Abstract
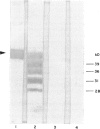
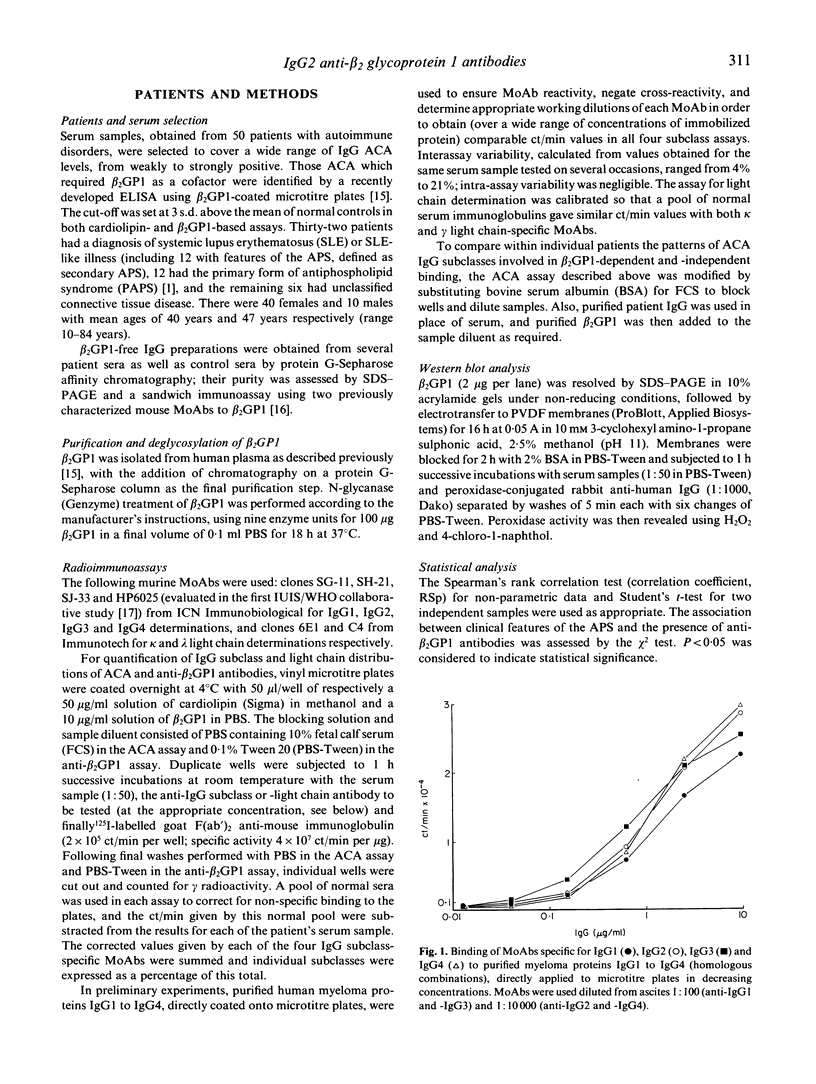
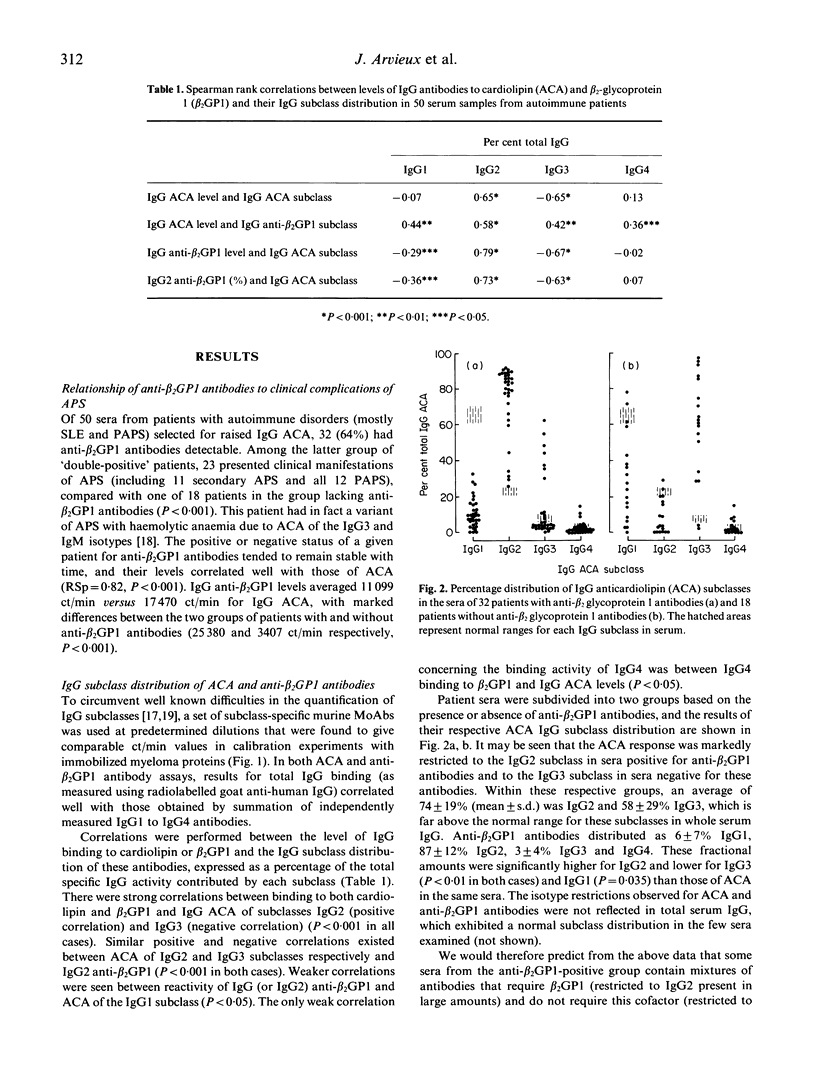
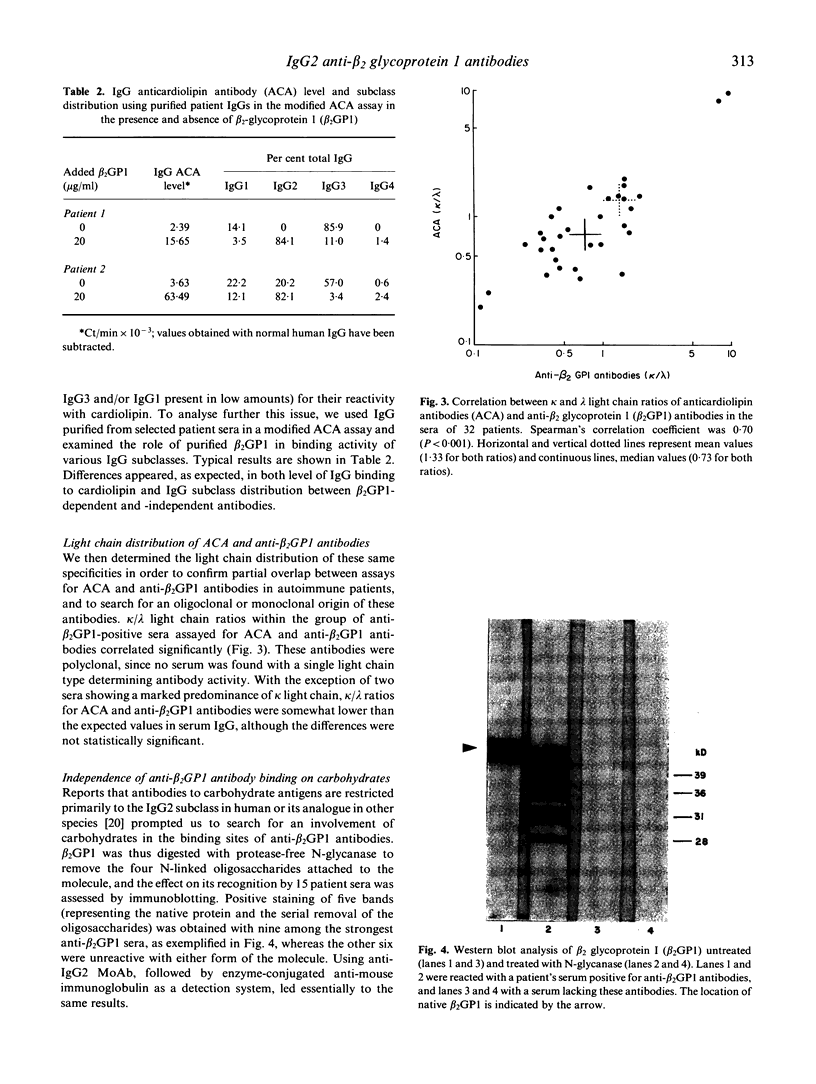
The IgG subclass and light chain distribution of antiphospholipid antibodies (aPL) occurring in autoimmune patients were determined by means of two radioimmunoassays using either cardiolipin- or beta 2 glycoprotein 1 (beta 2GP1)-coated microtitre plates and mouse MoAbs. Of 50 sera selected for positivity of anticardiolipin antibodies (ACA) of the IgG isotype, 32 (64%) possessed anti-beta 2GP1 antibodies and their presence was closely associated with clinical features of the antiphospholipid syndrome. Good correlations were found between ACA and anti-beta 2GP1 antibodies when considering antibody level and patterns of light chain and IgG subclass, suggesting that, overall, the same antibodies were being measured. Light chain analysis showed the polyclonal origin of these antibodies and, in most sera, a trend towards use of lambda chain. Among sera positive for anti-beta 2GP1 antibodies, IgG2 was the major subclass reactive with beta 2GP1 and cardiolipin (87% and 74% of the IgG antibody activity, respectively). In contrast, in the group of 18 sera lacking anti-beta 2GP1 antibodies, ACA were largely restricted to IgG3, with a lesser contribution by IgG1. A few selected sera from the anti-beta 2GP1-positive group were shown to contain mixtures of antibodies that required beta 2GP1 (restricted to IgG2 present in large amounts) and did not require this cofactor (restricted to IgG3 and/or IgG1 present in low amounts) for their reactivity with cardiolipin. There was no contribution of glycosylation to the epitopes recognized by anti-beta 2GP1 antibodies, even though human anti-carbohydrate antibodies are restricted to the IgG2 subclass. These findings further emphasize the intra- and interindividual heterogeneity of aPL, and should help to discriminate clinically relevant specificies.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arvieux J., Pouzol P., Roussel B., Jacob M. C., Colomb M. G. Lupus-like anticoagulant properties of murine monoclonal antibodies to beta 2-glycoprotein I. Br J Haematol. 1992 Aug;81(4):568–573. doi: 10.1111/j.1365-2141.1992.tb02993.x. [DOI] [PubMed] [Google Scholar]
- Arvieux J., Roussel B., Jacob M. C., Colomb M. G. Measurement of anti-phospholipid antibodies by ELISA using beta 2-glycoprotein I as an antigen. J Immunol Methods. 1991 Oct 25;143(2):223–229. doi: 10.1016/0022-1759(91)90047-j. [DOI] [PubMed] [Google Scholar]
- Arvieux J., Roussel B., Pouzol P., Colomb M. G. Platelet activating properties of murine monoclonal antibodies to beta 2-glycoprotein I. Thromb Haemost. 1993 Aug 2;70(2):336–341. [PubMed] [Google Scholar]
- Arvieux J., Schweizer B., Roussel B., Colomb M. G. Autoimmune haemolytic anaemia due to anti-phospholipid antibodies. Vox Sang. 1991;61(3):190–195. doi: 10.1111/j.1423-0410.1991.tb00945.x. [DOI] [PubMed] [Google Scholar]
- Bevers E. M., Galli M., Barbui T., Comfurius P., Zwaal R. F. Lupus anticoagulant IgG's (LA) are not directed to phospholipids only, but to a complex of lipid-bound human prothrombin. Thromb Haemost. 1991 Dec 2;66(6):629–632. [PubMed] [Google Scholar]
- Cabral A. R., Cabiedes J., Alarcón-Segovia D., Sánchez-Guerrero J. Phospholipid specificity and requirement of beta 2-glycoprotein-I for reactivity of antibodies from patients with primary antiphospholipid syndrome. J Autoimmun. 1992 Dec;5(6):787–801. doi: 10.1016/0896-8411(92)90193-t. [DOI] [PubMed] [Google Scholar]
- Dostal-Johnson D., Rote N. S., Branch D. W. IgG1 and IgG2 are the predominant subclasses of antiphospholipid antibody in women with the lupus anticoagulant. Clin Immunol Immunopathol. 1990 Feb;54(2):309–319. doi: 10.1016/0090-1229(90)90092-5. [DOI] [PubMed] [Google Scholar]
- Fishman P., Bakimer R., Blank M., Sredni D., Djaldetti M., Shoenfeld Y. The putative role of cytokines in the induction of primary anti-phospholipid syndrome in mice. Clin Exp Immunol. 1992 Nov;90(2):266–270. doi: 10.1111/j.1365-2249.1992.tb07940.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galli M., Comfurius P., Maassen C., Hemker H. C., de Baets M. H., van Breda-Vriesman P. J., Barbui T., Zwaal R. F., Bevers E. M. Anticardiolipin antibodies (ACA) directed not to cardiolipin but to a plasma protein cofactor. Lancet. 1990 Jun 30;335(8705):1544–1547. doi: 10.1016/0140-6736(90)91374-j. [DOI] [PubMed] [Google Scholar]
- Gharavi A. E., Harris E. N., Lockshin M. D., Hughes G. R., Elkon K. B. IgG subclass and light chain distribution of anticardiolipin and anti-DNA antibodies in systemic lupus erythematosus. Ann Rheum Dis. 1988 Apr;47(4):286–290. doi: 10.1136/ard.47.4.286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greenspan N. S., Cooper L. J. Intermolecular cooperativity: a clue to why mice have IgG3? Immunol Today. 1992 May;13(5):164–168. doi: 10.1016/0167-5699(92)90120-V. [DOI] [PubMed] [Google Scholar]
- Hamilton R. G. Human IgG subclass measurements in the clinical laboratory. Clin Chem. 1987 Oct;33(10):1707–1725. [PubMed] [Google Scholar]
- Hammarström L., Smith C. I. IgG subclasses in bacterial infections. Monogr Allergy. 1986;19:122–133. [PubMed] [Google Scholar]
- Hunt J. E., McNeil H. P., Morgan G. J., Crameri R. M., Krilis S. A. A phospholipid-beta 2-glycoprotein I complex is an antigen for anticardiolipin antibodies occurring in autoimmune disease but not with infection. Lupus. 1992 Feb;1(2):75–81. doi: 10.1177/096120339200100204. [DOI] [PubMed] [Google Scholar]
- Jefferis R., Reimer C. B., Skvaril F., de Lange G., Ling N. R., Lowe J., Walker M. R., Phillips D. J., Aloisio C. H., Wells T. W. Evaluation of monoclonal antibodies having specificity for human IgG sub-classes: results of an IUIS/WHO collaborative study. Immunol Lett. 1985;10(3-4):223–252. doi: 10.1016/0165-2478(85)90082-3. [DOI] [PubMed] [Google Scholar]
- Keeling D. M., Wilson A. J., Mackie I. J., Machin S. J., Isenberg D. A. Some 'antiphospholipid antibodies' bind to beta 2-glycoprotein I in the absence of phospholipid. Br J Haematol. 1992 Nov;82(3):571–574. doi: 10.1111/j.1365-2141.1992.tb06469.x. [DOI] [PubMed] [Google Scholar]
- Levy R. A., Gharavi A. E., Sammaritano L. R., Habina L., Qamar T., Lockshin M. D. Characteristics of IgG antiphospholipid antibodies in patients with systemic lupus erythematosus and syphilis. J Rheumatol. 1990 Aug;17(8):1036–1041. [PubMed] [Google Scholar]
- Loizou S., Cofiner C., Weetman A. P., Walport M. J. Immunoglobulin class and IgG subclass distribution of anticardiolipin antibodies in patients with systemic lupus erythematosus and associated disorders. Clin Exp Immunol. 1992 Dec;90(3):434–439. doi: 10.1111/j.1365-2249.1992.tb05864.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McNeil H. P., Simpson R. J., Chesterman C. N., Krilis S. A. Anti-phospholipid antibodies are directed against a complex antigen that includes a lipid-binding inhibitor of coagulation: beta 2-glycoprotein I (apolipoprotein H). Proc Natl Acad Sci U S A. 1990 Jun;87(11):4120–4124. doi: 10.1073/pnas.87.11.4120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pierangeli S. S., Harris E. N., Davis S. A., DeLorenzo G. Beta 2-glycoprotein 1 (beta 2GP1) enhances cardiolipin binding activity but is not the antigen for antiphospholipid antibodies. Br J Haematol. 1992 Nov;82(3):565–570. doi: 10.1111/j.1365-2141.1992.tb06468.x. [DOI] [PubMed] [Google Scholar]
- Sammaritano L. R., Lockshin M. D., Gharavi A. E. Antiphospholipid antibodies differ in aPL cofactor requirement. Lupus. 1992 Feb;1(2):83–90. doi: 10.1177/096120339200100205. [DOI] [PubMed] [Google Scholar]
- Snowden N., Wilson P., Pumphrey R. Anticardiolipin antibodies in systemic lupus erythematous. Ann Rheum Dis. 1989 Jan;48(1):80–82. doi: 10.1136/ard.48.1.80-b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stevens T. L., Bossie A., Sanders V. M., Fernandez-Botran R., Coffman R. L., Mosmann T. R., Vitetta E. S. Regulation of antibody isotype secretion by subsets of antigen-specific helper T cells. Nature. 1988 Jul 21;334(6179):255–258. doi: 10.1038/334255a0. [DOI] [PubMed] [Google Scholar]
- Tiikkainen U., Wangel A., Appleton S. L., Arthur D. Subclasses of IgG anticardiolipin antibodies in patients with systemic lupus erythematosus. Scand J Immunol. 1991 Sep;34(3):265–271. doi: 10.1111/j.1365-3083.1991.tb01546.x. [DOI] [PubMed] [Google Scholar]
- Tsutsumi A., Koike T., Ichikawa K., Shimada K., Takabayashi K., Tomioka H., Yoshida S. IgG subclass distribution of anticardiolipin antibody in patients with systemic lupus erythematosus. J Rheumatol. 1988 Dec;15(12):1764–1767. [PubMed] [Google Scholar]
- Wagenknecht D. R., McIntyre J. A. Changes in beta 2-glycoprotein I antigenicity induced by phospholipid binding. Thromb Haemost. 1993 Apr 1;69(4):361–365. [PubMed] [Google Scholar]
- Wall B. M., Crofton J. T., Share L., Cooke C. R. Chronic hyponatremia due to resetting of the osmostat in a patient with gastric carcinoma. Am J Med. 1992 Aug;93(2):223–228. doi: 10.1016/0002-9343(92)90055-g. [DOI] [PubMed] [Google Scholar]