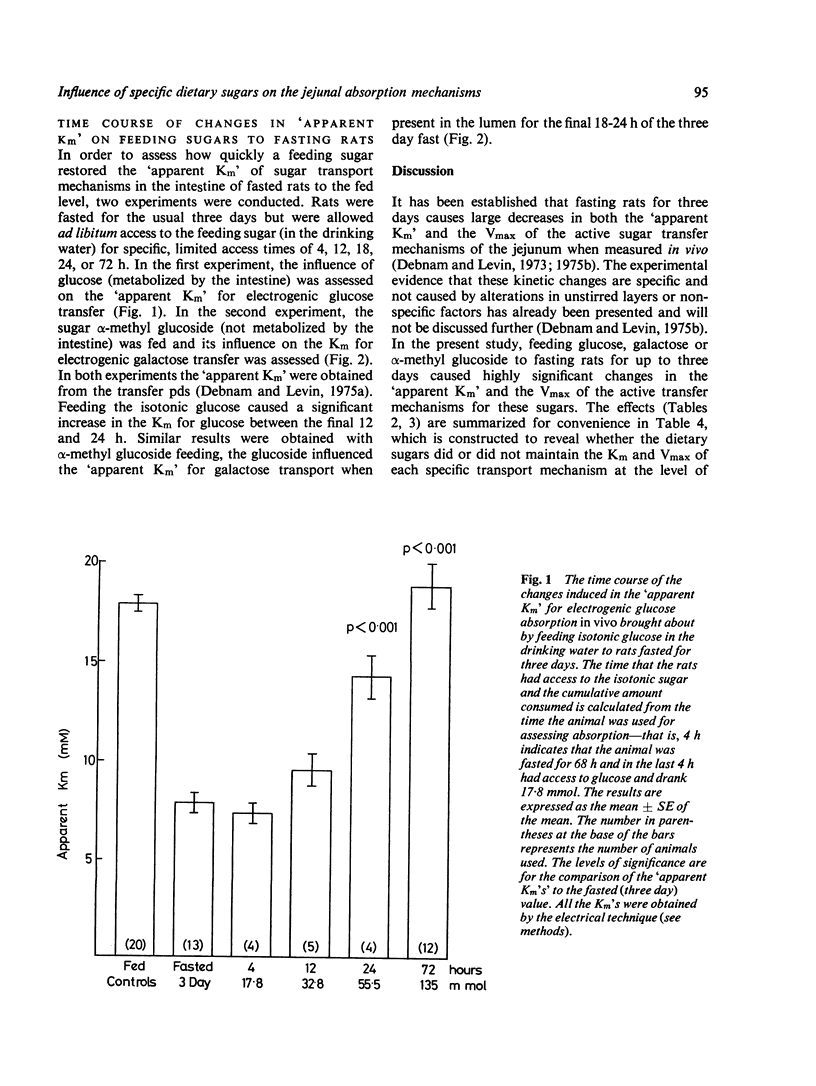
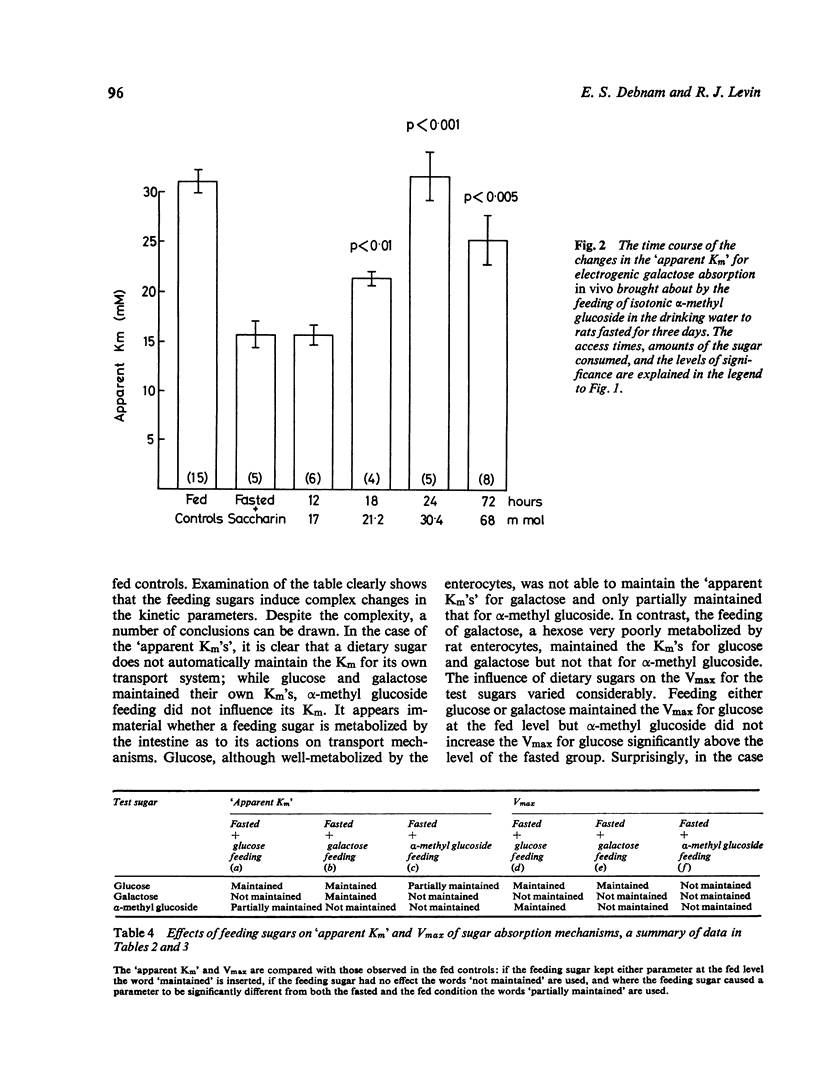
Abstract
The effects of feeding glucose, galactose, and alpha-methyl glucoside on the kinetics of absorption of these sugars in the jejunum of the fasting rat were assessed by recently developed chemical and electrical methods for characterizing the electrogenic active transfer mechanisms in vivo. The parameters of 'apparent Km' (an index of carrier affinity) and Vmax (an index of the maximum jejunal transfer capacity) were obtained from the kinetic data to describe quantitatively the active transport mechanisms in rats fed their normal solid diet, in rats deprived of solid food for three days, and in rats deprived of solid food but allowed ad libitum access to isotonic solutions of glucose, galactose, or alpha-methyl glucoside for up to three days. Feeding the sugars to fasting rats produced complex differential effects on the 'apparent Km's' and Vmax's of the various transport mechanisms. The changes, although complex, can best be explained by alterations in the transport mechanisms per se rather than by non-specific changes in intestinal morphology or metabolism. The differential effects induced by the feeding sugars are further evidence for the concept that jejunal enterocytes possess multiple carriers or mechansims for the absorption of actively transported sugars in vivo. The multiple forms of the sugar carriers (isocarriers?) MAY BE INVOLVED IN THE FACULTATIVE RESPONSES OF THE ENTEROCYTES TO CHANGES IN THE DIETARY LEVELS OF SUGAR AND MAY BE IMPORTANT IN UNDERSTANDING THE INFLUENCES OF DISEASE AND NUTRITIONAL STATUS ON ABSORPTION PROCESSES IN MAN.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anderson J. W., Herman R. H., Tyrrell J. B., Cohn R. M. Hexokinase: a compartmented enzyme. Am J Clin Nutr. 1971 Jun;24(6):642–650. doi: 10.1093/ajcn/24.6.642. [DOI] [PubMed] [Google Scholar]
- Anderson J. W., Tyrrell J. B. Hexokinase activity of rat intestinal mucosa: demonstration of four isozymes and of changes in subcellular distribution with fasting and refeeding. Gastroenterology. 1973 Jul;65(1):69–76. [PubMed] [Google Scholar]
- Cleland W. W. The statistical analysis of enzyme kinetic data. Adv Enzymol Relat Areas Mol Biol. 1967;29:1–32. doi: 10.1002/9780470122747.ch1. [DOI] [PubMed] [Google Scholar]
- Debnam E. S., Levin R. J. An experimental method of identifying and quantifying the active transfer electrogenic component from the diffusive component during sugar absorption measured in vivo. J Physiol. 1975 Mar;246(1):181–196. doi: 10.1113/jphysiol.1975.sp010885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Debnam E. S., Levin R. J. Assessment of the effects of starvation and of semi-starvation on the operational kinetic parameters of the active transfer of hexoses measured in vivo. J Physiol. 1973 May;231(1):21P–23P. [PubMed] [Google Scholar]
- Debnam E. S., Levin R. J. Effects of fasting and semistarvation on the kinetics of active and passive sugar absorption across the small intestine in vivo. J Physiol. 1975 Nov;252(3):681–700. doi: 10.1113/jphysiol.1975.sp011165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Debnam E. S., Levin R. J. Evidence for separate hexose carriers in the small intestine uncovered by fasting and glucose alimentation. J Physiol. 1971 Oct;218 (Suppl):38P–39P. [PubMed] [Google Scholar]
- Debnam E. S., Levin R. J. Proceedings: Dietary sugars and the maximum absorptive capacity of the small intestine; further evidence for multiple hexose transfer mechanisms. J Physiol. 1974 Apr;238(1):81P–82P. [PubMed] [Google Scholar]
- Genel M., London D., Holtzapple P. G., Segal S. Uptake of alpha-methylglucoside by normal and diabetic human jejunal mucosa. J Lab Clin Med. 1971 May;77(5):743–750. [PubMed] [Google Scholar]
- Honegger P., Gershon E. Further evidence for the multiplicity of carriers for free glucalogues in hamster small intestine. Biochim Biophys Acta. 1974 May 30;352(1):127–134. doi: 10.1016/0005-2736(74)90185-0. [DOI] [PubMed] [Google Scholar]
- Jones G. M., Mayer R. J. Glucose metabolism in the rat small intestine: the effect of glucose analogues on hexokinase activity. Biochem J. 1973 Jan;132(1):125–128. doi: 10.1042/bj1320125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levin R. J., Syme G. Differential changes in the 'apparent Km' and maximum potential difference of the hexose and amino acid electrogenic transfer mechanisms of the small intestine, induced by fasting and hypothyroidism. J Physiol. 1971 Mar;213(2):46P–48P. [PubMed] [Google Scholar]
- Malathi P., Ramaswamy K., Caspary W. F., Crane R. K. Studies on the transport of glucose from disaccharides by hamster small intestine in vitro. I. Evidence for a disaccharidase-related transport system. Biochim Biophys Acta. 1973 May 25;307(3):613–626. doi: 10.1016/0005-2736(73)90306-4. [DOI] [PubMed] [Google Scholar]
- Mayer R. J., Shakespeare P., Hübscher G. Glucose metabolism in the mucosa of the small intestine. Changes of hexokinase activity during perfusion of the proximal half of rat small intestine. Biochem J. 1970 Jan;116(1):43–48. doi: 10.1042/bj1160043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McMichael H. B. A second intestinal glucose carrier. Gut. 1973 May;14(5):428–429. [PubMed] [Google Scholar]
- McMichael H. B. Clinical studies of carbohydrate digestion and absorption. Biochem Soc Trans. 1975;3(2):223–227. doi: 10.1042/bst0030223. [DOI] [PubMed] [Google Scholar]
- McMichael H. B. Intestinal absorption of carbohydrates in man. Proc Nutr Soc. 1971 Dec;30(3):248–254. doi: 10.1079/pns19710048. [DOI] [PubMed] [Google Scholar]
- Meeuwisse G. W., Melin K. Glucose-galactose malabsorption. A clinical study of 6 cases. Acta Paediatr Scand. 1969;(Suppl):1+–1+. [PubMed] [Google Scholar]
- Menge H., Werner H., Lorenz-Meyer H., Riecken E. O. The nutritive effect of glucose on the structure and function of jejunal self-emptying blind loops in the rat. Gut. 1975 Jun;16(6):462–467. doi: 10.1136/gut.16.6.462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newey H., Sanford P. A., Smyth D. H. The effect of uranyl nitrate on intestinal transfer of hexoses. J Physiol. 1966 Oct;186(3):493–502. doi: 10.1113/jphysiol.1966.sp008051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roy C. C., Dubois R. S. Monosaccharide induction of 3-O-methyl glucose transport through the rat jejunum. Proc Soc Exp Biol Med. 1972 Mar;139(3):883–886. doi: 10.3181/00379727-139-36258. [DOI] [PubMed] [Google Scholar]
- Srivastava L. M., Hübscher G. Glucose metabolism in the mucosa of the small intestine. Glycolysis in subcellular preparations from the cat and rat. Biochem J. 1966 Aug;100(2):458–466. doi: 10.1042/bj1000458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van den Berg J. W.O., Hülsmann W. C. Insoluble hexokinase in the brush border region of rat intestinal epithelial cells. FEBS Lett. 1971 Jan 12;12(3):173–175. doi: 10.1016/0014-5793(71)80061-3. [DOI] [PubMed] [Google Scholar]


