Abstract
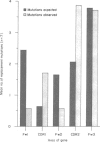
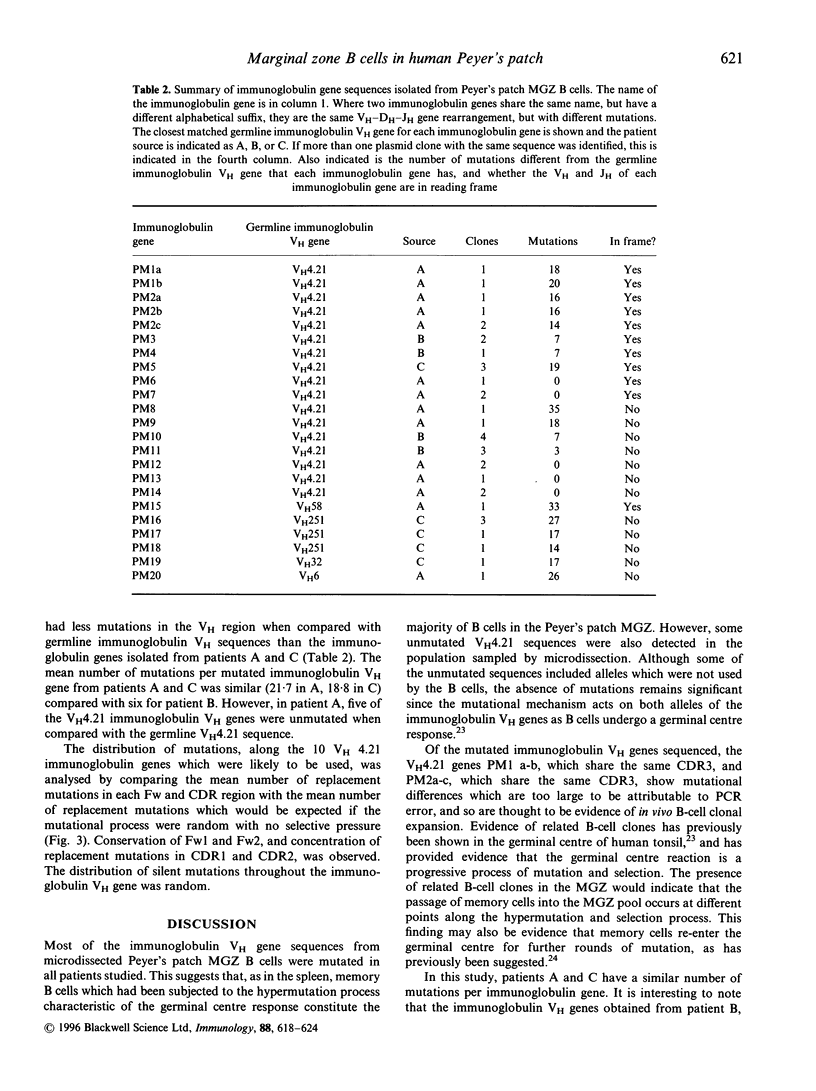
The Peyer's patches of the terminal ileum are a source of IgA plasma cells in the intestinal lamina propria of experimental animals. They are also thought to harbour IgA memory cells. However, the microanatomical location of Peyer's patch memory cells, and whether they are also present in man is not known. Human Peyer's patches have a pronounced marginal zone (MGZ) of sIgD-negative B cells. In this study we analysed the sequence of polymerase chain reaction-amplified, rearranged IgVH genes from microdissected MGZ B cells, to determine whether this is a site of B-cell memory in Peyer's patches. We observed that the majority of Peyer's patch MGZ B cells contain heavily mutated IgVH genes and are therefore clearly memory B cells. Sequences of rearranged mutated genes in the MGZ have a pattern of replacement and silent mutations expected of selected products of the affinity maturation process. Related clones, with identical CDR3 but different patterns of mutation, are seen. This suggests that either these memory cells are formed as the germinal centre selection process proceeds, or a memory cell has re-entered the germinal centre for further rounds of mutation. Interestingly, in one patient, the MGZ in the Peyer's patches also contains a proportion of B cells with unmutated IgVH 4.21 genes.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Altschul S. F., Gish W., Miller W., Myers E. W., Lipman D. J. Basic local alignment search tool. J Mol Biol. 1990 Oct 5;215(3):403–410. doi: 10.1016/S0022-2836(05)80360-2. [DOI] [PubMed] [Google Scholar]
- Amlot P. L., Hayes A. E. Impaired human antibody response to the thymus-independent antigen, DNP-Ficoll, after splenectomy. Implications for post-splenectomy infections. Lancet. 1985 May 4;1(8436):1008–1011. doi: 10.1016/s0140-6736(85)91613-7. [DOI] [PubMed] [Google Scholar]
- Andrew E., Hall J. G. IgA antibodies in the bile of rats. II. Evidence for immunological memory in secretory immunity. Immunology. 1982 Jan;45(1):177–182. [PMC free article] [PubMed] [Google Scholar]
- Berek C., Ziegner M. The maturation of the immune response. Immunol Today. 1993 Aug;14(8):400–404. doi: 10.1016/0167-5699(93)90143-9. [DOI] [PubMed] [Google Scholar]
- Campbell M. J., Zelenetz A. D., Levy S., Levy R. Use of family specific leader region primers for PCR amplification of the human heavy chain variable region gene repertoire. Mol Immunol. 1992 Feb;29(2):193–203. doi: 10.1016/0161-5890(92)90100-c. [DOI] [PubMed] [Google Scholar]
- Chang B., Casali P. The CDR1 sequences of a major proportion of human germline Ig VH genes are inherently susceptible to amino acid replacement. Immunol Today. 1994 Aug;15(8):367–373. doi: 10.1016/0167-5699(94)90175-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Craig S. W., Cebra J. J. Peyer's patches: an enriched source of precursors for IgA-producing immunocytes in the rabbit. J Exp Med. 1971 Jul 1;134(1):188–200. doi: 10.1084/jem.134.1.188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deane M., McCarthy K. P., Wiedemann L. M., Norton J. D. An improved method for detection of B-lymphoid clonality by polymerase chain reaction. Leukemia. 1991 Aug;5(8):726–730. [PubMed] [Google Scholar]
- Dunn-Walters D. K., Howe C. J., Isaacson P. G., Spencer J. Location and sequence of rearranged immunoglobulin genes in human thymus. Eur J Immunol. 1995 Feb;25(2):513–519. doi: 10.1002/eji.1830250231. [DOI] [PubMed] [Google Scholar]
- Dunn-Walters D. K., Isaacson P. G., Spencer J. Analysis of mutations in immunoglobulin heavy chain variable region genes of microdissected marginal zone (MGZ) B cells suggests that the MGZ of human spleen is a reservoir of memory B cells. J Exp Med. 1995 Aug 1;182(2):559–566. doi: 10.1084/jem.182.2.559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodglick L., Braun J. Revenge of the microbes. Superantigens of the T and B cell lineage. Am J Pathol. 1994 Apr;144(4):623–636. [PMC free article] [PubMed] [Google Scholar]
- Kepler T. B., Perelson A. S. Cyclic re-entry of germinal center B cells and the efficiency of affinity maturation. Immunol Today. 1993 Aug;14(8):412–415. doi: 10.1016/0167-5699(93)90145-B. [DOI] [PubMed] [Google Scholar]
- Klein U., Küppers R., Rajewsky K. Human IgM+IgD+ B cells, the major B cell subset in the peripheral blood, express V kappa genes with no or little somatic mutation throughout life. Eur J Immunol. 1993 Dec;23(12):3272–3277. doi: 10.1002/eji.1830231232. [DOI] [PubMed] [Google Scholar]
- Klein U., Küppers R., Rajewsky K. Human IgM+IgD+ B cells, the major B cell subset in the peripheral blood, express V kappa genes with no or little somatic mutation throughout life. Eur J Immunol. 1993 Dec;23(12):3272–3277. doi: 10.1002/eji.1830231232. [DOI] [PubMed] [Google Scholar]
- Kumararatne D. S., Bazin H., MacLennan I. C. Marginal zones: the major B cell compartment of rat spleens. Eur J Immunol. 1981 Nov;11(11):858–864. doi: 10.1002/eji.1830111103. [DOI] [PubMed] [Google Scholar]
- Lane P. J., Gray D., Oldfield S., MacLennan I. C. Differences in the recruitment of virgin B cells into antibody responses to thymus-dependent and thymus-independent type-2 antigens. Eur J Immunol. 1986 Dec;16(12):1569–1575. doi: 10.1002/eji.1830161216. [DOI] [PubMed] [Google Scholar]
- Lebman D. A., Griffin P. M., Cebra J. J. Relationship between expression of IgA by Peyer's patch cells and functional IgA memory cells. J Exp Med. 1987 Nov 1;166(5):1405–1418. doi: 10.1084/jem.166.5.1405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Y. J., Oldfield S., MacLennan I. C. Memory B cells in T cell-dependent antibody responses colonize the splenic marginal zones. Eur J Immunol. 1988 Mar;18(3):355–362. doi: 10.1002/eji.1830180306. [DOI] [PubMed] [Google Scholar]
- Lycke N., Holmgren J. Intestinal mucosal memory and presence of memory cells in lamina propria and Peyer's patches in mice 2 years after oral immunization with cholera toxin. Scand J Immunol. 1986 May;23(5):611–616. doi: 10.1111/j.1365-3083.1986.tb01995.x. [DOI] [PubMed] [Google Scholar]
- Morente M., Piris M. A., Orradre J. L., Rivas C., Villuendas R. Human tonsil intraepithelial B cells: a marginal zone-related subpopulation. J Clin Pathol. 1992 Aug;45(8):668–672. doi: 10.1136/jcp.45.8.668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pascual V., Liu Y. J., Magalski A., de Bouteiller O., Banchereau J., Capra J. D. Analysis of somatic mutation in five B cell subsets of human tonsil. J Exp Med. 1994 Jul 1;180(1):329–339. doi: 10.1084/jem.180.1.329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanz I., Kelly P., Williams C., Scholl S., Tucker P., Capra J. D. The smaller human VH gene families display remarkably little polymorphism. EMBO J. 1989 Dec 1;8(12):3741–3748. doi: 10.1002/j.1460-2075.1989.tb08550.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spencer J., Finn T., Isaacson P. G. A comparative study of the gut-associated lymphoid tissue of primates and rodents. Virchows Arch B Cell Pathol Incl Mol Pathol. 1986;51(6):509–519. doi: 10.1007/BF02899056. [DOI] [PubMed] [Google Scholar]
- Spencer J., Finn T., Isaacson P. G. Human Peyer's patches: an immunohistochemical study. Gut. 1986 Apr;27(4):405–410. doi: 10.1136/gut.27.4.405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stevenson F. K., Smith G. J., North J., Hamblin T. J., Glennie M. J. Identification of normal B-cell counterparts of neoplastic cells which secrete cold agglutinins of anti-I and anti-i specificity. Br J Haematol. 1989 May;72(1):9–15. doi: 10.1111/j.1365-2141.1989.tb07643.x. [DOI] [PubMed] [Google Scholar]
- Tomlinson I. M., Walter G., Marks J. D., Llewelyn M. B., Winter G. The repertoire of human germline VH sequences reveals about fifty groups of VH segments with different hypervariable loops. J Mol Biol. 1992 Oct 5;227(3):776–798. doi: 10.1016/0022-2836(92)90223-7. [DOI] [PubMed] [Google Scholar]
- Tseng J. Transfer of lymphocytes of Peyer's patches between immunoglobulin allotype congenic mice: repopulation of the IgA plasma cells in the gut lamina propria. J Immunol. 1981 Nov;127(5):2039–2043. [PubMed] [Google Scholar]