Abstract
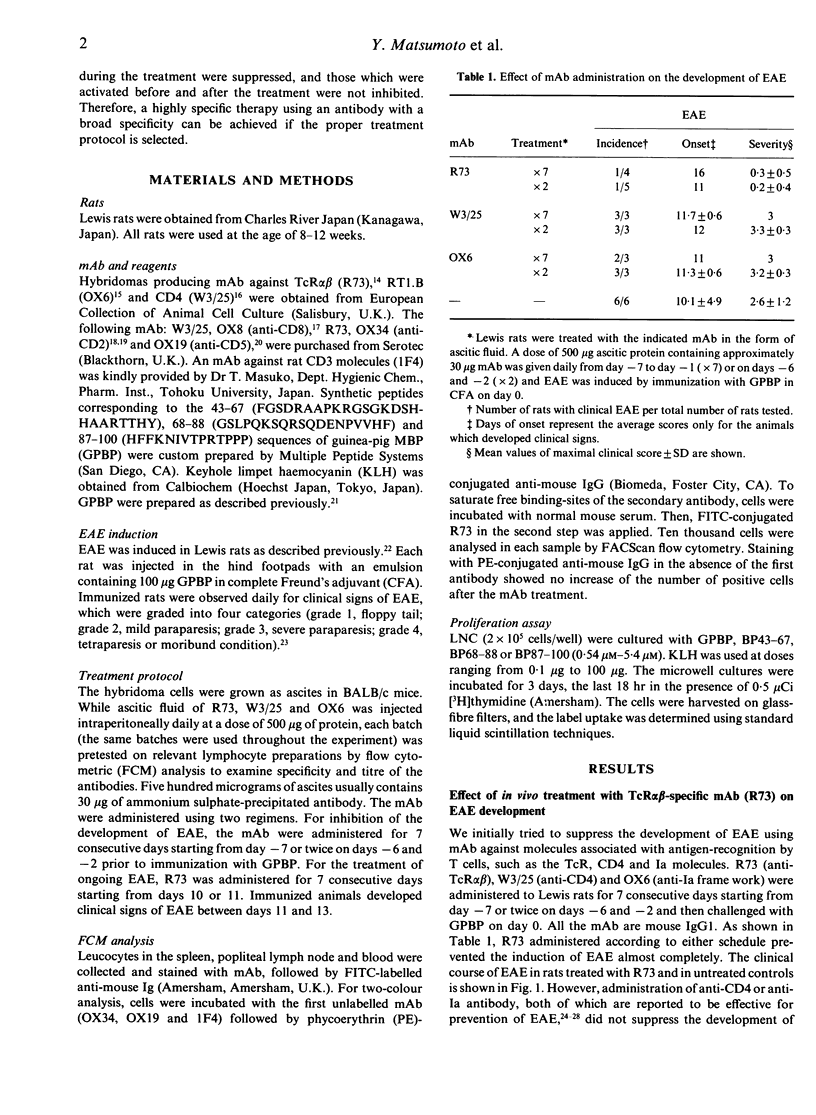
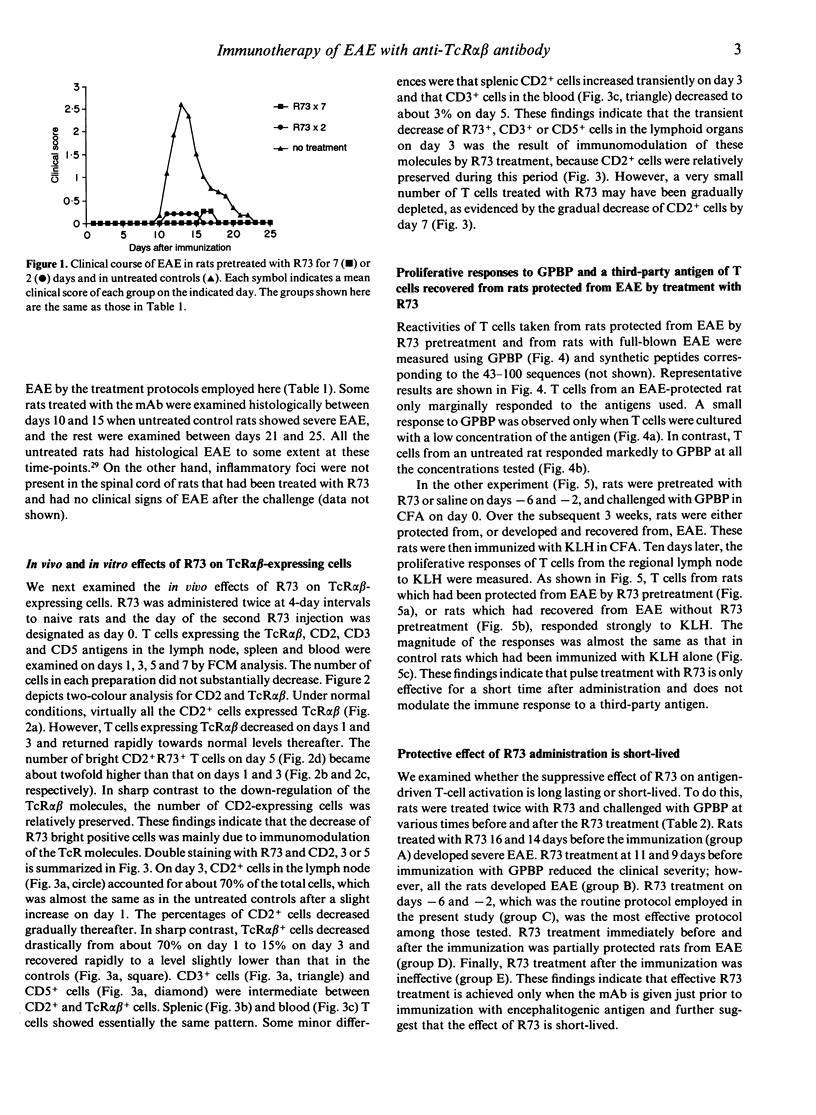
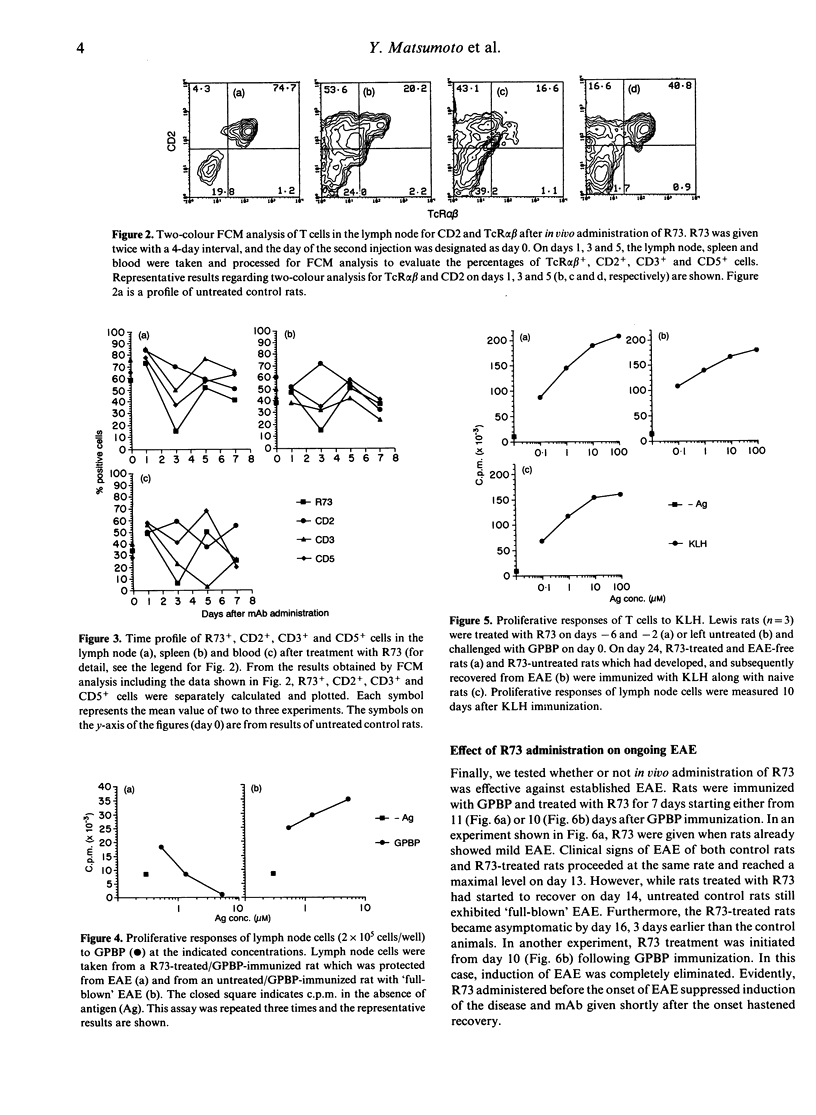
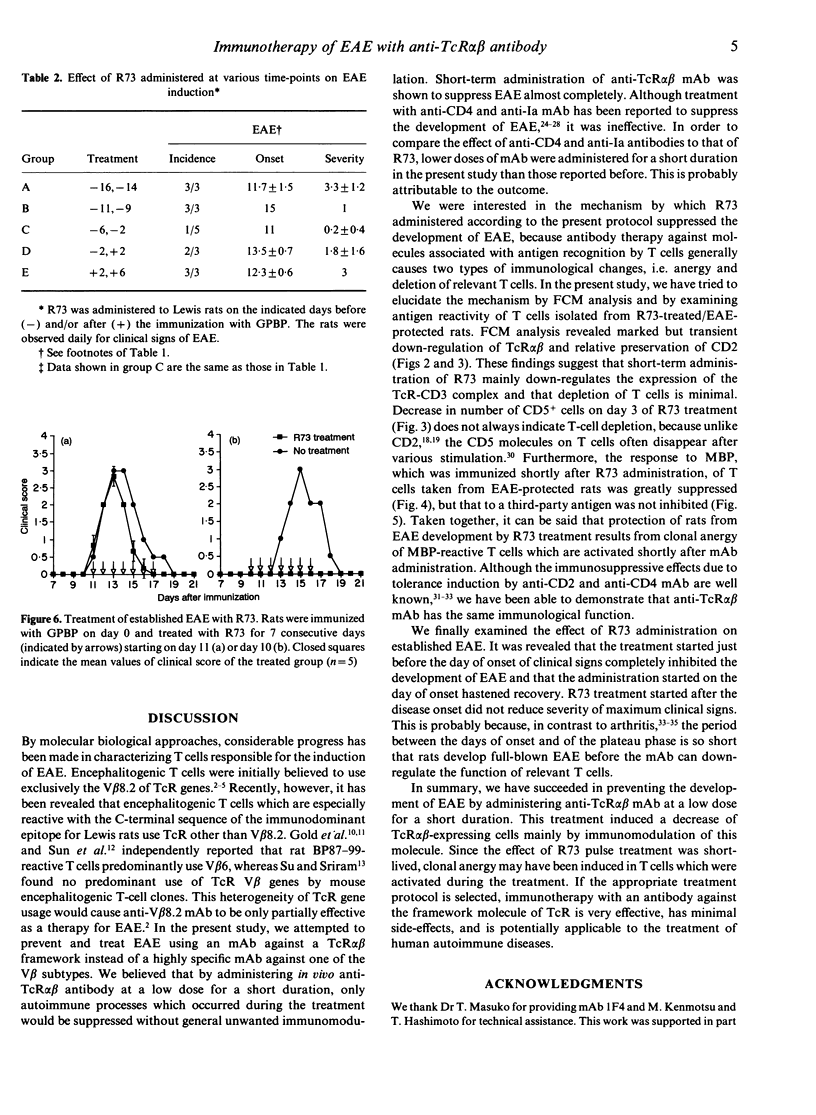
To identify an effective immunotherapy for T-cell-mediated autoimmune diseases, prevention and treatment of experimental autoimmune encephalomyelitis (EAE) induced in Lewis rats was attempted by administering a monoclonal antibody (mAb), R73, which is specific for rat T-cell receptor (TcR) alpha beta. Short-term administration of R73 at relatively low doses before immunization with encephalitogenic antigen, myelin basic protein (MBP), prevented the development of EAE. However, treatment with anti-CD4 and anti-Ia mAb in the same protocol was ineffective. Flow cytometric analysis demonstrated that short-term administration of R73 resulted in transient down-regulation of the TcR molecules, whereas the number of CD2-expressing T cells was well preserved. Furthermore, the response to MBP of T cells isolated from rats that were pretreated with R73 and then immunized with MBP was strongly suppressed. On the other hand, the T-cell response of R73-pretreated rats to a third-party antigen which was immunized at a later period was not inhibited. These findings suggest that in vivo administration of a low dose of R73 protects rats from EAE by inducing anergy of MBP-reactive encephalitogenic T cells. Furthermore, R73 treatment which started on day 10 of the immunization (shortly before the day of onset of clinical signs) completely suppressed the induction of EAE and that which started on day 11 (the day of onset) hastened recovery. Since the phenotypes of the TcR V beta chain of encephalitogenic T cells are not so limited as previously believed, immunotherapy with mAb against the TcR alpha beta framework may be one of the best methods for treatment of T-cell-mediated autoimmune diseases.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Acha-Orbea H., Mitchell D. J., Timmermann L., Wraith D. C., Tausch G. S., Waldor M. K., Zamvil S. S., McDevitt H. O., Steinman L. Limited heterogeneity of T cell receptors from lymphocytes mediating autoimmune encephalomyelitis allows specific immune intervention. Cell. 1988 Jul 15;54(2):263–273. doi: 10.1016/0092-8674(88)90558-2. [DOI] [PubMed] [Google Scholar]
- Alters S. E., Shizuru J. A., Ackerman J., Grossman D., Seydel K. B., Fathman C. G. Anti-CD4 mediates clonal anergy during transplantation tolerance induction. J Exp Med. 1991 Feb 1;173(2):491–494. doi: 10.1084/jem.173.2.491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bellgrau D., Talmage D. W. T cells can be cytotoxic without making interleukin 2: a model of separate pathways of induction. Proc Natl Acad Sci U S A. 1986 May;83(10):3412–3416. doi: 10.1073/pnas.83.10.3412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benjamin R. J., Waldmann H. Induction of tolerance by monoclonal antibody therapy. Nature. 1986 Apr 3;320(6061):449–451. doi: 10.1038/320449a0. [DOI] [PubMed] [Google Scholar]
- Brideau R. J., Carter P. B., McMaster W. R., Mason D. W., Williams A. F. Two subsets of rat T lymphocytes defined with monoclonal antibodies. Eur J Immunol. 1980 Aug;10(8):609–615. doi: 10.1002/eji.1830100807. [DOI] [PubMed] [Google Scholar]
- Brostoff S. W., Mason D. W. Experimental allergic encephalomyelitis: successful treatment in vivo with a monoclonal antibody that recognizes T helper cells. J Immunol. 1984 Oct;133(4):1938–1942. [PubMed] [Google Scholar]
- Chluba J., Steeg C., Becker A., Wekerle H., Epplen J. T. T cell receptor beta chain usage in myelin basic protein-specific rat T lymphocytes. Eur J Immunol. 1989 Feb;19(2):279–284. doi: 10.1002/eji.1830190210. [DOI] [PubMed] [Google Scholar]
- Clark S. J., Law D. A., Paterson D. J., Puklavec M., Williams A. F. Activation of rat T lymphocytes by anti-CD2 monoclonal antibodies. J Exp Med. 1988 Jun 1;167(6):1861–1872. doi: 10.1084/jem.167.6.1861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dallman M. J., Thomas M. L., Green J. R. MRC OX-19: a monoclonal antibody that labels rat T lymphocytes and augments in vitro proliferative responses. Eur J Immunol. 1984 Mar;14(3):260–267. doi: 10.1002/eji.1830140311. [DOI] [PubMed] [Google Scholar]
- Gold D. P., Vainiene M., Celnik B., Wiley S., Gibbs C., Hashim G. A., Vandenbark A. A., Offner H. Characterization of the immune response to a secondary encephalitogenic epitope of basic protein in Lewis rats. II. Biased T cell receptor V beta expression predominates in spinal cord infiltrating T cells. J Immunol. 1992 Mar 15;148(6):1712–1717. [PubMed] [Google Scholar]
- Gückel B., Berek C., Lutz M., Altevogt P., Schirrmacher V., Kyewski B. A. Anti-CD2 antibodies induce T cell unresponsiveness in vivo. J Exp Med. 1991 Nov 1;174(5):957–967. doi: 10.1084/jem.174.5.957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heber-Katz E., Acha-Orbea H. The V-region disease hypothesis: evidence from autoimmune encephalomyelitis. Immunol Today. 1989 May;10(5):164–169. doi: 10.1016/0167-5699(89)90174-6. [DOI] [PubMed] [Google Scholar]
- Howell M. D., Winters S. T., Olee T., Powell H. C., Carlo D. J., Brostoff S. W. Vaccination against experimental allergic encephalomyelitis with T cell receptor peptides. Science. 1989 Nov 3;246(4930):668–670. doi: 10.1126/science.2814489. [DOI] [PubMed] [Google Scholar]
- Matsumoto Y., Fujiwara M. Adoptively transferred experimental allergic encephalomyelitis in chimeric rats: identification of transferred cells in the lesions of the central nervous system. Immunology. 1988 Sep;65(1):23–29. [PMC free article] [PubMed] [Google Scholar]
- Matsumoto Y., Kawai K., Fujiwara M. Analysis of the T cell repertoire for myelin basic protein in thymus-grafted and other types of chimera: evidence that major histocompatibility complex molecules on accessory cells rather than T cell specificity mainly regulate susceptibility to autoimmune encephalomyelitis. Eur J Immunol. 1990 Sep;20(9):2119–2126. doi: 10.1002/eji.1830200934. [DOI] [PubMed] [Google Scholar]
- Matsumoto Y., Kawai K., Tomita Y., Fujiwara M. Limiting-dilution analysis of the frequency of myelin basic protein-reactive T cells in Lewis, PVG/c and BN rats. Implication for susceptibility to autoimmune encephalomyelitis. Immunology. 1990 Feb;69(2):215–221. [PMC free article] [PubMed] [Google Scholar]
- Matsumoto Y., Ohmori K., Fujiwara M. Microglial and astroglial reactions to inflammatory lesions of experimental autoimmune encephalomyelitis in the rat central nervous system. J Neuroimmunol. 1992 Mar;37(1-2):23–33. doi: 10.1016/0165-5728(92)90152-b. [DOI] [PubMed] [Google Scholar]
- Offner H., Hashim G. A., Vandenbark A. A. T cell receptor peptide therapy triggers autoregulation of experimental encephalomyelitis. Science. 1991 Jan 25;251(4992):430–432. doi: 10.1126/science.1989076. [DOI] [PubMed] [Google Scholar]
- Offner H., Vainiene M., Gold D. P., Celnik B., Wang R., Hashim G. A., Vandenbark A. A. Characterization of the immune response to a secondary encephalitogenic epitope of basic protein in Lewis rats. I. T cell receptor peptide regulation of T cell clones expressing cross-reactive V beta genes. J Immunol. 1992 Mar 15;148(6):1706–1711. [PubMed] [Google Scholar]
- Ohmori K., Hong Y., Fujiwara M., Matsumoto Y. In situ demonstration of proliferating cells in the rat central nervous system during experimental autoimmune encephalomyelitis. Evidence suggesting that most infiltrating T cells do not proliferate in the target organ. Lab Invest. 1992 Jan;66(1):54–62. [PubMed] [Google Scholar]
- Sriram S., Steinman L. Anti I-A antibody suppresses active encephalomyelitis: treatment model for diseases linked to IR genes. J Exp Med. 1983 Oct 1;158(4):1362–1367. doi: 10.1084/jem.158.4.1362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinman L., Rosenbaum J. T., Sriram S., McDevitt H. O. In vivo effects of antibodies to immune response gene products: prevention of experimental allergic encephalitis. Proc Natl Acad Sci U S A. 1981 Nov;78(11):7111–7114. doi: 10.1073/pnas.78.11.7111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinman L., Solomon D., Lim M., Zamvil S., Sriram S. Prevention of experimental allergic encephalitis with in vivo administration of anti I-A antibody. Decreased accumulation of radiolabelled lymph node cells in the central nervous system. J Neuroimmunol. 1983 Aug;5(1):91–97. doi: 10.1016/0165-5728(83)90029-2. [DOI] [PubMed] [Google Scholar]
- Su X. M., Sriram S. Analysis of TCR V beta gene usage and encephalitogenicity of myelin basic protein peptide p91-103 reactive T cell clones in SJL mice: lack of evidence for V gene hypothesis. Cell Immunol. 1992 May;141(2):485–495. doi: 10.1016/0008-8749(92)90165-l. [DOI] [PubMed] [Google Scholar]
- Sun D., Gold D. P., Smith L., Brostoff S., Coleclough C. Characterization of rat encephalitogenic T cells bearing non-V beta 8 T cell receptors. Eur J Immunol. 1992 Feb;22(2):591–594. doi: 10.1002/eji.1830220244. [DOI] [PubMed] [Google Scholar]
- Urban J. L., Kumar V., Kono D. H., Gomez C., Horvath S. J., Clayton J., Ando D. G., Sercarz E. E., Hood L. Restricted use of T cell receptor V genes in murine autoimmune encephalomyelitis raises possibilities for antibody therapy. Cell. 1988 Aug 12;54(4):577–592. doi: 10.1016/0092-8674(88)90079-7. [DOI] [PubMed] [Google Scholar]
- Vandenbark A. A., Hashim G., Offner H. Immunization with a synthetic T-cell receptor V-region peptide protects against experimental autoimmune encephalomyelitis. Nature. 1989 Oct 12;341(6242):541–544. doi: 10.1038/341541a0. [DOI] [PubMed] [Google Scholar]
- Waldor M. K., Sriram S., Hardy R., Herzenberg L. A., Herzenberg L. A., Lanier L., Lim M., Steinman L. Reversal of experimental allergic encephalomyelitis with monoclonal antibody to a T-cell subset marker. Science. 1985 Jan 25;227(4685):415–417. doi: 10.1126/science.3155574. [DOI] [PubMed] [Google Scholar]
- White R. A., Mason D. W., Williams A. F., Galfre G., Milstein C. T-lymphocyte heterogeneity in the rat: separation of functional subpopulations using a monoclonal antibody. J Exp Med. 1978 Sep 1;148(3):664–673. doi: 10.1084/jem.148.3.664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams A. F., Barclay A. N., Clark S. J., Paterson D. J., Willis A. C. Similarities in sequences and cellular expression between rat CD2 and CD4 antigens. J Exp Med. 1987 Feb 1;165(2):368–380. doi: 10.1084/jem.165.2.368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams A. F., Galfrè G., Milstein C. Analysis of cell surfaces by xenogeneic myeloma-hybrid antibodies: differentiation antigens of rat lymphocytes. Cell. 1977 Nov;12(3):663–673. doi: 10.1016/0092-8674(77)90266-5. [DOI] [PubMed] [Google Scholar]
- Yoshino S., Cleland L. G. Depletion of alpha/beta T cells by a monoclonal antibody against the alpha/beta T cell receptor suppresses established adjuvant arthritis, but not established collagen-induced arthritis in rats. J Exp Med. 1992 Apr 1;175(4):907–915. doi: 10.1084/jem.175.4.907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshino S., Cleland L. G., Mayrhofer G., Brown R. R., Schwab J. H. Prevention of chronic erosive streptococcal cell wall-induced arthritis in rats by treatment with a monoclonal antibody against the T cell antigen receptor alpha beta. J Immunol. 1991 Jun 15;146(12):4187–4189. [PubMed] [Google Scholar]
- Yoshino S., Schlipköter E., Kinne R., Hünig T., Emmrich F. Suppression and prevention of adjuvant arthritis in rats by a monoclonal antibody to the alpha/beta T cell receptor. Eur J Immunol. 1990 Dec;20(12):2805–2808. doi: 10.1002/eji.1830201241. [DOI] [PubMed] [Google Scholar]


