Abstract
Long-term potentiation (LTP) is thought to be particularly important in the acquisition of hippocampus-associated memory, in part because it develops quickly and persists for indefinite periods. Extracellular proteolysis has been hypothesized to contribute to LTP by modifying adhesive relations of synapses and thus the morphology of excitatory synapses. Here we report that neuropsin (NP), an extracellular serine protease, is critically involved in the formation of both the potentiation effect and hippocampus-dependent forms of memory. NP-knockout mice were significantly impaired in the Morris water maze and Y-mazes and failed to exhibit early phase LTP induced by a single tetanus. Potentiation was also impaired or completely blocked by in vivo application of a specific inhibitor or a neutralizing monoclonal antibody for NP. Intriguingly, recombinant (r-) NP alone, without tetanic stimulation, elicited either long-lasting potentiation or depression, depending on the applied dose. The r-NP-elicited potentiation was occluded by prior induction of LTP, while theta-burst-elicited LTP was occluded by application of r-NP alone, suggesting that the two forms of plasticity have a common signalling pathway. r-NP-elicited potentiation and depression increased phosphorylation at different sites on the GluR1 subunit of the AMPA receptor that had previously been associated with LTP or long-term depression. Thus, we conclude that NP is necessary for establishment of LTP and has a significant role in memory acquisition.
Hippocampal long-term potentiation (LTP) (Bliss & Lomo, 1973), a model for spatial and explicit memory, is widely held to involve early (E-LTP) and late (L-LTP) phases (Krug et al. 1984). The two phases occur independently and are regulated by different signalling systems: E-LTP requires activation of the N-methyl-d-aspartate (NMDA) receptor and calcium–calmodulin kinase II (CaMKII), while L-LTP requires activation of protein kinase A (PKA) followed by novel protein synthesis (Frey et al. 1993; Huang et al. 1994). E-LTP is particularly important in the reversal of LTP expression (Hesse & Teyler, 1976), an effect that occurs in vivo in the CA1 subfield of the hippocampus of free-moving rats when the animals enter a new environment (Xu et al. 1998). Reversal of LTP, which is thought to act with enhancements at selected synapses to allow the appropriate storage of new information, can only be obtained for a relatively short period (i.e. during E-LTP) following the induction of potentiation (Xu et al. 1998).
There is growing evidence that adhesion molecules play a critical role in stabilizing LTP, with interactions between synaptic membranes and the extracellular matrix (ECM) being particularly important. Ectodomain proteolysis of matrix and cell adhesion molecules (CAM) may play significant roles in dynamic synaptic rearrangement and mechanical plasticity (Matsumoto-Miyai et al. 2003). Several lines of evidence support the possibility that interactions between synaptic membranes and the ECM participate in certain types of synaptic plasticity, such as kindling, kainite-induced seizures, and LTP. Indeed, LTP is reduced by application of peptides and antibodies that block the extracellular interactions of adhesion receptors belonging to the CAM and integrin families (Luthl et al. 1994; Ronn et al. 1995; Bahr et al. 1997; Staubli et al. 1998). Moreover, tissue plasminogen activator (tPA), matrix metalloproteinases, and other extracellular proteases, either released or constitutively present, have been linked to LTP (Frey et al. 1996; Huang et al. 1996; Baranes et al. 1998). Neuropsin (NP; also referred to as KLK8) is of particular interest in this regard because of its unusual distribution and high concentration in the forebrain (Chen et al. 1995). This protease degrades the presynaptic L1 cell adhesion molecule (L1 cam) in an NMDA receptor-dependent fashion (Matsumoto-Miyai et al. 2003) and could thus serve as a link between the postsynaptic triggers for LTP and presynaptic adhesion changes needed for stable potentiation. There is also evidence that NP is involved in early phases of LTP (Momota et al. 1998; Yoshida & Shiosaka, 1999; Komai et al. 2000), unlike tPA, which predominantly affects L-LTP (Frey et al. 1996; Huang et al. 1996; Baranes et al. 1998), possibly via an action of the presynaptically located L1; the shedding of the extracellular domain of L1 is reported to play a significant role in establishing E-LTP (Matsumoto-Miyai et al. 2003).
These results strongly suggest that cleavage of an adhesion molecule by NP may be involved in converting sequential enhancement of a signalling pathway during E-LTP into stable memory acquisition. However, Davies et al. (2001) reported that NP-knockout mice had no significant impairment of spatial learning and displayed normal hippocampal LTP. In the present study therefore we re-examined behavioural memory and electrophysiological synaptic plasticity in NP-knockout mice, and then evaluated whether the protease-activated system alone can evoke potentiation that shares the same signalling mechanism with LTP. We found that learning and E-LTP were significantly impaired in NP-knockout mice, and that NP alone is necessary for the establishment of E-LTP.
Methods
Animals
A total of 156 male ddY mice (9–12 weeks old) and 45 male NP+/+, +/− and −/− mice (8–22 weeks old) were used in the present study. ddY mice were purchased from SLC Co. (Hamamatsu, Japan), and heterozygous and homozygous NP-mutant mice were produced as previously described (Hirata et al. 2001). NP−/− mice were backcrossed to C57BL/6J (n > 9), and C57BL/6J control mice (littermates) were used. Mice were maintained under a 12 h: 12 h light: dark cycle and fed ad libitum. At the end of the physiological experiments, mice were killed with an overdose of pentobarbital. All experimental protocols described below were performed according to the animal care and experimentation guidelines of the Nara Institute of Science and Technology and Physiological Society of Japan.
Morris water maze
A circular pool (diameter 120 cm, height 30 cm) was filled to a depth of 15 cm with water at room temperature (21–22°C). Four equally spaced points around the edge of the pool were designated as four starting positions: east (E), south (S), west (W) and north (N). A hidden circular platform (diameter 8 cm) was set 0.5 cm below the surface of the water in a fixed position in the middle of the NW quadrant. Mice were trained to find the platform for four consecutive days. Training consisted of four trials per day with an intertrial interval of 120 min. The mice were placed in the water, facing the wall, at one of the four starting positions in a random sequence; each position was used once each day. The time required to find the escape platform (escape latency) was measured during each trial. Having found and climbed onto the platform, the mice stayed there for 30 s and were then returned to their home cage. If the mice failed to find the platform within 100 s, they were placed on the platform by the experimenter and a maximum score of 100 s was assigned.
Y-maze
The Y-maze apparatus consisted of three arms. Each arm was 40 cm long, 10 cm high, 3 cm wide at the bottom and 10 cm wide at the top, and converged in an equilateral triangular central area. Each mouse, naive to the maze, was placed at the end of one arm and allowed to move freely through the maze during an 8 min session. The sequence of arm entries was recorded manually. Entry was considered complete when the hind paws of the mouse had completely entered the arm. A spontaneous alternation behaviour, which is regarded as a measure of spatial memory, was defined as successive entries into the three different arms (A, B or C) in overlapping triplet sets (e.g. ACBA BACBAB = 5). Percentage alternation was calculated as the ratio of actual to possible alternation (defined as the total number of arm entries minus two) multiplied by 100, as shown in the following equation:
The number of arm entries also serves as an indicator of activity.
Electrophysiology
ddY male mice were anaesthetized with urethane (1.25 g kg−1; i.p.) and placed in a stereotaxic frame with the skull horizontal. Rectal temperature was maintained at 37°C using a heated jacket pad (BWT-100, Bio Research Center, Nagoya, Japan). Extracellular field recording was carried out as previously described (Matsumoto-Miyai et al. 2003). A bipolar stainless-steel electrode (InterMedical Co., Tokyo, Japan) was used for stimulation in the two populations of Schaffer collateral–commissural projections (2.46 mm posterior and 2.30 mm lateral to bregma or 2.46 mm posterior and 0.4 mm lateral to bregma). Test pulses (100 μs duration) were collected every 30 s, and their intensity was adjusted to evoke field excitatory postsynaptic potential (fEPSP) and population spikes which were ∼50% of the maximal responses. Both were recorded with a monopolar tungsten electrode (InterMedical Co.) placed in the stratum radiatum or the stratum pyramidale of the CA1 region (2.46 mm posterior and 2.0 mm lateral to bregma). The slope of fEPSP was measured on the maximal initial slope, and the population spike amplitude was evaluated as the difference between the average of two positive peaks and the negative peak. All responses were expressed as a percentage change from the average response recorded during the 30 min immediately before inhibitor, antibody or r-NP injection, or LTP-inducing stimuli. E-LTP was induced using a single tetanic stimulation (100 Hz for 1 s). A theta-burst stimulation (TBS) protocol consisting of 10 burst-like trains (10 pulses at 200 Hz) at 5 Hz, applied four times every 30 s, was used in an attempt to elicit LTP. Long-term depression (LTD) was induced by low-frequency paired stimulation (LFPS; 700 μA, 150 μs pulses), comprising 900 pairs of pulses given at an interstimulus interval of 50 ms and an interpair interval of 1 s (Thiels et al. 1994). Paired-pulse facilitation (PPF) was tested after a 20 min period of stable responses and 3 h after terminating r-NP injection. Interpulse intervals of 25, 50, 75, 100 and 200 ms were investigated.
Administration of protease inhibitor, anti-NP antibody and r-NP
Intracerebroventricular infusion of an NP-specific inhibitor, NP stop (Taisho Pharmaceutical Co., Tokyo, Japan), and an activity-neutralizing anti-NP antibody (mAbB5; MBL, Nagoya, Japan) was performed as reported previously (Momota et al. 1998). NP stop (1 μg ml−1), mAbB5 (1.4 μg ml−1) or rat IgG diluted in saline was injected (0.22 mm anterior and 1.0 mm lateral to bregma; 2.3 mm ventral from surface of skull) at flow rates of 0.05 μl min−1 for 40 min or 0.5 μl min−1 for 10 min, using a Hamilton syringe driven by syringe pump (CFV-2100; Nihon Kohden, Tokyo, Japan). The r-NP was prepared as previously described (Shimizu et al. 1998); r-proNP, an inactive precursor form, was first obtained from insect cells (Shimizu et al. 1998) and then activated by lysyl endopeptidase (Kato et al. 2001). The enzymatic activity of r-NP was measured with t-Butyloxycarbonyl-l-Valyl-l-Prolyl-l-Arginine 4-Methyl-Coumaryl-7-Amide (Boc-Val-Pro-Arg-MCA), one unit being defined as the amount required to hydrolyse 1 μmol of this substrate per minute at 30°C. The r-NP was diluted with artificial cerebrospinal fluid (ACSF) consisting of 127 mm NaCl, 1.6 mm KCl, 1.24 mm KH2PO4, 1.3 mm MgSO4, 2.4 mm CaCl2, 26 mm NaHCO3 and 10 mm glucose. ACSF containing r-NP, or an equal volume of ACSF alone, was delivered for 10 min at a rate of 80 nl min−1 with a 10 μl Hamilton syringe in the hippocampus. For control experiments, r-NP (1.8 mU ml−1) was irreversibly inactivated by overnight incubation at 4°C with 3 mm phenyl methane sulphonyl fluoride (PMSF) (a serine protease inhibitor) or 5 μg ml−1 mAbB5, and used instead of active r-NP. The Hamilton syringe was connected to the infusion cannula with polyethylene tubing.
Western blot analysis
Hippocampi were removed from three ddY mice 90 min after r-NP injection, and the CA1 region around the inserted electrodes was microdissected. These samples were homogenized in a glass-Teflon homogenizer in ice-cold homogenization buffer (5 mm Hepes, pH 7.4, 320 mm sucrose, 5 mm EDTA, 5 mm EGTA, 50 mm NaF, 10 mm sodium pyrophosphate, 1 mm sodium orthovanadate, 1 mm PMSF, 1 μm okadaic acid and 5 μg ml−1 leupeptin), and then incubated on ice for 10 min. The homogenates were centrifuged at 1000 g for 10 min at 4°C to pellet the nuclei. The supernatants were centrifuged at 10 000 g for 15 min at 4°C, and the crude synaptosomal pellets were resuspended in homogenization buffer and centrifuged again at 10 000 g for another 15 min at 4°C. The washed crude synaptosomes were suspended in lysis buffer (50 mm Tris-HCl, pH 6.8, 150 mm NaCl, 5 mm EDTA, 5 mm EGTA, 50 mm NaF, 10 mm sodium pyrophosphate, 1 mm sodium orthovanadate, 1 mm PMSF, 1% SDS, 1 μm okadaic acid and 5 μg ml−1 leupeptin). Protein concentration was determined by BCA assay (Pierce, Rockford, IL, USA). Equal amounts of proteins were loaded onto SDS-PAGE gels (7.5%), and were then transferred to polyvinylidene difluoride (PVDF) membranes. Western blotting for the phosphorylated GluR1 subunit was performed as previously described (Lee et al. 1998). Briefly, the blotted PVDF membranes were blocked with 4% BSA and 0.05% Tween-20 in PBS for 1 h, and then incubated for 2 h with phosphorylation site-specific antibodies (1: 300; a gift from Dr K. Kameyama, National Institute of Bioscience and Human Technology, Japan). After being washed five times for 5 min each with 1% BSA and 0.05% Tween-20 in PBS, the membranes were incubated for 1 h with alkaline phosphatase-conjugated anti-rabbit immunoglobulin (1: 2000). Immunoreactive bands were detected using the Immun-Star chemiluminescent system (Bio-Rad, Hercules, CA, USA) and exposure to X-ray film. The antibodies were stripped with 62.5 mm Tris-HCl, pH 6.8, 2% SDS, and 100 mm 2-mercaptoethanol at 60°C for 30 min, after which the membranes were reblocked and reprobed with anti-C-terminal GluR1 antibodies (Upstate Biotechnology, Lake Placid, NY, USA) to estimate the total amount of GluR1. Band densities were measured using Scion Image (Scion Co., Frederick, MD, USA). Phospho-GluR1 immunoreactivity was normalized to total GluR1 using a phosphorylation-independent antibody.
Data analysis
All graphical data are represented as the mean ±s.e.m. Statistical analysis of group effects was performed using one-way or two-way analysis of variance (ANOVA) followed by Scheffé's test for post hoc comparison, and individual effects were measured by the unpaired t test. Significance was set at P < 0.05.
Results
Impairment of hippocampus-dependent learning in NP-knockout mice
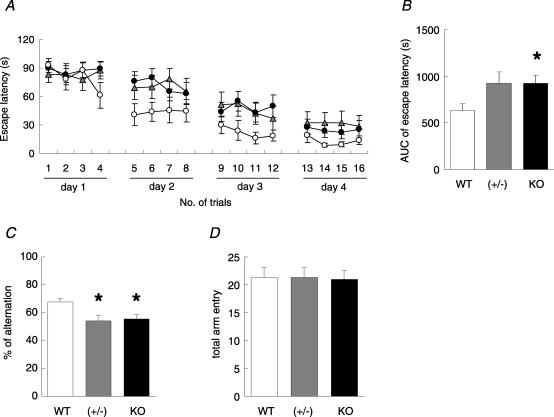
We evaluated hippocampus-dependent learning of NP-mutant mice in the Morris water maze task. During the hidden platform task, the escape latency period for the knockout mice was significantly longer than that for wild-type mice (two-way ANOVA: F(1,21)= 6.22, P= 0.021) and the latency period for heterozygous mice tended to be longer than for wild-type mice (two-way ANOVA: F(1,18)= 4.19, P= 0.056) (Fig. 1A and B). In a visual task, there was no difference between the knockout and the wild-type mice (data not shown).
Figure 1. Abnormalities of hippocampus-dependent learning and memory in NP-knockout mice as measured in the Morris water maze or the Y-maze.
A, escape latency to find a submerged platform by NP-knockout (filled circles, n= 13), heterozygous (grey triangles, n= 10) and wild-type mice (open circles, n= 10) in training session on days 1–4. B, total escape latency to find a submerged platform by NP-knockout (KO), heterozygous (+/−) and wild-type mice (WT). Knockout mice showed significantly longer escape latencies than wild-type mice (two-way repeated measures ANOVA: F(1,21)= 6.22, P= 0.022). C and D, spontaneous alternation behaviour of NP-knockout, heterozygous and wild-type mice in the Y-maze (column key as for B). NP-knockout and heterozygous mice displayed significantly lower ratios than wild-type mice (*P < 0.05), whereas there were no significant differences in the total number of arm entries among groups. All data are expressed as the mean ±s.e.m.
In the Y-maze, which is a less stressful hippocampus-dependent memory task than the water maze, wild-type mice showed a significant alternation ratio in arm choices, whereas heterozygous and knockout mice displayed no preference towards the novel arm (Fig. 1C). This working memory defect in the heterozygous and knockout mice is not caused by impairment of locomotor activity, because no difference was observed in the total number of arm entries among the three groups (Fig. 1D). Therefore, the NP-knockout mice were found to be impaired in spatial memory acquisition by both water maze and Y-maze tasks.
Despite the significant impairments in the learning tasks shown by the knockout mice, no difference was seen among NP−/−, NP+/− and NP+/+ mice in spontaneous motor activity examined by an infrared beam-based activity sensor apparatus during the 120 min test period (data not shown).
Deletion of the NP gene or inhibition of NP activity abolishes tetanus-elicited E-LTP and reduces theta-burst-elicited LTP in vivo
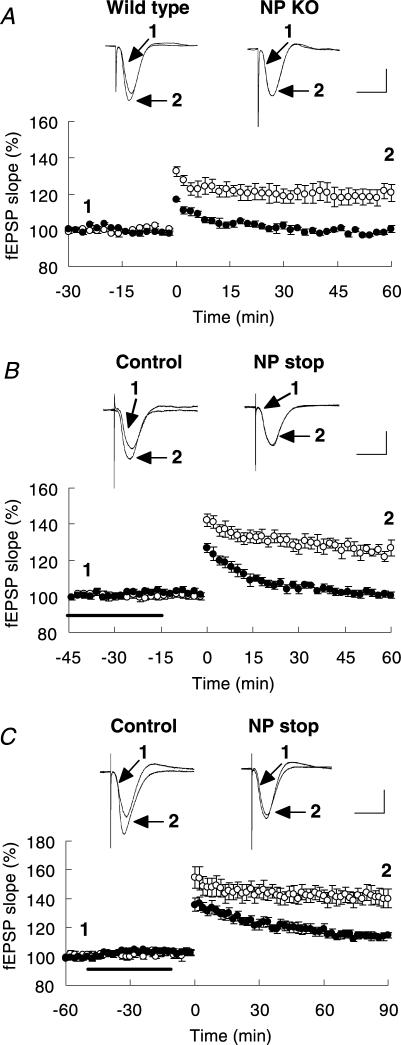
To examine the synaptic effects of NP, we first analysed hippocampal LTP using NP-deficient mice. E-LTP was severely impaired and almost disappeared in the homozygous mutants following a single tetanus, an E-LTP-specific stimulus (60 min after tetanus: wild-type, 120.5 ± 4.5% of baseline, n= 6; NP-knockout, 100.5 ± 2.1% of baseline, n= 6; t test, P < 0.003; Fig. 2A). Since NP is known to be activated during LTP in an NMDA receptor-dependent manner (Matsumoto-Miyai et al. 2003), we further determined whether inhibition of NP activity can abolish an induced LTP using the activity-neutralizing anti-NP monoclonal antibody mAbB5 or the NP-specific inhibitor NP stop. E-LTP was completely blocked by an intracerebroventricular infusion of either mAbB5 (60 min after tetanus: mAbB5, 103.7 ± 3.5% of baseline, n= 3; rat IgG, 125.1 ± 2.9% of baseline, n= 3; t test, P < 0.02) or NP stop (60 min after tetanus: NP stop, 100.5 ± 1.8% of baseline, n= 5; saline, 126.6 ± 4.3% of baseline, n= 5; t test, P < 0.0006; Fig. 2B). Consistent with our previous data (Komai et al. 2000), application of mAbB5 after 30 min of baseline recording and before TBS resulted in a significant decrease in the magnitude of LTP, most markedly in the early phase (data not shown). This was also confirmed by infusion of NP stop, where a significant TBS-associated reduction in LTP was found (90 min after TBS: NP stop, 114.7 ± 1.9% of baseline, n= 8; saline, 140.2 ± 6.6% of baseline, n= 6; t test, P < 0.002; Fig. 2C). Therefore, endogenous NP, and specifically its proteolytic activity, may play a critical role in the induction of E-LTP in vivo. NP stop, mAbB5 and rat IgG did not have any effect on baseline synaptic transmission.
Figure 2. Loss of NP gene or inhibition of NP abolished E-LTP and reduced theta-burst-induced LTP.
A, E-LTP induced by a single train of tetanus is impaired in NP-knockout mice. Representative waveforms of fEPSP slopes recorded before (1) or 60 min after (2) tetanization in NP-knockout (NP KO, •) and wild-type (○) mice are shown on the top. B and C, NP inhibitor, NP stop, significantly blocked theta-burst LTP and completely blocked E-LTP elicited by a single train of tetanus. NP stop (•) or saline (○) was applied intracerebroventricularly for 40 min before theta-burst stimuli (TBS) or tetanus. A stimulus was then applied after 15 min to the Schaffer collateral pathway. In each panel, representative waveforms of fEPSP slopes recorded before (1) or 90 min or 60 min after (2) TBS or tetanization in the presence of saline (Control) or NP stop are shown on the top. Horizontal bar denotes the period of delivery of saline or NP stop. Values are expressed as percentage of baseline. Scale bars, 2 mV and 10 ms. Symbols indicate the mean ±s.e.m.
r-NP alone can elicit LTP-like increases of the fEPSP slope and population spike amplitude in vivo
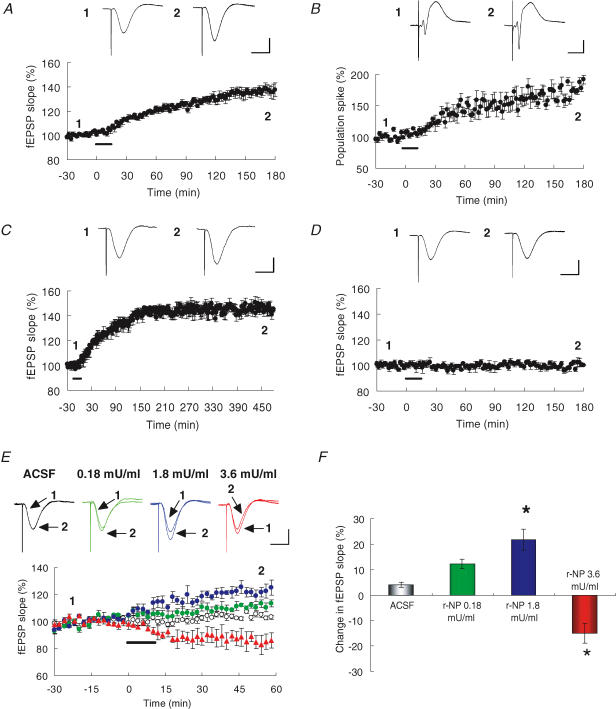
If NP has significant effects on E-LTP, exogenous application of recombinant (r-) NP alone should elicit LTP. The experiment was designed because the proteolytic activity of NP (which is usually inactive) is transiently activated by LTP-inducible neural stimuli and can be substituted by an exogenously applied active form of r-NP (Shimizu et al. 1998; Komai et al. 2000; Matsumoto-Miyai et al. 2003). Therefore, we measured the population spike amplitude and the fEPSP slope after intrahippocampal infusion of r-NP in vivo. Infusion led to an increase of the fEPSP slope (180 min after application of 1.8 mU ml−1 r-NP: 137.5 ± 5.8% of baseline, n= 5; Fig. 3A) and of the population spike amplitude in CA1 pyramidal neurones (180 min after application of 1.8 mU ml−1 r-NP: 191.7 ± 7.0% of baseline, n= 5; Fig. 3B). The enhancement reached a plateau ∼2.5 h after the injection, and was then sustained up to at least 8 h (480 min after application of 1.8 mU ml−1 r-NP: 145.2 ± 0.59% of baseline, n= 3; Fig. 3C). Neither PMSF- nor mAbB5-inactivated r-NP (see Methods) enhanced synaptic efficacy (180 min after application of 1.8 mU ml−1 r-NP: PMSF, 100.0 ± 0.47% of baseline, n= 3; Fig. 3D) (180 min after application of 1.8 mU ml−1 r-NP: mAbB5, 104.6 ± 1.7% of baseline, n= 4).
Figure 3. r-NP potentiates or depresses synaptic transmission on the Schaffer collateral pathway, depending on the applied concentration.
A and B, recombinant neuropsin (r-NP; 1.8 mU ml−1) elicited a stable increase in both fEPSP slope (n= 5) and population spike amplitude (n= 5). C, time course plots are shown from 8 h post-r-NP recording sessions (n= 3). D, PMSF-inactivated r-NP did not elicit an increase in fEPSP slope (n= 3). Each waveform represents an average of 4 sweeps, taken just before r-NP infusion (1) and 3 h after terminating infusion (2). r-NP was infused during the period indicated by the bars. Scale bars, 2 mV and 10 ms. E, representative waveforms before (1) and 60 min after (2) injection of ACSF or r-NP (low, middle, high). Time course of the fEPSP slope after infusion of ACSF (open circles, n= 4) or r-NP (green circles, low, n= 4; blue circles, medium, n= 5; red triangles, high, n= 4). ACSF or r-NP was infused during the period indicated by the bar. Values in A, B, C, D and E are expressed as percentage of baseline. Scale bars, 2 mV and 10 ms. F, the effects of r-NP are shown as average change in fEPSP slopes 60 min after infusion of ACSF (open) or low (green), middle (blue), or high (red) concentrations of r-NP. *P < 0.01 versus ACSF. Values in F are expressed as percentage change in fEPSP slope. Data represent mean ±s.e.m.
A low dose of r-NP elicits LTP, whereas a high dose elicits LTD
The effect of r-NP on the fEPSP slope was examined by infusion of low (0.18 mU ml−1), medium (1.8 mU ml−1) and high (3.6 mU ml−1) concentrations of r-NP. r-NP significantly changed the basal synaptic transmission in a dose-dependent manner (F(3,14)= 25.3, P < 0.001). The low dose had less enhancing effect on fEPSP than the medium dose (60 min after application of a low dose of r-NP: 112.3 ± 1.8% of baseline, n= 5; Fig. 3E and F). Intriguingly, the high dose of r-NP had the opposite effect to the low and medium doses, significantly depressing synaptic transmission (60 min after application of a high dose of r-NP: 85.0 ± 3.8% of baseline, n= 4; one-way ANOVA, P < 0.01 versus ACSF: 60 min after application of ACSF; 104.0 ± 1.1% of baseline, n= 4; Fig. 3E and F). Infused r-NP alone can therefore elicit not only LTP but also LTD in vivo, depending on the dose applied. Because the medium dose (1.8 mU ml−1) produced the maximum effect on LTP-like potentiation, this concentration was used for all subsequent experiments, unless otherwise stated.
Paired-pulse facilitation is not affected during r-NP-elicited synaptic potentiation
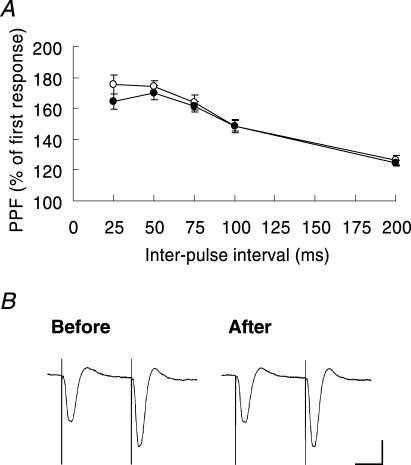
Paired-pulse facilitation (PPF) was examined to identify any presynaptic modification by infusing the medium dose of r-NP into the CA1 subfield. There was no difference between before and after r-NP injection at any of the interstimulus intervals tested (25, 50, 75, 100 and 200 ms, n= 9, two-way ANOVA, F(1,80)= 2.09, P= 0.15; Fig. 4A). The size of PPF taken before r-NP injection was largely consistent with that 3 h after injection (and reduction of stimulus intensity to match the pre-r-NP level) (Fig. 4B), indicating that the probability of neurotransmitter release is not affected by r-NP. Thus, synaptic potentiation by r-NP may be attributable to a change in postsynaptic gain.
Figure 4. Intrahippocampal infusion of r-NP has no significant effect on paired-pulse facilitation.
A, paired-pulse facilitation (PPF) ratios did not change at various interpulse intervals. The PPF ratio is expressed as the percentage of the second fEPSP slope relative to that of the first fEPSP slope. PPF profile 10 min before (○) and 3 h after (•) injection. B, representative waveforms showing PPF (50 ms interpulse intervals) recorded before and after reduction of stimulation strength at 3 h following the infusion of medium-concentration r-NP. Scale bars, 2 mV and 20 ms. Vertical bars on data points are ±s.e.m.
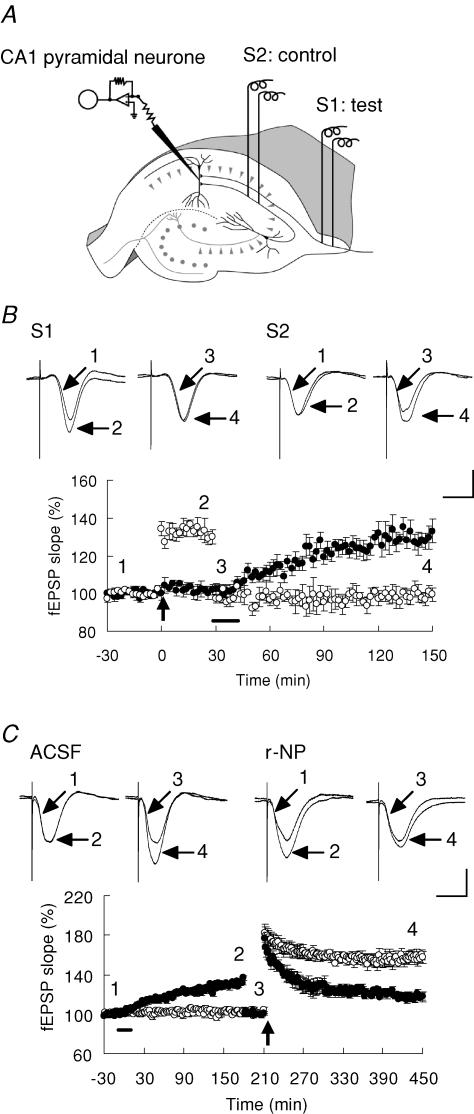
Mutual occlusion of electrically and r-NP-elicited synaptic enhancement
Since r-NP alone can elicit long-lasting synaptic potentiation or depression (Fig. 3E and F), we next focused on whether two forms of synaptic potentiation, electrically and r-NP-elicited, occlude each other. We monitored two populations of Schaffer collateral–commissural projections to the same population of postsynaptic neurones. LTP was induced using TBS in one of the pathways (S1), while the other pathway (S2) served as a control (Fig. 5A). Induction of stable LTP prevented the subsequent r-NP-induced synaptic potentiation, whereas application of r-NP led to an increase in the fEPSP slope on the control pathway (150 min after TBS: S1, 99.6 ± 3.5% of baseline, n= 5; S2, 133.1 ± 6.2% of baseline, n= 5, t test, P < 0.002 versus control; Fig. 5B).
Figure 5. TBS-elicited LTP occludes synaptic potentiation elicited by r-NP and vice versa.
A, two independent inputs (S1 and S2) onto the same population of postsynaptic neurones were alternately stimulated. LTP was induced using TBS of S1, and S2 served as the control. B, after TBS, the stimulus intensity was reduced to match the original baseline. Subsequent application of medium-concentration r-NP had no potentiation effect on the fEPSP slope but an enhancing effect on the control pathway. Representative waveforms (upper panels) were recorded at different times as indicated in the lower panel. Bar denotes the period of delivery of medium-concentration r-NP. C, after r-NP-induced LTP was saturated, the fEPSP slope was reduced to the initial level even when TBS was applied. Representative waveforms were taken at different times as indicated in the lower panel. Bar denotes the period of delivery of ACSF or medium-concentration r-NP. Arrow denotes the point of TBS. In B and C, ○ represents ACSF and • r-NP application. Scale bars in B and C, 2 mV and 10 ms. Percentage change data are presented as mean ±s.e.m.
Conversely, after r-NP-induced synaptic enhancement reached a plateau and stabilized for 30 min, significant attenuation of the TBS-induced LTP appeared, whereas clear LTP was induced in the ACSF-infused control mice (4 h after TBS: r-NP, 117.7 ± 5.6% of baseline, n= 5; ACSF, 157.5 ± 6.0% of baseline, n= 5, t test, P < 0.002 versus ACSF; Fig. 5C). These data strongly suggest that the two types of synaptic plasticity share a common cellular signalling mechanism.
r-NP-elicited change in fEPSP is responsible for phosphorylation of GluR1 subunit of AMPA receptor
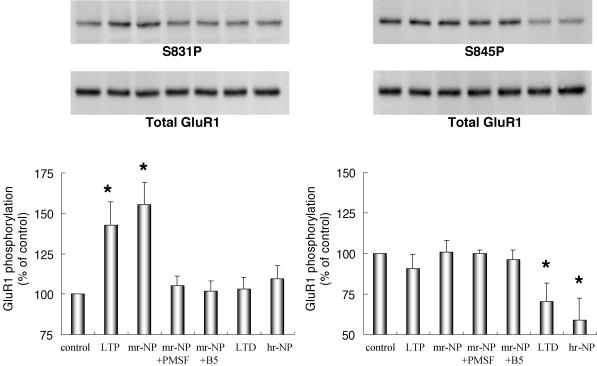
Western blot analysis using antibodies specific to the phosphorylated GluR1 subunit enabled us to examine LTP- or LTD-associated change in phosphorylation of the AMPA receptor (LTP: Ser-831; LTD: Ser-845). The phosphorylation profile of the receptor was examined after expression of r-NP-induced LTP- and LTD-like synaptic changes to verify a common cellular signalling mechanism for the two types of synaptic plasticity. As shown in Fig. 6, the medium dose of r-NP, which elicits maximum LTP-like potentiation, increased band density on Ser-831 (155.3 ± 13.8% of control phosphorylation, n= 5, t test, P < 0.004; Fig. 6) but not on Ser-845 (101.1 ± 7.0% of control phosphorylation, n= 5, t test, P > 0.8; Fig. 6). In contrast, the high dose of r-NP into the hippocampus, which induces LTD-like depression, significantly reduced band density on Ser-845 (58.9 ± 13.6% of control phosphorylation, n= 5, t test, P < 0.02; Fig. 6), with no change for Ser-831 (109.6 ± 8.1% of control phosphorylation, n= 5, t test, P > 0.2; Fig. 6). PMSF- or mAbB5-inactivated r-NP completely abolished the changes in phosphorylation of GluR1 (PMSF: Ser-831, 105.3 ± 6.0% of control phosphorylation, n= 3, t test, P > 0.4; Ser-845, 99.9 ± 2.2% of control phosphorylation, n= 3, t test, P > 0.9; mAbB5: Ser-831, 101.7 ± 6.3% of control phosphorylation, n= 3, t test, P > 0.7; Ser-845, 96.1 ± 6.1% of control phosphorylation, n= 3, t test, P > 0.5; Fig. 6). Taken together, these data show that r-NP-induced synaptic plasticity shares an identical cellular mechanism with conventional LTP and LTD elicited by electric stimuli.
Figure 6. Changes in LTP- and LTD-associated phosphorylation of the GluR1 subunit induced by intrahippocampal application of r-NP.
Infusion of medium-concentration r-NP (mr-NP) induced a significant increase in band density GluR1 subunit of AMPA receptor at Ser-831 (CAMK II site: S831P) but no change at Ser-845 (PKA site: S845P), while high-concentration r-NP (hr-NP) induced significant reduction in band density at Ser-845 but no change at Ser-831. The former is an LTP-specific change (LTP), and the latter is an LTD-specific change (LTD). Neither PMSF- (mr-NP + PMSF) nor mAbB5- (mr-NP + B5) inactivated mr-NP showed changed band densities. Control hippocampi infused with ACSF only (control) showed no change in band density. The blots were stripped and reprobed with anti-GluR1 C-terminal antibody (Total GluR1). Columns in the lower panels represent band densities of GluR1 at Ser-831 (left panel) and Ser-845 (right panel) before and after LTP or LTD induction and r-NP infusion (see Methods). *P < 0.05 versus control. Data represent mean ±s.e.m.
Discussion
LTP of synaptic transmission in the hippocampus is a well-studied experimental model for the synaptic changes that may be the mechanistic basis for memory. Brief periods of synchronous activity in the input fibres can trigger potentiation in postsynaptic intracellular signalling after the binding of an excitatory amino acid transmitter by its receptors (Bliss & Collingridge, 1993; Malenka & Nicoll, 1993). Recently, some investigators have suggested that plasticity change in long-lasting enhancement of synaptic transmission accompanies morphological change, synaptogenesis, disorganization and reorganization of the ECM and rearrangement of the cytoskeleton (Luthl et al. 1994; Cremer et al. 1998; Tang et al. 1998; Colicos et al. 2001). Peri-synaptic proteolysis of the ECM by extracellular proteases may be involved in such morphological changes and synaptogenesis. Ectodomain proteolysis by NP of bioactive proteins in the ECM and cell membrane may play significant roles in mechanical plasticity via processing or degradation of matrix and CAM. We have shown that NP has a strong modulatory effect on synaptic plasticity (Komai et al. 2000; Matsumoto-Miyai et al. 2003). In the present study, our NP-deficient mice had defects in hippocampus-dependent spatial memory notably during the non-stressful Y-maze task. This memory defect is strongly compatible with the finding that loss of the NP gene (see below) or inhibition of NP activity markedly impairs LTP in vivo. A potential role for NP in LTP might be as a major effecter situated upstream of a signalling cascade for synaptic plasticity, because r-NP alone can occlude TBS-induced LTP and induce LTP-associated phosphorylation of the AMPA receptor. In particular, the role of NP in LTP is critical in its early phase, since loss of NP or inhibition of NP completely abolished E-LTP induced by a single tetanus. Collectively, our findings suggest that the extracellular protease functions as an upstream regulator of AMPA receptor phosphorylation to determine synaptic efficacy in plasticity.
However, there are some inconsistencies between our data and those of Davies et al. (2001) for NP-deficient mice. Their knockout mice displayed normal LTP (presumably L-LTP), established by repeated tetanic stimulation, and a normal performance in the water maze test, whereas our data indicate significant impairment in both electrophysiological and behavioural studies. The present study clearly demonstrates that the effects of NP on synaptic plasticity are most marked, perhaps even critical, in the early phase of LTP, but less significant in the late phase. Because NP deficiency, or infusion of NP stop or anti-NP monoclonal antibody, strongly impaired E-LTP induced by a single tetanus, it is reasonable to assume that NP is involved in E-LTP rather than L-LTP. The same tendency was also found in the behavioural tests: NP-knockout mice are impaired not in memory retention but in memory acquisition (present study). Since stronger stimuli preferentially elicited late-phase potentiation, the water maze task, which is relatively stressful for mice, induced rather mild impairment in knockout mice. Davies et al. (2001) did not observe any aberration in the water maze task. The contradictory results might be attributable to differences in experimental conditions. The water maze test is particularly sensitive to water temperature, for example: animals trained in cooler water (19°C) perform better than those trained in the warmer water (25°C) which Davies et al. (2001) used (Akirav et al. 2001), and we used relatively cool water (21–22°C) for the water maze task in the present study. Other factors contributing to the inconsistency might be genetic background, the way in which the NP locus was disrupted in deficient mice, and different experimental conditions. To minimize genetic background influences, we exclusively used mutant mice of backcross generation (n > 9) on C57BL/6J and their littermates as a control. We also examined another memory task in addition to the water maze task. Spontaneous alternation behaviour in the Y-maze is regarded as a spatial working memory test. NP−/− and NP+/− mice exhibited a lower alternation ratio than the wild-type mice. Thus, the results of both memory tasks strongly support the idea that NP is involved in the acquisition of hippocampus-dependent learning processes, but is not so important in retention of memory.
The application of NP had no significant effect on paired-pulse facilitation, meaning that NP does not have any effect on presynaptic transmitter release. Therefore, NP-elicited plasticity in the hippocampus is a postsynaptic event, as are electrically evoked LTP and LTD. This interpretation is strongly supported by occlusion experiments and by activation of the AMPA receptor at specific phosphorylation sites for LTP and LTD. Thus, NP-dependent synaptic modification downstream of stimulus-triggered NMDA receptor signalling returns again to the postsynaptic elements, without modification of presynaptic elements. This turn-back signalling from outside to inside might multiply or strengthen the postsynaptic potentiation of signals. Since NP is diffusible in the extracellular space, it can be speculated that neighbouring boutons of L1cam-positive immature small synapses and not-yet-contacted L1cam-positive immature boutons (orphan boutons) are oscillatorily activated during LTP (Nakamura et al. unpublished observations).
We found that the active form of the protease NP alone can elicit a plastic change of intracellular signalling which is similar to electrically evoked LTP, and acts as a major extracellular modifier, particularly on E-LTP. The results imply that postsynaptic signalling subsequent to E-LTP-inducible stimuli returns to the extracellular space, where proNP (zymogen) is ready to activate (Shimizu et al. 1998; Matsumoto-Miyai et al. 2003). Activation of NP occurs by cleavage of a 4-amino acid peptide (QGSK) after a signal from downstream of the NMDA receptor (Shimizu et al. 1998; Matsumoto-Miyai et al. 2003). It is clear that this activation of NP is required for establishment of E-LTP and also partially of LTP induced by TBS, because inhibition or inactivation of NP blocked E-LTP completely and LTP induced by TBS partially (Komai et al. 2000; present study).
Finally, the present study has clearly demonstrated that NP-driven synaptic modification, presumably due to rearrangement of the ECM, is a major mechanism for synaptic plasticity, particularly in E-LTP. This mechanism also plays an important role in LTD. Peri-synaptic proteolysis of the ECM by NP might be essential in E-LTP and consequently in acquisition of memory.
Acknowledgments
We thank Professor N. Matsuki, Dr Y. Ikegaya, and Dr K. Nakao (Tokyo University) for technical advice on electrophysiology, Dr K. Kameyama (National Institute of Bioscience and Human Technology) for the gift of anti-phospho-GluR1 antibody, and Dr Ian Smith for help with the English. This work was supported partly by a Grant-in-Aid for 21st Century COE Research and KAKENHI (14658251) from the Ministry of Education, Culture, Sports, Science and Technology (Japan).
References
- Akirav I, Sandi C, Richter-Levin G. Differential activation of hippocampus and amygdala following spatial learning under stress. Eur J Neurosci. 2001;14:719–725. doi: 10.1046/j.0953-816x.2001.01687.x. [DOI] [PubMed] [Google Scholar]
- Bahr BA, Staubli U, Xiao P, Chun D, Ji ZX, Esteban ET, Lynch G. Arg-Gly-Asp-Ser-selective adhesion and the stabilization of long-term potentiation: pharmacological studies and the characterization of a candidate matrix receptor. J Neurosci. 1997;17:1320–1329. doi: 10.1523/JNEUROSCI.17-04-01320.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baranes D, Lederfein D, Huang YY, Chen M, Bailey CH, Kandel ER. Tissue plasminogen activator contributes to the late phase of LTP and to synaptic growth in the hippocampal mossy fiber pathway. Neuron. 1998;21:813–825. doi: 10.1016/s0896-6273(00)80597-8. [DOI] [PubMed] [Google Scholar]
- Bliss TV, Collingridge GL. A synaptic model of memory: long-term potentiation in the hippocampus. Nature. 1993;361:31–39. doi: 10.1038/361031a0. [DOI] [PubMed] [Google Scholar]
- Bliss TV, Lomo T. Long-lasting potentiation of synaptic transmission in the dentate area of the anaesthetized rabbit following stimulation of the perforant path. J Physiol. 1973;232:331–356. doi: 10.1113/jphysiol.1973.sp010273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen ZL, Yoshida S, Kato K, Momota Y, Suzuki J, Tanaka T, et al. Expression and activity-dependent changes of a novel limbic-serine protease gene in the hippocampus. J Neurosci. 1995;15:5088–5097. doi: 10.1523/JNEUROSCI.15-07-05088.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colicos MA, Collins BE, Sailor MJ, Goda Y. Remodeling of synaptic actin induced by photoconductive stimulation. Cell. 2001;107:605–616. doi: 10.1016/s0092-8674(01)00579-7. [DOI] [PubMed] [Google Scholar]
- Cremer H, Chazal G, Carleton A, Goridis C, Vincent JD, Lledo PM. Long-term but not short-term plasticity at mossy fiber synapses is impaired in neural cell adhesion molecule-deficient mice. Proc Natl Acad Sci U S A. 1998;95:13242–13247. doi: 10.1073/pnas.95.22.13242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davies B, Kearns IR, Ure J, Davies CH, Lathe R. Loss of hippocampal serine protease BSP1/neuropsin predisposes to global seizure activity. J Neurosci. 2001;21:6993–7000. doi: 10.1523/JNEUROSCI.21-18-06993.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frey U, Huang YY, Kandel ER. Effects of cAMP simulate a late stage of LTP in hippocampal CA1 neurons. Science. 1993;260:1661–1664. doi: 10.1126/science.8389057. [DOI] [PubMed] [Google Scholar]
- Frey U, Muller M, Kuhl D. A different form of long-lasting potentiation revealed in tissue plasminogen activator mutant mice. J Neurosci. 1996;16:2057–2063. doi: 10.1523/JNEUROSCI.16-06-02057.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hesse GW, Teyler TJ. Reversible loss of hippocampal long term potentiation following electronconvulsive seizures. Nature. 1976;264:562–564. doi: 10.1038/264562a0. [DOI] [PubMed] [Google Scholar]
- Hirata A, Yoshida S, Inoue N, Matsumoto-Miyai K, Ninomiya A, Taniguchi M, et al. Abnormalities of synapses and neurons in the hippocampus of neuropsin-deficient mice. Mol Cell Neurosci. 2001;17:600–610. doi: 10.1006/mcne.2000.0945. [DOI] [PubMed] [Google Scholar]
- Huang YY, Bach ME, Lipp HP, Zhuo M, Wolfer DP, Hawkins RD, et al. Mice lacking the gene encoding tissue-type plasminogen activator show a selective interference with late-phase long-term potentiation in both Schaffer collateral and mossy fiber pathways. Proc Natl Acad Sci U S A. 1996;93:8699–8704. doi: 10.1073/pnas.93.16.8699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang YY, Li XC, Kandel ER. cAMP contributes to mossy fiber LTP by initiating both a covalently mediated early phase and macromolecular synthesis-dependent late phase. Cell. 1994;79:69–79. doi: 10.1016/0092-8674(94)90401-4. [DOI] [PubMed] [Google Scholar]
- Kato K, Kishi T, Kamachi T, Akisada M, Oka T, Midorikawa R, et al. Serine proteinase inhibitor 3 and murinoglobulin I are potent inhibitors of neuropsin in adult mouse brain. J Biol Chem. 2001;276:14562–14571. doi: 10.1074/jbc.M010725200. [DOI] [PubMed] [Google Scholar]
- Komai S, Matsuyama T, Matsumoto K, Kato K, Kobayashi M, Imamura K, Yoshida S, Ugawa S, Shiosaka S. Neuropsin regulates an early phase of schaffer-collateral long-term potentiation in the murine hippocampus. Eur J Neurosci. 2000;12:1479–1486. doi: 10.1046/j.1460-9568.2000.00035.x. [DOI] [PubMed] [Google Scholar]
- Krug M, Lossner B, Ott T. Anisomycin blocks the late phase of long-term potentiation in the dentate gyrus of freely moving rats. Brain Res Bull. 1984;13:39–42. doi: 10.1016/0361-9230(84)90005-4. [DOI] [PubMed] [Google Scholar]
- Lee HK, Kameyama K, Huganir RL, Bear MF. NMDA induces long-term synaptic depression and dephosphorylation of the GluR1 subunit of AMPA receptors in hippocampus. Neuron. 1998;21:1151–1162. doi: 10.1016/s0896-6273(00)80632-7. [DOI] [PubMed] [Google Scholar]
- Luthl A, Laurent JP, Figurov A, Muller D, Schachner M. Hippocampal long-term potentiation and neural cell adhesion molecules L1 and NCAM. Nature. 1994;372:777–779. doi: 10.1038/372777a0. [DOI] [PubMed] [Google Scholar]
- Malenka RC, Nicoll RA. NMDA-receptor-dependent synaptic plasticity: multiple forms and mechanisms. Trends Neurosci. 1993;16:521–527. doi: 10.1016/0166-2236(93)90197-t. [DOI] [PubMed] [Google Scholar]
- Matsumoto-Miyai K, Ninomiya A, Yamasaki H, Tamura H, Nakamura Y, Shiosaka S. NMDA-dependent proteolysis of presynaptic adhesion molecule L1 in the hippocampus by neuropsin. J Neurosci. 2003;23:7727–7736. doi: 10.1523/JNEUROSCI.23-21-07727.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Momota Y, Yoshida S, Ito J, Shibata M, Kato K, Sakurai K, Matsumoto K, Shiosaka S. Blockade of neuropsin, a serine protease, ameliorates kindling epilepsy. Eur J Neurosci. 1998;10:760–764. doi: 10.1046/j.1460-9568.1998.00068.x. [DOI] [PubMed] [Google Scholar]
- Ronn LC, Bock E, Linnemann D, Jahnsen H. NCAM-antibodies modulate induction of long-term potentiation in rat hippocampal CA1. Brain Res. 1995;677:145–151. doi: 10.1016/0006-8993(95)00147-i. [DOI] [PubMed] [Google Scholar]
- Shimizu C, Yoshida S, Shibata M, Kato K, Momota Y, Matsumoto K, et al. Characterization of recombinant and brain neuropsin, a plasticity-related serine protease. J Biol Chem. 1998;273:11189–11196. doi: 10.1074/jbc.273.18.11189. [DOI] [PubMed] [Google Scholar]
- Staubli U, Chun D, Lynch G. Time-dependent reversal of long-term potentiation by an integrin antagonist. J Neurosci. 1998;18:3460–3469. doi: 10.1523/JNEUROSCI.18-09-03460.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang L, Hung CP, Schuman EM. A role for the cadherin family of cell adhesion molecules in hippocampal long-term potentiation. Neuron. 1998;20:1165–1175. doi: 10.1016/s0896-6273(00)80497-3. [DOI] [PubMed] [Google Scholar]
- Thiels E, Barrionuevo G, Berger TW. Excitatory stimulation during postsynaptic inhibition induces long-term depression in hippocampus in vivo. J Neurophysiol. 1994;72:3009–3016. doi: 10.1152/jn.1994.72.6.3009. [DOI] [PubMed] [Google Scholar]
- Xu L, Anwyl R, Rowan MJ. Spatial exploration induces a persistent reversal of long-term potentiation in rat hippocampus. Nature. 1998;394:891–894. doi: 10.1038/29783. [DOI] [PubMed] [Google Scholar]
- Yoshida S, Shiosaka S. Plasticity-related serine proteases in the brain. Int J Mol Med. 1999;3:405–409. doi: 10.3892/ijmm.3.4.405. [DOI] [PubMed] [Google Scholar]