Abstract
Deletion of the nef gene from simian immunodeficiency virus (SIV) strain SIVmac239 yields a virus that undergoes attenuated growth in rhesus macaques and offers substantial protection against a subsequent challenge with some SIV wild-type viruses. We used a recently described model to identify sites in which the SIVΔnef vaccine strain replicates and elicits immunity in vivo. A high dose of SIVΔnef was applied to the palatine and lingual tonsils, where it replicated vigorously in this portal of entry at 7 days. Within 2 weeks, the virus had spread and was replicating actively in axillary lymph nodes, primarily in extrafollicular T-cell-rich regions but also in germinal centers. At this time, large numbers of perforin-positive cells, both CD8+ T cells and CD3-negative presumptive natural killer cells, were found in the tonsil and axillary lymph nodes. The number of infected cells and perforin-positive cells then fell. When autopsy studies were carried out at 26 weeks, only 1 to 3 cells hybridized for viral RNA per section of lymphoid tissue. Nevertheless, infected cells were detected chronically in most lymphoid organs, where the titers of infectious virus could exceed by a log or more the titers in blood. Immunocytochemical labeling at the early active stages of infection showed that cells expressing SIVΔnef RNA were CD4+ T lymphocytes. A majority of infected cells were not in the active cell cycle, since 60 to 70% of the RNA-positive cells in tissue sections lacked the Ki-67 cell cycle antigen, and both Ki-67-positive and -negative cells had similar grain counts for viral RNA. Macrophages and dendritic cells, identified with a panel of monoclonal antibodies to these cells, were rarely infected. We conclude that the attenuated growth and protection observed with the SIVΔnef vaccine strain does not require that the virus shift its characteristic site of replication, the CD4+ T lymphocyte. In fact, this immunodeficiency virus can replicate actively in CD4+ T cells prior to being contained by the host, at least in part by a strong killer cell response that is generated acutely in the infected lymph nodes.
The simian immunodeficiency virus (SIV), when deleted of its nef gene (SIVΔnef), exhibits two major features in the rhesus macaque. First, virus replication is greatly attenuated in most monkeys (5, 18–20). Second, infection with the Δnef strain of SIVmac239 protects most monkeys against challenge with SIVmac239 and -251 (7, 8, 19, 30). Nevertheless, the SIVΔnef vaccine strain has considerable replicative potential. In vitro, the vaccine strain replicates similarly to wild-type SIV in activated T cells and T cells that are cocultured with mature dendritic cells (17, 22). In vivo, upon vaccination with SIVΔnef, the virus reaches infectious titers that are 1 to 10% of those found with wild-type SIV; these titers then subside to a low set point (5).
We have now investigated the cellular sites of acute and chronic infection with SIVΔnef in vivo to better understand its lower level of replication relative to the wild type and its capacity to act as a vaccine. We wanted to determine if the absence of the nef gene alters the site of SIV replication in vivo from CD4+ T cells to dendritic cells and macrophages and whether this resulted in less active virus replication but more effective immune stimulation. SIV-specific CD4+ (11, 30) and CD8+ (38) immune responses have been documented in the blood during chronic infection with SIVΔnef, and furthermore, temporary depletion of CD8+ T cells results in a rapid increase in viral RNA in the plasma (23). Therefore, infection with this attenuated vaccine strain most likely represents a “battleground” in which there is ongoing virus replication and immune-based resistance, but where is the virus replicating? Chakrabarti et al. showed that infected cells are found in lymphoid tissues (5), but these cells remain to be defined.
To pursue these questions, we have used a recently described system in which infection is initiated by atraumatic application of SIV to the oral mucosa-associated lymphoid tissue (MALT), both the palatine and lingual tonsils (31). It is known that SIV is highly infectious via the oral route (3, 27). By applying virus directly to the tonsil, it becomes feasible to directly analyze the site of primary infection and its spread to other lymphoid organs. Application of vaccines to the oral MALT may also facilitate vaccination of adult and pediatric populations, with the potential to produce stronger mucosal immunity. Elsewhere (unpublished data) we will show that tonsillar application of SIVΔnef in fact provides protection against a subsequent tonsillar challenge with SIVmac251.
Here we studied two animals at early time points, 4 and 7 days after tonsillar application of SIVΔnef, and three animals at both 2 and 26 weeks to identify sites in which the vaccine virus replicates and spreads. During acute infection and spread, many infected cells were evident in lymphoid tissues. CD4+ T cells were clearly the dominant sites for vaccine replication, but in these infected lymph nodes, there was a marked expansion of perforin-positive, cytolytic lymphocytes. Chronically, infectious virus was recovered from most lymphoid tissues, and infected cells were observed in tissue sections. These findings add to the evidence that an infected host can contain a significant level of virus replication in T cells, even after an initial period of active acute SIV replication and spread.
MATERIALS AND METHODS
Animals and virus inoculation.
Juvenile and young adult rhesus monkeys (Macaca mulatta) of Indian origin were bred at the German Primate Centre (Deutsches Primatenzentrum) or imported from the United States (Laboratory Animal Breeder & Services, Yemassee, N.C.). Animal care was in accordance with guidelines of the German Primate Centre. Monkeys were of either sex, had a body weight of 4.5 to 6 kg, and were antibody negative for simian T-lymphotropic virus type 1, simian D-type retrovirus, and SIV. Animals were caged individually and monitored as described (30).
Virus inoculation, physical examination, and bleeding were performed under ketamine anesthesia, while removal of peripheral lymph nodes used deeper anesthesia via a combined injection of ketamine, xylazine, and atropine. The infecting virus was the attenuated SIV nef deletion mutant SIVΔNU, in which 513 bp had been deleted from the nef gene and the U3 region of SIVmac239 (15). For the preparation of our virus stock, supernatant from freshly transfected CEMx174 cells was harvested and used to infect fresh monkey peripheral blood mononuclear cells (PBMCs). The virus stock had an in vitro titer of 106.3 median tissue culture infectious doses (TCID50) in the human T-cell line C81-66. Application of SIVΔNU to the palatine and lingual tonsils was performed as described (31), using ≈105 TCID50.
Determination of virus load and serology.
Cell-associated virus loads were determined in a limiting dilution coculture assay with mononuclear cells from blood and lymphoid organs as described before (30, 31). Viral RNA in plasma was determined by a quantitative RNA-PCR (33). The detection limit of this assay is 40 RNA equivalents per ml of plasma. SIV-specific serum antibodies were detected by Western blotting (32).
In situ hybridization.
The in situ hybridization was performed on either paraffin or cryostat sections as described previously (34). Briefly, 5-μm sections were cut onto slides coated with 3-aminopropyltriethosilane. Frozen sections were fixed in 4% paraformaldehyde for 30 min and subjected to in situ hybridization. Dewaxed paraffin sections were either boiled in a domestic pressure cooker in citrate buffer (pH 6.0) for 5 min or treated with proteinase K (0.01 mg/ml) for 8 min at room temperature and subjected to in situ hybridization to detect viral RNA.
We used a 35S-labeled, single-stranded antisense RNA probe of SIVmac239 (Lofstrand Labs, Gaithersburg, Md.). The probe was composed of fragments of 1.4 to 2.7 kb, which collectively represent approximately 90% of the SIV genome. The specific activity of the probe was 2 × 106 dpm of probe/ml. The hybridization was performed overnight at 45°C in a moist chamber. The slides were washed, digested with RNase (Boehringer Mannheim GmbH, Mannheim, Germany) at 37°C for 40 min, and washed again. Then the slides were dipped into photo emulsion (NTB2; Kodak, Rochester, N.Y.), exposed for 3 to 7 days, developed, counterstained with Hemalaun, and mounted. As a positive control, cytospin preparations of SIV-infected PBMCs were used. As a negative control, sections were hybridized with a 35S-labeled sense probe. The sections were examined with a microscope equipped with epiluminescent illumination (Axiophot; Carl Zeiss Inc., Jena, Germany). Cells were considered positive for viral gene expression if the grain count was more than six times the background.
Immunocytochemistry.
Immunocytochemistry was performed by the alkaline phosphatase-anti-alkaline phosphatase (APAAP) and peroxidase methods. Paraffin-treated or frozen sections were incubated with primary antibodies to defined cellular antigens and perforin as summarized in Table 1. The immunolabeling was performed before in situ hybridization. To identify the perforin-positive cells, immunohistochemical double labeling was made on paraffin-treated or frozen sections. Dewaxed paraffin sections were placed in a domestic pressure cooker containing 1 mM EDTA (pH 8), boiled for 7 min, and chilled to room temperature. The sections were incubated with the primary antibody, anti-CD4 (Novocastra), anti-CD3, or Ki-67, overnight. Binding of anti-CD3 was detected with the peroxidase-antiperoxidase technique, and binding of the other antibodies was detected with the APAAP method using either New Fuchsin as the red chromogen or Fast Blue salt, giving a blue reaction product. The sections were than heat treated again for 15 min and incubated with antibody to perforin overnight, followed by visualization with the APAAP method.
TABLE 1.
Antibodies used in the present study
| Antibody (clone) | Main specificities in lymphoid tissuea | Dilution
|
Sourceb (reference) | |
|---|---|---|---|---|
| Frozen | Paraffin | |||
| CD3 (Leu4) | T cells | 1:50 | Becton Dickinson | |
| CD3 (polyclonal) | T cells | 1:50 | Dakopatts | |
| CD4 (NCL-CD4-1F6) | T helper, DC, macrophages | 1:40 | Novocastra | |
| CD4 (Leu3a) | T helper, DC, macrophages | 1:40 | Becton Dickinson | |
| CD4 (M-T477) | T helper, DC, macrophages | 1:50 | Becton Dickinson | |
| CD8 (Leu2a) | T suppressor/cytotoxic cells | 1:70 | Becton Dickinson | |
| CD8 (CB/144B) | T suppressor/cytotoxic cells | 1:70 | Dakopatts | |
| Perforin (PI-8) | Activated CTL, NK cells | 1:100 | 1:70 | Dakopatts |
| Granzyme B (GrB-7) | Activated CTL, NK cells | 1:20 | Dakopatts | |
| TIA-1 | 15-kDa protein in cytotoxic cells | 1:500 | 1:500 | Coulter |
| CD1a (Leu 6) | Subset of DC and T cells | 1:20 | Becton Dickinson | |
| CD207 (Langerin) | Langerhans-type cells | 1:20 | 1:10 | S. Saeland (35, 36) |
| CD208 (DC-LAMP) | Mature DC | 1:400 | S. Saeland (9) | |
| CD83 (HB15A) | DC, activated lymphocytes | 1:200 | Immunotech | |
| CD68 (KP1) | Macrophages, some DC | 1:70 | 1:20 | Dakopatts |
| CD169 (ID2) | Sialoadhesin, macrophages | 1:200 | P. Crocker (16) | |
| CD20 (L26) | B cells | 1:50 | 1:50 | Dakopatts |
| R4/23 | Follicular DC | 1:30 | Dakopatts | |
DC, dendritic cells; CTL, cytotoxic T cells; NK, natural killer cells.
Becton Dickinson or BD-PharMingen, San Diego, Calif.; Novocastra, Newcastle upon Tyne, United Kingdom; Dakopatts, Copenhagen, Denmark; Coulter Corp, Krefeld, Germany; Immunotech, Marseille, France; S. Saeland, Schering-Plough, Dardilly, France; P. R. Crocker, University of Dundee, Scotland.
The perforin-containing CD8+ T-cell subset was detected on frozen sections. These were fixed in acetone and treated with a ready-to-use peroxidase blocking reagent (Dako) for 10 min. After rinsing in phosphate-buffered saline, the sections were incubated with a mixture of mouse anti-CD8 and rat antiperforin antibodies. The sections were fixed in 4% paraformaldehyde and incubated with a biotinylated rabbit anti-mouse immunoglobulin (Ig) (Dako). After incubation with the StreptAB complex/horseradish peroxidase (Dako), the CD8+ cells were visualized with 3-amino-9-ethylcarbazole (Sigma-Aldrich, St. Louis, Mo.). To detect binding of antiperforin, the sections were incubated with an anti-rat Ig secondary antibody followed by the tertiary antibody (rat APAAP; Dako) and visualized with Fast Blue salt.
RESULTS
Recovery of infectious virus from the lymphoid tissues of monkeys vaccinated with SIVΔnef via the tonsillar route.
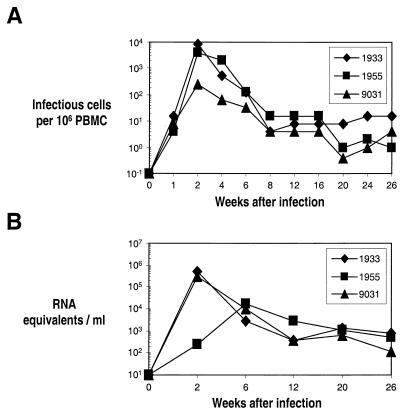
A high dose of SIVΔnef (≈105 infectious units) was applied to the palatine tonsils and the back of the tongue (lingual tonsils) of five animals. Three of five animals underwent peripheral lymph node biopsies (axillary lymph nodes) at 2 weeks and blood sampling at several time points, and the animals were euthanized at 26 weeks. In these monkeys, we observed an acute burst of virus replication (Fig.. 1A), peaking at a level that was similar to the amount of infectious cell-associated virus found in animals infected with wild-type virus. By contrast, peak levels of viral RNA in plasma were about 10% of those measured in SIV wild-type-infected animals (Fig.. 1B). The results with the tonsillar route of vaccination were similar to those observed after intravenous SIVΔnef infection, i.e., a burst of replication followed by a decay in viremia to a low set point (5, 8).
FIG. 1.
Virus load over time in the blood of rhesus macaques after tonsillar infection with SIVΔnef. (A) Cell-associated virus in PBMCs was determined by a limiting-dilution coculture technique, and the endpoint was calculated. Virus loads are expressed as the number of infectious cells per 106 PBMCs. (B) Levels of viral RNA in plasma measured by quantitative reverse transcription-PCR and expressed as RNA equivalents per ml. The detection limit of this assay is 40 RNA equivalents per ml. Individual monkeys are indicated by four-digit numbers.
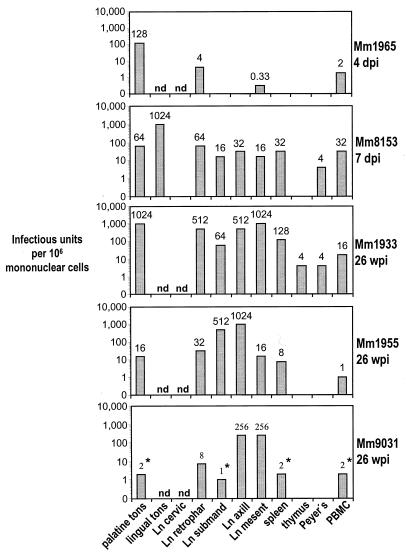
The lymphoid tissues were examined from the three chronically infected animals as well as two monkeys euthanized acutely 4 and 7 days after infection (Fig. 2). In the latter animals, the highest titers of infectious virus were measured primarily at the portal of entry, the tonsils (Fig. 2). Actually, the infection of the tonsils was focal in nature, since separate portions of the tonsil could have very different extents of infection, as assessed by virologic and histologic (in situ hybridization of viral RNA) criteria (not shown). For the chronically infected monkeys assayed at 26 weeks, infectious virus was found in almost all peripheral lymphoid tissues (Fig. 2). The titers of virus in chronically infected animals varied considerably from one organ to another, but it was evident that infection could be comparably active in the tonsillar site of entry and the sites to which the virus had spread. Furthermore, the titers in lymphoid tissues were generally much higher than in blood (compare Fig. 2 with Fig. 1A). We conclude that a high dose of the SIVΔnef vaccine strain undergoes active replication acutely, spreads from one lymphoid organ to another, and replicates chronically at low levels in lymphoid tissues. However, the extent of replication of the vaccine is much greater in lymphoid tissues than in blood.
FIG. 2.
Cell-associated virus load in lymphoid organs and blood of rhesus macaques during acute infection (4 and 7 days postinfection, upper two panels) with attenuated SIVΔnef and in the chronic stage (26 weeks, lower three panels). Mononuclear cell suspensions from each organ and the blood were prepared, and infectious units were determined as described for Fig. 1. Missing bars mean that no virus could be isolated; nd means the organ was not obtained; ne means the culture could not be evaluated because of contamination; the asterisks indicate that the endpoint was not reached. dpi, days postinfection; wpi, weeks postinfection; Ln, lymph node; tons, tonsil; cervic, cervical; retrophar, retropharyngeal; submand, submandibular; axill, axillary; mesent, mesenteric; Peyer’s, Peyer’s patches. Individual monkeys are indicated on the right of each panel by Mm (for Macaca mulatta) and four-digit numbers.
Tempo and spread of the SIVΔnef vaccine strain in sections of lymphoid tissues. (i) Studies at 4 and 7 days.
Following in situ hybridization of tonsil sections with radiolabeled antisense RNA probes, the day 4 animal showed only a few infected cells (Table 2). In contrast, prior studies with much lower doses (about 2%) of wild-type SIV showed active infection at the portal of entry by day 4 (31) (Table 2). This is consistent with the fact that SIVΔnef exhibits attenuated growth properties in vivo (15) even in the highly permissive tonsil. At day 7, extensive infection (19 SIV RNA+ cells/mm2 of tissue) with the vaccine strain was evident both in the germinal centers of the tonsil and in extrafollicular regions (Table 2, Fig.. 3A). Since day 7 is well before the time of seroconversion (3 to 4 weeks), germinal center cells can be infected in the absence of antibody-trapped virus.
TABLE 2.
Frequency of SIVΔnef or SIVmac251 RNA-positive cells in lymphoid tissue
| Tissuea | No. of RNA-positive cells/mm2 of lymphoid tissueb
|
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 7205 (wt, 4 dpi) | 1882 (wt, 7 dpi) | 1965 (Δnef, 4 dpi) | 8153 (Δnef, 7 dpi) | 1933 (Δnef)
|
1955 (Δnef)
|
9031 (Δnef)
|
||||
| 2 wpi | 26 wpi | 2 wpi | 26 wpi | 2 wpi | 26 wpi | |||||
| Palatine tonsil | 18.40 | 15.18 | 0.48 | 19 | 1 | 0.70 | 0 | |||
| Lingual tonsil | 12.13 | 13.5 | 0 | 0.20 | 2.11 | 0.90 | 0 | |||
| Retropharyngeal LN | 0 | 1.80 | 0.27 | 0.25 | 0.90 | |||||
| Submandibular LN | 0 | 1.47 | 0.20 | 0.13 | 0.75 | 0.34 | 0.11 | |||
| Cervical LN | 0.25 | 0 | 0 | 0 | 0.40 | 0.20 | 0 | |||
| Axillary LN | 0.08 | 0.60 | 0.03 | 0.08 | 10.1 | 1.58 | 21.5 | 0.50 | 1.16 | 0.4 |
| Inguinal LN | 0 | 2.43 | 0 | 0.02 | 0.70 | 0.50 | 0.30 | |||
| Mesenteric LN | 0.06 | 1.04 | 0.16 | 0.34 | 0.91 | 0.25 | 0.10 | |||
| Paragastric LN | 0.18 | 0 | 0 | 0 | 0.22 | 0.10 | ||||
| Spleen | 0.05 | 0.55 | 0 | 0.14 | 0.74 | 0.12 | 0.03 | |||
| GALT | 0 | 0.12 | 0 | 0 | 1–2c | 1–2c | 1–2c | |||
LN, lymph node; GALT, gut-associated lymphoid tissue.
The animal number, infecting strain (wt, SIVmac251; Δnef, SIVΔnef), and time postinfection (dpi, days postinfection; wpi, weeks postinfection:) are indicated.
1 to 2 RNA+ cells in four sections.
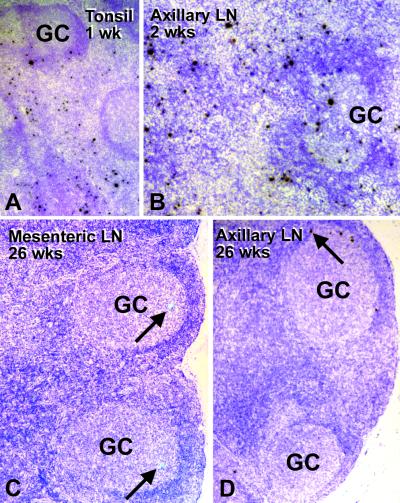
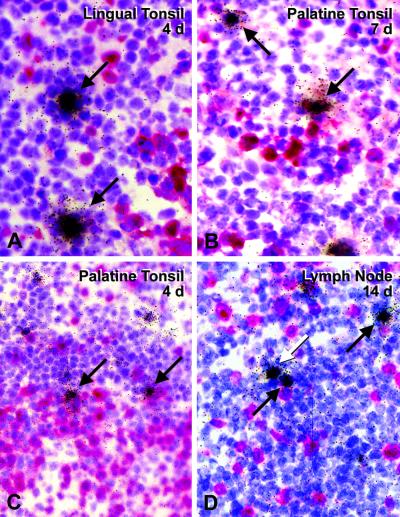
FIG. 3.
Active replication and spread of SIVΔnef following infection via the tonsil. Lymphoid tissues were examined by in situ hybridization with 35S-labeled antisense probes (labeled cells were not found with the control sense probe; not shown). Cells expressing viral RNA appear black (A, B, and D) or green (C). (A) The tonsil during acute infection, 1 week after infection with SIVΔnef. Many RNA -positive cells (black) are in the extrafollicular T-cell-rich regions but are also found in germinal centers (GC). (B) Axillary lymph nodes, to which infection has spread at 2 weeks, contain many infected cells, including in the germinal centers. (C and D) Mesenteric node and auxiliary node 26 weeks after infection contain a few but clear-cut (arrows) RNA-positive cells, typically in the germinal centers.
We observed some spread of SIVΔnef to lymph nodes as early as day 4 by histology and also with the virus isolation coculture technique. However, 10-fold-less virus, based on the number of positive cells by in situ hybridization, was present in the retropharyngeal lymph nodes that drain the tonsil than at the portal of virus entry. More distant lymph nodes, the spleen, and MALT showed either no signs of virus replication or only minimal virus production (<0.1 cell/mm2).
(ii) Studies at 2 weeks.
For the three long-term-infected animals, we took axillary lymph node biopsies at 2 weeks to look for the early spread of infection prior to seroconversion. All three axillary nodes were infected, with two of them having numerous RNA-positive cells by in situ hybridization (Table 2, Fig. 3B). The replication was similar in magnitude to that observed in the tonsil at day 7 (Table 2).
(iii) Studies at 26 weeks.
At autopsy at 26 weeks, infected cells were noted in many peripheral lymphoid tissues, including the spleen and gut. These cells were mainly found in the germinal centers (Table 2 and Fig. 3C and D, arrows). One chronically infected monkey even replicated infectious virus in the thymus at a low level. This animal presented with the highest virus load in lymphoid tissue compared to that observed in the others. No infection was found in the liver, kidneys, or lamina propria of the intestine. The frequency of infected cells in the lymph nodes was much less than that observed acutely at the site of entry (tonsils) or spread (axillary lymph nodes) (compare Fig. 3C and D with Fig. 3A and B). Morphologically, every virus-positive cell resembled a lymphocyte. The remarkable drop in infected CD4+ T cells means that, following application of SIVΔnef, the replication and spread of virus from CD4+ lymphocytes were successfully contained by the host.
Of interest was the nearly complete absence of virus or RNA trapping by follicular dendritic cells. Bound SIVΔnef RNA was absent in the three animals studied at 26 weeks and extensively sampled (tonsils, lymph nodes, spleen, and MALT; Fig. 3C and D). The absence of trapping was in part unexpected, since all animals seroconverted and remained persistently seropositive during the course of this study. Also, plasma virus levels were above the limit of detection (Fig. 1B), and a low level of virus replication was evident in some of the germinal centers (Fig. 3C and D).
Another feature of infection with the SIVΔnef vaccine strain was its focal nature in chronically infected lymph nodes. Occasional germinal centers could have several infected cells, while most others in the field had none (Fig. 3D). This is consistent with prior data that immunodeficiency viruses seed germinal centers under limiting dilution conditions, and then virus spreads to other cells locally within the environment of the germinal center (6).
Identification by immunolabeling of the types of cells infected with SIVΔnef.
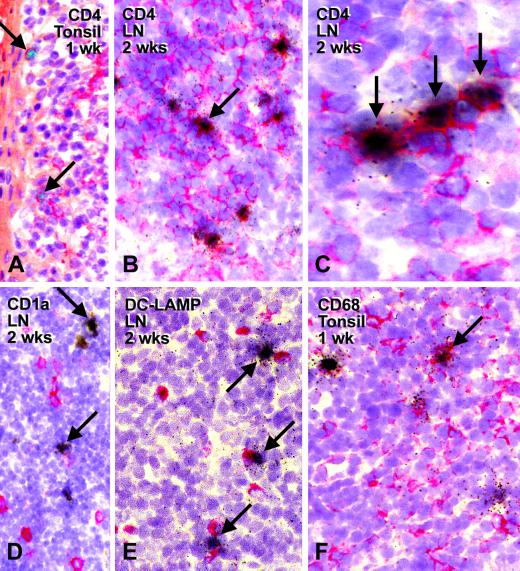
Tissue sections were immunolabeled with a panel of monoclonal antibodies (Table 1) prior to in situ hybridization for SIV RNA. The antibodies recognized T cells (CD4), macrophages (sialoadhesin/CD169 and macrosialin/CD68), and dendritic cells (Langerin/CD207, DC-LAMP/CD208, CD1a, and CD83). At the site of entry, i.e., the crypt lymphoepithelium of the tonsils, the infected cells were CD4+ and had lymphoid morphology (Fig. 4A), which was also the case in the lymph nodes to which the infection had spread acutely at 2 weeks (Fig. 4B and C).
FIG. 4.
SIVΔnef primarily replicates in CD4+ T cells in lymphoid nodes. Tissue sections from lymph node (LN) and tonsil were labeled with the indicated monoclonal antibodies using an immunoperoxidase method (red color) and by in situ hybridization (black) to visualize RNA-positive cells. (A) Two infected CD4+ cells (arrows) in the lymphoepithelium of the acutely infected tonsil at 1 week. (B and C) Low- and high-power views of peripheral lymph nodes at 2 weeks to show labeling of infected cells for the CD4 T-cell marker (arrows). (D to F) Double labeling for CD1a (D), DC-LAMP/CD208 (E), and CD68 (F). Most infected cells are negative. Infected T cells can be found close to noninfected dendritic cells (E, arrows). An occasional CD68-positive cell is infected (F, arrow).
As illustrated in Fig. 4D to F, it was difficult to identify infected cells that were labeled with antibodies to dendritic cells and macrophages, although in rare instances infected cells were found to be labeled for either CD1a, CD68 (Fig.. 4F, arrow) or CD169. Interestingly, infected cells could be juxtaposed to noninfected, more mature dendritic cells carrying the DC-LAMP marker (Fig.. 4E, arrows), consistent with in vitro data that mature dendritic cells are able to drive the replication of SIVΔnef in T cells (17, 22). In summary, the predominant site for replication at the portals of entry and spread of the SIV vaccine strain is the CD4+ T cell, much the same as wild-type SIV (31).
Majority of infected CD4+ T cells do not express the Ki-67 cell cycle antigen.
It has been suggested that the nef gene product facilitates virus replication by increasing the state of cellular activation (1, 14). We explored the activation status of the infected T cells by double labeling the specimens for the cell cycle antigen Ki-67 (13, 28). This antigen produces a strong and discrete nuclear stain. However, wild-type SIV replicated primarily in cells that were not double labeled for Ki-67 (Fig. 5A and B). This confirms prior data with HIV-1 infection (37). The same finding was observed with SIVΔnef. 65 to 70% of the RNA positive cells were not labeled for Ki-67 (Fig. 5C and D) and therefore not clearly in cell cycle at all. In both Ki-67-positive and -negative T cells, the grain counts generated from the 35S-labeled probe ranged from 60 to 300 per cell (white and black arrows, respectively, in Fig.. 5D), indicating that the levels of virus production per infected cell were similar in cycling and noncycling cells. Thus, the absence of a nef gene does not substantially alter the capacity of SIV to replicate in noncycling T cells.
FIG. 5.
SIV wild type (A and B) and SIVΔnef (C and D) often replicate in cells that do not immunolabel for the cell cycle antigen Ki-67 (red). Black arrows indicate Ki-67-negative RNA-infected cells, while white arrows indicate Ki-67-positive infected cells.
Marked expansion of perforin-positive, CD3+, and CD3− cells in acutely infected lymph nodes.
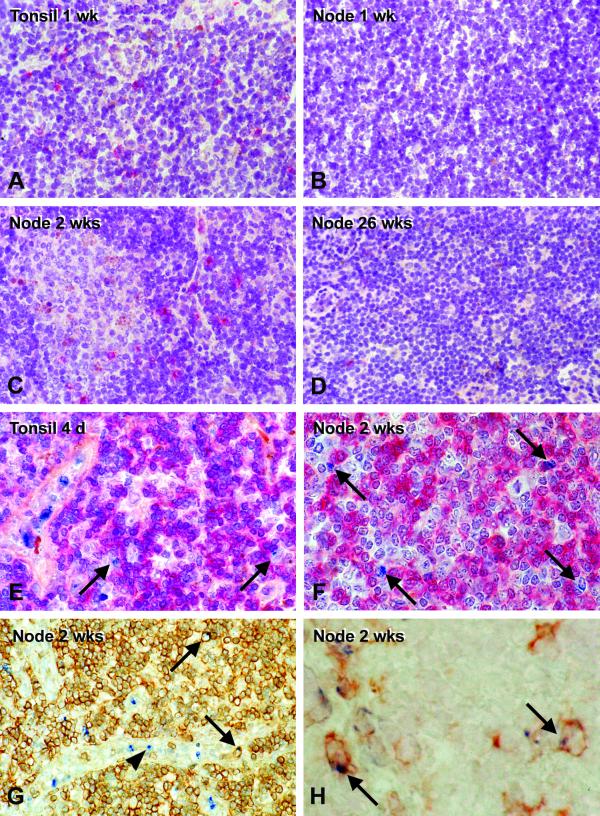
It is known that significant antiviral, CD8+ T-cell responses develop during infection with SIVΔnef (23). To gain evidence for such a response in vivo, we stained the specimens for lysosomal granules containing the pore-forming protein perforin, a key component of CD8+ cytolytic T lymphocytes and natural killer (NK) cells. Perforin-positive lymphocytes were rare in the lymph node and the tonsil of uninfected macaques. In contrast, perforin-positive lymphocytes were abundant (7 to 14 cells per field with a 40× objective) in the acutely infected tonsil (day 7, Fig. 6A) but not in the poorly infected retropharyngeal lymph node at that time (Fig. 6B). When the lymph nodes became infected (week 2), perforin-positive cells were abundant, including in germinal centers (Fig. 6C). At later time points (26 weeks), perforin-positive cells were difficult to detect (Fig. 6D). Therefore, it appears that lytic cells are most abundant early in response to the vaccine strain.
FIG. 6.
Detection of perforin-positive lymphocytes in tissue sections following tonsillar infection of rhesus macaques with SIVΔnef. Single-color immunolabeling (red) at 1 week in the heavily infected tonsil (A) and weakly infected retropharyngeal lymph node (B) and at 2 weeks (C) and 26 weeks (D) in strongly infected and weakly infected nodes. Two-color immunolabeling, with perforin in blue, shows the cells to be CD4 negative (red color in E and F), either CD3+ (arrows, G) or CD3− (arrowheads, G), or CD8+ (arrows, H).
We assessed the lineage of the perforin-positive cells by immunohistochemical double labeling. The majority of perforin-positive cells in the acutely infected tonsil (Fig. 6E) and lymph nodes (Fig. 6F) did not display CD4. However, a significant percentage (25 to 32%) were CD3+ (arrows, Fig. 6G), suggesting that they were cytolytic T lymphocytes. Likewise, about 30% of the perforin-positive cells double labeled for the CD8 killer T-cell marker (arrows, Fig. 6H). Most perforin-positive cells were CD3 negative and were most likely NK cells (Fig. 6G).
Additional findings that are not shown are as follows. Perforin-positive cells were similarly abundant in the infected lymphoid tissues of animals given wild-type SIVmac251 through the tonsillar route (31), again within CD3-negative and CD3+ lymphocyte subsets. In both wild-type- and SIVΔnef vaccine strain-infected monkeys, some of the perforin-positive cells were double labeled for Ki-67, the cell cycle antigen. We found many perforin-positive cells in the lumen of the high endothelial venules of the T-dependent zone as well as the medullary sinuses, indicating that the killer cells were able to migrate out of the lymphoid tissues into the blood. These results indicate that there is a strong cytolytic cell response in the acutely infected lymphoid tissues, with both T cells and NK cells participating.
DISCUSSION
The SIVΔnef vaccine strain has limited but clear replicative potential in vivo, as assessed by measurements of infectious units and viral RNA in blood. By applying the SIVΔnef vaccine strain directly to the tonsils of rhesus macaques, we demonstrated significant virus replication at the site of inoculation at 1 week and shortly thereafter in peripheral lymph nodes. Then the virus is brought under control, though it continues to replicate at lower levels in most lymphoid tissues.
Here we stress two new findings. First, the vaccinated animals are containing a substantial primary and spreading infection with SIVΔnef in the CD4+ T-cell compartment. The SIVΔnef vaccine strain therefore is not less virulent, because it shifts its site of infection from T cells to non-T cells, such as macrophages or dendritic cells. Second, there is a major expansion of perforin-positive killer cells, including CD8+ and CD3+ T cells, in lymph nodes that are supporting viral replication. The SIVΔnef vaccine strain therefore seems to elicit a strong local immune response, even though killer cells can be difficult to detect in the bloodstream (38). SIV-specific CD8+ T-cell responses become more evident when one challenges the vaccinated animals with wild-type SIV (38).
In designing this study, we wanted to explore the possibility that the efficacy of the SIVΔnef vaccine strain reflected a relative increase in virus replication in dendritic cells, leading to more effective antigen presentation and antiviral immunity. However, we could not demonstrate viral RNA by in situ hybridization in cells that were double labeled with dendritic cell markers. These included Langerin/CD207, a constituent of the Birbeck granules of Langerhans cells (35, 36); DC-LAMP/CD208, a lysosome-associated membrane protein of more mature dendritic cells (9); CD1a, a marker for Langerhans cells as well as dendritic cells in the external and crypt epithelia of the tonsils (31); and CD83 (39). Also, we only rarely found RNA-positive cells that were double labeled for sialoadhesin/CD169 (16) and macrosialin/CD68 (25), two markers that are abundant in resident and inflammatory macrophages.
Instead, the site for virus replication in vivo, acutely and chronically, proved to be the CD4+ T cell. More than half of the infected cells were not in cell cycle at the time of examination, as indicated by Ki-67 staining. This also occurs with infection with human immunodeficiency virus (HIV) and wild-type SIV (37). These findings might be explained as a composite of several elements in the virus-host interaction. Initially SIVΔnef is able to replicate in T cells that are interacting with mature dendritic cells, as occurs in tissue culture (22), perhaps through the aegis of the HIV-1 and SIV binding lectin DC-SIGN (12, 24). DC-SIGN is proposed to mediate the known capacity of dendritic cells to capture virus and transmit infection in trans to T cells. In contrast, replication of SIVΔnef is compromised when immature dendritic cells are carrying the vaccine (22). When an immune response develops, especially a CD8+ T-cell response that is known to dampen the levels of infection in vivo (23), virus replication is contained.
The new data in this paper on the number and types of infected cells in SIVΔnef-vaccinated monkeys indicate that the host can contain or resist a substantial early infection of CD4+ T cells with an immunodeficiency virus. Elsewhere we will document that monkeys vaccinated via the tonsillar route are indeed protected against a challenge with the pathogenic SIVmac251. Perhaps infection with SIVΔnef has some parallels with what takes place when SIVagm infects its natural African green monkey host.
During acute infection with SIVagm, there are abundant infected cells in lymphoid tissues, and then viral RNA and DNA levels fall to a low set point (10). The resistance that the host can mount to infected T cells also is reminiscent of individuals (26) and of macaques (21, 29) treated with highly active antiretroviral therapy early in acute infection with HIV-1 or SIV. In these individuals, the virus does take hold initially, but early treatment seems to allow the immune system or other defenses to control the infection. Conceivably, the initial virus infection interferes or retards further progression of infection, although our observations raise the possibility that T-cell and NK cell activity in the lymphoid tissue, especially at the portal of virus entry, may be playing a containment role. These lines of evidence are consistent with recent SIV-HIV hybrids results in macaques (2, 4), concluding that vaccines for immunodeficiency viruses need not provide sterilizing immunity against infection of CD4+ T cells, but rather need to contain the infection, allowing the host to establish a low viral set point compatible with immunocompetence.
Acknowledgments
This work was supported by grant QLRT-PL 1999-01215 from the European Community and by grants AI40045 and AI40874 from the NIH and Direct Effect.
We thank Gudrun Grossschupff, Petra Mayer, and Birgit Raschdorff for excellent technical assistance.
REFERENCES
- 1.Alexander, L., Z. Du, M. Rosenzweig, J. U. Jung, and R. C. Desrosiers. 1997. A role for natural simian immunodeficiency virus and human immunodeficiency virus type 1 nef alleles in lymphocyte activation. J. Virol. 71:6094–6099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Amara, R. R., F. Villinger, J. D. Altman, S. L. Lydy, S. P. O’Neil, S. I. Staprans, D. C. Montefiori, Y. Xu, J. G. Herndon, L. S. Wyatt, M. A. Candido, N. L. Kozyr, P. L. Earl, J. M. Smith, H.-L. Ma, B. D. Grimm, M. L. Hulsey, J. Miller, H. M. McClure, J. M. McNicholl, B. Moss, and H. L. Robinson. 2001. Control of mucosal challenge and prevention of AIDS by a multiprotein DNA/MVA vaccine. Science 292:69–74. [DOI] [PubMed] [Google Scholar]
- 3.Baba, T. W., A. M. Trichel, L. An, V. Liska, L. N. Martin, M. Murphey-Corb, and R. M. Ruprecht. 1996. Infection and AIDS in adult macaques after nontraumatic oral exposure to cell-free SIV. Science 272:1486–1489. [DOI] [PubMed] [Google Scholar]
- 4.Barouch, D. H., S. Santra, J. E. Schmitz, M. J. Kuroda, T. M. Fu, W. Wagner, M. Bilska, A. Craiu, X. X. Zheng, G. R. Krivulka, K. Beaudry, M. A. Lifton, C. E. Nickerson, W. L. Trigona, K. Punt, D. C. Freed, L. Guan, S. Dubey, D. Casimiro, A. Simon, M. E. Davies, M. Chastain, T. B. Strom, R. S. Gelman, D. C. Montefiori, and M. G. Lewis. 2000. Control of viremia and prevention of clinical AIDS in rhesus monkeys by cytokine-augmented DNA vaccination. Science 290:486–492. [DOI] [PubMed] [Google Scholar]
- 5.Chakrabarti, L., V. Baptiste, E. Khatissian, M.-C. Cumont, A.-M. Aubertin, L. Montagnier, and B. Hurtel. 1995. Limited viral spread and rapid immune response in lymph nodes of macaques inoculated with attenuated simian immunodeficiency virus. Virology 213:535–548. [DOI] [PubMed] [Google Scholar]
- 6.Cheynier, R., S. Henrichwark, F. Hadida, E. Pelletier, E. Oksenhendler, B. Autran, and S. Wain-Hobson. 1994. HIV and T cell expansion in splenic white pulps is accompanied by infiltration of HIV-specific cytotoxic T lymphocytes. Cell 78:373–387. [DOI] [PubMed] [Google Scholar]
- 7.Cranage, M. P., A. M. Whatmore, S. A. Sharpe, N. Cook, N. Polyanskaya, S. Leech, J. D. Smith, E. W. Rud, M. J. Dennis, and G. A. Hall. 1997. Macaques infected with live attenuated SIVmac are protected against superinfection via the rectal mucosa. Virology 229:143–154. [DOI] [PubMed] [Google Scholar]
- 8.Daniel, M. D., F. Kirchhoff, S. C. Czajak, P. K. Sehgal, and R. C. Desrosiers. 1992. Protective effects of a live attenuated SIV vaccine with a deletion in the nef gene. Science 258:1938–1941. [DOI] [PubMed] [Google Scholar]
- 9.de Saint-Vis, B., J. Vincent, S. Vandenabeele, B. Vanbervliet, J.-J. Pin, S. Ait-Yahia, S. Patel, M.-G. Mattei, J. Banchereau, S. Zurawski, J. Davoust, C. Caux, and S. Lebecque. 1998. A novel lysosome-associated membrane glycoprotein, DC-LAMP, induced upon DC maturation, is transiently expressed in MHC class II compartment. Immunity 9:325–336. [DOI] [PubMed] [Google Scholar]
- 10.Diop, O. M., A. Gueye, M. Dias-Tavares, C. Kornfeld, A. Faye, P. Ave, M. Huerre, S. Corbet, F. Barre-Sinoussi, and M. C. Muller-Trutwin. 2000. High levels of viral replication during primary simian immunodeficiency virus SIVagm infection are rapidly and strongly controlled in African green monkeys. J. Virol. 74:7538–7547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gauduin, M. C., R. L. Glickman, S. Ahmad, T. Yilma, and R. P. Johnson. 1999. Immunization with live attenuated simian immunodeficiency virus induces strong type 1 T helper responses and beta-chemokine production. Proc. Natl. Acad. Sci. USA 96:14031–14036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Geijtenbeek, T. B. H., D. S. Kwon, R. Torensma, S. J. van Vliet, G. C. F. van Duijnhoven, J. Middel, I. L. M. H. A. Cornelissen, H. S. L. M. Nottet, V. N. KewalRamani, D. R. Littman, C. G. Figdor, and Y. van Kooyk. 2000. DC-SIGN, a dendritic cell specific HIV-1 binding protein that enhances trans-infection of T cells. Cell 100:587–597. [DOI] [PubMed] [Google Scholar]
- 13.Gerdes, J., H. Lemke, H. Baisch, H. H. Wacker, U. Schwabb, and H. Stein. 1984. Cell cycle analysis of a cell proliferation-associated human nuclear antigen defined by the monoclonal antibody Ki-67. J. Immunol. 133:1710–1715. [PubMed] [Google Scholar]
- 14.Glushakova, S., J. C. Grivel, K. Suryanarayana, P. Meylan, J. D. Lifson, R. Desrosiers, and L. Margolis. 1999. Nef enhances human immunodeficiency virus replication and responsiveness to interleukin-2 in human lymphoid tissue ex vivo. J. Virol. 73:3968–3974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gundlach, B. R., H. Linhart, U. Dittmer, S. Sopper, S. Reiprich, D. Fuchs, B. Fleckenstein, G. Hunsmann, C. Stahl-Hennig, and K. Überla. 1997. Construction, replication, and immunogenic properties of a simian immunodeficiency virus expressing interleukin-2. J. Virol. 71:2225–2232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hartnell, A., J. Steel, H. Turley, M. Jones, D. G. Jackson, and P. R. Crocker. 2001. Characterization of human sialoadhesin, a sialic acid binding receptor expressed by resident and inflammatory macrophage populations. Blood 97:288–296. [DOI] [PubMed] [Google Scholar]
- 17.Ignatius, R., F. Isdell, U. O’Doherty, and M. Pope. 1998. Dendritic cells from skin and blood of macaques both promote SIV replication with T cells from different anatomical sites. J. Med. Primatol. 27:121–128. [DOI] [PubMed] [Google Scholar]
- 18.Johnson, R. P., J. D. Lifson, S. C. Czajak, K. S. Cole, K. H. Manson, R. Glickman, J. Yang, D. C. Montefiori, R. Montelaro, M. S. Wyand, and R. C. Desrosiers. 1999. Highly attenuated vaccine strains of simian immunodeficiency virus protect against vaginal challenge: inverse relationship of degree of protection with level of attenuation. J. Virol. 73:4952–4961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Johnson, R. P., and R. C. Desrosiers. 1998. Protective immunity induced by live attenuated simian immunodeficiency virus. Curr. Opin. Immunol. 10:436–443. [DOI] [PubMed] [Google Scholar]
- 20.Kestler, H.W., 3rd, D. J. Ringler, K. Mori, D. L. Panicali, P. K. Sehgal, M. D. Daniel, and R. C. Desrosiers. 1992. Importance of the nef gene for maintenance of high virus loads and for development of AIDS. Cell 65:651–662. [DOI] [PubMed] [Google Scholar]
- 21.Lori, F., M. G. Lewis, J. Xu, G. Varga, D. E. Zinn, Jr., C. Crabbs, W. Wagner, J. Greenhouse, P. Silvera, J. Yalley-Ogunro, C. Tinelli, and J. Lisziewicz. 2000. Control of SIV rebound through structured treatment interruptions during early infection. Science 290:1591–1593. [DOI] [PubMed] [Google Scholar]
- 22.Messmer, D., R. Ignatius, C. Santisteban, R. M. Steinman, and M. Pope. 2000. The decreased replicative capacity of SIVmac239Δnef is manifest in cultures of immature dendritic cells and T cells. J. Virol. 74:2406–2413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Metzner, K. J., X. Jin, F. V. Lee, A. Gettie, D. E. Bauer, M. Di Mascio, A. S. Perelson, P. A. Marx, D. D. Ho, L. G. Kostrikis, and R. I. Connor. 2000. Effects of in vivo CD8 (+) T cell depletion on virus replication in rhesus macaques immunized with a live, attenuated simian immunodeficiency virus vaccine. J. Exp. Med. 191:1921–1932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Pohlmann, S., E. J. Soilleux, F. Baribaud, G. J. Leslie, L. S. Morris, J. Trowsdale, B. Lee, N. Coleman, and R. W. Doms. 2001. DC-SIGNR, a DC-SIGN homologue expressed in endothelial cells, binds to human and simian immunodeficiency viruses and activates infection in trans. Proc. Natl. Acad. Sci. USA 98:2670–2675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Rabinowitz, S. S., and S. Gordon. 1991. Macrosialin, a macrophage-restricted membrane sialoprotein differentially glycosylated in response to inflammatory stimuli. J. Exp. Med. 174:827–836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Rosenberg, E., M. Altfed. S. Poon, M. Phillips, B. Wilkes, R. Eldridge, G. Robbins, R. D’Aquila, P. Goulder, and B. Walker. 2000. Immune control of HIV-1 following early treatment of acute infection. Nature 407:523–526. [DOI] [PubMed] [Google Scholar]
- 27.Ruprecht, R. M., T. W. Baba, V. Liska, N. B. Ray, L. N. Martin, M. Murphey-Corb, T. A. Rizvi, B. J. Bernacky, M. E. Keeling, H. M. McClure, and J. Andersen. 1999. Oral transmission of primate lentiviruses. J. Infect. Dis. 179(Suppl. 3):S408–412. [DOI] [PubMed] [Google Scholar]
- 28.Schluter, C., M. Duchrow, C. Wohlenberg, M. G. Becker, G. Key, H.-D. Flad, and J. Gerdes. 1993. The cell proliferation-associated antigen of antibody Ki-67: a very large, ubiquitous nuclear protein with numerous repeated elements, representing a new kind of cell cycle-maintaining protein. J. Cell Biol. 123:513–522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Spring, M., C. Stahl-Hennig, N. Stolte, N. Bischofberger, J. Heeney, P. Ten Haaft, K. Tenner-Racz, P. Racz, D. Lorenzen, G. Hunsmann, and U. Dittmer. 2001. Enhanced cellular immune response and reduced CD8+ lymphocyte apoptosis in acutely SIV-infected rhesus macaques after short-term antiretroviral treatment. Virology 279:221–232. [DOI] [PubMed] [Google Scholar]
- 30.Stahl-Hennig, C., U. Dittmer, T. Nisslein, H. Petry, E. Jurkiewicz, D. Fuchs, H. Wachter, K. Maetz-Rensing, E.-M. Kuhn, F.-J. Kaup, E. W. Rud, and G. Hunsmann. 1996. Rapid development of vaccine protection in macaques by live-attenuated simian immunodeficiency virus. J. Gen. Virol. 77:2969–2981. [DOI] [PubMed] [Google Scholar]
- 31.Stahl-Hennig, C., R. M. Steinman, K. Tenner-Racz, M. Pope, N. Stolte, K. Matz-Rensing, G. Grobschupff, B. Raschdorff, G. Hunsmann, and P. Racz. 1999. Rapid infection of oral mucosal-associated lymphoid tissue with simian immunodeficiency virus. Science 285:1261–1265. [DOI] [PubMed] [Google Scholar]
- 32.Stahl-Hennig, C., G. Voss, S. Nick, H. Perty, D. Fuchs, H. Wachter, C. Coulibaly, W. Luke, and G. Hunsmann. 1992. Immunization with Tween-ether-treated SIV adsorbed onto aluminum hydroxide protects monkeys against experimental SIV infection. Virology 186:588–596. [DOI] [PubMed] [Google Scholar]
- 33.Ten Haaft, P., B. Verstrepen, K. Überla, B. Rosenwirth, and J. Heeney. 1998. A pathogenic threshold of virus load defined in simian immunodeficiency virus-or simian-human immunodeficiency virus-infected macaques. J. Virol. 72:10281–10285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tenner-Racz, K., H.-J. Stellbrink, J. van Lunzen, C. Schneider, J.-P. Jacobs, B. Raschdorff, G. Groβschupff, R. M. Steinman, and P. Racz. 1998. The unenlarged lymph nodes of HIV-1 infected, asymptomatic patients with high CD4 T cell counts are sites for virus replication and CD4 T cell proliferation: the impact of active antiretroviral therapy. J. Exp. Med. 187:949–959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Valladeau, J., V. Duvert-Frances, J. J. Pin, C. Dezutter-Dambuyant, C. Vincent, C. Massacrier, J. Vincent, K. Yoneda, J. Banchereau, C. Caux, J. Davoust, and S. Saeland. 1999. The monoclonal antibody DCGM4 recognizes Langerin, a protein specific of Langerhans cells, and is rapidly internalized from the cell surface. Eur. J. Immunol. 29:2695–2704. [DOI] [PubMed] [Google Scholar]
- 36.Valladeau, J., O. Ravel, C. Dezutter-Dambuyant, K. Moore, M. Kleijmeer, Y. Liu, V. Duvert-Frances, C. Vincent, D. Schmitt, J. Davoust, C. Caux, S. Lebecque, and S. Saeland. 2000. Langerin, a novel C-type lectin specific to Langerhans cells, is an endocytic receptor that induces the formation of Birbeck granules. Immunity 12:71–81. [DOI] [PubMed] [Google Scholar]
- 37.Zhang, Z., T. Schuler, M. Zupancic, S. Wietgrefe, K. A. Staskus, K. A. Reimann, T. A. Reinhart, M. Rogan, W. Cavert, C. J. Miller, R. S. Veazey, D. Notermans, S. Little, S. A. Danner, D. D. Richman, D. Havlir, J. Wong, H. L. Jordan, T. W. Schacker, P. Racz, K. Tenner-Racz, N. L. Letvin, S. Wolinsky, and A. T. Haase. 1999. Sexual transmission and propagation of SIV and HIV in resting and activated CD4(+) T cells. Science 286:1353–1357. [DOI] [PubMed] [Google Scholar]
- 38.Zhong, L., A. Granelli-Piperno, M. Pope, R. Ignatius, M. Lewis, S. S. Frankel, and R. M. Steinman. 2000. Presentation of SIVgag to monkey T cells using dendritic cells transfected with a recombinant adenovirus. Eur. J. Immunol. 30:3281–3290. [DOI] [PubMed] [Google Scholar]
- 39.Zhou, L.-J., and T. F. Tedder. 1995. Human blood dendritic cells selectively express CD83, a member of the immunoglobulin superfamily. J. Immunol. 154:3821–3835. [PubMed] [Google Scholar]