Abstract
A critical aspect of the efforts to relate the results of short-term genetic toxicity tests with those from long-term rodent tests for carcinogens is the quality and consistency of the studies conducted by the National Toxicology Program. Analysis of the results in relationship to chemical structure has shown that mutagenic potential is a primary risk factor for carcinogen identification. Chemicals positive in the Salmonella assay-generally possess "structural alerts" for electrophilic interactions, are predominantly represented among chemicals producing trans-species carcinogenic effects in rodents, and among those identified as carcinogenic to humans. Current efforts are aimed at defining toxicological, structural, and mechanistic properties of nonmutagens that are carcinogenic in rodents.
Full text
PDF







Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ahrens LA, Aronson SH, Connolly PL, Gibbard BG, Murtagh MJ, Murtagh SJ, Terada S, White DH, Callas JL, Cutts D. Determination of the neutrino fluxes in the Brookhaven wide-band beams. Phys Rev D Part Fields. 1986 Jul 1;34(1):75–84. doi: 10.1103/physrevd.34.75. [DOI] [PubMed] [Google Scholar]
- Ames B. N., Durston W. E., Yamasaki E., Lee F. D. Carcinogens are mutagens: a simple test system combining liver homogenates for activation and bacteria for detection. Proc Natl Acad Sci U S A. 1973 Aug;70(8):2281–2285. doi: 10.1073/pnas.70.8.2281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ames B. N., Gold L. S. Too many rodent carcinogens: mitogenesis increases mutagenesis. Science. 1990 Aug 31;249(4972):970–971. doi: 10.1126/science.2136249. [DOI] [PubMed] [Google Scholar]
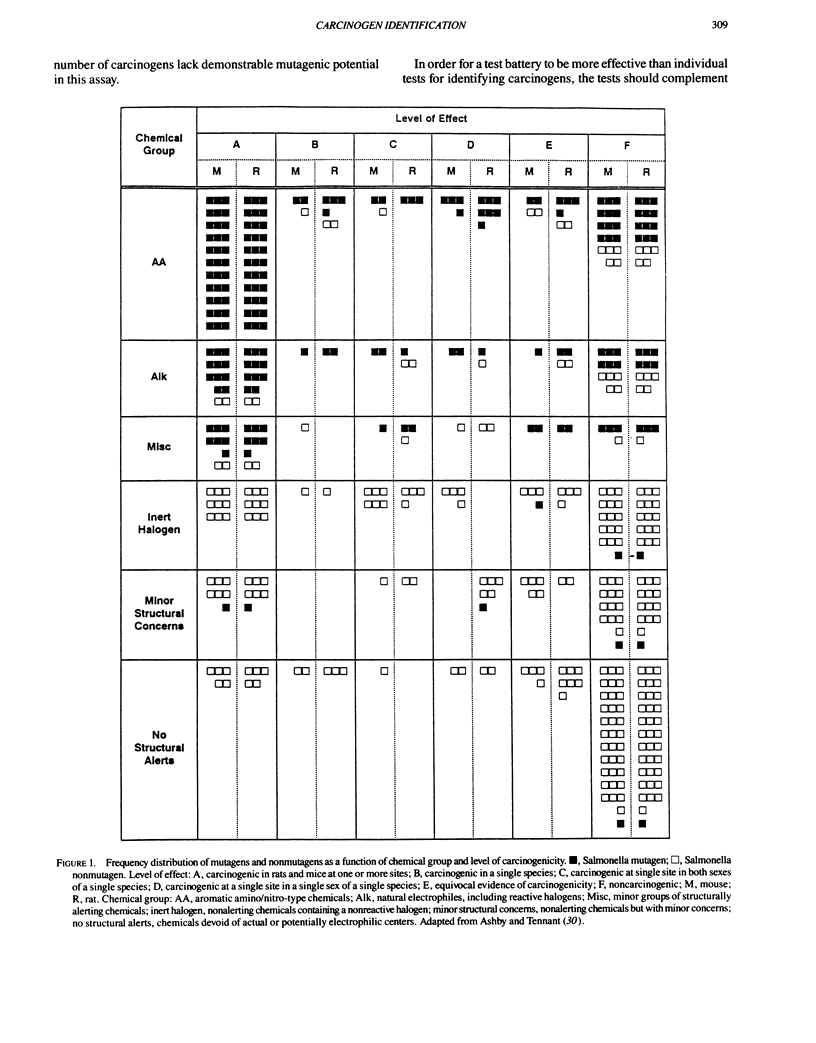
- Ashby J., Tennant R. W. Chemical structure, Salmonella mutagenicity and extent of carcinogenicity as indicators of genotoxic carcinogenesis among 222 chemicals tested in rodents by the U.S. NCI/NTP. Mutat Res. 1988 Jan;204(1):17–115. doi: 10.1016/0165-1218(88)90114-0. [DOI] [PubMed] [Google Scholar]
- Ashby J., Tennant R. W. Definitive relationships among chemical structure, carcinogenicity and mutagenicity for 301 chemicals tested by the U.S. NTP. Mutat Res. 1991 May;257(3):229–306. doi: 10.1016/0165-1110(91)90003-e. [DOI] [PubMed] [Google Scholar]
- Ashby J., Tennant R. W., Zeiger E., Stasiewicz S. Classification according to chemical structure, mutagenicity to Salmonella and level of carcinogenicity of a further 42 chemicals tested for carcinogenicity by the U.S. National Toxicology Program. Mutat Res. 1989 Jun;223(2):73–103. doi: 10.1016/0165-1218(89)90037-2. [DOI] [PubMed] [Google Scholar]
- Bishop J. M. Molecular themes in oncogenesis. Cell. 1991 Jan 25;64(2):235–248. doi: 10.1016/0092-8674(91)90636-d. [DOI] [PubMed] [Google Scholar]
- Caspary W. J., Daston D. S., Myhr B. C., Mitchell A. D., Rudd C. J., Lee P. S. Evaluation of the L5178Y mouse lymphoma cell mutagenesis assay: interlaboratory reproducibility and assessment. Environ Mol Mutagen. 1988;12 (Suppl 13):195–229. doi: 10.1002/em.2860120506. [DOI] [PubMed] [Google Scholar]
- Caspary W. J., Lee Y. J., Poulton S., Myhr B. C., Mitchell A. D., Rudd C. J. Evaluation of the L5178Y mouse lymphoma cell mutagenesis assay: quality-control guidelines and response categories. Environ Mol Mutagen. 1988;12 (Suppl 13):19–36. doi: 10.1002/em.2860120503. [DOI] [PubMed] [Google Scholar]
- Dunkel V. C., Zeiger E., Brusick D., McCoy E., McGregor D., Mortelmans K., Rosenkranz H. S., Simmon V. F. Reproducibility of microbial mutagenicity assays: II. Testing of carcinogens and noncarcinogens in Salmonella typhimurium and Escherichia coli. Environ Mutagen. 1985;7 (Suppl 5):1–248. doi: 10.1002/em.2860070902. [DOI] [PubMed] [Google Scholar]
- Farber E., Rubin H. Cellular adaptation in the origin and development of cancer. Cancer Res. 1991 Jun 1;51(11):2751–2761. [PubMed] [Google Scholar]
- Fearon E. R., Vogelstein B. A genetic model for colorectal tumorigenesis. Cell. 1990 Jun 1;61(5):759–767. doi: 10.1016/0092-8674(90)90186-i. [DOI] [PubMed] [Google Scholar]
- Galloway S. M., Bloom A. D., Resnick M., Margolin B. H., Nakamura F., Archer P., Zeiger E. Development of a standard protocol for in vitro cytogenetic testing with Chinese hamster ovary cells: comparison of results for 22 compounds in two laboratories. Environ Mutagen. 1985;7(1):1–51. doi: 10.1002/em.2860070102. [DOI] [PubMed] [Google Scholar]
- Gulati D. K., Wojciechowski J. P., Kaur P. Comparison of single-, double- or triple-exposure protocols for the rodent bone marrow/peripheral blood micronucleus assay using 4-aminobiphenyl and treosulphan. Mutat Res. 1990 Jun-Aug;234(3-4):135–139. doi: 10.1016/0165-1161(90)90005-9. [DOI] [PubMed] [Google Scholar]
- Haseman J. K., Clark A. M. Carcinogenicity results for 114 laboratory animal studies used to assess the predictivity of four in vitro genetic toxicity assays for rodent carcinogenicity. Environ Mol Mutagen. 1990;16 (Suppl 18):15–31. doi: 10.1002/em.2850160503. [DOI] [PubMed] [Google Scholar]
- Kier L. D., Brusick D. J., Auletta A. E., Von Halle E. S., Brown M. M., Simmon V. F., Dunkel V., McCann J., Mortelmans K., Kier L. E. The Salmonella typhimurium/mammalian microsomal assay. A report of the U.S. Environmental Protection Agency Gene-Tox Program. Mutat Res. 1986 Sep;168(2):69–240. doi: 10.1016/0165-1110(86)90002-3. [DOI] [PubMed] [Google Scholar]
- Luke C. A., Tice R. R. Effect of processing time on the quality of mouse bone-marrow metaphase preparations. Mutat Res. 1989 Sep;227(1):59–62. doi: 10.1016/0165-7992(89)90069-9. [DOI] [PubMed] [Google Scholar]
- MacGregor J. T., Wehr C. M., Henika P. R., Shelby M. D. The in vivo erythrocyte micronucleus test: measurement at steady state increases assay efficiency and permits integration with toxicity studies. Fundam Appl Toxicol. 1990 Apr;14(3):513–522. doi: 10.1016/0272-0590(90)90255-i. [DOI] [PubMed] [Google Scholar]
- Margolin B. H., Collings B. J., Mason J. M. Statistical analysis and sample-size determinations for mutagenicity experiments with binomial responses. Environ Mutagen. 1983;5(5):705–716. doi: 10.1002/em.2860050509. [DOI] [PubMed] [Google Scholar]
- Margolin B. H., Kaplan N., Zeiger E. Statistical analysis of the Ames Salmonella/microsome test. Proc Natl Acad Sci U S A. 1981 Jun;78(6):3779–3783. doi: 10.1073/pnas.78.6.3779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Margolin B. H., Resnick M. A., Rimpo J. Y., Archer P., Galloway S. M., Bloom A. D., Zeiger E. Statistical analyses for in vitro cytogenetic assays using Chinese hamster ovary cells. Environ Mutagen. 1986;8(2):183–204. doi: 10.1002/em.2860080203. [DOI] [PubMed] [Google Scholar]
- Margolin B. H., Risko K. J., Shelby M. D., Zeiger E. Sources of variability in Ames Salmonella typhimurium tester strains: analysis of the International Collaborative Study on 'genetic drift'. Mutat Res. 1984 Feb;130(1):11–25. doi: 10.1016/0165-1161(84)90003-7. [DOI] [PubMed] [Google Scholar]
- Maron D. M., Ames B. N. Revised methods for the Salmonella mutagenicity test. Mutat Res. 1983 May;113(3-4):173–215. doi: 10.1016/0165-1161(83)90010-9. [DOI] [PubMed] [Google Scholar]
- Mason J. M., Valencia R., Woodruff R. C., Zimmering S. Genetic drift and seasonal variation in spontaneous mutation frequencies in Drosophila. Environ Mutagen. 1985;7(5):663–676. doi: 10.1002/em.2860070506. [DOI] [PubMed] [Google Scholar]
- McFee A. F., Tice R. R. Influence of treatment to sacrifice time and the presence of BrdUrd on chemically-induced aberration rates in mouse marrow cells. Mutat Res. 1990 May;241(1):95–108. doi: 10.1016/0165-1218(90)90112-f. [DOI] [PubMed] [Google Scholar]
- Murphy S. A., Caspary W. J., Margolin B. H. A statistical analysis for the mouse lymphoma cell forward mutation assay. Mutat Res. 1988 Jun;203(3):145–154. doi: 10.1016/0165-1161(88)90100-8. [DOI] [PubMed] [Google Scholar]
- Murphy S. A., Tice R. R., Smith M. G., Margolin B. H. Contributions to the design and statistical analysis of in vivo SCE experiments. Mutat Res. 1992 Feb;271(1):39–48. doi: 10.1016/0165-1161(92)90030-p. [DOI] [PubMed] [Google Scholar]
- Shelby M. D., Bishop J. B., Mason J. M., Tindall K. R. Fertility, reproduction, and genetic disease: studies on the mutagenic effects of environmental agents on mammalian germ cells. Environ Health Perspect. 1993 Apr;100:283–291. doi: 10.1289/ehp.93100283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shelby M. D., Zeiger E. Activity of human carcinogens in the Salmonella and rodent bone-marrow cytogenetics tests. Mutat Res. 1990 Jun-Aug;234(3-4):257–261. doi: 10.1016/0165-1161(90)90022-g. [DOI] [PubMed] [Google Scholar]
- Solomon E., Borrow J., Goddard A. D. Chromosome aberrations and cancer. Science. 1991 Nov 22;254(5035):1153–1160. doi: 10.1126/science.1957167. [DOI] [PubMed] [Google Scholar]
- Tennant R. W., Ashby J. Classification according to chemical structure, mutagenicity to Salmonella and level of carcinogenicity of a further 39 chemicals tested for carcinogenicity by the U.S. National Toxicology Program. Mutat Res. 1991 May;257(3):209–227. doi: 10.1016/0165-1110(91)90002-d. [DOI] [PubMed] [Google Scholar]
- Tennant R. W., Elwell M. R., Spalding J. W., Griesemer R. A. Evidence that toxic injury is not always associated with induction of chemical carcinogenesis. Mol Carcinog. 1991;4(6):420–440. doi: 10.1002/mc.2940040604. [DOI] [PubMed] [Google Scholar]
- Tennant R. W., Margolin B. H., Shelby M. D., Zeiger E., Haseman J. K., Spalding J., Caspary W., Resnick M., Stasiewicz S., Anderson B. Prediction of chemical carcinogenicity in rodents from in vitro genetic toxicity assays. Science. 1987 May 22;236(4804):933–941. doi: 10.1126/science.3554512. [DOI] [PubMed] [Google Scholar]
- Tennant R. W., Spalding J., Stasiewicz S., Ashby J. Prediction of the outcome of rodent carcinogenicity bioassays currently being conducted on 44 chemicals by the National Toxicology Program. Mutagenesis. 1990 Jan;5(1):3–14. doi: 10.1093/mutage/5.1.3. [DOI] [PubMed] [Google Scholar]
- Tennant R. W. The genetic toxicity database of the National Toxicology Program: evaluation of the relationships between genetic toxicity and carcinogenicity. Environ Health Perspect. 1991 Dec;96:47–51. doi: 10.1289/ehp.919647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tice R. R., Erexson G. L., Hilliard C. J., Huston J. L., Boehm R. M., Gulati D., Shelby M. D. Effect of treatment protocol and sample time on the frequencies of micronucleated polychromatic erythrocytes in mouse bone marrow and peripheral blood. Mutagenesis. 1990 Jul;5(4):313–321. doi: 10.1093/mutage/5.4.313. [DOI] [PubMed] [Google Scholar]
- Tice R. R., Erexson G. L., Shelby M. D. The induction of micronucleated polychromatic erythrocytes in mice using single and multiple treatments. Mutat Res. 1990 Jun-Aug;234(3-4):187–193. doi: 10.1016/0165-1161(90)90014-f. [DOI] [PubMed] [Google Scholar]
- Totter J. R. Spontaneous cancer and its possible relationship to oxygen metabolism. Proc Natl Acad Sci U S A. 1980 Apr;77(4):1763–1767. doi: 10.1073/pnas.77.4.1763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weinberg R. A. Tumor suppressor genes. Science. 1991 Nov 22;254(5035):1138–1146. doi: 10.1126/science.1659741. [DOI] [PubMed] [Google Scholar]
- Zeiger E. An overview of genetic toxicity testing in the National Toxicology Program. Ann N Y Acad Sci. 1983;407:387–394. doi: 10.1111/j.1749-6632.1983.tb47844.x. [DOI] [PubMed] [Google Scholar]
- Zeiger E., Haseman J. K., Shelby M. D., Margolin B. H., Tennant R. W. Evaluation of four in vitro genetic toxicity tests for predicting rodent carcinogenicity: confirmation of earlier results with 41 additional chemicals. Environ Mol Mutagen. 1990;16 (Suppl 18):1–14. doi: 10.1002/em.2850160502. [DOI] [PubMed] [Google Scholar]


