Abstract
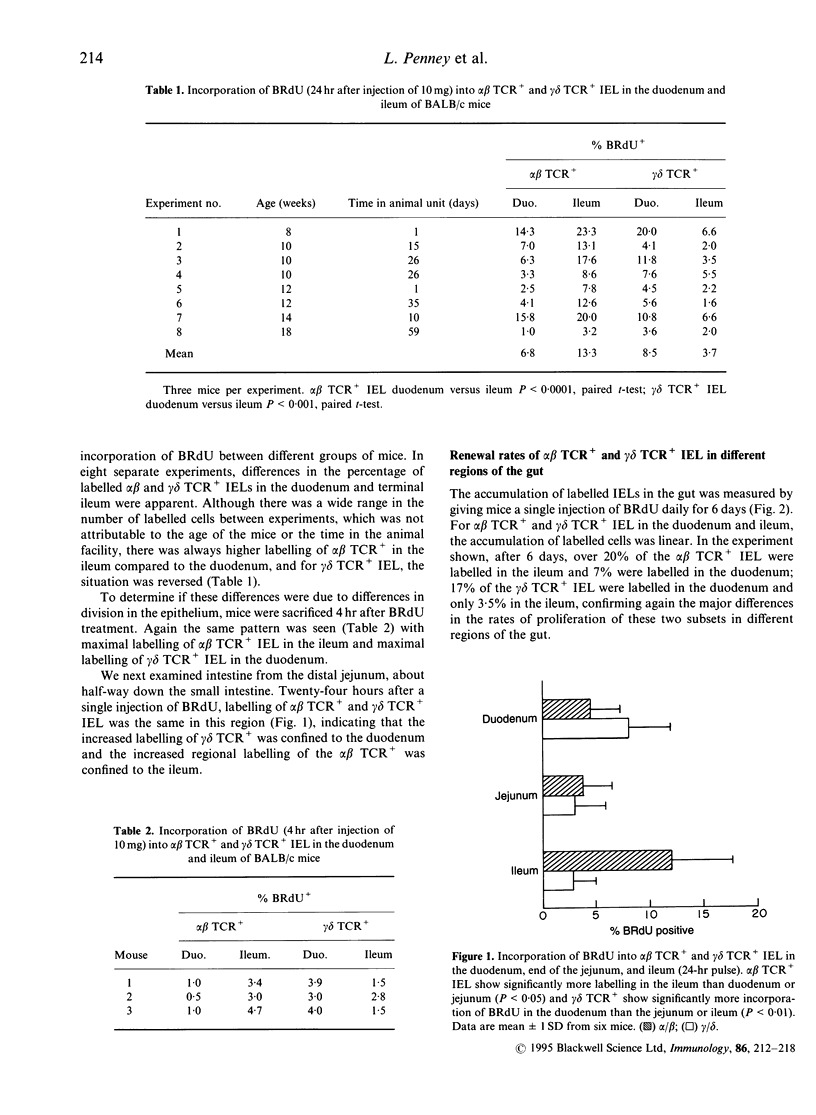
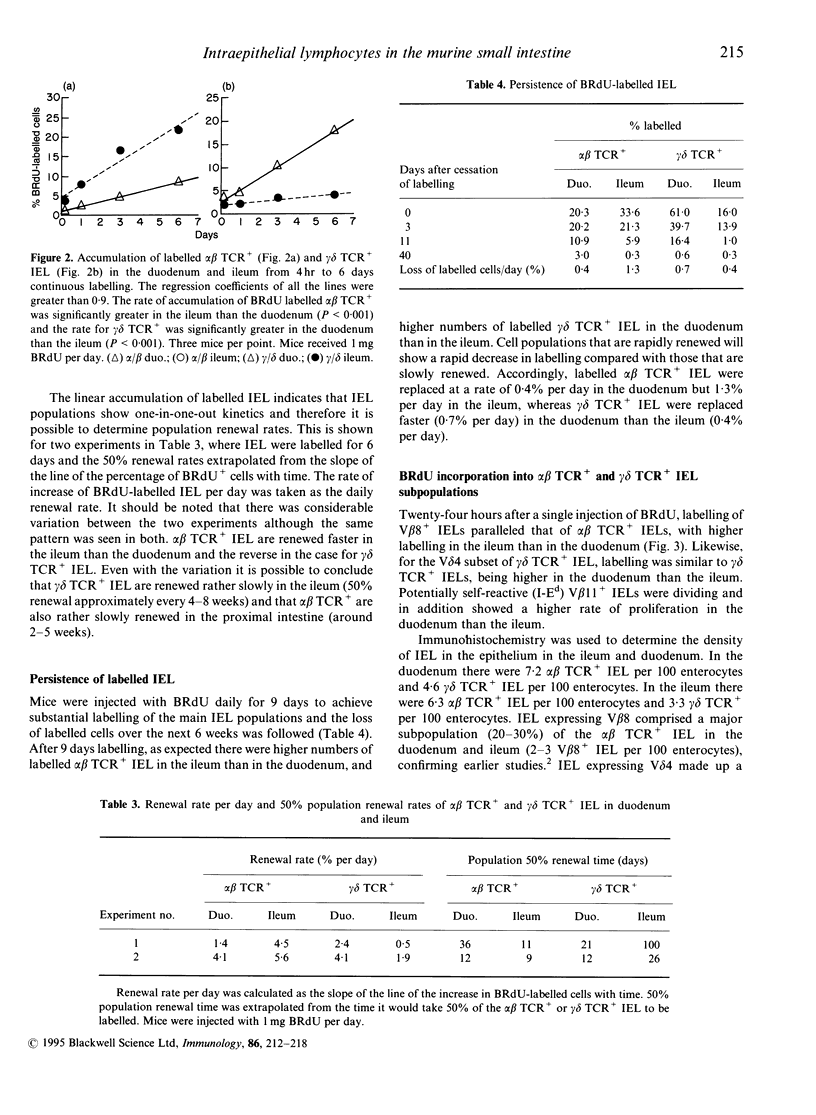
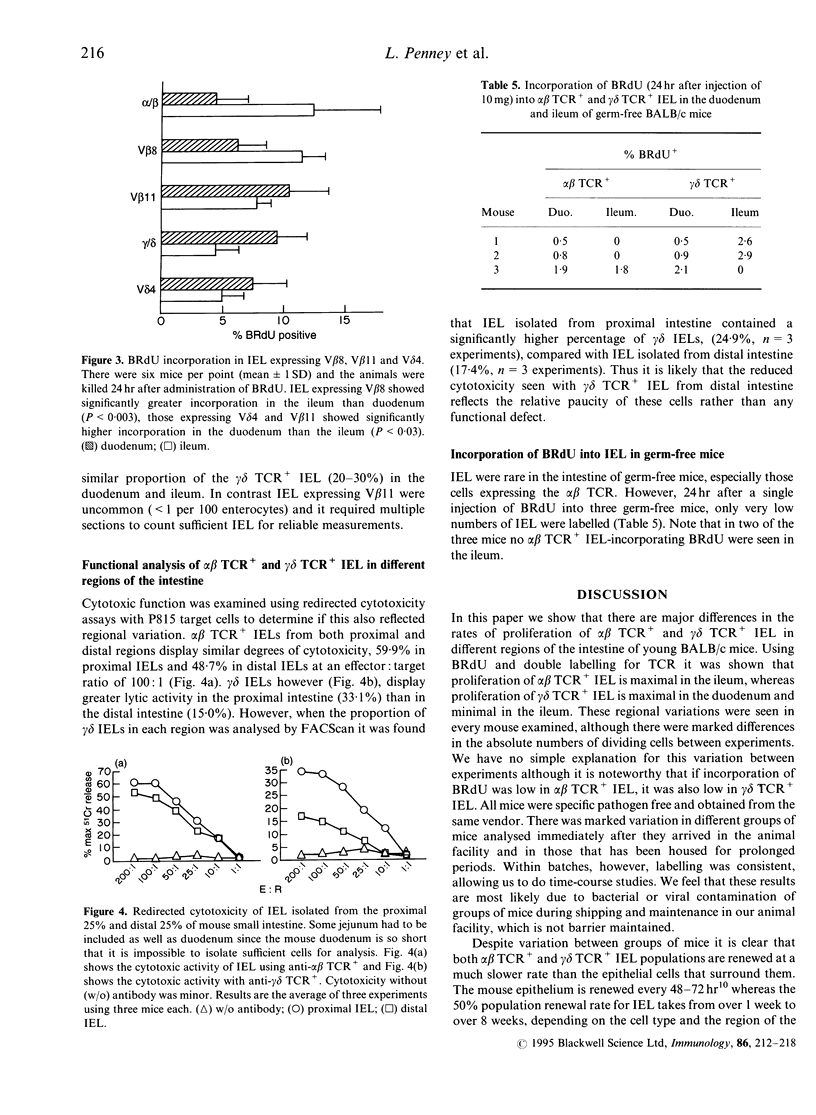
Using double staining for T-cell receptor (TCR) and 5-bromo-2'-deoxyuridine (BRdU) we have examined the proliferation rates and lifespan of murine intraepithelial lymphocytes (IEL's) in vivo. After a 24-hr pulse of BRdU the number of labelled alpha beta TCR+ IEL was significantly higher in the ileum than the duodenum. In contrast, incorporation of BRdU into gamma delta TCR+ IEL was significantly higher in the duodenum than the ileum. This regional variation was also seen after a 4-hr pulse of BRdU indicating that the differences probably reflect local rates of proliferation in the epithelium. Over a 6-day labelling period, the accumulation of labelled alpha beta TCR+ and gamma delta TCR+ IEL was linear, which allowed IEL lifespan to be calculated. There was considerable variation between groups of mice but the 50% population renewal time for alpha beta TCR+ IEL was 12-36 days in the duodenum and 9-11 days in the ileum, and for gamma delta TCR+ IEL was 12-21 days in the duodenum and 26-100 days in the ileum. The incorporation of BRdU into V beta 8+ IEL showed the same regional variation as alpha beta TCR+ IEL and the V delta 4 population behaved like the total gamma delta TCR+ IEL population. In contrast V beta 11+, potentially self-reactive IEL, showed a regional pattern of labelling like gamma delta TCR+ IEL. Incorporation of BRdU into both alpha beta TCR+ and gamma delta TCR+ IEL in germ-free mice was very low and did not show marked regional variation. alpha beta TCR+ and gamma delta TCR+ IEL from both proximal and distal bowel were cytotoxic. Therefore alpha beta TCR+ and gamma delta TCR+ IEL show different rates of division in different sections of the gut, perhaps reflecting responses to different antigens. Both alpha beta TCR+ and gamma delta TCR+ IEL reside in the epithelium for weeks during which time the gut epithelial population will have been renewed many times.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bandeira A., Itohara S., Bonneville M., Burlen-Defranoux O., Mota-Santos T., Coutinho A., Tonegawa S. Extrathymic origin of intestinal intraepithelial lymphocytes bearing T-cell antigen receptor gamma delta. Proc Natl Acad Sci U S A. 1991 Jan 1;88(1):43–47. doi: 10.1073/pnas.88.1.43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barrett T. A., Delvy M. L., Kennedy D. M., Lefrancois L., Matis L. A., Dent A. L., Hedrick S. M., Bluestone J. A. Mechanism of self-tolerance of gamma/delta T cells in epithelial tissue. J Exp Med. 1992 Jan 1;175(1):65–70. doi: 10.1084/jem.175.1.65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bill J., Kanagawa O., Woodland D. L., Palmer E. The MHC molecule I-E is necessary but not sufficient for the clonal deletion of V beta 11-bearing T cells. J Exp Med. 1989 Apr 1;169(4):1405–1419. doi: 10.1084/jem.169.4.1405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Camerini V., Panwala C., Kronenberg M. Regional specialization of the mucosal immune system. Intraepithelial lymphocytes of the large intestine have a different phenotype and function than those of the small intestine. J Immunol. 1993 Aug 15;151(4):1765–1776. [PubMed] [Google Scholar]
- Cuff C. F., Cebra C. K., Rubin D. H., Cebra J. J. Developmental relationship between cytotoxic alpha/beta T cell receptor-positive intraepithelial lymphocytes and Peyer's patch lymphocytes. Eur J Immunol. 1993 Jun;23(6):1333–1339. doi: 10.1002/eji.1830230622. [DOI] [PubMed] [Google Scholar]
- De Geus B., Van den Enden M., Coolen C., Nagelkerken L., Van der Heijden P., Rozing J. Phenotype of intraepithelial lymphocytes in euthymic and athymic mice: implications for differentiation of cells bearing a CD3-associated gamma delta T cell receptor. Eur J Immunol. 1990 Feb;20(2):291–298. doi: 10.1002/eji.1830200210. [DOI] [PubMed] [Google Scholar]
- Dillon S. B., MacDonald T. T. Functional properties of lymphocytes isolated from murine small intestinal epithelium. Immunology. 1984 Jul;52(3):501–509. [PMC free article] [PubMed] [Google Scholar]
- Dubos R. J., Savage D. C., Schaedler R. W. The indigenous flora of the gastrointestinal tract. Dis Colon Rectum. 1967 Jan-Feb;10(1):23–34. doi: 10.1007/BF02617382. [DOI] [PubMed] [Google Scholar]
- Fujihashi K., Taguchi T., Aicher W. K., McGhee J. R., Bluestone J. A., Eldridge J. H., Kiyono H. Immunoregulatory functions for murine intraepithelial lymphocytes: gamma/delta T cell receptor-positive (TCR+) T cells abrogate oral tolerance, while alpha/beta TCR+ T cells provide B cell help. J Exp Med. 1992 Mar 1;175(3):695–707. doi: 10.1084/jem.175.3.695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodman T., Lefrancois L. Intraepithelial lymphocytes. Anatomical site, not T cell receptor form, dictates phenotype and function. J Exp Med. 1989 Nov 1;170(5):1569–1581. doi: 10.1084/jem.170.5.1569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guy-Grand D., Cerf-Bensussan N., Malissen B., Malassis-Seris M., Briottet C., Vassalli P. Two gut intraepithelial CD8+ lymphocyte populations with different T cell receptors: a role for the gut epithelium in T cell differentiation. J Exp Med. 1991 Feb 1;173(2):471–481. doi: 10.1084/jem.173.2.471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guy-Grand D., Griscelli C., Vassalli P. The mouse gut T lymphocyte, a novel type of T cell. Nature, origin, and traffic in mice in normal and graft-versus-host conditions. J Exp Med. 1978 Dec 1;148(6):1661–1677. doi: 10.1084/jem.148.6.1661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guy-Grand D., Vanden Broecke C., Briottet C., Malassis-Seris M., Selz F., Vassalli P. Different expression of the recombination activity gene RAG-1 in various populations of thymocytes, peripheral T cells and gut thymus-independent intraepithelial lymphocytes suggests two pathways of T cell receptor rearrangement. Eur J Immunol. 1992 Feb;22(2):505–510. doi: 10.1002/eji.1830220232. [DOI] [PubMed] [Google Scholar]
- Haskins K., Hannum C., White J., Roehm N., Kubo R., Kappler J., Marrack P. The antigen-specific, major histocompatibility complex-restricted receptor on T cells. VI. An antibody to a receptor allotype. J Exp Med. 1984 Aug 1;160(2):452–471. doi: 10.1084/jem.160.2.452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kubo R. T., Born W., Kappler J. W., Marrack P., Pigeon M. Characterization of a monoclonal antibody which detects all murine alpha beta T cell receptors. J Immunol. 1989 Apr 15;142(8):2736–2742. [PubMed] [Google Scholar]
- LEBLOND C. P., MESSIER B. Renewal of chief cells and goblet cells in the small intestine as shown by radioautography after injection of thymidine-H3 into mice. Anat Rec. 1958 Nov;132(3):247–259. doi: 10.1002/ar.1091320303. [DOI] [PubMed] [Google Scholar]
- Lefrançois L., Olson S. A novel pathway of thymus-directed T lymphocyte maturation. J Immunol. 1994 Aug 1;153(3):987–995. [PubMed] [Google Scholar]
- Lin T., Matsuzaki G., Kenai H., Nakamura T., Nomoto K. Thymus influences the development of extrathymically derived intestinal intraepithelial lymphocytes. Eur J Immunol. 1993 Aug;23(8):1968–1974. doi: 10.1002/eji.1830230836. [DOI] [PubMed] [Google Scholar]
- Lin T., Matsuzaki G., Kenai H., Nomoto K. Progenies of fetal thymocytes are the major source of CD4-CD8+ alpha alpha intestinal intraepithelial lymphocytes early in ontogeny. Eur J Immunol. 1994 Aug;24(8):1785–1791. doi: 10.1002/eji.1830240810. [DOI] [PubMed] [Google Scholar]
- McMenamin C., McKersey M., Kühnlein P., Hünig T., Holt P. G. Gamma delta T cells down-regulate primary IgE responses in rats to inhaled soluble protein antigens. J Immunol. 1995 May 1;154(9):4390–4394. [PubMed] [Google Scholar]
- Meader R. D., Landers D. F. Electron and light microscopic observations on relationships between lymphocytes and intestinal epithelium. Am J Anat. 1967 Nov;121(3):763–773. doi: 10.1002/aja.1001210318. [DOI] [PubMed] [Google Scholar]
- Mosley R. L., Styre D., Klein J. R. Differentiation and functional maturation of bone marrow-derived intestinal epithelial T cells expressing membrane T cell receptor in athymic radiation chimeras. J Immunol. 1990 Sep 1;145(5):1369–1375. [PubMed] [Google Scholar]
- Murosaki S., Yoshikai Y., Ishida A., Nakamura T., Matsuzaki G., Takimoto H., Yuuki H., Nomoto K. Failure of T cell receptor V beta negative selection in murine intestinal intra-epithelial lymphocytes. Int Immunol. 1991 Oct;3(10):1005–1013. doi: 10.1093/intimm/3.10.1005. [DOI] [PubMed] [Google Scholar]
- Penit C. In vivo thymocyte maturation. BUdR labeling of cycling thymocytes and phenotypic analysis of their progeny support the single lineage model. J Immunol. 1986 Oct 1;137(7):2115–2121. [PubMed] [Google Scholar]
- Poussier P., Edouard P., Lee C., Binnie M., Julius M. Thymus-independent development and negative selection of T cells expressing T cell receptor alpha/beta in the intestinal epithelium: evidence for distinct circulation patterns of gut- and thymus-derived T lymphocytes. J Exp Med. 1992 Jul 1;176(1):187–199. doi: 10.1084/jem.176.1.187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rocha B., Vassalli P., Guy-Grand D. The V beta repertoire of mouse gut homodimeric alpha CD8+ intraepithelial T cell receptor alpha/beta + lymphocytes reveals a major extrathymic pathway of T cell differentiation. J Exp Med. 1991 Feb 1;173(2):483–486. doi: 10.1084/jem.173.2.483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Röpke C., Everett N. B. Kinetics of intraepithelial lymphocytes in the small intestine of thymus-deprived mice and antigen-deprived mice. Anat Rec. 1976 May;185(1):101–108. doi: 10.1002/ar.1091850110. [DOI] [PubMed] [Google Scholar]
- Röpke C., Everett N. B. Proliferative kinetics of large and small intraepithelial lymphocytes in the small intestine of the mouse. Am J Anat. 1976 Mar;145(3):395–408. doi: 10.1002/aja.1001450307. [DOI] [PubMed] [Google Scholar]
- Sidhu N. K., Wright G. M., Markham R. J., Ireland W. P., Singh A. Quantitative regional variation in the expression of major histocompatibility class II antigens in enterocytes of the mouse small intestine. Tissue Cell. 1992;24(2):221–228. doi: 10.1016/0040-8166(92)90095-o. [DOI] [PubMed] [Google Scholar]
- Sydora B. C., Mixter P. F., Houlden B., Hershberg R., Levy R., Comay M., Bluestone J., Kronenberg M. T-cell receptor gamma delta diversity and specificity of intestinal intraepithelial lymphocytes: analysis of IEL-derived hybridomas. Cell Immunol. 1993 Dec;152(2):305–322. doi: 10.1006/cimm.1993.1293. [DOI] [PubMed] [Google Scholar]
- Viney J. L., Kilshaw P. J., MacDonald T. T. Cytotoxic alpha/beta+ and gamma/delta+ T cells in murine intestinal epithelium. Eur J Immunol. 1990 Jul;20(7):1623–1626. doi: 10.1002/eji.1830200734. [DOI] [PubMed] [Google Scholar]
- Viney J. L., MacDonald T. T., Kilshaw P. J. T-cell receptor expression in intestinal intra-epithelial lymphocyte subpopulations of normal and athymic mice. Immunology. 1989 Apr;66(4):583–587. [PMC free article] [PubMed] [Google Scholar]


