Abstract
The locus of enterocyte effacement (LEE) is a large multigene chromosomal segment encoding gene products responsible for the generation of attaching and effacing lesions in many diarrheagenic Escherichia coli strains. A recently sequenced LEE harboring a pathogenicity island (PAI) from a Shiga toxin E. coli serotype O26 strain revealed a LEE PAI (designated LEE O26) almost identical to that obtained from a rabbit-specific enteropathogenic O15:H− strain. LEE O26 comprises 59,540 bp and is inserted at 94 min within the mature pheU tRNA locus. The LEE O26 PAI is flanked by two direct repeats of 137 and 136 bp (DR1 and DR2), as well as a gene encoding an integrase belonging to the P4 integrase family. We examined LEE O26 for horizontal gene transfer. By generating mini-LEE plasmids harboring only DR1 or DR2 with or without the integrase-like gene, we devised a simple assay to examine recombination processes between these sequences. Recombination was shown to be integrase dependent in a ΔrecA E. coli K-12 strain background. Recombinant plasmids harboring a single direct repeat cloned either with or without the LEE O26 integrase gene were found to insert within the chromosomal pheU locus of E. coli K-12 strains with equal efficiency, suggesting that an endogenous P4-like integrase can substitute for this activity. An integrase with strong homology to the LEE O26 integrase was detected on the K-12 chromosome associated with the leuX tRNA locus at 97 min. Strains deleted for this integrase demonstrated a reduction in the insertion frequency of plasmids harboring only the DR into the pheU locus. These results provide strong evidence that LEE-harboring elements are indeed mobile and suggest that closely related integrases present on the chromosome of E. coli strains contribute to the dynamics of PAI mobility.
Shiga toxin-producing Escherichia coli (STEC) strains are well-known enteropathogens that have recently emerged, causing serious forms of diarrheagenic disease and acute renal failure (4, 15). The pathogenicity of these strains is due to different independent elements such as phage-encoded toxins (Shiga toxins) (34), plasmids (5), and chromosomal elements (18, 33). A pathogenicity island (PAI) known as the locus of enterocyte effacement (LEE) encodes the required bacterial products for the production of the attaching and effacing (A/E) lesions (29). The A/E lesion is characterized by the loss of host cell microvilli (effacement) and intimate attachment of the bacterium to the host membrane on a pedestal of polymerized cytoskeletal elements (13, 25).
The LEE region, which is not present in the K-12 chromosome (3), shows some differences in length and location between different E. coli groups. In enteropathogenic E. coli (EPEC) O127:H7 it is 35.637 kb in length and is inserted at ca. 82 min, next to the locus of tRNA for selenocysteine (9, 30). The LEE region in STEC O157:H7 strain EDL933 shows the same genes found in the EPEC LEE at the same location but also possesses a fragment that encodes for a cryptic prophage at the 3′ end that is not observed in EPEC (36). The LEE region in STEC serotype O26:H− strain 413/89-1 (NCBI accession number AJ277443) has a structure different from those of the two loci described above. Referred to as LEE O26, it is located at 94 min on the K-12 chromosome, adjacent to the pheU tRNA locus, and has a total length of 59.5 kb. This LEE PAI is virtually identical in sequence and length and shares the same point of insertion on the E. coli chromosome as a LEE PAI characterized from an enteropathogenic rabbit serotype O15:H− (REPEC) isolate (45). The LEE O26 sequences are flanked by two direct repeats of 137 and 136 bp (DR1 and DR2) at its boundaries. An integrase gene with homology to the P4 family of integrases is present immediately adjacent to the DR2 repeat close to the pheU gene. The 137-bp DR1 repeat sequence is also present, albeit as a single copy in the chromosome of E. coli K-12 (3).
An integrase is a site-specific recombinase that mediates a wide range of biological outcomes by the sequential exchange of two pairs of DNA strands (14, 16). The well-known integrase family of tyrosine recombinases comprises 81 members, including P4 integrase (12). Three boxes (A, B, and C) characterize the family, the members of which share a high degree of sequence similarity at the amino acid level, as well as a conserved tyrosine residue close to the C terminus. The P4 integrase promotes integration and excision of the phage genome from that of the host (26) and thus regulates the life cycle of the temperate phage. All of these processes involve the conservative site-specific recombination of two DNA partners (donor and recipient). Without these specific sequences, integrases are not able to act on their target DNA (19).
A common feature of PAIs is that they are often flanked by small direct-repeat (DR) DNA sequences resembling attachment (att) sites for phage integrases and contain potential mobility genes, such as transposase and integrase genes (17). PAIs possessing this general structure are unstable and have been described for many organisms, including E. coli, Staphylococcus aureus, Shigella flexneri, and Yersinia pseudotuberculosis (1, 27, 36, 48). In contrast to these PAIs, the LEE loci of STEC that have been characterized to date are bereft of elements that indicate mobility (i.e., flanking homologous sequences and the presence of active integrases) (36). Thus, these elements appear to be stable entities residing within the E. coli genome. For the LEE region of REPEC serotypes O15:H− and O103:H2, an active integrase directly associated with this PAI was identified that could mediate excision and site-specific integration at the pheU tRNA locus of E. coli DH1 at low frequency (45).
The presence of an integrase, as well as two defined homologous regions, in O26 LEE suggests that this element is capable of mobility and indicates a possible mode for the insertion of this genetic element via site-specific recombination. A more general interpretation of this hypothesis suggests that in an earlier evolutionary step these sequences were used to integrate the LEE locus into a progenitor chromosome, generating strains with a new A/E phenotype. In the present study, we looked for evidence of mobility by generating mini-LEE O26 substrates comprising the direct repeat sequences (DR1 and DR2) and the integrase. Our data indicate that the integrase encoded on the LEE O26 element is indeed active and mediates site-specific recombination using DR1/DR2. Furthermore, we show that other elements present in the K-12 chromosome play an active role in absence of the LEE O26 integrase by promoting integration of LEE O26 substrates in the E. coli chromosome.
MATERIALS AND METHODS
Bacterial strains and growth media.
The bacterial strains and plasmids used are summarized in Table 1. Bacteria were routinely grown at 37°C in Luria-Bertani (LB) broth. The medium was supplemented with ampicillin (100 μg ml−1), kanamycin (50 μg ml−1), or chloramphenicol (20 μg ml−1) as required.
TABLE 1.
Strains and plasmids used in this study
| Strain or plasmid | Descriptiona | Source or reference |
|---|---|---|
| Strains | ||
| 413/89-1 | Stx-producing E. coli O26:H− wild-type, bovine isolate | 47 |
| INVαF′ | E. coli K-12 F′ endA1 recA1 hsdR17 (rK− mK−) supE44 thi-1 gyrA96 relA1 φ80 lacZΔM15 Δ(lacZYA-argF)U169 deoR λ− | Invitrogen |
| S17-λpir::RP4 | E. coli Tpr SmrrecA thi pro hsdRM+ RP4:2-Tc:Mu:Km Tn7 λ− pir | 21 |
| MG1655 | E. coli K-12 recA+ | 3 |
| MG1655recA56 srl300::Tn10 | E. coli MG1655 derivative harboring the recA56 mutant allele; P1 transduction | This study |
| MG1655Δb4271 | E. coli MG1655 derivative containing an in-frame deletion in the P4 integrase gene (b4271) located in the leuX locus | This study |
| MG1655Δb4271 recA56 srl300::Tn10 | E. coli MG1655 derivative containing an in-frame deletion in the integrase gene and recA56 allele | This study |
| E1294 | E. coli INVαF′ harboring plasmid pMMG-8; Apr | This study |
| Plasmids | ||
| pBAD/Myc-HisC | Apr | Invitrogen |
| pTSC29 | Cmr | 37 |
| pGP704 | Apr; ori R64 mob RP4 | 21 |
| pWH36 | pBluescript II SK derivative; Apr | This study |
| pWSK129 | Kmr | 46 |
| pMMG-4 | pGP704 harboring a 1.65-kb EcoRI fragment that corresponds to the initial sequence from LEE O26 containing direct repeat 1 (LEE O26-1) | This study |
| pMMG-5 | pGP704 harboring a 1.45-kb EcoRI fragment that corresponds to the final sequence from LEE O26 containing direct repeat 2 and 500 bp of LEE O26 integrase (LEE O26-2) | This study |
| pMMG-7 | pWSK129 harboring a 1.45-kb EcoRI fragment that corresponds to the final sequence from LEE O26 containing direct repeat 2 and LEE O26 integrase (LEE O26-2) | This study |
| pMMG-8 | pBAD/Myc-HisC, harboring an NcoI/BglII 1.19-kb fragment containing LEE O26 integrase gene | This study |
| pMMG-12 | pGP704 harboring an 860-bp EcoRI fragment containing only direct repeat 2 | This study |
| pMMG-15 | pGP704 harboring a EcoRI 1.19-kb fragment containing LEE O26 integrase gene | This study |
| pMMG-16 | pGP704 harboring a EcoRI 2.1-kb fragment containing LEE O26 integrase gene and direct repeat 2 | This study |
Cmr, chloramphenicol resistance; Smr, streptomycin resistance; Tpr, trimethoprim resistance; Apr, ampicillin resistance; Kmr, kanamycin resistance.
DNA manipulations.
Plasmid DNA isolation, restriction endonuclease digestion, ligation, transformation, agarose gel electrophoresis, and other standard DNA techniques were performed as previously described (40). PCR amplifications were performed by using the Expand High Fidelity PCR Kit (Boehringer Mannheim, Germany). The oligonucleotides used to amplify the different sequences from the O26 LEE O26 region to confirm chromosomal integration and to construct the nonpolar mutants are summarized in Table 2. Extraction of PCR products and cloning experiments were performed according to standard protocols (40). Restriction and modification enzymes were purchased from New England Biolabs (Schwalbach, Germany). Electroporation was carried out by using a Gene Pulser (Bio-Rad Laboratories) as described by O'Callaghan and Charbit (35). The sequencing of all recombinant clones was performed by standard procedures and analyzed on an automatic sequencing apparatus MegaBace1000. All sequences obtained were analyzed in contigs by using the SeqMan software of DNAStar 93-98. The sequence of the O26 LEE has been deposited at GenBank under the accession number AJ277443.
TABLE 2.
Oligonucleotides used in this study
| Name | Nucleotide sequencea | Amplimer |
|---|---|---|
| ML1up | TATGGACGGAGTAACAAGCC | LEE O26-1: 1.65-kb amplimer that corresponds to the first fragment of the LEE O26 locus containing direct repeat 1 |
| ML1lp | CGTCAATCGTCTGGTAGAACA | |
| ML1upEco | TTGGTTTATGGAATCTGGAGTAACAAGCCAAC | Same as above with restriction cutting sites for EcoRI and BglII, respectively |
| ML1lpBgl | CGTCAATCGAGATCTGGTAGAACATAATCTTCA | |
| ML2up | AGCAGGATTATATCTGATGAT | LEE O26-2: 1.45-kb amplimer that corresponds to the final fragment from LEE O26 containing direct repeat 2 and a 500-bp fragment of the LEE O26 integrase |
| ML2lp | ATGGTTGCTGAACGTGTGGATT | |
| ML2upBgl | AGCAGGATTAGATCTGATGATGCCAGACTGTAC | Same as above with restriction cutting sites for BglII and EcoRI, respectively |
| ML2lpEco | ATGGTTGCTGAATTCCGTGTGGATTATCCACTG | |
| Intup-Bgl | GCATTTGCCCGGAGATCTGATTACTGTTTTAATGGAGC | 1.19-kb fragment containing LEE O26 integrase; restriction cutting sites for BglII and NcoI, respectively |
| Intlp-Nco | GCGCCATGGGCCTGCTGGTACATCCTAATGG | |
| Intup-Eco | GCATTTGAATTCCCGGGATGATTACTGTTTTA | 1.19-kb fragment containing LEE O26 integrase; restriction cutting sites for EcoRI |
| Intlp-Eco | GCGCGGAATTCGCCTGCTGGTACATCCTAATGG | |
| DR2up.Eco | AGTTCAGCCGAATTCTGAATGCGTTAAAGAGGGTAC | 860-bp fragment containing repeat sequence 2; restriction cutting sites for EcoRI |
| ML2lp-Eco | ATGGTTGCTGAATTCCGTGTGGATTATCCACTG | |
| PheUup | TTGGTTTATGGACGGAGTAACAA | Used to evaluate chromosomal insertion in E. coli K-12 |
| PheUlp | GCAACGTGAAGATGTACTGGGAGA | Used to evaluate chromosomal insertion in E. coli K-12 |
| DP-ΔInt1 | CACAACGAATTCGCATACCTCTTCAGTGC | Construction of MG1655Δb4271 mutant located at locus leuX; restriction cutting sites for EcoRI and BglII, respectively |
| DP-ΔInt2 | AGATCTACAAGCAGATGCATACC | |
| DP-ΔInt3 | AGATCTGTTTATCAGTCCATTTGAATATG | |
| DP-ΔInt4 | GGTTCAGAATTCTGCTGAACAATTGCGATG |
Restriction sites are indicated in boldface.
Site-specific recombination.
Recombinant plasmids described in Table 1 were used to transform chemically competent cells as previously described (40). Transformation was performed by using precisely 200 ng of each plasmid, and the entire mixture was plated. After overnight incubation at 37°C, plates were examined for the generation of cointegrate structures or for chromosomal integration after recombination via the repeat sequences (Fig. 1 and 2). The recombination frequency was monitored and is expressed as the number of recombinants resulting from the transformation per 200 ng of plasmid DNA.
FIG. 1.
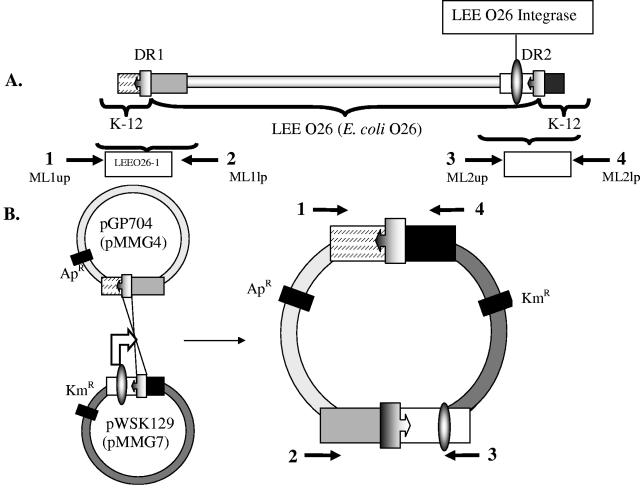
(A) Schematic map of the E. coli O26 LEE O26 locus indicating the position of the direct repeats (DR1 and DR2) and the integrase gene. Adjacent E. coli K-12 regions are indicated, as well as the PCR-amplified regions that were cloned to construct plasmids pMMG-4 and pMMG-7, respectively. (B) Schematic diagram of plasmids pMMG-4 (pGP704 derivative containing DR1) and pMMG-7 (pWSK129 derivative containing DR2 and the LEE integrase) indicating the mechanism of homologous recombination between the DRs and the resulting cointegrate structure. Shown also are the primer concentrations used to monitor site-specific cointegration.
FIG. 2.
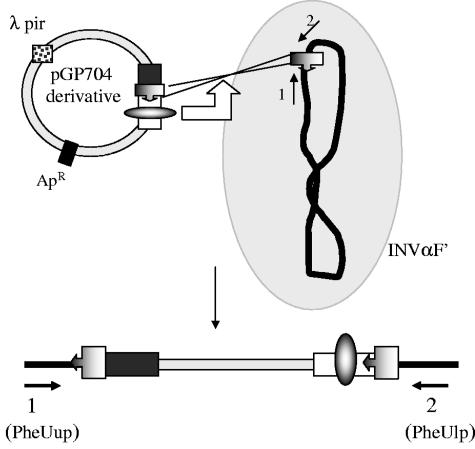
Schematic diagram demonstrating the mechanism of homologous recombination between the pGP704-derivative plasmids containing a copy of the LEE DR and the identical copy of the DR sequence on the E. coli K-12 chromosome. The resultant chromosomal integration structure is also shown, together with the primer set used to monitor this insertion event.
Monitoring of cointegrate formation.
Plasmids pMMG-4 and pMMG-7 were simultaneously transformed into E. coli INVαF′. Transformant colonies that were kanamycin resistant (Kmr) and ampicillin resistant (Apr) were isolated and checked for the presence of cointegrates as follows. Transformants were pooled into groups of 10 colonies, resuspended in 50 μl of LB broth, and lysed by using an established procedure (22). The suspension was centrifuged, and 10 μl of the supernatant was used as a template to perform a PCR with the primer pairs ML1up-ML2lp and ML2up-ML1lp (to amplify the expected cointegrate fragments of 1.76 and 1.34 kb, respectively) (Fig. 1). Pools from which positive cointegrates were identified were screened to identify individual positive colonies. Cointegrate formation was further identified by restriction analysis with EcoRI and finally by DNA sequencing.
To evaluate the role of different genetic elements in cointegrate formation, pMMG-7 was transformed simultaneously with either pMMG-4, pMMG-5, pMMG-12, pMMG-15, or pMMG-16 (Table 1). These plasmids contain different fragments originating from the ends of the LEE O26 PAI (see Table 1 and Fig. 3). Precisely 200 ng of plasmid pMMG-7 was simultaneously transformed into E. coli INVαF′ with 200 ng of each of the pGP704 derivative plasmids. The number of transformant colonies that were Kmr and Apr was counted to evaluate the frequency of the homologous recombination between plasmid pMMG-7 and the different pGP704 plasmid derivatives. In addition, five colonies from each independent experiment were selected for further analysis. Plasmid minipreps were prepared from overnight cultures, and these were analyzed for cointegrate formation by restriction endonuclease digestion and PCR. The PCR strategy used to amplify the recombinant sequence contained in each cointegrate involved two reactions. The first reaction was performed with the forward primer of the LEE O26-DR2 sequence (ML2up) and the reverse primer for each respective sequence from the pGP704 derivative plasmids. The second reaction was performed with the reverse primer of the LEE O26-DR2 sequence (ML2lp) and the forward primer for each respective sequence from the pGP704 derivative plasmids. The formation of a cointegrate structure was assumed when both PCR products showed the expected size in the two reactions. The final conformation was provided by direct DNA sequencing of the PCR products.
FIG. 3.
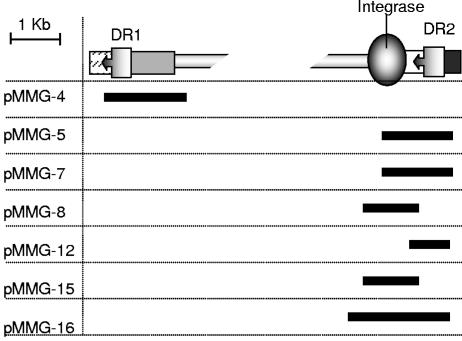
Schematic map of the end regions of the E. coli O26 LEE O26 locus indicating the extent of the segments cloned in each plasmid construct.
Chromosomal insertion.
Chromosomal insertion of the pGP704 derivative plasmids was achieved by transforming precisely 200 ng of each plasmid into E. coli INVαF′ as described above. Transformant colonies that were Apr were isolated and checked for the chromosomal insertion of plasmid DNA by PCR. The primers used were PheUup/ML2lp and PheUlp/ML2up for the integration of plasmid pMMG-5, PheUup/ML2lp and PheUlp/DR2upEco for the integration of plasmid pMMG-12, and PheUup/ML2lp-Eco and PheUlp/Intup-Eco for the integration of plasmid pMMG-16. The identity of each amplification product was verified by DNA sequencing. The total number of colonies obtained was enumerated, and the efficiency of the integration between different plasmids was compared. Chromosomal insertions originating from the cotransformation of pMMG-4 and pMMG-7 were detected by PCR with the primer pairs PheUup/ML1lp and PheUlp/ML1up.
Cloning and expression of the LEE integrase gene.
The LEE integrase was PCR amplified with the primers Intup-Bgl and Intlp-Nco (Table 2) and cloned into the NcoI-BglII sites of plasmid pBAD/Myc-HisC (Invitrogen). This resulted in a plasmid construct (pMMG-8) in which the LEE integrase was positioned immediately downstream of the Para inducible promoter. Different final concentrations of arabinose (ranging from 0.002 to 0.2%) were used to optimize integrase expression as described by the manufacturer (Invitrogen). The results were visualized by sodium dodecyl sulfate-polyacrylamide gel electrophoresis and Coomassie blue staining. The optimum concentration of arabinose required for the maximum expression of the cloned LEE integrase was found to be 0.2%.
Site-specific recombination following integrase induction.
To evaluate the role of the LEE O26 integrase on DR-mediated recombination, plasmid pMMG-8 was transformed into E. coli INVαF′ to generate the strain E1294. Competent cells of this strain were then prepared after arabinose induction to ensure optimum intracellular expression levels of the LEE integrase. To assess the frequency of site-specific integration between plasmid-located DR sites, E1294 cells were simultaneously cotransformed with precisely 200 ng of plasmid pMMG-7 and 200 ng of pMMG-4, pMMG-5, pMMG-12, pMMG-15, or pMMG-16. The transformation mix was grown as two replicate cultures of 50 ml. After 20 min, arabinose (0.2%) was added to one of these cultures, and both were then incubated for an additional 4 h before they were plated onto LB containing ampicillin (Ap) and kanamycin (Km). The frequency of site-specific chromosomal integration was assessed essentially as described above, except that the transformations were performed only with the pGP704 derivative plasmids (pMMG-4, pMMG-5, pMMG-12, pMMG-15, and pMMG-16). For each experiment, the total number of clones obtained was enumerated, and the results obtained with or without integrase were compared. Five clones from each experiment were tested by PCR to confirm the formation of the expected cointegrate. We note that since ampicillin resistance is encoded by both the pGP704 derivative plasmids and pMMG-8, it was not possible to distinguish the recombinants by antibiotic selection alone. However, the frequency of homologous recombination and chromosomal insertion was determined by comparing the number of colonies obtained with or without arabinose induction and by comparing the colony numbers to controls transformed with only plasmid pMMG-8.
Construction of mutants.
Strains used in this assay were made recA56 by P1 transduction. P1 transductions (42) were carried out as previously described. Defined integrase (b4271) and integrase (b4271)-recA deletion mutants of E. coli K-12 strain MG1655 were constructed by using the temperature-sensitive gene replacement system as previously described (37). To remove the integrase locus, we initially PCR amplified the 5′ and 3′ ends of the integrase gene by using the primer combinations ΔInt1-ΔInt2 and ΔInt3-ΔInt4. These amplification products were cloned into the temperature-sensitive plasmid pTSC29, which contains an origin of replication that permits it to replicate only at temperatures ≤30°C. The resultant suicide plasmid was transformed by electroporation into MG1655, and the recombinant strain was grown overnight at 30°C on 20 μg ml−1 cloramphenicol plates. After restreaking at 30°C for 2 days, single colonies were streaked out on 20 μg ml−1 chloramphenicol plates and placed at 42°C to select for single crossover events. Chloramphenicol-resistant colonies were then grown at 42°C in liquid LB medium containing 20 μg of chloramphenicol ml−1. Overnight cultures were diluted 1:1,000 in liquid LB medium without antibiotics, grown to an optical density at 600 nm of 0.5, and then plated out on LB plates. Colonies from these plates were replica plated on 20 μg ml−1 chloramphenicol plates and LB plates. Chloramphenicol-sensitive colonies were tested for the deletion of the integrase locus by PCR. A colony that had a deletion in the integrase locus was selected and named MG1655Δb4271. A double mutant was constructed by introducing the deletion into the MG1655recA56 srl300::Tn10 strain, thus yielding MG1655Δb4271 recA56 srl300::Tn10.
Chromosomal insertion in the recA and integrase mutants.
To evaluate the role of the integrase and recA genes in site-specific chromosomal integration, the three strains (MG1655 and its isogenic derivatives Δb4271 and the Δb4271 recA56 srl300::Tn10 double mutant) described above were transformed with 200 ng of plasmid pMMG-12 or pMMG-16. E. coli strain MG1655 was also included as a control. To account for differences in transformation efficiency between the four strains, an ampicillin-resistant pBluescript II derivative plasmid (pWH36) was also transformed into each of the strains. This provided a reference for each transformation. The chromosomal integration frequency for plasmids pMMG-12 and pMMG-16 was then calculated for each strain by counting the number of ampicillin-resistant colonies and expressing this number as a percentage of colonies grown with respect to the number of transformants obtained with plasmid pWH36.
RESULTS
Generation of mini-LEE O26 substrates.
In order to evaluate the mobility of the O26 LEE O26 PAI, we generated smaller derivates comprising the primary elements required for homologous recombination, i.e., the integrase and the DR1/DR2 direct repeats. The LEE O26-DR1 region was cloned in the suicide plasmid pGP704 to generate plasmid pMMG-4 (see Table 1 for a description of recombinant plasmids). This plasmid carries a kanamycin resistance gene marker and is unable to replicate in an INVαF′ recA56 strain (21). The LEE O26-DR2 and integrase were cloned in the Apr plasmid pWSK129 to generate plasmid pMMG-7. We hypothesized that the cotransformation of both plasmids should allow homologous recombination between DR1 and DR2 mediated by the integrase and thus result in the formation of a cointegrate structure (Fig. 1) in the transformed E. coli strain.
Plasmids pMMG-4 and pMMG-7 were cotransformed into E. coli strain INVαF′ and transformant colonies resistant to both ampicillin and kanamycin were selected. Plasmids were isolated from individual transformant clones and analyzed by PCR. The results showed a 1.76-kb amplification product using the primers ML1up and ML2lp and a 1.34-kb amplification product using the primers ML2up and ML1lp, both of which correspond to the specific fragments of a cointegrate structure (Fig. 1 and 4). DNA sequence analysis confirmed that the PCR products were indeed the result of cointegrate structures formed by homologous recombination between DR1 and DR2. We note that we were never able to generate a single plasmid harboring DR1-DR2-integrase and attribute this to integrase-mediated spontaneous homologous recombination at the DR1 and DR2 sites.
FIG. 4.
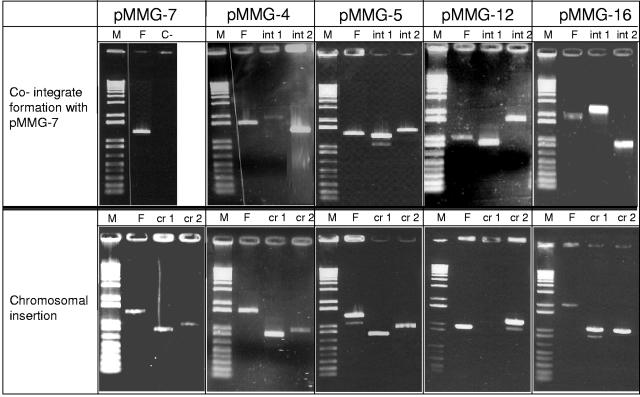
PCR amplimers obtained to confirm cointegrate formation obtained either with the respective recombinant plasmid or after insertion into the chromosome. In the upper panel, the PCR amplimers obtained confirm the cointegrate formation with plasmid pMMG-7. Lanes: M, molecular weight marker; F, original fragment cloned in the respective vector; int1 and int2 correspond to the two segments formed in the derivative plasmids after homologous recombination; C−, negative control. In the lower panel, the PCR amplimers obtained to confirm insertion in the K-12 chromosome are depicted. Lanes: M and F are as described for the upper panel; cr1 and cr2 correspond to the two amplimers obtained with the primers PheUup and PheUlp, together with the respective primer set used to verify the formation of the chromosomal integration structure. Molecular weight marker sizes (in kilobases): 0.1, 0.2, 0.3, 0.4, 0.5, 0.65, 0.85, 1.0, 1.65, 2.0, 3.0, 4.0, 5.0, etc., up to 12.
Cointegrate formation occurs between plasmid substrates containing the DR elements.
To determine the generation of cointegrates in a more quantitative manner, we constructed a series of plasmids containing different sectors of the LEE PAI ends (Fig. 3). Each of these plasmids (pMMG-4, pMMG-5, pMMG-12, pMMG-15, and pMMG-16) was then cotransformed together with plasmid pMMG-7 (LEE O26-DR2 and integrase) into E. coli strain INVαF′. This enabled us to evaluate the frequency of homologous recombination between sites, the results of which are summarized in Table 3. We observed that recombination was obtained when one direct repeat was present (pMMG-4, pMMG-5, pMMG-12, and pMMG-16), whereas no recombination occurred when it was absent (pMMG-15 or control pGP704).
TABLE 3.
Frequency of recombination events for the different plasmids used in this study
| Recombinant event | Avg recombination frequency (no. of doubly resistant colonies)a
|
|||||
|---|---|---|---|---|---|---|
| pMMG-4 | pMMG-5 | pMMG-12 | pMMG-15 | pMMG-16 | pGP704 | |
| Cointegrate formation with pMMG-7 | 56.8 | 121.5 | 31.6 | 0 | 178.2 | 0 |
| Chromosomal insertion | 56.2 | 75.8 | 41.4 | 0 | 86.5 | 0 |
That is, the number of doubly resistant colonies (to kanamycin and ampicillin) after transformation of 200 ng of each plasmid. The data represent averages from three independent experiments.
Homologous recombination with the LEE DRs can occur at both plasmid and chromosomal sites.
To investigate the location of cointegrate formation in more detail, we isolated 20 ampicillin-kanamycin-resistant colonies originating from a single cotransformation experiment with the plasmids pMMG-4 and pMMG-7. Plasmid DNA was isolated from each of the clones and examined by restriction endonuclease digestion and PCR. Among these clones, 14 contained plasmids with the expected recombined fragments described for the cointegrate structure. However, six of the clones contained only plasmid pMMG-7. Since plasmid pMMG-4 is not able to replicate in an INVαF′ strain (21), we hypothesized that the observed resistance to ampicillin was due to chromosomal insertion of the plasmid into the E. coli chromosome (Fig. 2). Subsequent experiments indicated that for each cointegration experiment with plasmids pMMG-4 and pMMG-7 chromosomal insertion occurred at a frequency of approximately one in four clones.
Detection and characterization of chromosomal insertion.
To examine this chromosomal insertion event in more detail, we transformed our plasmid derivatives containing various regions of LEE O26 (pMMG-4, pMMG-5, pMMG-12, pMMG-15, and pMMG-16) into INVαF′ (see Fig. 2, for example). The results of the transformation are summarized in Table 3. The role of the DR sequences in mediating chromosomal insertion was clear since no transformants were obtained from plasmids lacking these sequences (i.e., plasmids pMMG-15 and the vector control plasmid pGP704). Ampicillin-resistant transformants indicative of site-specific homologous recombination and chromosomal insertion were obtained with the plasmids pMMG-4, pMMG-5, pMMG-12, and pMMG-16. In the case of plasmid pMMG-16 this was expected since it contains DR2 and the complete integrase gene. Plasmids pMMG-4 (DR1 alone) and pMMG-12 (DR2 alone) do not contain the LEE integrase. The identification of chromosomal insertion events upon transformation with these plasmids is indicative of an additional chromosomally encoded recombinase that can mediate this process. Site-specific integration was also observed with plasmid pMMG-5, which contains DR2 and a truncated version of the integrase gene. Our data suggest that the product of this truncated gene possesses partial activity, since the frequency of recombination for this plasmid was higher than that obtained in the absence of the LEE integrase but lower than that for pMMG-16, which contains the complete LEE integrase gene.
The 137-bp LEE-DR1 sequence is present as a single copy on the E. coli K-12 chromosome (3). An analysis of clones containing chromosomal insertions by PCR (Fig. 4) and sequencing revealed that all were integrated at this site in the pheU tRNA locus. This included integrates derived from the LEE O26 integrase as well as in its absence. Taken together, our data demonstrate that plasmid-derived LEE-DR insertion occurs via homologous recombination with an identical DR found in the K-12 chromosome. Furthermore, this site-specific insertion event is catalyzed at high frequency by the LEE integrase but can also occur at lower frequencies in its absence.
Integrase activity.
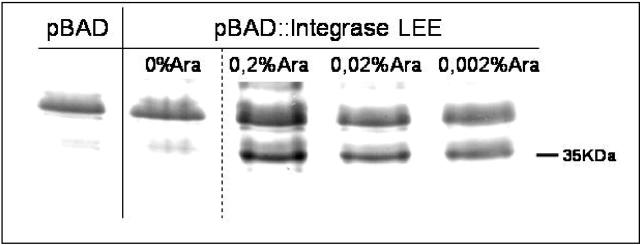
Since the LEE O26 integrase appeared to play an active role in the recombination process, we examined the influence of higher levels of the protein in the system. To perform this, the integrase gene was cloned behind the arabinose inducible Para promoter (plasmid pMMG-8). Optimal expression of a 35-kDa integrase protein was achieved with a 0.2% final concentration of arabinose (Fig. 5). Thus, this was the concentration used to induce the production of integrase for all remaining experiments.
FIG. 5.
Coomassie brilliant blue-stained sodium dodecyl sulfate-polyacrylamide gel of cell lysates of E. coli strain TOP10 harboring pBAD/Myc-HisC (pBAD) or pMMG-8 (pBAD::integrase LEE). The strain harboring pMMG-8 was grown without arabinose induction or with 0.2, 0.02, or 0.002% arabinose. The position of the 35-kDa LEE integrase protein is indicated. Optimal expression of the LEE integrase from pMMG-8 was observed after 1 h induction with 0.2% arabinose.
To observe the effect of increased amounts of LEE integrase on site-specific integration at both plasmid and chromosomal sites, the following approach was used. First, pMMG-8 was transformed into E. coli TOP10 to generate strain E1294. Competent cells of this strain were then prepared after 0.2% arabinose induction to ensure optimal integrase production. These cells were then transformed with our series of plasmid derivatives containing various regions of LEE O26 (with or without pMMG-7). This method contains an additional level of complexity since ampicillin resistance is encoded by both the pGP704 derivative plasmids and pMMG-8. Thus, it is not possible to distinguish the recombinants by antibiotic selection alone. However, the frequency of homologous recombination and chromosomal insertion could be determined by comparing the number of colonies obtained with or without arabinose induction and by comparing colony numbers to controls transformed with only plasmid pMMG-8.
The results are summarized in Table 4. Plasmids pMMG-4 (DR1 alone), pMMG-12 (DR2 alone), and pMMG-5 (DR2 plus truncated integrase gene) displayed a higher level of homologous recombination when integrase expression was induced in the recipient cells. This effect could be observed for both the chromosomal insertion process and the generation of plasmid cointegrates. When plasmid pMMG-16 (DR2 plus complete integrase gene) was assessed for recombination efficiency, no increase was observed after arabinose induction of the recipient cells. This suggests that recombination mediated by the LEE integrase is already highly efficacious when the integrase is expressed from its endogenous promoter. As observed previously, the plasmid derivative lacking the direct repeat sequence (pMMG-15) was unable to mediate either chromosomal integration or the formation of plasmid cointegrates. Thus, the numbers of resistant bacteria after transformation were essentially the same as the background level obtained with the control plasmids pBAD/Myc-HisC and pGP704. Taken together, the data demonstrate that overexpression of the LEE O26 integrase increases the frequency of the recombination between DR sites. Furthermore, as observed previously, site-specific recombination can occur in the absence of the LEE integrase, indicating the presence of an additional factor(s) intrinsic to E. coli K-12 MG1655 that mediates this process. However, irrespective of the source of integrase, homologous recombination occurs only when the specific direct repeat is present in both the donor and the recipient, indicating that these integrases have strong affinities for the same DR sequences.
TABLE 4.
Increase of recombinant events after arabinose induction for the different plasmids used in this study
| Recombination event | Avg recombination frequency (no. of doubly resistant colonies)a
|
|||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| pBAD vector onlyb
|
pGP704 derivativec
|
|||||||||||||
| pMMG-4
|
pMMG-5
|
pMMG-12
|
pMMG-15
|
pMMG-16
|
pGP704
|
|||||||||
| − | + | − | + | − | + | − | + | − | + | − | + | − | + | |
| Cointegrate formation with pMMG-7 | 8.3 | 9.7 | 30.4 | 85.7 | 39.5 | 96.0 | 64.0 | 221.2 | 7.7 | 7.9 | 87.5 | 110.5 | 8.7 | 8.3 |
| Chromosomal insertion | 10.1 | 11.8 | 216.2 | 593.0 | 224.2 | 693.7 | 237.0 | 421.5 | 11.1 | 12.3 | 112.2 | 119.5 | 9.8 | 11.0 |
That is, the number of doubly resistant colonies (to kanamycin and ampicillin) after transformation of 200 ng of each plasmid. Values are averages from three independent experiments.
That is, strain E1294 harboring pBAD vector. +, Induction with 0.2% arabinose; −, no induction with arabinose.
That is, strain E1294 harboring the relevant indicated plasmid. +, induction with 0.2% arabinose; −, no induction with arabinose.
Chromosomal insertion in an MG1655Δb4271 recA56 mutant.
Two main candidate genes were identified from the genome sequence of E. coli K-12 that could contribute to site-specific recombination of the DR-containing plasmids. A P4-like integrase with strong overall homology to the LEE O26 integrase is located adjacent to the leuX tRNA locus at 97 min on the E. coli K-12 MG1655 chromosome (b4271 [3]). The recA gene encodes the major bacterial protein involved in homologous recombination, DNA repair, the SOS response, and prophage induction (39). In order to assess the contribution of these two proteins on site-specific recombination of the DR-containing plasmids, a series of mutants were constructed. In the first instance, mutant strains of MG1655 were created by mutation of the respective genes individually (i.e., MG1655ΔrecA56 and MG1655Δb4271). Finally, a double MG1655Δb4271 recA56 mutant was also constructed. These strains were then transformed with either plasmid pMMG-12 (DR2 alone) or pMMG-16 (DR2 plus intact LEE integrase). The results are presented in Table 5. A reduction in the number of viable colonies corresponding to reduced chromosomal insertion of the respective plasmids was observed in all three mutants. As expected, the chromosomal integration frequencies were higher for plasmid pMMG16, which harbors DR2 and an intact copy of the LEE integrase. From the data it appears that the P4-like integrase (b4271) may be more efficient at promoting site specific recombination between the DR elements. Surprisingly, chromosomal integration was not completely prevented in the MG1655Δb4271 recA56 double mutant. Background recombination frequencies of ca. 20% were obtained with both plasmids and suggest that an additional unknown element(s) may contribute to this phenomenon. Integrase proteins with related sequences and presumed common ancestry can recognize diverse core sites (6, 14). The E. coli K-12 MG1655 genome contains several P4-related integrases, such as the CP4 and SF6 integrases (12), that may account for the residual recombination activity observed in the MG1655Δb4271 recA56 double mutant. However, the data indicate that both the P4-like integrase (b4271) and the RecA protein play a role in site-specific recombination of the DR-containing plasmids.
TABLE 5.
Chromosomal insertion of plasmids pMMG-12 and pMMG-16 in the different mutants used in this study
| Plasmid | Strain | % Integrationa |
|---|---|---|
| pMMG-12 | Wild type | 43.3 |
| recA56 | 24.8 | |
| Δb4271 | 17.2 | |
| recA56 Δb4271 | 23.9 | |
| pMMG-16 | Wild type | 88.4 |
| recA56 | 43.3 | |
| Δb4271 | 20.7 | |
| recA56 Δb4271 | 31.3 |
That is, the percentage of chromosomal integration calculated with each strain using, as a reference of growth on LB medium plus ampicillin, the same strain transformed with plasmid pWH36 (Apr). Values represent an average of three independent experiments.
DISCUSSION
In this study we have shown that the LEE O26 integrase is highly active and uses the DR1/DR2 sequences to mediate site-specific recombination. The positioning of the element between two short direct repeats is characteristic of mobile elements, including temperate bacteriophages (7). The bacteriophage P4 integrase gene is also a feature that is characteristic of many other PAIs (20, 32) and has been suggested as a tool of insertion and excision of chromosomal sequences (1). A prerequisite for site-specific recombination is the presence of a target DNA sequence specifically recognized by the recombinase. In the PAIs of several bacterial species, direct repeats are present at each extremity; this is the case for Helicobacter pylori (8), Vibrio cholerae (24), and Staphylococcus aureus (28), among others.
The sequencing of several E. coli genomes has revealed the widespread occurrence of multiple integrases with similar specificities. We present evidence here to demonstrate that these integrases provide an additional level for dissemination and spread of PAIs. Several insertion loci have been described for the LEE PAIs and include selC and pheU and, more recently, pheV (23, 45). Adding a further level of complexity to this is the observation that the same insertion locus can sometimes be altered by the insertion of additional foreign DNA (2). The pheU locus of the E. coli serotype O6 strain J96 has been the site of insertion of a different PAI (designated PAI V), which harbors the hlyCABD gene cluster as well as other virulence genes. A natural hemolysis-negative variant has been described for strain J96, which has resulted from the excision of the complete PAI V locus (44). It is of great interest to note that the regions comprising the point of insertion, the DR1/DR2, and the integrase genes are identical between J96 and 413/89-1 (44; unpublished data), suggesting that different genetic loads have been introduced onto naturally occurring mobile elements whose basic composition may not vary significantly from that of the mini-LEE O26 elements generated in the present study.
Differences in the LEE region have been reported among E. coli strain types (9, 17, 23, 45). A cloned LEE element from EPEC is able to confer the A/E phenotype and Esp secretion upon E. coli K-12 (30), although the LEE from O157 is unable to fulfill this function (10). These studies and several others (11, 43) which further confirm genetic differences between EPEC and EHEC LEE suggest an evolutive development of pathogenic E. coli by insertion of foreign genetic elements from different origins.
The formation of cointegrates between DR elements has also been described for the B13 integrase of Pseudomonas. Plasmids containing attB1 and attP plus int-B13 gave rise to stable cointegrates when cotransformed into E. coli DH5α (38). Our results are in accordance with these findings, since an intact integrase gene was required for precise recombination. Ravatn et al. (38) also reported a lower but significant recombination when the integrase was absent but did not provide a plausible explanation for this. We have now shown that several related proteins encoded by recombinase or integrase genes present on the E. coli MG1655 K-12 genome may perform this process. This background level of recombination and/or insertion potential could be important for the acquisition of foreign DNA elements by bacteria and most likely also accounts for the low frequency recombination previously observed in Pseudomonas in the absence of specific integrase (38).
The LEE O26 PAI occurs more widely in STEC bacteria than the previously described LEE PAIs in O127:H− EPEC and O157:H7 STEC, suggesting that it has been repeatedly introduced into many clonal types by horizontal transmission. A feature of LEE O26 is also its apparent stability, i.e., it is not lost by spontaneous deletion from the chromosome (unpublished observations), suggesting that either the integrase or the flanking DR sequences harbor mutations rendering them inactive. However, the results of the analysis presented here provide strong evidence that LEE O26 is located on a mobile genetic element. This also indicates that this LEE PAI may have been acquired from a bacteriophage via horizontal transfer since the integrase has a phage origin. To support this hypothesis, one should assume the existence of a large phage able to incorporate the complete LEE region. Other authors have suggested that a normal P4 prophage may have initially integrated into the pheU tRNA locus and created a suitable environment for the integration of other mobile elements (41, 45). In agreement with this hypothesis are several studies showing the association between PAI O122 and the LEE region, which appear to be contiguous in some strains (31). During the evolution of some PAI elements, it also appears that deletions within key sectors required for prophage lysis have resulted in some sequences being stably incorporated into the chromosome. Our results demonstrate that although a complete prophage is not essential for the integration of mobile elements, the presence of the direct repeats is crucial for the chromosomal integration (and excision). LEE sequences that have lost one or both direct repeats would therefore no longer be mobile.
It is interesting to reflect that neither of the previously described LEE PAIs inserted at the selC locus harbor direct repeats and an integrase-like gene has only been detected within the LEE locus of the O157:H7 EDL933 strain. Furthermore, all of these LEE loci have been described as nonmobile. In the present study we have demonstrated two important facets of PAI mobility: (i) the flanking direct repeats are instrumental for horizontal transmission of PAI elements and (ii) integrases with similar specificities may function in trans to promote the insertion of foreign DNA segments. Finally, this implies that the LEE PAIs located at selC may represent an evolutionary dead end and that other virulence properties have been independently acquired. Further studies will be required to elucidate the molecular and epidemiological differences in these strains to explain their occurrence and virulence potential.
Acknowledgments
This study was supported by the Forschungsgemeinschaft (SFB 535 TPA6) and the Australian National Health and Medical Research Council (301163). M.M. was a recipient of a grant from the Alexander von Humboldt Foundation.
Editor: V. J. DiRita
REFERENCES
- 1.Al Hasani, K., K. Rajakumar, D. Bulach, R. Robins-Browne, B. Adler, and H. Sakellaris. 2001. Genetic organization of the she pathogenicity island in Shigella flexneri 2a. Microb. Pathog. 30:1-8. [DOI] [PubMed] [Google Scholar]
- 2.Bertin, Y., K. Boukhors, V. Livrelli, and C. Martin. 2004. Localization of the insertion site and pathotype determination of the locus of enterocyte effacement of Shiga toxin-producing Escherichia coli strains. Appl. Environ. Microbiol. 70:61-68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Blattner, F. R., G. Plunkett III, C. A. Bloch, N. T. Perna, V. Burland, M. Riley, J. Collado-Vides, J. D. Glasner, C. K. Rode, G. F. Mayhew, J. Gregor, N. W. Davis, H. A. Kirkpatrick, M. A. Goeden, D. J. Rose, B. Mau, and Y. Shao. 1997. The complete genome sequence of Escherichia coli K-12. Science 277:1453-1474. [DOI] [PubMed] [Google Scholar]
- 4.Boyce, T. G., D. L. Swerdlow, and P. M. Griffin. 1995. Escherichia coli O157:H7 and the hemolytic-uremic syndrome. N. Engl. J. Med. 333:364-368. [DOI] [PubMed] [Google Scholar]
- 5.Brunder, B., H. Schmidt, and H. Karch. 1996. KatP, a novel catalase-peroxidase encoded by the large plasmid of enterhemorrhagic Escherichia coli O157:H7. Microbiology 142:3305-3315. [DOI] [PubMed] [Google Scholar]
- 6.Campbell, A. M. 1992. Chromosomal insertion sites for phages and plasmids. J. Bacteriol. 174:7495-7499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Campbell, A. 1994. Comparative molecular biology of lambdoid phages. Annu. Rev. Microbiol. 48:193-222. [DOI] [PubMed] [Google Scholar]
- 8.Censini, S., C. Lange, Z. Xiang, J. E. Crabtree, P. Ghiara, M. Borodovsky, R. Rappuoli, and A. Covacci. 1996. cag, a pathogenicity island of Helicobacter pylori, encodes type I-specific and disease-associated virulence factors. Proc. Natl. Acad. Sci. USA 93:14648-14653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Elliott, S. J., L. A. Wainwright, T. K. McDaniel, K. G. Jarvis, Y. K. Deng, L. C. Lai, B. P. McNamara, M. S. Donnenberg, and J. B. Kaper. 1998. The complete sequence of the locus of enterocyte effacement (LEE) from enteropathogenic Escherichia coli E2348/69. Mol. Microbiol. 28:1-4. [DOI] [PubMed] [Google Scholar]
- 10.Elliott, S. J., J. Yu, and J. B. Kaper. 1999. The cloned locus of enterocyte effacement from enterohemorrhagic Escherichia coli O157:H7 is unable to confer the attaching and effacing phenotype upon E. coli K-12. Infect. Immun. 67:4260-4263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Elliott, S. J., S. W. Hutcheson, M. S. Dubois, J. L. Mellies, L. A. Wainwright, M. Batchelor, G. Frankel, S. Knutton, and J. B. Kaper. 1999. Identification of CesT, a chaperone for the type III secretion of Tir in enteropathogenic Escherichia coli. Mol. Microbiol. 33:1176-1189. [DOI] [PubMed] [Google Scholar]
- 12.Esposito, D., and J. Scocca. 1997. The integrase family of tyrosine recombinases: evolution of a conserved active site domain. Nucleic Acids Res. 18:3605-3614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Frankel, G., A. D. Phillips, I. Rosenshine, G. Dougan, J. B. Kaper, and S. Knutton. 1998. Enteropathogenic and enterohemorrhagic E. coli: more subversive elements. Mol. Microbiol. 30:911-921. [DOI] [PubMed] [Google Scholar]
- 14.Grainge, I., and M. Jayaram. 1999. The integrase family of recombinases: organization and function of the active site. Mol. Microbiol. 33:449-456. [DOI] [PubMed] [Google Scholar]
- 15.Griffin, P. M., and R. V. Tauxe. 1991. The epidemiology of infections caused by Escherichia coli O157:H7, other enterohemorrhagic E. coli, and the associated hemolytic-uremic syndrome. Epidemiol. Rev. 13:60-98. [DOI] [PubMed] [Google Scholar]
- 16.Groth, A. C., and M. P. Calos. 2004. Phage integrases: biology and applications. J. Mol. Biol. 335:667-678. [DOI] [PubMed] [Google Scholar]
- 17.Hacker, J., G. Blum-Oehler, I. Muhldorfer, and H. Tschape. 1997. Pathogenicity islands of virulent bacteria: structure, function, and impact on microbial evolution. Mol. Microbiol. 23:1089-1097. [DOI] [PubMed] [Google Scholar]
- 18.Hacker, J., and J. B. Kaper. 2000. Pathogenicity islands and the evolution of microbes. Annu. Rev. Microbiol. 54:641-679. [DOI] [PubMed] [Google Scholar]
- 19.Hallet, B., and D. J. Sherrat. 1997. Transposition and site-specific recombination: adapting DNA cut-and-paste mechanisms to a variety of genetic rearrangements. FEMS Microbiol. Rev. 21:157-178. [DOI] [PubMed] [Google Scholar]
- 20.Hare, J. M., A. K. Wagner, and K. A. McDonough. 1999. Independent acquisition and insertion into different chromosomal locations of the same pathogenicity island in Yersinia pestis and Yersinia pseudotuberculosis. Mol. Microbiol. 31:291-303. [DOI] [PubMed] [Google Scholar]
- 21.Herrero, M., V. de Lorenzo, and K. N. Timmis. 1990. Transposon vectors containing non-antibiotic resistance selection markers for cloning and stable chromosomal insertion of foreign genes in gram-negative bacteria. J. Bacteriol. 172:6557-6567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hofmann, M. A., and D. A. Brian. 1991. Sequencing PCR DNA amplified directly from a bacterial colony. BioTechniques 11:30-31. [PubMed] [Google Scholar]
- 23.Jores, J., L. Rumer, S. Kiessling, J. B. Kaper, and L. H. Wieler. 2001. A novel locus of enterocyte effacement (LEE) pathogenicity island inserted at pheV in bovine Shiga toxin-producing Escherichia coli strain O103:H2. FEMS Microbiol. Lett. 204:75-79. [DOI] [PubMed] [Google Scholar]
- 24.Karaolis, D. K., J. A. Johnson, C. C. Bailey, E. C. Boedeker, J. B. Kaper, and P. R. Reeves. 1998. A Vibrio cholerae pathogenicity island associated with epidemic and pandemic strains. Proc. Natl. Acad. Sci. USA 95:3134-3139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kresse, A. U., K. Schulze, C. Deibel, F. Ebel, M. Rohde, T. Chakraborty, and C. A. Guzman. 1998. Pas, a novel protein required for protein secretion and attaching and effacing activities of enterohemorrhagic Escherichia coli. J. Bacteriol. 180:4370-4379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Landy, A. 1999. Coming or going it's another pretty picture for the lambda-Int family album. Proc. Natl. Acad. Sci. USA 96:7122-7124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lesic, B., S. Bach, J. M. Ghigo, U. Dobrindt, J. Hacker, and E. Carniel. 2004. Excision of the high-pathogenicity island of Yersinia pseudotuberculosis requires the combined actions of its cognate integrase and Hef, a new recombination directionality factor. Mol. Microbiol. 52:1337-1348. [DOI] [PubMed] [Google Scholar]
- 28.Lindsay, J. A., A. Ruzin, H. F. Ross, N. Kurepina, and R. P. Novick. 1998. The gene for toxic shock toxin is carried by a family of mobile pathogenicity islands in Staphylococcus aureus. Mol. Microbiol. 29:527-543. [DOI] [PubMed] [Google Scholar]
- 29.McDaniel, T. K., K. G. Jarvis, M. S. Donnenberg, and J. B. Kaper. 1995. A genetic locus of enterocyte effacement conserved among diverse enterobacterial pathogens. Proc. Natl. Acad. Sci. USA 92:1664-1668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.McDaniel, T. K., and J. B. Kaper. 1997. A cloned pathogenicity island from enteropathogenic Escherichia coli confers the attaching and effacing phenotype on E. coli K-12. Mol. Microbiol. 23:399-407. [DOI] [PubMed] [Google Scholar]
- 31.Morabito, S., R. Tozzoli, E. Oswald, and A. Caprioli. 2003. A mosaic pathogenicity island made up of the locus of enterocyte effacement and a pathogenicity island of Escherichia coli O157:H7 is frequently present in attaching and effacing E. coli. Infect. Immun. 71:3343-3348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Moss, J. E., T. J. Cardozo, A. Zychlinsky, and E. A. Groisman. 1999. The selC-associated SHI-2 pathogenicity island of Shigella fexneri. Mol. Microbiol. 33:74-83. [DOI] [PubMed] [Google Scholar]
- 33.Nataro, J. P., and J. B. Kaper. 1998. Diarrheagenic Escherichia coli. Clin. Microbiol. Rev. 11:142-201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.O'Brien, A. D., J. W. Newland, S. F. Miller, R. K. Holmes, H. W. Smith, and S. B. Formal. 1984. Shiga-like toxin-converting phages from Escherichia coli strains that cause hemorrhagic colitis or infantile diarrhea. Science 226:694-696. [DOI] [PubMed] [Google Scholar]
- 35.O'Callaghan, D., and A. Charbit. 1990. High efficiency transformation of Salmonella typhimurium and Salmonella typhi by electroporation. Mol. Gen. Genet. 223:156-158. [DOI] [PubMed] [Google Scholar]
- 36.Perna, N. T., G. F. Mayhew, G. Posfai, S. J. Elliot, M. S. Donnenberg, J. B. Kaper, and F. R. Blattner. 1998. Molecular evolution of a pathogenicity island from enterohemorrhagic Escherichia coli O157:H7. Infect. Immun. 66:3810-3817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Phillips, G. J. 1999. New cloning vectors with temperature-sensitive replication. Plasmid 41:78-81. [DOI] [PubMed] [Google Scholar]
- 38.Ravatn, R., S. Studer, A. J. B. Zehnder, and J. R. Van der Meer. 1998. Int-B13, an unusual site-specific recombinase of the bacteriophage P4 integrase family, is responsible for chromosomal insertion of the 105-kilobase clc element of Pseudomonas sp. strain B13. J. Bacteriol. 180:5505-5514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Roca, A. I., and M. M. Cox. 1997. RecA protein: structure, function, and role in recombinational DNA repair. Prog. Nucleic Acids Res. Mol. Biol. 56:129-223. [DOI] [PubMed] [Google Scholar]
- 40.Sambrook, J., E. F. Fritsch, and T. Maniatis. 1989. Molecular cloning: a laboratory manual, 2nd ed. Cold Spring Harbor Laboratory, Cold Spring Harbor, N.Y.
- 41.Sandner, L., L. E. Eguiarte, A. Navarro, A. Cravioto, and V. Souza. 2001. The elements of the locus of enterocyte effacement in human and wild mammal isolates of Escherichia coli: evolution by assemblage or disruption? Microbiology 147:3149-3158. [DOI] [PubMed] [Google Scholar]
- 42.Silhavy, T., M. Berman, and L. Enquist. 1984. Experiments with gene fusions. Cold Spring Harbor Laboratory, Cold Spring Harbor, N.Y.
- 43.Sperandio, V., J. L. Mellies, W. Nguyen, S. Shin, and J. B. Kaper. 1999. Quorum sensing controls expression of the type III secretion gene transcription and protein secretion in enterohemorrhagic and enteropathogenic Escherichia coli. Proc. Natl. Acad. Sci. USA 96:15196-15201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Swenson, D. L., N. O. Bukanov, D. E. Berg, and R. A. Welch. 1996. Two pathogenicity islands in uropathogenic Escherichia coli J96: cosmid cloning and sample sequencing. Infect. Immun. 64:3736-3743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Tauschek, M., R. A. Strugnell, and R. M. Robins-Browne. 2002. Characterization and evidence of mobilization of the LEE pathogenicity island of rabbit-specific strains of enteropathogenic Escherichia coli. Mol. Microbiol. 44:1533-1550. [DOI] [PubMed] [Google Scholar]
- 46.Wang, R., and S. R. Kushner. 1991. Construction of versatile low-copy-number vectors for cloning, sequencing, and gene expression in Escherichia coli. Gene 100:195-199. [PubMed] [Google Scholar]
- 47.Wieler, L. H., M. Tigges, F. Ebel, S. Schäferkordt, S. Djafari, T. Schlapp, G. Baljer, and T. Chakraborty. 1996. The enterohemolysin phenotype of Shiga-like toxin-producing Escherichia coli (SLTEC) is encoded by the EHEC-hemolysin gene. Vet. Microbiol. 52:153-164. [DOI] [PubMed] [Google Scholar]
- 48.Yarwood, J. M., J. K. McCormick, M. L. Paustian, P. M. Orwin, V. Kapur, and P. M. Schlievert. 2002. Characterization and expression analysis of Staphylococcus aureus pathogenicity island III: implications for the evolution of staphylococcal pathogenicity islands. J. Biol. Chem. 277:13138-13147. [DOI] [PubMed] [Google Scholar]