. D'Ari, and S. Gottesman. 1984. Cell-division control in Escherichia coli: specific induction of the SOS function SfiA protein is sufficient to block septation. Proc. Natl. Acad. Sci. USA 81:4490-4494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jeanjean, R., S. Bédu, M. Havaux, H. C. P. Matthijs, and F. Joset. 1998. Salt-induced photosystem I cyclic electron transfer restores growth on low inorganic carbon in a type 1 NAD(P)H dehydrogenase deficient mutant of Synechocystis PCC6803. FEMS Microbiol. Lett. 167:131-137. [Google Scholar]
- 23.Khudyakov, I. Y., and J. W. Golden. 2004. Different functions of HetR, a master regulator of heterocyst differentiation in Anabaena sp. PCC 7120, can be separated by mutation. Proc. Natl. Acad. Sci. USA 101:16040-16045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Koksharova, O. A., and C. P. Wolk. 2002. A novel gene that bears a DnaJ motif influences cyanobacterial cell division. J. Bacteriol. 184:5524-5528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kuhn, I., L. Peng, S. Bedu, and C.-C. Zhang. 2000. Developmental regulation of the cell division protein FtsZ in Anabaena sp. strain PCC 7120, a cyanobacterium capable of terminal differentiation. J. Bacteriol. 182:4640-4643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Laurent, S., H. Chen, S. Bédu, F. Ziarelli, L. Peng, and C.-C. Zhang. 2005. Nonmetabolizable analogue of 2-oxoglutarate elicits heterocyst differentiation under repressive conditions in Anabaena sp. strain PCC 7120. Proc. Natl. Acad. Sci. USA 102:9907-9912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Leganés, F., A. Blanco-Rivero, F. Fernández-Pinãs, M. Redondo, E. Fernández-Valente, Q. Fan, S. Lechno-Yossef, and C. P. Wolk. 2005. Wide variation in the cyanobacterial complement of presumptive penicillin-binding proteins. Arch. Microbiol. 184:234-248. [DOI] [PubMed] [Google Scholar]
- 28.Lucet, I., A. Feucht, M. D. Yudkin, and J. Errington. 2000. Direct interaction between the cell division protein FtsZ and the cell differentiation protein SpoIIE. EMBO J. 19:1467-1475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Margalit, D., L. Romberg, R. B. Mets, A. M. Hebert, T. J. Mitchison, M. W. Kirschner, and D. RayChaudhuri. 2004. Targeting cell division: small-molecule inhibitors of FtsZ GTPase perturb cytokinetic ring assembly and induce bacterial lethality. Proc. Natl. Acad. Sci. USA 101:11821-11826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Mazouni, K., F. Domain, C. Cassier-Chauvat, and F. Chauvat. 2004. Molecular analysis of the key cytokinetic components of cyanobacteria: FtsZ, ZipN and MinCDE. Mol. Microbiol. 52:1145-1158. [DOI] [PubMed] [Google Scholar]
- 31.Meeks, J. C., and J. Elhai. 2002. Regulation of cellular differentiation in filamentous cyanobacteria in free-living and plant-associated symbiotic growth states. Microbiol. Mol. Biol. Rev. 66:94-121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Mitchison, G. J., and M. Wilcox. 1972. Rule governing cell division in Anabaena. Nature 239:110-111. [Google Scholar]
- 33.Mitchison, G. J., M. Wilcox, and R. J. Smith. 1976. Measurement of an inhibitory zone. Science 191:866-867. [DOI] [PubMed] [Google Scholar]
- 34.Miyagishima, S.-y., C. P. Wolk, and K. W. Osteryoung. 2005. Identification of cyanobacterial cell division genes by comparative and mutational analyses. Mol. Microbiol. 56:126-143. [DOI] [PubMed] [Google Scholar]
- 35.Mizukami, Y. 2001. A matter of size: developmental control of organ size in plants. Curr. Opin. Plant Biol. 4:533-539. [DOI] [PubMed] [Google Scholar]
- 36.Mukherjee, A., C. Cao, and J. Lutkenhaus. 1998. Inhibition of FtsZ polymerization by SulA, an inhibitor of septation in Escherichia coli. Proc. Natl. Acad. Sci. USA 95:2885-2890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Osteryoung, K. W., and J. Nunnari. 2003. The division of endosymbiotic organelles. Science 302:1698-1704. [DOI] [PubMed] [Google Scholar]
- 38.Pisabarro, A. G., R. Prats, D. Vaquez, and A. Rodriguez-Tebar. 1986. Activity of penicillin-binding protein 3 from Escherichia coli. J. Bacteriol. 168:199-206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Quardokus, E. M., and Y. V. Brun. 2003. Cell cycle timing and developmental checkpoints in Caulobacter crescentus. Curr. Opin. Microbiol. 6:541-549. [DOI] [PubMed] [Google Scholar]
- 40.Spratt, B. G. 1975. Distinct penicillin-binding proteins involved in the division, elongation and shape of Escherichia coli K12. Proc. Natl. Acad. Sci. USA 72:2999-3003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Spratt, B. G. 1977. Temperature sensitive cell division mutants of Escherichia coli with thermolabile penicillin-binding proteins. J. Bacteriol. 131:293-305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Tanigawa, R., M. Shirokane, S. I. Maeda, T. Omata, K. Tanaka, and H. Takahashi. 2002. Transcriptional activation of NtcA-dependent promoters of Synechococcus sp. PCC 7942 by α-ketoglutarate in vitro. Proc. Natl. Acad. Sci. USA 99:4251-4255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Trusca, D., S. Scott, C. Thompson, and D. Bramhill. 1998. Bacterial SOS checkpoint protein SulA inhibits polymerization of purified FtsZ cell division protein. J. Bacteriol. 180:3946-3953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Vázquez-Bermúdez, M. F., A. Herrero, and E. Flores. 2002. α-Ketoglutarate increases the binding affinity of the NtcA (nitrogen control) transcription factor for the Synechococcus glnA promoter. FEBS Lett. 512:71-74. [DOI] [PubMed] [Google Scholar]
- 45.Wei, T. F., T. S. Ramasubramanian, and J. W. Golden. 1994. Anabaena sp. strain PCC 7120 ntcA gene required for growth on nitrate and heterocyst development. J. Bacteriol. 176:4473-4482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Weiss, D. S. 2004. Bacterial cell division and the septal ring. Mol. Microbiol. 54:588-597. [DOI] [PubMed] [Google Scholar]
- 47.Wilcox, M., G. J. Mitchison, and R. J. Smith. 1973. Pattern formation in the blue-green alga Anabaena. II. Controlled proheterocyst regression. J. Cell Sci. 13:637-649. [DOI] [PubMed] [Google Scholar]
- 48.Wolk, C. P., Y. Cai, L. Cardemil, E. Flores, R. Hohn, M. Murry, G. Schmetterer, B. Schrautemeier, and R. Wilson. 1988. Isolation and complementation of mutants of Anabaena sp. strain PCC 7120 unable to grow aerobically on dinitrogen. J. Bacteriol. 170:1239-1244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Wu, X., D. Liu, M. H. Lee, and J. W. Golden. 2004. patS minigenes inhibit heterocyst development of Anabaena sp. strain PCC 7120. J. Bacteriol. 186:6422-6429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Xu, X. D., and C. P. Wolk. 2001. Role for hetC in the transition to a nondividing state during heterocyst differentiation in Anabaena sp. J. Bacteriol. 183:393-396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Yoon, H. S., and J. W. Golden. 1998. Heterocyst pattern formation controlled by a diffusible peptide. Science 282:935-938. [DOI] [PubMed] [Google Scholar]
- 52.Yoon, H. S., and J. W. Golden. 2001. PatS and products of nitrogen fixation control heterocyst pattern. J. Bacteriol. 183:2605-2613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Zhang, C.-C., S. Huguenin, and A. Friry. 1995. Analysis of genes encoding the cell division protein FtsZ and a glutathione synthetase homologue in the cyanobacterium Anabaena PCC 7120. Res. Microbiol. 146:445-455. [DOI] [PubMed] [Google Scholar]
- 54.Zhang, C.-C., S. Laurent, S. Sakr, L. Peng, and S. Bédu. 2006. Heterocyst differentiation and pattern formation in cyanobacteria: a chorus of signals. Mol. Microbiol. 59:367-375. [DOI] [PubMed] [Google Scholar]
- 55.Zhou, R., W. Wei, N. Jiang, H. Li, Y. Dong, K. L. Hsi, and J. Zhao. 1998. Evidence that HetR protein is an unusual serine-type protease. Proc. Natl. Acad. Sci. USA 95:4959-4963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Zhu, J., R. Kong, and C. P. Wolk. 1998. Regulation of hepA of Anabaena sp. strain PCC 7120 by elements 5′ from the gene and by hepK. J. Bacteriol. 180:4233-4242. [DOI] [PMC free article] [PubMed] [Google Scholar]