Abstract
The Bloom's syndrome helicase, BLM, is a member of the highly conserved RecQ family, and possesses both DNA unwinding and DNA strand annealing activities. BLM also promotes branch migration of Holliday junctions. One role for BLM is to act in conjunction with topoisomerase IIIα to process homologous recombination (HR) intermediates containing a double Holliday junction by a process termed dissolution. However, several lines of evidence suggest that BLM may also act early in one or more of the recombination pathways to eliminate illegitimate or aberrantly paired DNA joint molecules. We have investigated whether BLM can disrupt DNA displacement loops (D-loops), which represent the initial strand invasion step of HR. We show that mobile D-loops created by the RecA recombinase are a highly preferred substrate for BLM with the invading strand being displaced from the duplex. We have identified structural features of the D-loop that determine the efficiency with which BLM promotes D-loop dissociation. We discuss these results in the context of models for the role of BLM as an ‘anti-recombinase’.
INTRODUCTION
Bloom's syndrome (BS) is an extremely rare genetic disorder, characterized by proportional dwarfism, a sun-sensitive facial erythema, male sterility and female sub-fertility and non-insulin-dependent diabetes mellitus in young patients. The most striking feature, however, is the high incidence of malignancies of most types (1). Cells carrying mutations in the BS gene (designated BLM) display decreased plating efficiency and various signs of genomic instability; namely, chromosomal rearrangements, an elevated frequency of homologous recombination (HR), and sensitivity to certain DNA damaging agents and replication inhibitors [(2) and references therein]. The hallmark feature of these cells, which is used in the diagnosis of the disorder, is an elevated frequency of sister-chromatid exchanges (SCEs) (3).
The BS gene product, BLM, is a DNA helicase belonging to the highly conserved RecQ family (4). BLM is a DNA structure-specific helicase that displays 3′→5′ directionality (5). The RecQ family of helicases includes four additional members in humans: WRN, the gene defective in Werner's syndrome (WS) (6,7); RECQ4, associated with some cases of Rothmund–Thomson syndrome (8) and RAPADILINO (9); RECQ1 (10,11); and RECQ5 (12). Unicellular organisms generally contain only one representative of the family, such as the founding member in Escherichia coli, RecQ (13), and Sgs1 in the budding yeast, Saccharomyces cerevisiae (14,15).
Although the precise function of any RecQ helicase is yet to be established, several lines of evidence suggest that they play roles in HR. HR is important for the repair of DNA double strand breaks (DSBs), for the restoration of productive DNA synthesis following disruption of DNA replication forks, and for meiosis, where it is essential for creation of genetic diversity and for faithful chromosome segregation. Although the biochemical details of HR are not fully worked out, several proteins are required to process DSBs to create a 3′ ssDNA tail that can invade a homologous duplex to create a three-stranded, branched structure called a displacement loop (D-loop). The key catalytic factor in D-loop formation is the Rad51 recombinase. It polymerizes on the ssDNA tail generated by resection of the DSB to create an extended filament in which the search for homology is conducted. This function requires the action of several accessory factors in yeast, including the Rad50/Mre11/Xrs2 complex, replication protein A (RPA), Rad52, Rad54 and the Rad51-related proteins, Rad55 and Rad57. In mammals, this basic process in conserved, although it requires some additional factors; most notably, the BRCA2 protein that is associated with some cases of hereditary breast and ovarian cancer (16,17).
RecQ helicase mutants frequently display an elevated rate of HR, which in BS cells is manifested as an increase in the frequency of SCEs and inter-homologue recombination (3). BLM-defective cells of mouse or chicken origin show an elevated frequency of gene targeting by HR, as well as loss of heterozygosity (18–20), which can arise when crossing-over events occur between recombining homologous chromosomes. Similarly, E.coli recQ mutants (21) and S.cerevisiae sgs1 mutants (22) show excessive illegitimate recombination, suggestive of a failure to suppress inappropriate recombination occurring between non-identical sequences. The presence of a constitutively high level of RAD51-containing nuclear foci in BS cells, even outside of S-phase where they normally exist, also indicates the presence of a constitutively high number of sites of ongoing or incomplete HR events (23,24). Moreover, RecQ helicases show direct physical interactions with HR proteins; e.g. BLM and Sgs1 each bind to the Rad51 protein in their respective organisms through a conserved C-terminal interaction domain (23).
There is evidence of roles for RecQ helicases both early and late in the HR pathway. Rockmill et al. (25) showed that Sgs1 is required for the suppression of meiotic cross-overs by decreasing the number of sites of so-called axial associations, which are sites for reciprocal exchange of recombining homologues. This would suggest an early role in eliminating unwanted or aberrant recombination sites, at least in meiosis, and is consistent with the hyper-recombination phenotype seen in many RecQ helicase mutants. Nevertheless, there are also several reports that point to a late role for RecQ helicases in HR. First, certain defects in yeast RecQ helicase mutants are at least partially suppressed by deletion of RAD51 or other early acting HR proteins (23,26,27). Second, sgs1 mutants exposed to the DNA damaging agent methyl methane sulphonate during S-phase accumulate Rad51-dependent, X-shaped structures, suggestive of a failure to resolve a DNA recombination intermediate (28). Third, BLM and topoisomerase IIIα together promote the so-called dissolution of a double Holliday junction structure, a model for a late recombination intermediate (29). These apparently conflicting conclusions concerning the stage during HR that RecQ helicases might act could reflect a dual role for these enzymes in HR. One plausible explanation is that RecQ helicases act alone at early stages in HR to disrupt paired DNA intermediates deemed inappropriate for one reason or another, and that they also act late, this time alongside topoisomerase IIIα, to resolve recombination intermediates.
In this study, we set out to test the hypothesis that the BLM helicase can perform an anti-recombinase role through the disruption of the D-loop recombination intermediate. For this, we specifically utilized RecA-generated mobile (or migratable) D-loop structures. We report that BLM is highly efficient at displacing the invading strand of mobile D-loops, and have identified structural features of the D-loop that direct BLM to act in this manner.
MATERIALS AND METHODS
Proteins
Recombinant BLM was purified as described previously (30).
Oligonucleotides
Oligonucleotide sequences used in this study are listed in Table 1. Additionally, the X12 four-way junction, X12 replication fork and OX-1T G-quadruplex substrates described by Mohaghegh et al. (31) were also generated. The oligonucleotides were either custom synthesized by Sigma–Genosys (Sigma–Aldrich House, Haverhill, UK), or provided by the oligonucleotide synthesis service of Cancer Research UK (CR-UK). Reverse polarity oligonucleotides were synthesized using 5′-CE phosphoramidites and 5′-supports (Glen Research, Sterling, VA) by the CR-UK oligonucleotide synthesis service. All oligonucleotides were purified on a 10% denaturing polyacrylamide gel before use. Labelling was carried out using either T4 polynucleotide kinase (New England Biolabs, Beverly, MA) and [γ-32P]ATP (Amersham Biosciences, Chalfont St.Giles, UK), or terminal deoxynucleotidyl transferase (New England Biolabs) and [α-32P]ddATP (Amersham) according to the manufacturer's instructions. Activity of the labelled oligonucleotides was measured and specific activity was calculated.
Table 1.
Sequences and description of D-loop forming oligonucleotides used in this study
Yellow highlighting denotes the invading, complementary region. Blue highlighting marks the region of scrambled pUC18 sequences that can form a bubble when aligned to pUCbottom. White letters on a black background denote positions of G/T mismatches.
Construction of substrates
The oligonucleotide-based D-loops were prepared by the sequential annealing of the constituent oligonucleotides, as described by Opresko et al. (32). The X12 four-way junction, replication fork and OX-1T G4 substrates were generated as described previously (31). The plasmid-based D-loop substrates were generated as described by McIlwraith et al. (33). Briefly, the invading oligonucleotide (Table 1) was coated with RecA (New England Biolabs), for 5 min, and then supercoiled pUC18 plasmid was added to initiate strand invasion. The strand invasion reaction was allowed to progress for 3 min. The DNA was then deproteinized using proteinase K in the presence of SDS for 30 min. Substrates were purified essentially as described previously (31), but using 5% polyacrylamide gels to achieve better recovery of large D-loop molecules. The purified substrates were stored in 10 mM Tris–HCl (pH 7.5) and 10 mM MgCl2 (for D-loops and standard B-form DNA substrates) or 100 mM NaCl, 10 mM Tris–HCl (pH 8.0), 1 mM EDTA (for G4 DNA). Activity was measured and, using the specific activity of the corresponding oligonucleotide, the substrate concentration was calculated.
Helicase assays
Helicase assays were carried out in a 10 µl reaction volume containing 1× helicase buffer [33 mM Tris-acetate (pH 7.8); 1 mM Mg-acetate; 66 mM Na-acetate; 0.1 mg/ml BSA; 1 mM DTT; 1 mM ATP], 56 pM substrate and the indicated amount of enzyme for 15 min at 37°C, unless otherwise stated. The reaction products were separated by non-denaturing PAGE. The gels were dried, and radiolabelled DNA was visualized using a Storm 840 PhosphorImager. Band intensities were quantified using ImageQuant software (Amersham). Quantitative results were expressed as percent unwinding normalized to activities in the untreated and denatured controls, as described previously (31). Enzyme concentrations required for 50% unwinding, as well as inhibitor concentrations required for 50% inhibition (IC50 values), were determined after fitting a sigmoid curve with the non-linear regression model in the SPSS software package.
RESULTS
We set out to test the hypothesis that one mechanism by which BLM acts as an antirecombinase is through its ability to disrupt the DNA D-loop that forms at the initial strand invasion step of HR. In this way, BLM could abort HR reactions that are deemed inappropriate; e.g. because the recombining sequences are not identical. Previous studies indicated that RecQ family helicases are able to disrupt oligonucleotide-based mimics of a D-loop created by the annealing of three partially complementary oligonucleotides (32,34,35). We considered it unlikely, however, that these D-loop mimics represent an ideal model for the D-loop structure generated in vivo for several reasons. First, oligonucleotide-based D-loops are ‘static’ and are not capable of branch migration along the DNA, in contrast to genuine ‘mobile’ D-loops created enzymatically by the RecA recombinase or one of its eukaryotic orthologues. Second, the unwinding of oligonucleotide-based D-loops generates products that can be re-annealed by the ssDNA annealing activity of BLM (36,37), potentially generating artifactual products that could never arise when mobile D-loops are employed. Third, oligonucleotide-based D-loops cannot be generated in negatively-supercoiled molecules, unlike mobile D-loops, and do not, therefore, model the in vivo situation. Fourth, oligonucleotide-based D-loops present recognition features for helicases that are not evident in mobile D-loops or in vivo; in particular, two free dsDNA ends.
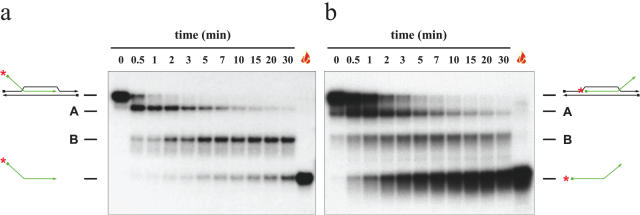
We considered the possibility that oligonucleotide-based D-loops might be disrupted by a mechanism different from that employed for the disruption of mobile D-loops. We anticipated that the physiologically-relevant mechanism of disruption would entail the expulsion of the invading ssDNA from the recipient duplex in a single step. If such a mechanism were not obviously in evidence during disruption of oligonucleotide-based D-loops, then this would suggest that studies of D-loop unwinding by helicases should focus exclusively on mobile D-loop substrates. Indirect evidence to suggest that this proposal might have some credence came from our analysis of the data presented previously by van Brabant et al. (34). This analysis suggested that the ‘invading’ oligonucleotide was not simply being expelled from the duplex in a single step because reaction intermediates were evident in the data presented by these authors (34). Using substrate oDL3 (see Materials and Methods and Figure 1), we confirmed the results of van Brabant et al. (34) indicating that BLM can disrupt oligonucleotide-based D-loops (Figure 2). Moreover, we also observed at least two reaction intermediates that contained the 32P-labelled oligonucleotide, but had a slower mobility on PAGE than did the free ‘invading’ oligonucleotide. These intermediate species are designated A and B on Figure 2. From the time-course experiments presented in Figure 2a it is clear that the oligonucleotide-based D-loop substrate was unwound in a multi-step manner. Consistently, we found that species A appeared first over time, and was then converted to species B. Subsequently, species B gave rise to the radiolabelled single-stranded oligonucleotide. Using another substrate, this time with a 5′ invading end (oDL5; Figure 1), the pattern of appearance of products was different from that seen with oDL3. In this case, the single-stranded product was generated preferentially (Figure 2b), but again species A appeared as the first product. Species B was also observed; however, the timing of its appearance did not obviously precede that of the free single-stranded oligonucleotide. Species B had a mobility identical to that of a partially unwound intermediate comprising the bottom strand of the duplex annealed to the invading oligonucleotide (data not shown). These results would indicate that the unwinding of an oligonucleotide-based, static D-loop can proceed via the denaturation of the duplex portion of the substrate (equivalent to the recipient duplex in strand invasion during HR) to create a partially unwound intermediate that can be denatured in a second phase by BLM to yield the free oligonucleotide. This order of events is clearly not representative of the physiological process of D-loop disruption.
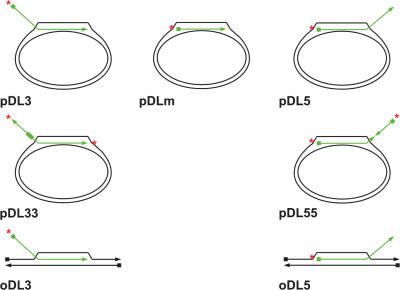
Figure 1.
Schematic representation of the different D-loop molecule species used in this study. The radiolabelled invading strand is shown in green. The triangles denote the 3′ end, while the squares denote the 5′ end of each oligonucleotide. The position of the radioactive label is indicated by a red asterisk.
Figure 2.
Kinetics of unwinding of oligonucleotide-based D-loops in a time-course experiment. Unwinding was carried out in the presence of 0.64 nM BLM for the period of time indicated above the lanes. The position of the substrate and fully unwound single-stranded oligonucleotide products, together with reaction intermediates A and B, are shown on each side. The flame symbol denotes heat-denatured DNA.
Generation of ‘mobile’ D-loops
Given the above considerations, we set out to generate mobile D-loops employing the established method of McIlwraith et al. (33). In this, RecA is used to catalyse the invasion of an 90mer single-stranded oligonucleotide into a negatively-supercoiled pUC18 plasmid DNA that contains a 54 bp region of sequence identity with the 3′ end of the oligonucleotide (Table 1 and Figure 1). The 5′ region of the oligonucleotide used was not complementary to the pUC18 and can form a 36 nt single-stranded tail protruding from the D-loop. By minor modification of the sequence of the oligonucleotide, it was a straightforward task to create D-loops incorporating one or more single base mismatches between the oligonucleotide and the recipient plasmid. This permitted us to analyse the influence of MMR factors on the ability of BLM to process the D-loop structure (see Discussion).
BLM catalyses efficient D-loop disruption
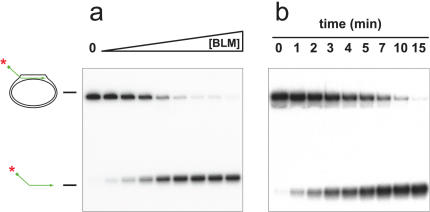
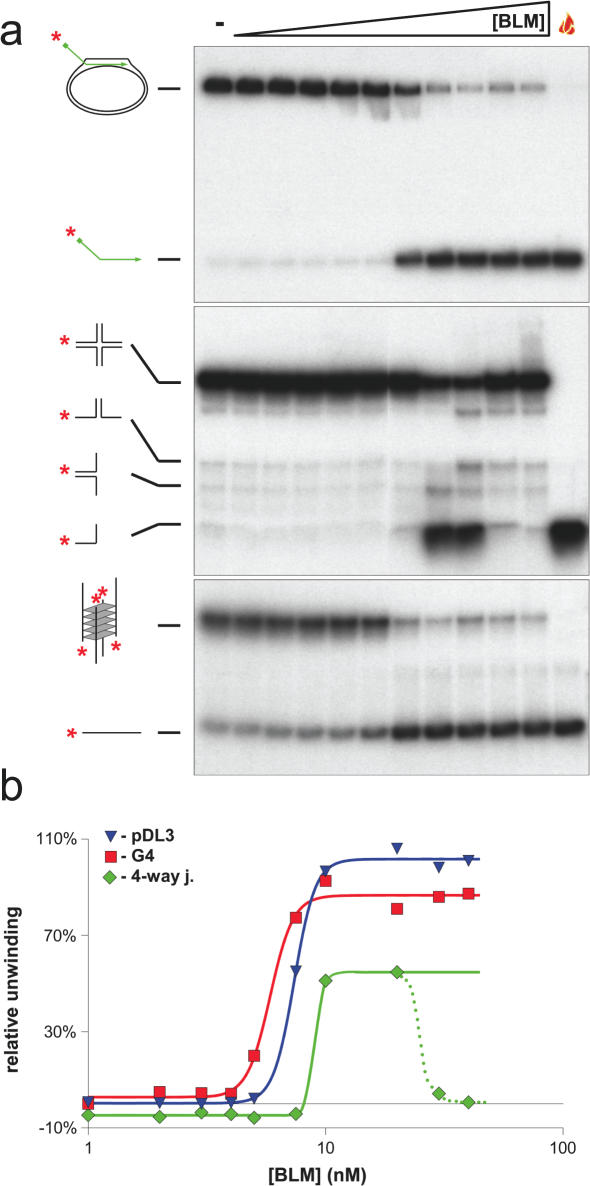
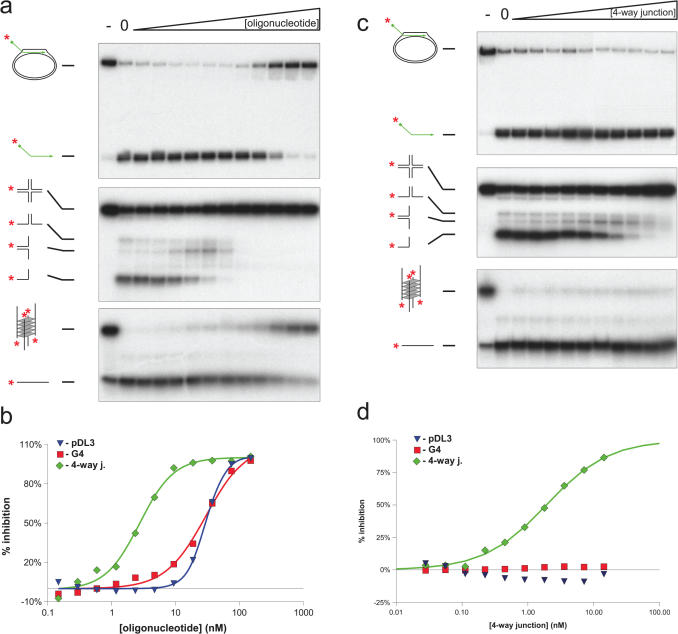
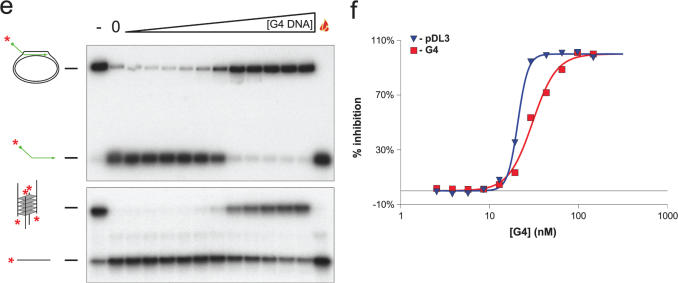
Using a conventional D-loop (pDL3), in which the 3′ end of the oligonucleotide was base paired with the pUC18 plasmid and the 32P-labelled 5′ tail was protruding, we tested whether BLM could disrupt the D-loop by displacing the invading strand. Using either a titration of increasing BLM concentration, or a time course at a fixed BLM concentration, we found that BLM could catalyse displacement of the 5′ labelled oligonucleotide from the D-loop (Figure 3). Based on our previous analyses of the unwinding of other DNA substrates by BLM, it appeared that the D-loop was very efficiently disrupted by BLM. To study this formally, we undertook additional analyses. First, we preformed a head-to-head comparison of the ability of BLM to disrupt the D-loop and two other DNA structures that have been shown previously to be efficient substrates for BLM; a four-way junction modelling the Holliday junction and a G-quadruplex structure created from sequences of the Oxytricha telomere repeat region. G-quadruplexes are acknowledged as the best substrate found to date not only for BLM, but also for most other RecQ helicases analysed (31,38–40). As shown in Figure 4, the BLM concentration required to disrupt the D-loop and the G-quadruplex were very similar (50% unwinding at 7.38 and 5.84 nM, respectively). In contrast, disruption of the four-way junction DNA consistently required a higher BLM concentration. Moreover, the extent of unwinding of this substrate peaked at a value below 100% and declined at high BLM concentrations. This phenomenon has been described previously and reflects the known DNA strand annealing activity of BLM (36,37) (discussed more in detail below).
Figure 3.
A plasmid-based, mobile D-loop is a substrate for BLM. (a) BLM-concentration dependent unwinding of pDL3 in a 15 min reaction. The enzyme concentration was varied (as indicated by the triangle above) between 0.7 and 5.6 nM in steps of 0.7. ‘0’ denotes a control sample in the absence of BLM, which was otherwise treated identically. (b) Unwinding of pDL3 in a time-course experiment. Samples were taken at the time points indicated above the lanes. The positions of the substrate and the free radiolabelled oligonucleotide are indicated on the left.
Figure 4.
Comparison of the efficiency of unwinding of different substrates by BLM. BLM enzyme-concentration dependent unwinding of the pDL3 mobile D-loop (upper panel), the X12 four-way junction (middle panel) and G4 (lower panel) substrates. The enzyme-concentration was varied between 1–40 nM as indicated above by the triangle. Reactions lacking BLM are denoted by ‘−’ above. The flame symbol denotes heat-denatured DNA. The substrates and the products of unwinding are shown on the left of each panel. (b) Quantification of the data from (a). Blue triangle, green diamond and red square symbols depict the pDL3, four-way junction and G4 DNA substrates, respectively. Analysis of the concentration-dependence curve indicated that 50% unwinding of pDL3, four-way junction and G4 DNA is achieved at 7.38, 8.97 and 5.84 nM, respectively. At high concentrations of BLM, the strand annealing activity of the enzyme becomes dominant over the unwinding activity, resulting in a diminution in the level of unwound product for the four-way junction substrate [middle panel of (a), green triangles and dotted line of (b)]. The last two concentration points (30 and 40 nM) were, therefore, omitted from the non-linear regression analysis and for determination of the 50% unwinding concentration (see Materials and Methods).
The above data suggested that the D-loop is very efficiently disrupted by BLM, and that it may rank alongside the G-quadruplex as a preferred substrate for BLM. To analyse this issue more quantitatively, competition experiments were performed. For this, increasing concentrations of unlabelled (cold) competitor DNA were added to helicase assays to determine the concentration of competitor DNA that reduced the level of unwinding of the D-loop, G-quadruplex or four-way junction DNA to 50% of control levels seen in the absence of competitor. The cold competitor DNAs analysed were an 80 nt oligonucleotide of unrelated sequences to any of the substrates, and the four-way junction and Oxytricha telomeric G-quadruplex described above. Unwinding of the four-way junction DNA was inhibited by the non-specific oligonucleotide competitor at concentrations of oligonucleotide ∼10-fold below those required to inhibit unwinding of the G-quadruplex or D-loop substrates (IC50 values of 2.73 nM for the four-way junction compared to 29.88 and 29.8 nM, respectively, for the G-quadruplex and D-loop;Figure 5a and b). Consistent with the G-quadruplex and D-loop molecules being significantly better substrates for BLM than was the four-way junction, competition experiments using cold four-way junction revealed efficient inhibition of unwinding of the labelled four-way junction substrate, but not of the G-quadruplex or D-loop substrates at the concentrations of competitor used (Figure 5c and d). If, as indicated by the above results, the D-loop is comparable to the G-quadruplex in being a highly efficient substrate for BLM, it would be predicted that cold G-quadruplex competitor DNA would inhibit unwinding of the labelled G-quadruplex essentially as efficiently as it would inhibit unwinding of the D-loop. We found, indeed, that this was the case, although it should be noted that the transition in the sigmoidal inhibition curve was sharper for the D-loop substrate than for the G-quadruplex substrate (Figure 5e and f). Taken together, these data indicate that BLM can disrupt a mobile D-loop with very high efficiency.
Figure 5.
Competition of BLM-dependent unwinding of different substrates. (a) Competition with a cold, unrelated, 80 nt oligonucleotide in reactions containing the pDL3 D-loop (upper panel), the X12 four-way junction (middle panel) and G4 DNA (lower panel). Lanes marked ‘0’ contained BLM but no competitor. (b) % inhibition expressed as a function of inhibitor concentration. A total of 50% inhibition of unwinding of pDL3, four-way junction and G4 was achieved at 29.8, 2.73 and 29.88 nM, respectively. (c) Competition with cold four-way junction. (d) % inhibition expressed as a function of inhibitor concentration. A total of 50% inhibition of unwinding of the radiolabelled four-way junction substrate occurred at 1.91 nM. No significant inhibition of unwinding of either the plasmid-based D-loop or G4 DNA was found in the concentration range used. (e) Competition with cold G4 DNA. (f) % inhibition expressed as a function of inhibitor concentration. A total of 50% inhibition of unwinding of the pDL3 (blue triangle) and G4 DNA (red square) substrates occurred at 20.78 and 30.39 nM concentration, respectively.
Structural features of D-loops that are recognized by BLM
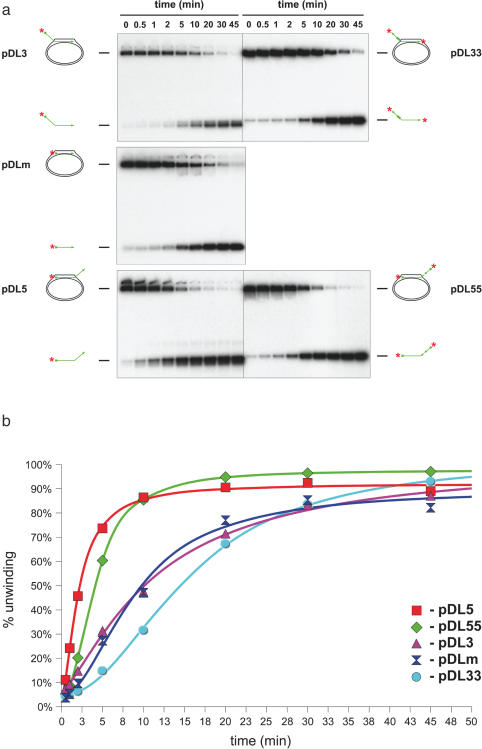
The D-loop substrate described above mimics an HR event in which the 3′ end of the ssDNA has invaded the homologous duplex, and the 5′ end is left protruding from the D-loop (designated pDL3; Figure 1). According to our current understanding, this is the situation that occurs during HR in vivo, because it permits priming of DNA synthesis from the 3′ end of the invading DNA. To address the key structural features of the D-loop that direct efficient BLM-mediated unwinding, we created two additional mobile D-loops: one with no protruding tail (pDLm) and one in which the 5′ end of the oligonucleotide had invaded and the 3′ end was protruding (pDL5). Analysis of the kinetics of unwinding of these three D-loop substrates indicated that the pDL5 molecule was unwound at a significantly faster rate than either the pDLm or pDL3 substrates (Figure 6). These data indicate that, while a protruding tail is not essential for BLM-mediated D-loop disruption, the orientation of the invading DNA must somehow influence the efficiency of D-loop unwinding.
Figure 6.
Analysis of the recognition features important for efficient unwinding of plasmid-based D-loops. (a) Head-to-head comparison of the efficiency of unwinding of the different plasmid-based D-loops in a time-course study in the presence of 0.52 nM BLM. Samples were taken at the time points indicated above the lanes. (b) Quantification of data from (a). Symbols used were: red square, pDL5; green diamond, pDL55; blue double-triangle, pDLm; purple triangle, pDL3; cyan circle, pDL33.
While a protruding tail may not be an essential element for unwinding of a D-loop, it appeared possible that the 3′ tail of pDL5 provided an additional recognition feature for BLM that would explain why this substrate was unwound more efficiently than was pDL3 or pDLm. To analyse this, we created two additional D-loop substrates in which the invading oligonucleotide contained a 25 nt stretch of reverse polarity (Figure 1). In pDL33, the oligonucleotide contained a 3′ invading end and a 3′ terminus on the protruding tail. In pDL55, the oligonucleotide had two 5′ ends (see Materials and Methods and Figure 1). The data in Figure 6 show that pDL55 was unwound as rapidly as pDL5, and at a significantly faster rate than was pDL33 (which was comparable in efficiency to pDL3). Taken together, these data indicate that neither the presence nor the polarity of the protruding tail are important features in determining the efficiency by which BLM disrupts mobile D-loops.
DISCUSSION
We have shown that the BLM helicase can disrupt both mobile D-loops generated enzymatically by a bona fide recombination initiating protein, and static D-loops created from oligonucleotides. Mobile D-loops are disrupted with high efficiency by BLM and rank alongside G-quadruplexes as the preferred substrate for the unwinding activity of BLM. We have also identified structural features of mobile D-loops that influence BLM helicase activity and have presented evidence that mobile and static D-loops are disrupted via different mechanisms.
There are a number of possible explanations for why mobile D-loops are such good substrates for BLM. First, and perhaps most logically, D-loops may represent one of the major functionally relevant in vivo substrates for BLM. This possibility has been proposed previously by others on the basis of genetic analyses in model organisms. For example, the data of Rockmill et al. (25) are consistent with a role for the yeast BLM orthologue, Sgs1, in disruption of an early, strand-invasion intermediate in meiotic recombination. Moreover, during a study of the role of the Drosophila BLM orthologue in DNA DSB repair, Adams et al. (41) provided evidence that DmBLM was involved in the process of synthesis-dependent strand annealing, which essentially requires the propagation of a D-loop intermediate along DNA concomitant with DNA synthesis. A second possible reason for the D-loop appearing to be such a good substrate is that it is not influenced by the single stranded DNA (ssDNA) annealing activity of BLM. The unwinding of more widely studied helicase substrates, such as forked duplexes and 3′-tailed duplexes, generates products that can be re-annealed to re-create the starting substrate. As a result, there is competition between the forward (helicase) and reverse (annealing) reactions as the concentrations of substrate and products change over time. This creates a characteristic bell shaped curve if the concentration of ssDNA product is plotted against BLM concentration or time (Figure 4b) (36). Hence, there can be an underestimation of the true efficiency with which BLM unwinds any substrate where the products are susceptible to re-annealing. This issue is irrelevant where mobile D-loop substrates are concerned, because BLM cannot re-generate the initial substrate from the reaction products as it lacks an ability to catalyse DNA strand invasion. Simple re-annealing would not be sufficient to re-generate the D-loop. Moreover, the equally efficiently unwound G-quadruplex substrate is not subject to re-annealing, as the re-generation of the quadruplex would require specific incubation conditions not provided during a helicase reaction. Although this ‘resistance’ to strand annealing may at least partially explain why D-loops and G-quadruplexes appear to be such good substrates for BLM, we would argue that the competition experiments described both in our study and previously during the study of G-quadruplexes by Maizels and co-workers (38,39) indicate that these DNA structures are intrinsically good substrates for BLM.
There are several features of D-loops that could influence the mechanism by which BLM effects their disruption. These include the distortion of the duplex in the region where strand invasion has occurred, the 3-stranded nature of the D-loop, and the presence of an ssDNA tail emerging from the D-loop. The finding that the ‘tail-less’ pDLm substrate can be unwound by BLM argues strongly against the tail providing a fundamentally important recognition feature. Of course, in vivo, although there is ssDNA invading a homologous duplex, this ssDNA is continuous and would not terminate with a free end soon after emerging from the duplex, as is seen with the mimics of D-loops necessarily used in vitro experiments. Hence, it was never likely that the nature of the terminus of the ssDNA would be a critical recognition feature. Nevertheless, BLM can distinguish between different D-loops depending on whether the ssDNA has a 5′ or a 3′ invading end. As reported previously using oligonucleotide-based D-loops (34), BLM prefers to unwind D-loops with a 5′ invading terminus, and we have confirmed this using mobile D-loops. What we have shown additionally through the analysis of the reverse polarity oligonucleotides is that this preference is not determined by the presence of a 3′ terminus on the tail of the oligonucleotide, since a substrate with two 5′ ends is unwound as efficiently as is a conventional 5′ invading oligonucleotide with a 3′ tail.
The human orthologue of RecA, RAD51, interacts physically with BLM (23). RAD51 differs from RecA in that it promotes strand invasion from a 5′ ssDNA end more efficiently than it does from a 3′ ssDNA end. Since 5′ end invasions are unproductive because they cannot be extended by DNA polymerases, some mechanisms must exist in vivo to limit their occurrence. Recently, the BRCA2 homologue of Ustilago maydis (42) has been shown to control the polarity of strand invasion. We suggest that BLM might impose a second barrier to unproductive 5′ strand invasions through its preferential unwinding of this form of D-loop. We propose that BLM has the intrinsic ability to recognize the orientation of the invading strand at the transition between the three-stranded and double stranded regions of the D-loop. Assuming that this junction proves to be a critical recognition feature, this opens up the possibility that mobile D-loops are not disrupted by conventional helicase action at all, but rather through branch migration of the three-stranded junction. Two pieces of evidence are consistent with this possibility. First, BLM is known to promote branch migration of four-stranded molecules over long distances (Kbps). Second, in the absence of RPA, BLM can unwind only very short partial duplexes, with the efficiency of unwinding decreasing markedly as the length of the duplex portion of the substrate increases (43). In contrast, BLM unwinds the D-loop very efficiently in the absence of RPA.
Recently, BLM, together with its interacting partner topoisomerase IIIα, has been shown to resolve a double Holliday junction molecule, a mimic of a late recombination structure (29). It might, therefore, be expected that the two proteins also act together in the early stages of recombination, and that TOPOIIIα might modulate the D-loop unwinding activity of BLM. We attempted to address this possibility experimentally; however, this proved to be impossible because TOPOIIIα alone was able to cause the release of the oligonucleotide from the plasmid-based D-loop (data not shown). This release was, however, the consequence of the nicking of the plasmid backbone by the topoisomerase and concomitant relaxation of the negative supercoiling that is important for stabilization of the D-loop. A similar effect can be achieved simply by cutting the backbone with a restriction enzyme.
We propose that BLM disrupts mobile D-loops and oligonucleotide-based D-loops via fundamentally different mechanisms. The most direct evidence to substantiate this proposal comes from an analysis of the order of appearance of the products of unwinding reactions in each case. In the presence of BLM, at least three discernible products are generated from the static D-loop substrate, two of which represent reaction intermediates that do not co-migrate with the eventual, end-labelled ssDNA product. In contrast, the mobile D-loop is disrupted apparently in a single step, as would be predicted. While it is formally possible that one or both of the reaction products generated from the oligonucleotide-based D-loops is not a genuine reaction intermediate but instead represents the product of a ssDNA annealing reaction, analysis of the order of appearance of the products would argue otherwise. These products clearly appear at earlier time points than does the free ssDNA oligonucleotide. A similar conclusion can be derived from an analysis of the data in Figure 5 of van Brabant et al. (34).
The studies described herein were initially aimed at testing the hypothesis that BLM and MMR factors co-operate to disrupt aberrant HR reactions. While our data are consistent with such a role for BLM, we obtained no evidence for a combined role for BLM and MMR proteins in D-loop disruption through studying the efficiency by which BLM and the MMR proteins together disrupt D-loops containing one, two or four G:T type mismatches (Table 1; C. Z. Bachrati, R. H. Borts and I. D. Hickson, unpublished data). This does not, in any way, argue against a key role for MMR proteins in ‘quality controlling’ HR processes; there is ample evidence to indicate such a role. The reason(s) why we failed to identify a role for MMR proteins at the D-loop stage of HR may reflect any of several possibilities. First, they may not play any role during this step of HR, but instead act later. Second, we may not have placed the mismatches in the appropriate location(s) in the D-loop. Third, factors in addition to BLM and MMR proteins are required to form a complex in order to promote abortion of mismatched D-loops. Further analyses will be required to analyse whether any of these possibilities is true.
In summary, we have presented evidence that mobile D-loops are a highly preferred substrate for the BLM helicase. One phenotype of BLM-deficient cells is an elevated frequency of recombination between homologous sequences. Moreover, cells from lower organisms deficient in BLM orthologues display hyper-recombination and a failure to suppress homologous recombination. We propose that a plausible explanation, at least in part, for these phenotypes is that BLM (and perhaps other RecQ helicases) can prevent D-loop structures from being converted into mature recombinants. Clearly, it would not be advantageous for BLM to effect this role indiscriminately in vivo; otherwise, all recombination reactions would be abortive. The identification of factors that modulate the function of BLM in the process of HR will likely identify factors that are crucial for the maintenance of genome stability and suppression of tumorigenesis in man.
Acknowledgments
The authors wish to thank members of the Genome Integrity Group for useful discussions, Drs P. McHugh and L. Wu for critical reading of the manuscript, and Miss P. White for preparation of the manuscript. This work was supported by the Cancer Research UK and by a Marie Curie Fellowship of the European Union to C.Z.B (HPMF-CT-2000-00952).
Conflict of interest statement. None declared.
REFERENCES
- 1.German J. Bloom syndrome: a mendelian prototype of somatic mutational disease. Medicine (Baltimore) 1993;72:393–406. [PubMed] [Google Scholar]
- 2.Bachrati C.Z., Hickson I.D. RecQ helicases: suppressors of tumorigenesis and premature aging. Biochem. J. 2003;374:577–606. doi: 10.1042/BJ20030491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chaganti R.S.K., Schonberg S., German J. A many-fold increase in sister chromatid exchanges in Bloom's syndrome lymphocytes. Proc. Natl Acad. Sci. USA. 1974;71:4508–4512. doi: 10.1073/pnas.71.11.4508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ellis N.A., Groden J., Ye T.Z., Straughen J., Lennon D.J., Ciocci S., Proytcheva M., German J. The Bloom's syndrome gene product is homologous to RecQ helicases. Cell. 1995;83:655–666. doi: 10.1016/0092-8674(95)90105-1. [DOI] [PubMed] [Google Scholar]
- 5.Karow J.K., Chakraverty R.K., Hickson I.D. The Bloom's syndrome gene product is a 3′–5′ DNA helicase. J. Biol. Chem. 1997;272:30611–30614. doi: 10.1074/jbc.272.49.30611. [DOI] [PubMed] [Google Scholar]
- 6.Yu C.E., Oshima J., Fu Y.H., Wijsman E.M., Hisama F., Alisch R., Matthews S., Nakura J., Miki T., Ouais S., et al. Positional cloning of the Werner's syndrome gene. Science. 1996;272:258–262. doi: 10.1126/science.272.5259.258. [DOI] [PubMed] [Google Scholar]
- 7.Gray M.D., Shen J.C., Kamath-Loeb A.S., Blank A., Sopher B.L., Martin G.M., Oshima J., Loeb L.A. The Werner syndrome protein is a DNA helicase. Nature Genet. 1997;17:100–103. doi: 10.1038/ng0997-100. [DOI] [PubMed] [Google Scholar]
- 8.Kitao S., Shimamoto A., Goto M., Miller R.W., Smithson W.A., Lindor N.M., Furuichi Y. Mutations in RECQL4 cause a subset of cases of Rothmund–Thomson syndrome. Nature Genet. 1999;22:82–84. doi: 10.1038/8788. [DOI] [PubMed] [Google Scholar]
- 9.Siitonen H.A., Kopra O., Kaariainen H., Haravuori H., Winter R.M., Saamanen A.M., Peltonen L., Kestila M. Molecular defect of RAPADILINO syndrome expands the phenotype spectrum of RECQL diseases. Hum. Mol. Genet. 2003;12:2837–2844. doi: 10.1093/hmg/ddg306. [DOI] [PubMed] [Google Scholar]
- 10.Puranam K.L., Blackshear P.J. Cloning and characterization of RECQL, a potential human homologue of the Escherichia coli DNA helicase RecQ. J. Biol. Chem. 1994;269:29838–29845. [PubMed] [Google Scholar]
- 11.Seki M., Miyazawa H., Tada S., Yanagisawa J., Yamaoka T., Hoshino S., Ozawa K., Eki T., Nogami M., Okumura K., et al. Molecular cloning of cDNA encoding human DNA helicase Q1 which has homology to Escherichia coli RecQ helicase and localization of the gene at chromosome 12p12. Nucleic Acids Res. 1994;22:4566–4573. doi: 10.1093/nar/22.22.4566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sekelsky J.J., Brodsky M.H., Rubin G.M., Hawley R.S. Drosophila and human RecQ5 exist in different isoforms generated by alternative splicing. Nucleic Acids Res. 1999;27:3762–3769. doi: 10.1093/nar/27.18.3762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Nakayama K., Irino N., Nakayama H. The RecQ gene of Escherichia coli K12: molecular cloning and isolation of insertion mutants. Mol. Gen. Genet. 1985;200:266–271. doi: 10.1007/BF00425434. [DOI] [PubMed] [Google Scholar]
- 14.Gangloff S., McDonald J.P., Bendixen C., Arthur L., Rothstein R. The yeast type I topoisomerase Top3 interacts with Sgs1, a DNA helicase homolog: a potential eukaryotic reverse gyrase. Mol. Cell. Biol. 1994;14:8391–8398. doi: 10.1128/mcb.14.12.8391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Watt P.M., Hickson I.D., Borts R.H., Louis E.J. SGS1, a homologue of the Bloom's and Werner's syndrome genes, is required for maintenance of genome stability in Saccharomyces cerevisiae. Genetics. 1996;144:935–945. doi: 10.1093/genetics/144.3.935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jasin M. Homologous repair of DNA damage and tumorigenesis: the BRCA connection. Oncogene. 2002;21:8981–8993. doi: 10.1038/sj.onc.1206176. [DOI] [PubMed] [Google Scholar]
- 17.Shin D.S., Chahwan C., Huffman J.L., Tainer J.A. Structure and function of the double-strand break repair machinery. DNA Repair (Amst) 2004;3:863–873. doi: 10.1016/j.dnarep.2004.03.022. [DOI] [PubMed] [Google Scholar]
- 18.Luo G., Santoro I.M., McDaniel L.D., Nishijima I., Mills M., Youssoufian H., Vogel H., Schultz R.A., Bradley A. Cancer predisposition caused by elevated mitotic recombination in Bloom mice. Nature Genet. 2000;26:424–429. doi: 10.1038/82548. [DOI] [PubMed] [Google Scholar]
- 19.Wang W., Seki M., Narita Y., Sonoda E., Takeda S., Yamada K., Masuko T., Katada T., Enomoto T. Possible association of BLM in decreasing DNA double strand breaks during DNA replication. EMBO J. 2000;19:3428–3435. doi: 10.1093/emboj/19.13.3428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Imamura O., Fujita K., Itoh C., Takeda S., Furuichi Y., Matsumoto T. Werner and Bloom helicases are involved in DNA repair in a complementary fashion. Oncogene. 2002;21:954–963. doi: 10.1038/sj.onc.1205143. [DOI] [PubMed] [Google Scholar]
- 21.Hanada K., Ukita T., Kohno Y., Saito K., Kato J., Ikeda H. RecQ DNA helicase is a suppressor of illegitimate recombination in Escherichia coli. Proc. Natl Acad. Sci. USA. 1997;94:3860–3865. doi: 10.1073/pnas.94.8.3860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yamagata K., Kato J., Shimamoto A., Goto M., Furuichi Y., Ikeda H. Bloom's and Werner's syndrome genes suppress hyperrecombination in yeast sgs1 mutant: implication for genomic instability in human diseases. Proc. Natl Acad. Sci. USA. 1998;95:8733–8738. doi: 10.1073/pnas.95.15.8733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wu L., Davies S.L., Levitt N.C., Hickson I.D. Potential role for the BLM helicase in recombinational repair via a conserved interaction with RAD51. J. Biol. Chem. 2001;276:19375–19381. doi: 10.1074/jbc.M009471200. [DOI] [PubMed] [Google Scholar]
- 24.Pichierri P., Franchitto A., Mosesso P., Palitti F. Werner's syndrome protein is required for correct recovery after replication arrest and DNA damage induced in S-phase of cell cycle. Mol. Biol. Cell. 2001;12:2412–2421. doi: 10.1091/mbc.12.8.2412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Rockmill B., Fung J.C., Branda S.S., Roeder G.S. The Sgs1 helicase regulates chromosome synapsis and meiotic crossing over. Curr. Biol. 2003;13:1954–1962. doi: 10.1016/j.cub.2003.10.059. [DOI] [PubMed] [Google Scholar]
- 26.Murray J.M., Lindsay H.D., Munday C.A., Carr A.M. Role of Schizosaccharomyces pombe RecQ homolog, recombination, and checkpoint genes in UV damage tolerance. Mol. Cell. Biol. 1997;17:6868–6875. doi: 10.1128/mcb.17.12.6868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wang S.W., Goodwin A., Hickson I.D., Norbury C.J. Involvement of Schizosaccharomyces pombe Srs2 in cellular responses to DNA damage. Nucleic Acids Res. 2001;29:2963–2972. doi: 10.1093/nar/29.14.2963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Liberi G., Maffioletti G., Lucca C., Chiolo I., Baryshnikova A., Cotta-Ramusino C., Lopes M., Pellicioli A., Haber J.E., Foiani M. Rad51-dependent DNA structures accumulate at damaged replication forks in sgs1 mutants defective in the yeast ortholog of BLM RecQ helicase. Genes Dev. 2005;19:339–350. doi: 10.1101/gad.322605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wu L., Hickson I.D. The Bloom's syndrome helicase suppresses crossing over during homologous recombination. Nature. 2003;426:870–874. doi: 10.1038/nature02253. [DOI] [PubMed] [Google Scholar]
- 30.Karow J.K., Newman R.H., Freemont P.S., Hickson I.D. Oligomeric ring structure of the Bloom's syndrome helicase. Curr. Biol. 1999;9:597–600. doi: 10.1016/s0960-9822(99)80264-4. [DOI] [PubMed] [Google Scholar]
- 31.Mohaghegh P., Karow J.K., Brosh R.M., Jr, Bohr V.A., Hickson I.D. The Bloom's and Werner's syndrome proteins are DNA structure-specific helicases. Nucleic Acids Res. 2001;29:2843–2849. doi: 10.1093/nar/29.13.2843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Opresko P.L., Otterlei M., Graakjaer J., Bruheim P., Dawut L., Kolvraa S., May A., Seidman M.M., Bohr V.A. The Werner syndrome helicase and exonuclease cooperate to resolve telomeric D loops in a manner regulated by TRF1 and TRF2. Mol. Cell. 2004;14:763–774. doi: 10.1016/j.molcel.2004.05.023. [DOI] [PubMed] [Google Scholar]
- 33.McIlwraith M.J., Hall D.R., Stasiak A.Z., Stasiak A., Wigley D.B., West S.C. RadA protein from Archaeoglobus fulgidus forms rings, nucleoprotein filaments and catalyses homologous recombination. Nucleic Acids Res. 2001;29:4509–4517. doi: 10.1093/nar/29.22.4509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.van Brabant A.J., Ye T., Sanz M., German J., Ellis N.A., Holloman W.K. Binding and melting of D-loops by the Bloom syndrome helicase. Biochemistry (Mosc). 2000;39:14617–14625. doi: 10.1021/bi0018640. [DOI] [PubMed] [Google Scholar]
- 35.Orren D.K., Theodore S., Machwe A. The Werner syndrome helicase/exonuclease (WRN) disrupts and degrades D-loops in vitro. Biochemistry (Mosc). 2002;41:13483–13488. doi: 10.1021/bi0266986. [DOI] [PubMed] [Google Scholar]
- 36.Cheok C.F., Wu L., Garcia P.L., Janscak P., Hickson I.D. The Bloom's syndrome helicase promotes the annealing of complementary single-stranded DNA. Nucleic Acids Res. 2005;33:3932–3941. doi: 10.1093/nar/gki712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Machwe A., Xiao L., Groden J., Matson S.W., Orren D.K. RECQ family members combine strand pairing and unwinding activities to catalyze strand exchange. J. Biol. Chem. 2005;280:23397–23407. doi: 10.1074/jbc.M414130200. [DOI] [PubMed] [Google Scholar]
- 38.Huber M.D., Lee D.C., Maizels N. G4 DNA unwinding by BLM and Sgs1p: substrate specificity and substrate-specific inhibition. Nucleic Acids Res. 2002;30:3954–3961. doi: 10.1093/nar/gkf530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sun H., Karow J.K., Hickson I.D., Maizels N. The Bloom's syndrome helicase unwinds G4 DNA. J. Biol. Chem. 1998;273:27587–27592. doi: 10.1074/jbc.273.42.27587. [DOI] [PubMed] [Google Scholar]
- 40.Sun H., Bennett R.J., Maizels N. The Saccharomyces cerevisiae Sgs1 helicase efficiently unwinds G-G paired DNAs. Nucleic Acids Res. 1999;27:1978–1984. doi: 10.1093/nar/27.9.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Adams M.D., McVey M., Sekelsky J.J. Drosophila BLM in double-strand break repair by synthesis-dependent strand annealing. Science. 2003;299:265–267. doi: 10.1126/science.1077198. [DOI] [PubMed] [Google Scholar]
- 42.Yang H., Li Q., Fan J., Holloman W.K., Pavletich N.P. The BRCA2 homologue Brh2 nucleates RAD51 filament formation at a dsDNA-ssDNA junction. Nature. 2005;433:653–657. doi: 10.1038/nature03234. [DOI] [PubMed] [Google Scholar]
- 43.Brosh R.M., Jr, Li J.L., Kenny M.K., Karow J.K., Cooper M.P., Kureekattil R.P., Hickson I.D., Bohr V.A. Replication protein A physically interacts with the Bloom's syndrome protein and stimulates its helicase activity. J. Biol. Chem. 2000;275:23500–23508. doi: 10.1074/jbc.M001557200. [DOI] [PubMed] [Google Scholar]
- 44.Mazin A.V., Zaitseva E., Sung P., Kowalczykowski S.C. Tailed duplex DNA is the preferred substrate for Rad51 protein-mediated homologous pairing. EMBO J. 2000;19:1148–1156. doi: 10.1093/emboj/19.5.1148. [DOI] [PMC free article] [PubMed] [Google Scholar]