Abstract
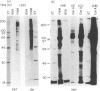
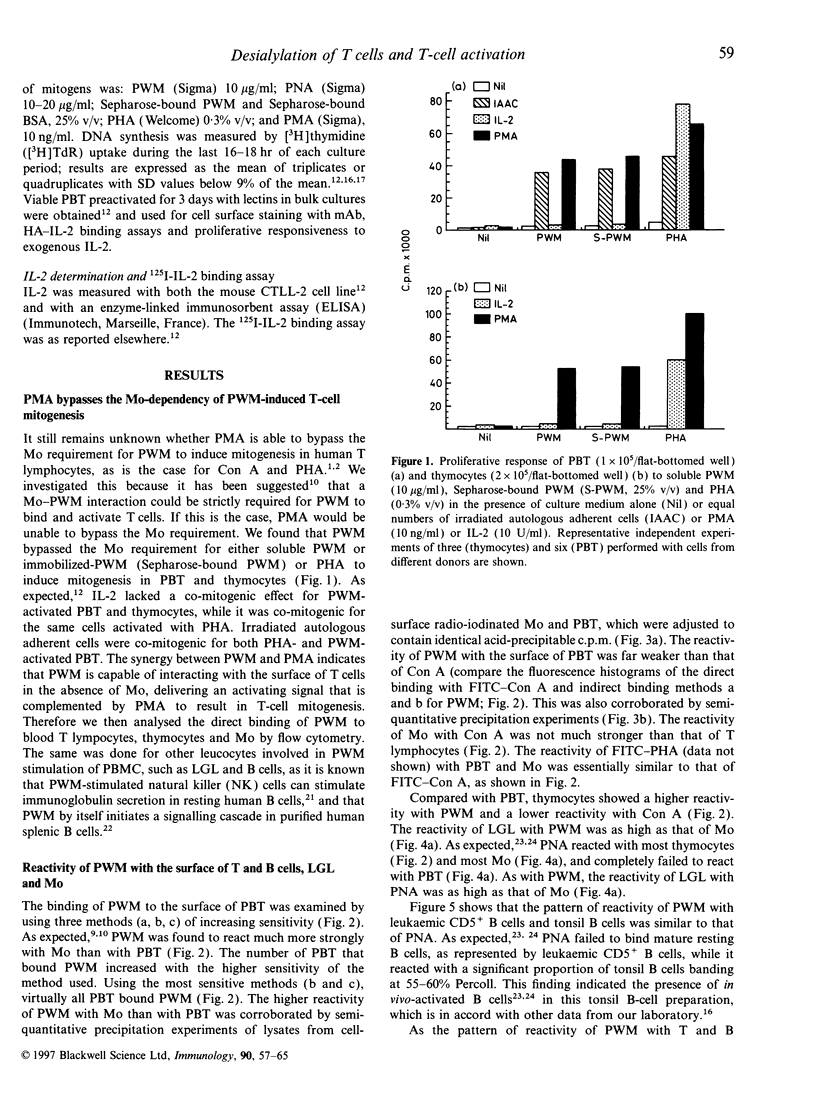
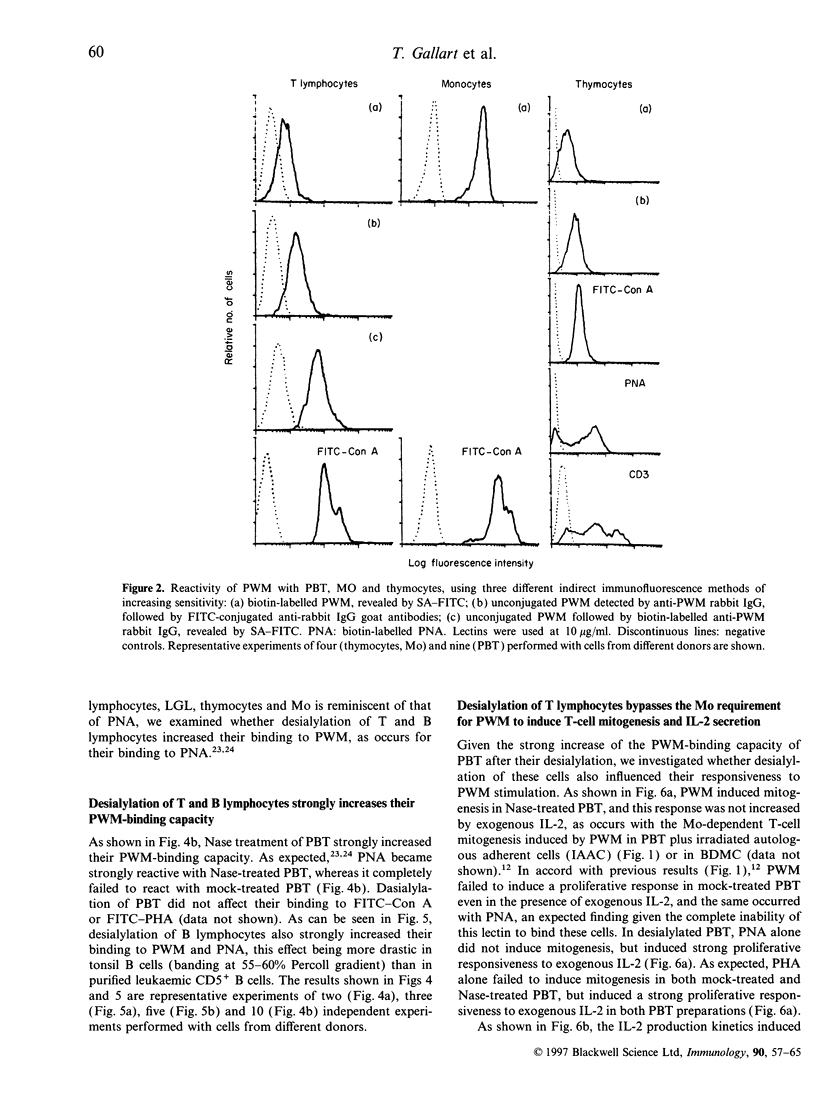
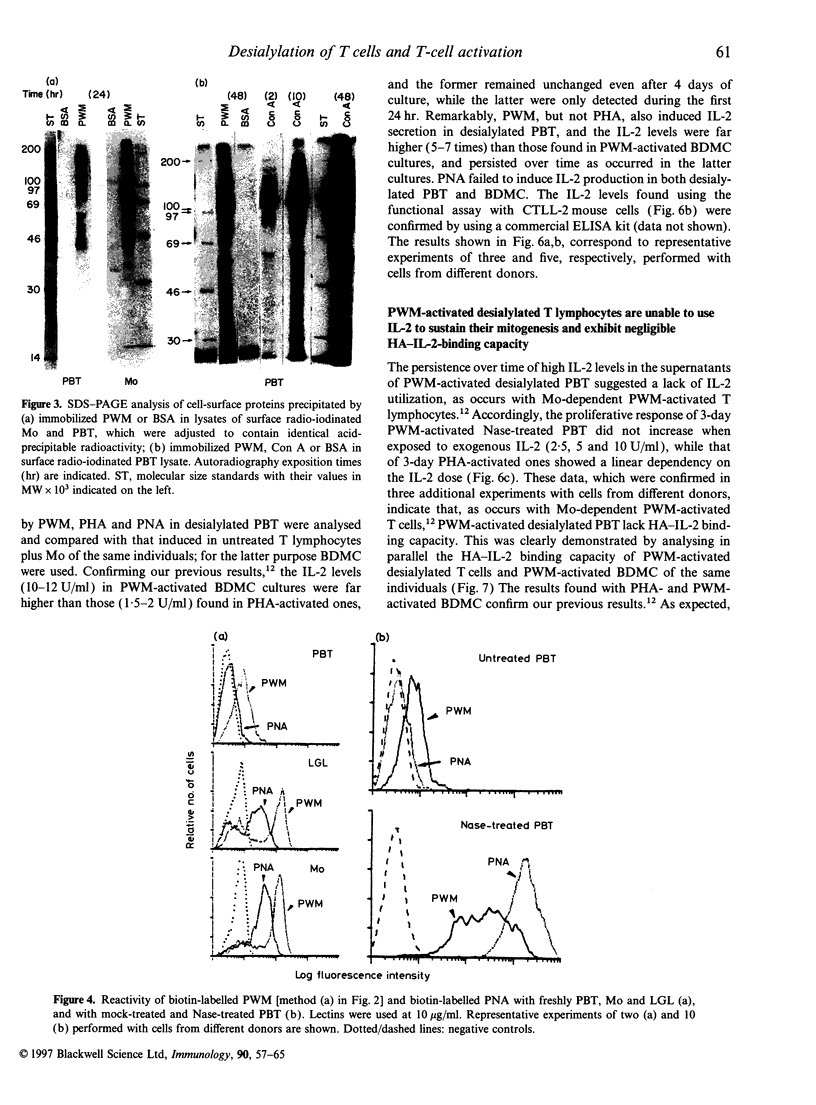
Activation of T lymphocytes by pokeweed mitogen (PWM) is strictly monocyte (Mo)-dependent and results in T-cell mitogenesis and interleukin-2 (IL-2) secretion, coupled with an inability to utilize IL-2 due to an impaired expression of functional IL-2 receptor (IL-2R). Such IL-2R impairment could arise in PWM-activated T cells themselves or, alternatively, be the result of Mo-derived influences, as it is known that PWM binds Mo strongly and does not or poorly binds lymphocytes, and Mo becomes rapidly destroyed in PWM-stimulated cultures of blood mononuclear cells or T cells plus Mo. The present study investigated these possibilities. The results show for the first time that desialylation of T lymphocytes strongly increases their PWM-binding capacity and, in addition, overcomes the Mo requirement for PWM to induce T-cell mitogenesis and IL-2 secretion. Such secreted IL-2 levels were even higher that those found in cultures of Mo-dependent PWM-activated T lymphocytes but similarly to the latter, PWM-activated desialylated purified T lymphocytes exhibited negligible high-affinity IL-2 binding capacity and an inability to utilize the IL-2 they produced. These effects were not due to desialylation itself, as indicated by data obtained with peanut agglutinin, a lectin that becomes strongly reactive with desialylated T lymphocytes. The data clearly indicate the existence of PWM-related events capable of impairing the expression of functional IL-2R without affecting IL-2 secretion, and indicate that such events are due to mechanisms arising at the level of PWM-activated T cells themselves.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alberola-Ila J., Places L., de la Calle O., Romero M., Yagüe J., Gallart T., Vives J., Lozano F. Stimulation through the TCR/CD3 complex up-regulates the CD2 surface expression on human T lymphocytes. J Immunol. 1991 Feb 15;146(4):1085–1092. [PubMed] [Google Scholar]
- Altman A., Coggeshall K. M., Mustelin T. Molecular events mediating T cell activation. Adv Immunol. 1990;48:227–360. doi: 10.1016/s0065-2776(08)60756-7. [DOI] [PubMed] [Google Scholar]
- Ceuppens J. L., Meurs L., Baroja M. L., Van Wauwe J. P. Effect of T3 modulation on pokeweed mitogen-induced T cell activation: evidence for an alternative pathway of T cell activation. J Immunol. 1986 May 1;136(9):3346–3350. [PubMed] [Google Scholar]
- Chauhan D., Kharbanda S. M., Uchiyama H., Urashima M., Anderson K. C. Activation of pp90rsk and early growth response-1 gene expression by pokeweed mitogen in human B cells. Leuk Res. 1995 May;19(5):337–344. doi: 10.1016/0145-2126(94)00132-t. [DOI] [PubMed] [Google Scholar]
- Childs R. A., Kapadia A., Feizi T. Expression of blood group I and i active carbohydrate sequences on cultured human and animal cell lines assessed by radioimmunoassays with monoclonal cold agglutinins. Eur J Immunol. 1980 May;10(5):379–384. doi: 10.1002/eji.1830100512. [DOI] [PubMed] [Google Scholar]
- De Vries E., Van der Weij J. P., Van der Veen C. J., Doekes G., Steven M. M., Cats A. Electronmicroscopical observations on monocyte-lymphocyte interactions upon stimulation with pokeweed mitogen latex conjugate. Eur J Immunol. 1986 Feb;16(2):122–128. doi: 10.1002/eji.1830160203. [DOI] [PubMed] [Google Scholar]
- Engel P., Inglés J., de la Calle O., Gallart T. Cellular activation without proliferation to B cell growth factor and interleukin 2 in chronic lymphocytic leukaemia B cells stimulated with phorbol ester plus calcium ionophore. Clin Exp Immunol. 1989 Apr;76(1):61–67. [PMC free article] [PubMed] [Google Scholar]
- Feuerstein N., Huang D., Prystowsky M. B. Rapamycin selectively blocks interleukin-2-induced proliferating cell nuclear antigen gene expression in T lymphocyte. Evidence for inhibition of CREB/ATF binding activities. J Biol Chem. 1995 Apr 21;270(16):9454–9458. doi: 10.1074/jbc.270.16.9454. [DOI] [PubMed] [Google Scholar]
- Geppert T. D., Davis L. S., Gur H., Wacholtz M. C., Lipsky P. E. Accessory cell signals involved in T-cell activation. Immunol Rev. 1990 Oct;117:5–66. doi: 10.1111/j.1600-065x.1990.tb00566.x. [DOI] [PubMed] [Google Scholar]
- Gray J. D., Horwitz D. A. Activated human NK cells can stimulate resting B cells to secrete immunoglobulin. J Immunol. 1995 Jun 1;154(11):5656–5664. [PubMed] [Google Scholar]
- Inglés J., Engel P., De La Calle O., Gallart T. Differential responsiveness of human B lymphocytes to phorbol ester and calcium ionophore based on their state of activation. Immunology. 1989 Jul;67(3):359–364. [PMC free article] [PubMed] [Google Scholar]
- Johnson K. W., Smith K. A. cAMP regulation of IL-2 receptor expression. Selective modulation of the p75 subunit. J Immunol. 1990 Aug 15;145(4):1144–1151. [PubMed] [Google Scholar]
- Karasuno T., Kanayama Y., Nishiura T., Nakao H., Yonezawa T., Tarui S. Glycosidase inhibitors (castanospermine and swainsonine) and neuraminidase inhibit pokeweed mitogen-induced B cell maturation. Eur J Immunol. 1992 Aug;22(8):2003–2008. doi: 10.1002/eji.1830220808. [DOI] [PubMed] [Google Scholar]
- Kierszenbaum F., Sztein M. B., Beltz L. A. Decreased human IL-2 receptor expression due to a protozoan pathogen. Immunol Today. 1989 Apr;10(4):129–131. doi: 10.1016/0167-5699(89)90246-6. [DOI] [PubMed] [Google Scholar]
- Landolfi N. F., Leone J., Womack J. E., Cook R. G. Activation of T lymphocytes results in an increase in H-2-encoded neuraminidase. Immunogenetics. 1985;22(2):159–167. doi: 10.1007/BF00563513. [DOI] [PubMed] [Google Scholar]
- Miedema F., Van Oostveen J. W., Sauerwein R. W., Terpstra F. G., Aarden L. A., Melief C. J. Induction of immunoglobulin synthesis by interleukin 2 is T4+/T8- cell dependent. A role for interleukin 2 in the pokeweed mitogen-driven system. Eur J Immunol. 1985 Feb;15(2):107–112. doi: 10.1002/eji.1830150202. [DOI] [PubMed] [Google Scholar]
- Minami Y., Kono T., Miyazaki T., Taniguchi T. The IL-2 receptor complex: its structure, function, and target genes. Annu Rev Immunol. 1993;11:245–268. doi: 10.1146/annurev.iy.11.040193.001333. [DOI] [PubMed] [Google Scholar]
- Muraguchi A., Hirano T., Tang B., Matsuda T., Horii Y., Nakajima K., Kishimoto T. The essential role of B cell stimulatory factor 2 (BSF-2/IL-6) for the terminal differentiation of B cells. J Exp Med. 1988 Feb 1;167(2):332–344. doi: 10.1084/jem.167.2.332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakagawa N., Nakagawa T., Volkman D. J., Ambrus J. L., Jr, Fauci A. S. The role of interleukin 2 in inducing Ig production in a pokeweed mitogen-stimulated mononuclear cell system. J Immunol. 1987 Feb 1;138(3):795–801. [PubMed] [Google Scholar]
- Novogrodsky A., Lotan R., Ravid A., Sharon N. Peanut agglutinin, a new mitogen that binds to galactosyl sites exposed after neuraminidase treatment. J Immunol. 1975 Nov;115(5):1243–1248. [PubMed] [Google Scholar]
- Poyet J. L., Radom J., Hoeveler A. Isolation and characterization of a cDNA clone encoding the pokeweed antiviral protein II from Phytolacca americana and its expression in E. coli. FEBS Lett. 1994 Jun 27;347(2-3):268–272. doi: 10.1016/0014-5793(94)00565-6. [DOI] [PubMed] [Google Scholar]
- Pryjma J., Zembala M., Pituch-Noworolska A., Ernst M., Van der Bosch J., Flad H. D. Monocyte-T-cell interactions in pokeweed mitogen-activated cultures. Immunology. 1990 Nov;71(3):397–403. [PMC free article] [PubMed] [Google Scholar]
- Rose M. L., Birbeck M. S., Wallis V. J., Forrester J. A., Davies A. J. Peanut lectin binding properties of germinal centres of mouse lymphoid tissue. Nature. 1980 Mar 27;284(5754):364–366. doi: 10.1038/284364a0. [DOI] [PubMed] [Google Scholar]
- Sherblom A. P., Sathyamoorthy N., Decker J. M., Muchmore A. V. IL-2, a lectin with specificity for high mannose glycopeptides. J Immunol. 1989 Aug 1;143(3):939–944. [PubMed] [Google Scholar]
- Sleasman J. W., Morimoto C., Schlossman S. F., Tedder T. F. The role of functionally distinct helper T lymphocyte subpopulations in the induction of human B cell differentiation. Eur J Immunol. 1990 Jun;20(6):1357–1366. doi: 10.1002/eji.1830200623. [DOI] [PubMed] [Google Scholar]
- Stevenson H. C., Miller P. J., Waxdal M. J., Haynes B. F., Thomas C. A., Fauci A. S. Interaction of pokeweed mitogen with monocytes in the activation of human lymphocytes. Immunology. 1983 Aug;49(4):633–640. [PMC free article] [PubMed] [Google Scholar]
- Takeshita T., Goto Y., Tada K., Nagata K., Asao H., Sugamura K. Monoclonal antibody defining a molecule possibly identical to the p75 subunit of interleukin 2 receptor. J Exp Med. 1989 Apr 1;169(4):1323–1332. doi: 10.1084/jem.169.4.1323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weinberg A. D., Swain S. L. IL-2 receptor (Tac antigen) protein expression is down-regulated by the 5'-untranslated region of the mRNA. J Immunol. 1990 Jun 15;144(12):4712–4720. [PubMed] [Google Scholar]
- de la Calle-Martín O., Alberola-Ila J., Engel P., Inglés J., Fabregat V., Barceló J. J., Lozano F., Gallart T. Impaired post-transcriptional expression of interleukin-2 receptor in pokeweed mitogen-activated T cells. Eur J Immunol. 1992 Apr;22(4):897–902. doi: 10.1002/eji.1830220403. [DOI] [PubMed] [Google Scholar]