Abstract
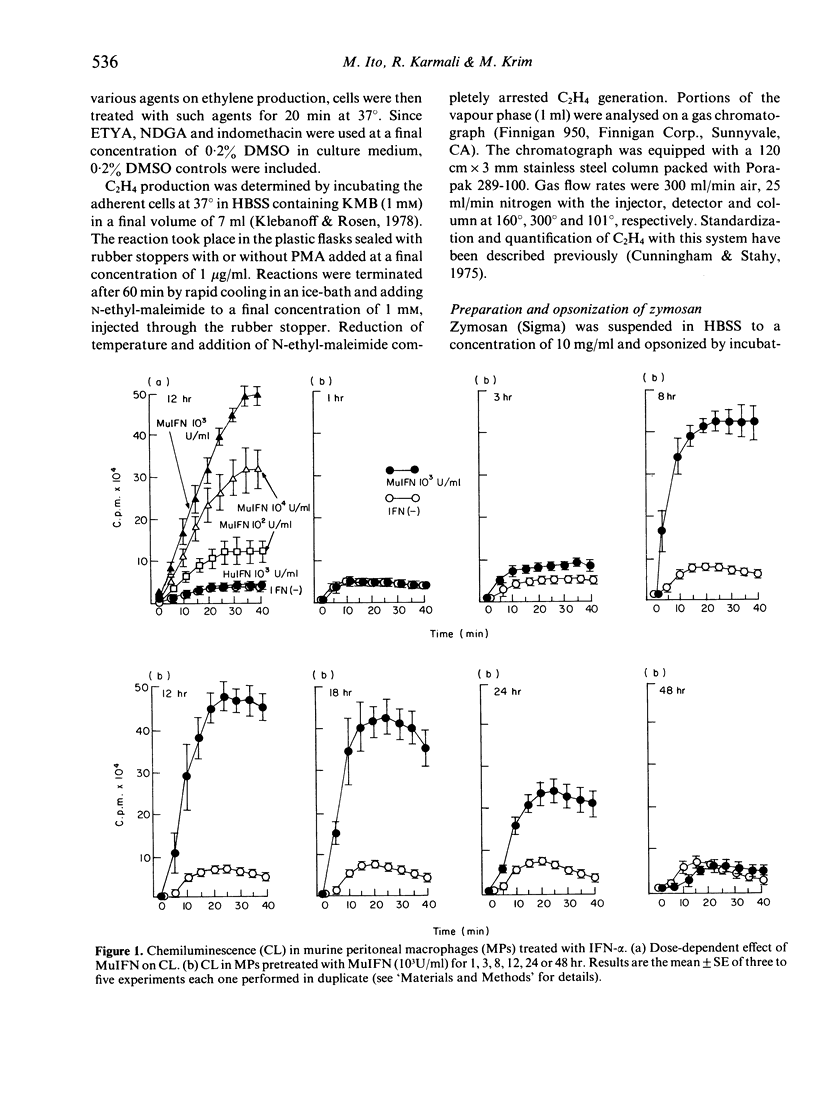
Considerably augmented chemiluminescence (CL) occurred when murine peritoneal resident macrophages (MPs), pretreated with murine interferon (MuIFN)-alpha within 24 hr, were stimulated by 4-beta-phorbol, 12-beta-myristate, 13-beta-acetate (PMA). Augmentation of CL generation ceased when incubation in the presence of MuIFN was continued for 48 hr. As 12 hr preincubation with MuIFN procured optimal CL generation, the various reactive oxygen species (OH, O2-., H2O2) were measured at that point. The hydroxyl radical (OH.) level in MuIFN-treated MPs was 19.44 times higher than in MuIFN-untreated MPs. However, the levels of O2-. and H2O2 generation were the same in both MuIFN-treated and untreated MPs. Moreover, by using the inhibitors lipoxygenase and cyclo-oxygenase, we established clearly that CL and OH. generation in MuIFN-treated MPs is due to the lipoxygenase pathway of arachidonic acid metabolism.
Full text
PDF







Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Boraschi D., Ghezzi P., Pasqualetto E., Salmona M., Nencioni L., Soldateschi D., Villa L., Tagliabue A. Interferon decreases production of hydrogen peroxide by macrophages: correlation with reduction of suppressive capacity and of anti-microbial activity. Immunology. 1983 Nov;50(3):359–368. [PMC free article] [PubMed] [Google Scholar]
- Boraschi D., Ghezzi P., Salmona M., Tagliabue A. IFN-beta-induced reduction of superoxide anion generation by macrophages. Immunology. 1982 Apr;45(4):621–628. [PMC free article] [PubMed] [Google Scholar]
- Cantell K., Hirvonen S. Large-scale production of human leukocyte interferon containing 10(8) units per ml. J Gen Virol. 1978 Jun;39(3):541–543. doi: 10.1099/0022-1317-39-3-541. [DOI] [PubMed] [Google Scholar]
- Dianzani F., Salter L., Fleischmann W. R., Jr, Zucca M. Immune interferon activates cells more slowly than does virus-induced interferon. Proc Soc Exp Biol Med. 1978 Oct;159(1):94–97. doi: 10.3181/00379727-159-40291. [DOI] [PubMed] [Google Scholar]
- Hatch G. E., Gardner D. E., Menzel D. B. Chemiluminescence of phagocytic cells caused by N-formylmethionyl peptides. J Exp Med. 1978 Jan 1;147(1):182–195. doi: 10.1084/jem.147.1.182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iwakura Y., Yonehara S., Kawade Y. Purification of mouse L cell interferon. Essentially pure preparations with associated cell growth inhibitory activity. J Biol Chem. 1978 Jul 25;253(14):5074–5079. [PubMed] [Google Scholar]
- Johnston R. B., Jr, Godzik C. A., Cohn Z. A. Increased superoxide anion production by immunologically activated and chemically elicited macrophages. J Exp Med. 1978 Jul 1;148(1):115–127. doi: 10.1084/jem.148.1.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnston R. B., Jr, Lehmeyer J. E., Guthrie L. A. Generation of superoxide anion and chemiluminescence by human monocytes during phagocytosis and on contact with surface-bound immunoglobulin G. J Exp Med. 1976 Jun 1;143(6):1551–1556. doi: 10.1084/jem.143.6.1551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karmali R. A., Muse P., Allen G., Louis T. Macrophage production of prostaglandins: effects of fetal calf serum and diazepam. Use of an improved method for extracting 6-keto-PGF1 alpha. Prostaglandins Leukot Med. 1982 Jun;8(6):565–577. [PubMed] [Google Scholar]
- Klebanoff S. J. Oxygen metabolism and the toxic properties of phagocytes. Ann Intern Med. 1980 Sep;93(3):480–489. doi: 10.7326/0003-4819-93-3-480. [DOI] [PubMed] [Google Scholar]
- Klebanoff S. J., Rosen H. Ethylene formation by polymorphonuclear leukocytes. Role of myeloperoxidase. J Exp Med. 1978 Aug 1;148(2):490–506. doi: 10.1084/jem.148.2.490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuehl F. A., Jr, Egan R. W. Prostaglandins, arachidonic acid, and inflammation. Science. 1980 Nov 28;210(4473):978–984. doi: 10.1126/science.6254151. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Murray H. W., Cohn Z. A. Macrophage oxygen-dependent antimicrobial activity. III. Enhanced oxidative metabolism as an expression of macrophage activation. J Exp Med. 1980 Dec 1;152(6):1596–1609. doi: 10.1084/jem.152.6.1596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nathan C. F., Murray H. W., Cohn Z. A. The macrophage as an effector cell. N Engl J Med. 1980 Sep 11;303(11):622–626. doi: 10.1056/NEJM198009113031106. [DOI] [PubMed] [Google Scholar]
- Nathan C. F., Murray H. W., Wiebe M. E., Rubin B. Y. Identification of interferon-gamma as the lymphokine that activates human macrophage oxidative metabolism and antimicrobial activity. J Exp Med. 1983 Sep 1;158(3):670–689. doi: 10.1084/jem.158.3.670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosen H., Klebanoff S. J. Chemiluminescence and superoxide production by myeloperoxidase-deficient leukocytes. J Clin Invest. 1976 Jul;58(1):50–60. doi: 10.1172/JCI108458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schultz R. M., Papamatheakis J. D., Chirigos M. A. Interferon: an inducer of macrophage activation by polyanions. Science. 1977 Aug 12;197(4304):674–676. doi: 10.1126/science.877584. [DOI] [PubMed] [Google Scholar]
- Smith R. L., Weidemann M. J. Reactive oxygen production associated with arachidonic acid metabolism by peritoneal macrophages. Biochem Biophys Res Commun. 1980 Dec 16;97(3):973–980. doi: 10.1016/0006-291x(80)91472-2. [DOI] [PubMed] [Google Scholar]
- Smolen J. E., Weissmann G. Effects of indomethacin, 5,8,11,14-eicosatetraynoic acid, and p-bromophenacyl bromide on lysosomal enzyme release and superoxide anion generation by human polymorphonuclear leukocytes. Biochem Pharmacol. 1980 Feb 15;29(4):533–538. doi: 10.1016/0006-2952(80)90373-1. [DOI] [PubMed] [Google Scholar]
- Stevens P., Winston D. J., Van Dyke K. In vitro evaluation of opsonic and cellular granulocyte function by luminol-dependent chemiluminescence: utility in patients with severe neutropenia and cellular deficiency states. Infect Immun. 1978 Oct;22(1):41–51. doi: 10.1128/iai.22.1.41-51.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suthanthiran M., Solomon S. D., Williams P. S., Rubin A. L., Novogrodsky A., Stenzel K. H. Hydroxyl radical scavengers inhibit human natural killer cell activity. Nature. 1984 Jan 19;307(5948):276–278. doi: 10.1038/307276a0. [DOI] [PubMed] [Google Scholar]
- Weiss S. J., Rustagi P. K., LoBuglio A. F. Human granulocyte generation of hydroxyl radical. J Exp Med. 1978 Feb 1;147(2):316–323. doi: 10.1084/jem.147.2.316. [DOI] [PMC free article] [PubMed] [Google Scholar]


