Abstract
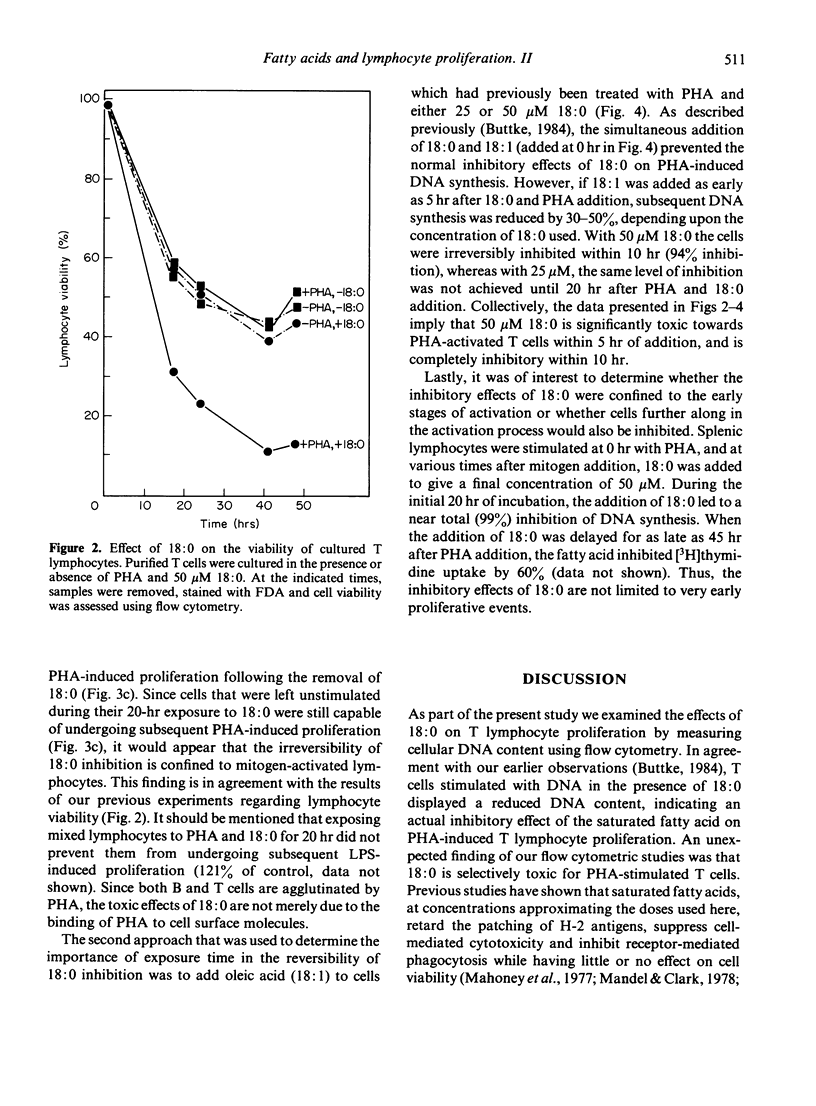
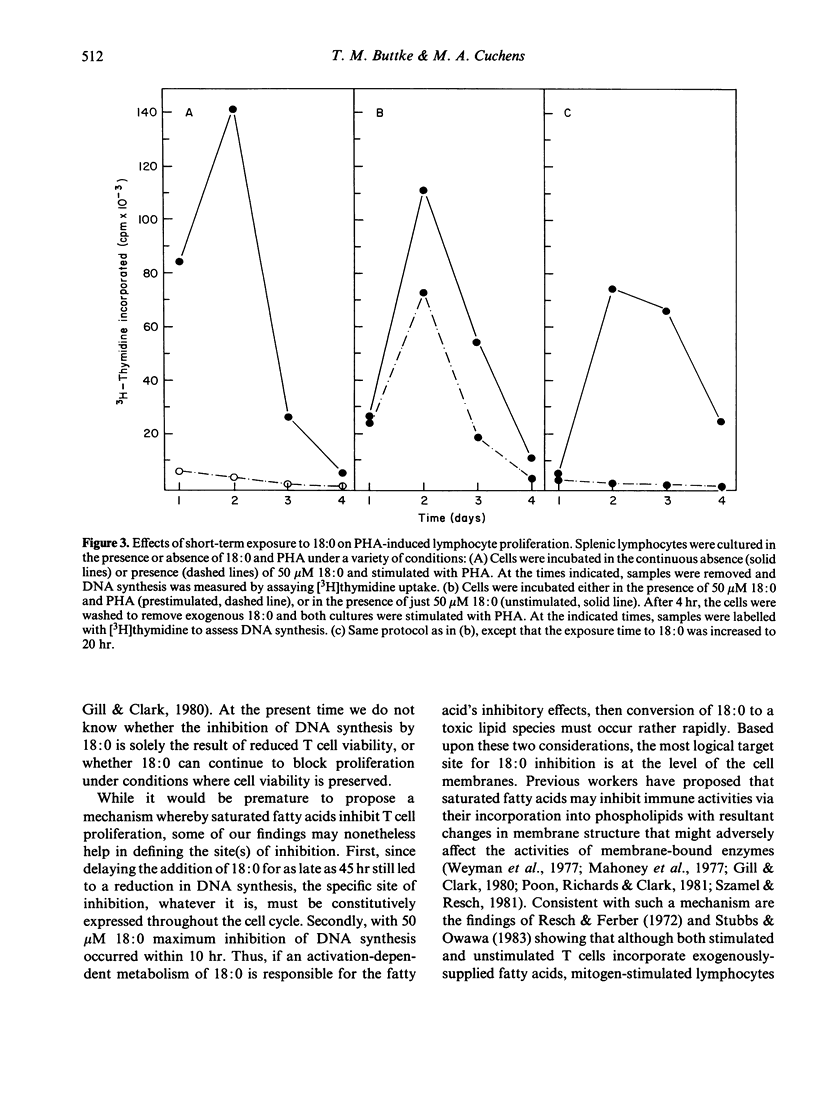
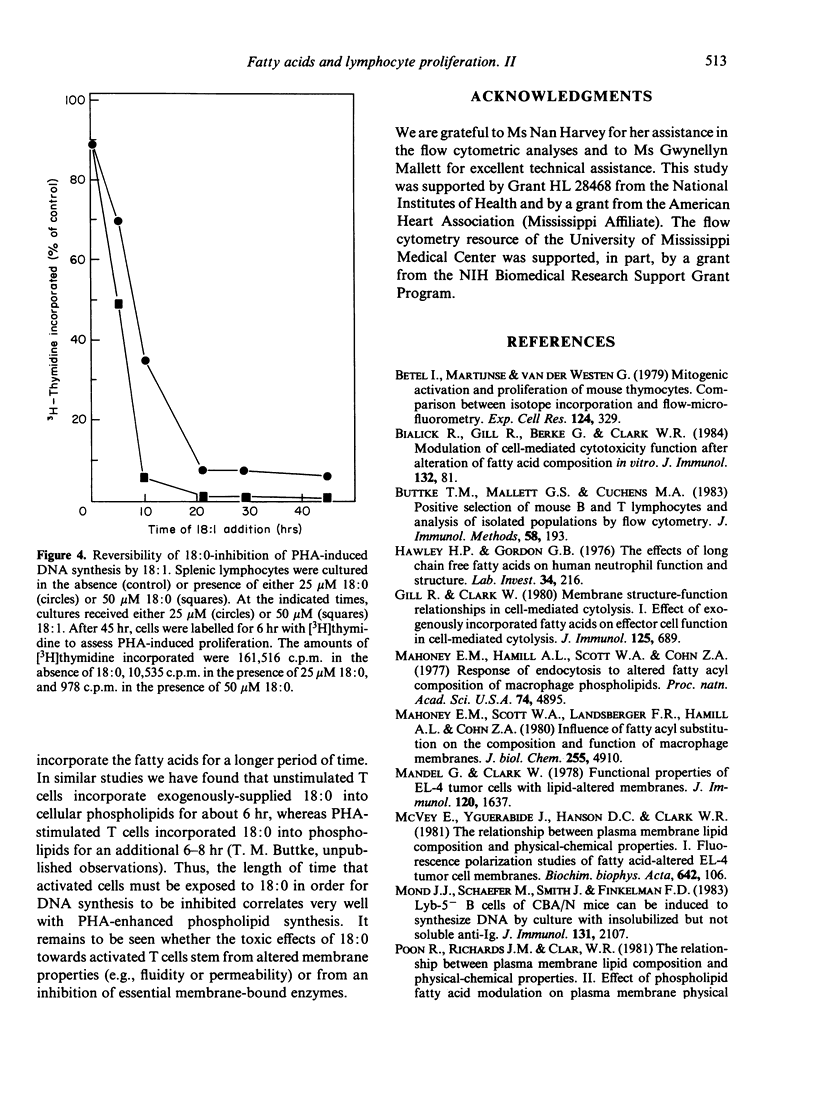
Studies were performed to further characterize the effects of saturated fatty acids on murine T lymphocyte proliferation. Flow cytometry was used to show that the inhibitory effects of stearic acid (18:0) on [3H]thymidine uptake can be correlated with changes in cellular DNA content. Additional studies using flow cytometry and fluorescein diacetate as a viability stain showed that exogenous 18:0 was toxic for phytohemagglutinin (PHA)-stimulated T cells, whereas the viability of unstimulated T cells was less affected by 18:0. The inhibitory effects of 18:0 on T cell proliferation were evident as early as 4 hr after fatty acid addition and after a 10-hr exposure, the effects of 18:0 could not be reversed by washing the cells or by adding oleic acid (18:1). It is proposed that the inhibitory effects of 18:0 are dependent upon PHA-induced changes in T cell lipid metabolism.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Betel I., Martijnse J., van der Westen G. Mitogenic activation and proliferation of mouse thymocytes. Comparison between isotope incorporation and flow-microfluorometry. Exp Cell Res. 1979 Dec;124(2):329–337. doi: 10.1016/0014-4827(79)90208-8. [DOI] [PubMed] [Google Scholar]
- Bialick R., Gill R., Berke G., Clark W. R. Modulation of cell-mediated cytotoxicity function after alteration of fatty acid composition in vitro. J Immunol. 1984 Jan;132(1):81–87. [PubMed] [Google Scholar]
- Buttke T. M., Mallett G. S., Cuchens M. A. Positive selection of mouse B and T lymphocytes and analysis of isolated populations by flow cytometry. J Immunol Methods. 1983 Mar 11;58(1-2):193–207. doi: 10.1016/0022-1759(83)90275-2. [DOI] [PubMed] [Google Scholar]
- Gill R., Clark W. Membrane structure-function relationships in cell-mediated cytolysis. I. Effect of exogenously incorporated fatty acids on effector cell function in cell-mediated cytolysis. J Immunol. 1980 Aug;125(2):689–695. [PubMed] [Google Scholar]
- Hawley H. P., Gordon G. B. The effects of long chain free fatty acids on human neutrophil function and structure. Lab Invest. 1976 Feb;34(2):216–222. [PubMed] [Google Scholar]
- Mahoney E. M., Hamill A. L., Scott W. A., Cohn Z. A. Response of endocytosis to altered fatty acyl composition of macrophage phospholipids. Proc Natl Acad Sci U S A. 1977 Nov;74(11):4895–4899. doi: 10.1073/pnas.74.11.4895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mahoney E. M., Scott W. A., Landsberger F. R., Hamill A. L., Cohn Z. A. Influence of fatty acyl substitution on the composition and function of macrophage membranes. J Biol Chem. 1980 May 25;255(10):4910–4917. [PubMed] [Google Scholar]
- Mandel G., Clark W. Functional properties of EL-4 tumor cells with lipid-altered membranes. J Immunol. 1978 May;120(5):1637–1643. [PubMed] [Google Scholar]
- McVey E., Yguerabide J., Hanson D. C., Clark W. R. The relationship between plasma membrane lipid composition and physical-chemical properties. I. Fluorescence polarization studies of fatty acid-altered EL4 tumor cell membranes. Biochim Biophys Acta. 1981 Mar 20;642(1):106–118. doi: 10.1016/0005-2736(81)90142-5. [DOI] [PubMed] [Google Scholar]
- Mond J. J., Schaefer M., Smith J., Finkelman F. D. Lyb-5- B cells of CBA/N mice can be induced to synthesize DNA by culture with insolubilized but not soluble anti-Ig. J Immunol. 1983 Nov;131(5):2107–2109. [PubMed] [Google Scholar]
- Resch K., Ferber E. Phospholipid metabolism of stimulated lymphocytes. Effects of phytohemagglutinin, concanavalin A and anti-immunoglobulin serum. Eur J Biochem. 1972 May;27(1):153–161. doi: 10.1111/j.1432-1033.1972.tb01821.x. [DOI] [PubMed] [Google Scholar]
- Rotman B., Papermaster B. W. Membrane properties of living mammalian cells as studied by enzymatic hydrolysis of fluorogenic esters. Proc Natl Acad Sci U S A. 1966 Jan;55(1):134–141. doi: 10.1073/pnas.55.1.134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steen H. B., Lindmo T. Initiation of the blastogenic response of lymphocytes by hyperoptimal concentrations of concanavalin A. Eur J Immunol. 1979 Jun;9(6):434–439. doi: 10.1002/eji.1830090604. [DOI] [PubMed] [Google Scholar]
- Stubbs C. D., Osawa T. The effect of concanavalin A on the incorporation of fatty acids into lymphocyte phospholipids. J Pharmacobiodyn. 1983 Mar;6(3):217–224. doi: 10.1248/bpb1978.6.217. [DOI] [PubMed] [Google Scholar]
- Stubbs C. D., Tsang W. M., Belin J., Smith A. D., Johnson S. M. Incubation of exogenous fatty acids with lymphocytes. Changes in fatty acid composition and effects on the rotational relaxation time of 1,6-diphenyl-1,3,5-hexatriene. Biochemistry. 1980 Jun 10;19(12):2756–2762. doi: 10.1021/bi00553a034. [DOI] [PubMed] [Google Scholar]
- Szamel M., Resch K. Modulation of enzyme activities in isolated lymphocyte plasma membranes by enzymatic modification of phospholipid fatty acids. J Biol Chem. 1981 Nov 25;256(22):11618–11623. [PubMed] [Google Scholar]
- Weyman C., Morgan S. J., Belin J., Smith A. D. Phytohaemagglutinin stimulation of human lymphocytes: effect of fatty acids on uridine uptake and phosphoglyceride fatty acid profile. Biochim Biophys Acta. 1977 Jan 24;496(1):155–166. doi: 10.1016/0304-4165(77)90123-4. [DOI] [PubMed] [Google Scholar]


