Abstract
We show here that Helicobacter pylori broth culture supernatants disrupt the actin cytoskeleton of epithelial cell lines, leading to cell rounding and apoptosis through anoikis. We demonstrate that there are marked quantitative differences between strains and that there are different cell line sensitivities. By constructing VacA null isogenic mutants, we show that the effect is not due to the vacuolating cytotoxin.
Helicobacter pylori is the main cause of peptic ulceration and the most important risk factor for distal gastric adenocarcinoma and primary gastric lymphoma (22, 23, 26). Although about 30% of the population in developed countries and more than 60% of the population in developing countries are infected with H. pylori, most individuals do not develop significant disease. Who develops disease depends on host genetic factors, (11) environmental factors (19), and bacterial virulence determinants, such as vacuolating cytotoxin (VacA) activity and possession of the cag pathogenicity island (1, 4, 5). However, the pathogenesis of H. pylori-associated diseases remains uncertain. One much-studied area is H. pylori-induced epithelial cell apoptosis (6, 14, 20, 28, 30). Apoptotic cell loss could be a factor in both gastroduodenal ulceration and gastric atrophy; the latter is a known precursor of gastric adenocarcinoma (7). Disruption of the proliferation-apoptosis balance could also be important in affecting the number of surviving cells with DNA replication errors and so in modulating cancer risk. Isogenic mutant studies have shown that the cag pathogenicity island may play a role in inducing epithelial cell apoptosis (16), although cag-negative strains can also induce apoptosis (30). VacA is another strong inducer of apoptosis, as shown by experiments in which bacterial cell-free culture supernatants from wild-type strains and isogenic VacA-negative mutants were compared (14).
While studying VacA-induced epithelial cell apoptosis using bacterial culture supernatants, we noticed that among the rounded apoptotic cells were other cells, which, although rounded, did not show features of apoptosis. In this study we characterized this newly described effect of H. pylori on epithelial cells, showed that it is bacterial strain and epithelial cell line dependent, and proved, using an isogenic mutant approach, that it is not VacA dependent.
Preparation of H. pylori broth culture supernatants.
We used the H. pylori strains described in Table 1 (3, 4, 10, 17). We prepared concentrated broth culture supernatants of H. pylori from 48-h cultures in sulfite-free brucella broth (2) supplemented with 0.2% β-cyclodextrin and incubated microaerobically (Campypak-Plus; Becton-Dickenson) with shaking at 37°C. We adjusted the optical density at 550 of uncontaminated broth media to 0.8, removed bacterial cells by centrifugation, concentrated filtered supernatants 20-fold by ultrafiltration with Centriprep-30 filters (Amicon), and filtered (pore size, 0.2 μm) the preparations before dividing them into aliquots and freezing them at −20°C.
TABLE 1.
H. pylori strains used
| Straina | cagA status | vacA genotypeb | Reference |
|---|---|---|---|
| 60190 (= ATCC 49503) | + | sl/ml | 17 |
| 60190v1 | + | Isogenic vacA insertion mutant of 60190 | This study |
| Tx30a (= ATCC 51392) | − | s2/m2 | 17 |
| J116 | + | sl/m2 | 4 |
| M99 | + | sl/m1 | This study |
| M99v1 | + | Isogenic vacA insertion mutant of M99 | This study |
Strain J116 was cultured from a patient with duodenal ulceration from Nashville, Tenn. Strain M99 (originally isolated from a rhesus monkey without peptic ulceration) was a kind gift from Andre Dubois. Similar isolates have previously been confirmed to be H. pylori (10), and we confirmed that this isolate was H. pylori by extended 16s rRNA nucleotide sequence analysis.
See reference 4.
H. pylori supernatants cause epithelial cell apoptosis but also induce a rounded nonapoptotic phenotype.
We used the following cell lines from the European tissue culture collection: AGS (human gastric adenocarcinoma), HeLa (human cervical adenocarcinoma), and RK13 (rabbit kidney epithelium). We diluted our stored concentrated broth culture supernatants fivefold and added them to subconfluent epithelial cells on 96-well plates (Costar) in progressive doubling dilutions so that the maximum final concentration of the broth culture supernatant on cells was fourfold concentrated. We then placed the plates in a 5% CO2 incubator and examined them with a light microscope after various time intervals. Fresh and freeze-thawed supernatants had similar effects on cells. We used identically prepared uninoculated broth media as controls.
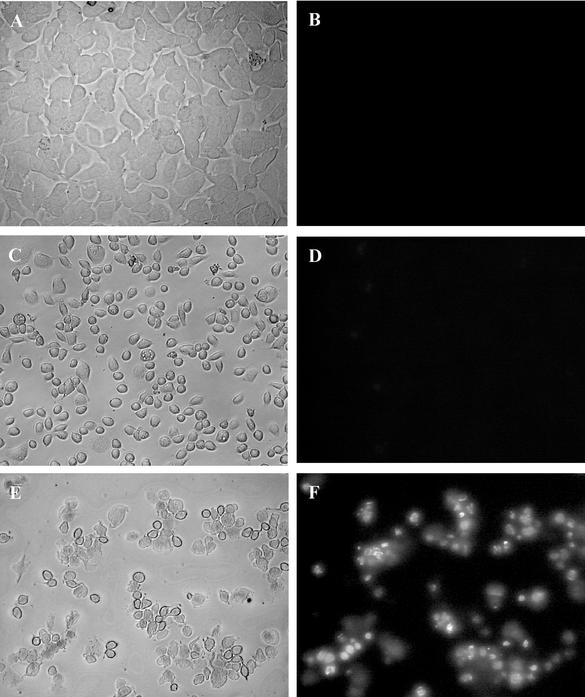
Addition of H. pylori concentrated broth culture supernatants to epithelial cells induced a rounded phenotype, which in the case of toxigenic vacA s1/m1 strain 60190 was accompanied by characteristic vacuolation (17). Many rounded cells were clearly apoptotic; they were stained with Hoechst no. 33258 stain (0.1 mg/ml; Sigma-Aldrich) and showed nuclear fragmentation. However, many other rounded cells showed no Hoechst staining or other signs of apoptosis at 3 h (Fig. 1C and D) or 24 h (Fig. 1E and F). To confirm definitively that many rounded cells were not apoptotic, we grew AGS cells in culture dishes, fixed them in 2.5% gluteraldehyde, processed them (27), and examined ultrathin sections by transmission electron microscopy (Jeol 1200 EX; Jeol, Welwyn, United Kingdom). Many rounded AGS cells exhibited ultrastructural changes typical of apoptosis, but many others showed no such features.
FIG. 1.
Hoechst staining of H. pylori-treated AGS cells. (A and B) Sulfite-free brucella broth-treated AGS cells (A) (bright field) show no uptake of Hoechst no. 33258 stain (B). (C and D) AGS cells treated for 3 h with fourfold-concentrated M99 broth supernatant show altered morphology with a rounded appearance (C), and Hoechst staining shows that very few of the cells stain positively (D), indicating that there was a lack of apoptosis. (E and F) At 24 h significantly more AGS cells show apoptotic changes. Magnification, ×10.
H. pylori-induced nonapoptotic cell rounding is partially reversible but is associated with cytoskeletal changes.
We next concentrated on the nonapoptotic rounded cell population. These cells excluded trypan blue, proving that they were viable. By 3 h, many AGS cells adopted a rounded phenotype, but less than 3% were stained with Hoechst stain, whereas at 24 h distinct populations of rounded apoptotic (68%) and nonapoptotic (32%) cells were present. To assess whether the nonapoptotic rounding was reversible, we removed media from rounded cells 24 h after addition of bacterial supernatant, washed the cells twice, and replaced the media with fresh supernatant-free media. As controls, we used rounded cells without the media removed. Most rounded cells remained rounded over the next 48 h, but approximately 20% reverted to a nonrounded phenotype, showing that rounding could be reversed and did not inevitably lead to apoptosis.
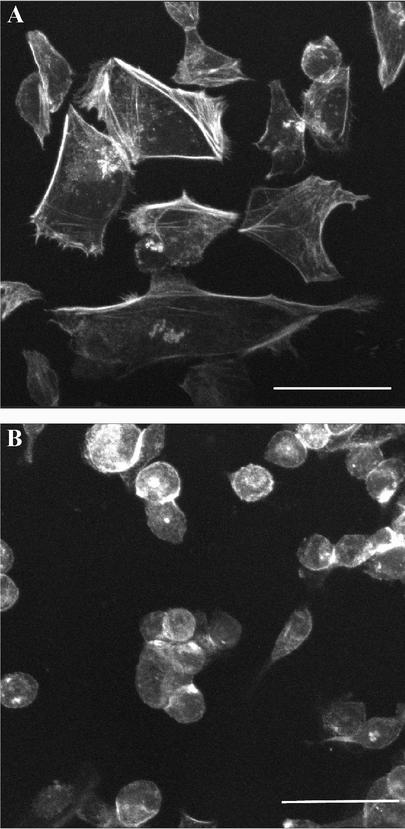
To define whether rounding resulted from cytoskeletal changes, we assessed the actin cytoskeleton in AGS cells. We fixed H. pylori culture supernatant-treated and control-treated cells on Lab-Tek chamber slides in 3.7% paraformaldehyde, permeabilized them with 0.1% Triton X-100, blocked them in phosphate-buffered saline (PBS)-5% bovine serum albumin, stained them with fluorescein isothiocyanate-labeled phalloidin (5 μg/ml in PBS; Sigma-Aldrich), washed them five times in PBS-0.1% bovine serum albumin, mounted them with cover slips and ProLong Antifade (Molecular Probes), and viewed them using confocal microscopy (Leica SP2). Cells that had been treated with H. pylori culture supernatant showed rearrangement and disruption of the actin cytoskeleton and, in particular, a lack of actin stress fibers (Fig. 2).
FIG. 2.
Fluorecein isothiocyanate Phalloidin staining of AGS cells. (A) AGS cells treated with uninoculated sulfite-free brucella broth (fourfold concentrated) show cortical actin staining with occasional intracellular stress fibers. (B) AGS cells treated with M99 supernatant (fourfold concentrated) show rearrangement and disruption of the actin cytoskeleton. Cells were fixed and stained after 24 h of incubation with supernatants. Bars = 50 μm.
H. pylori-induced cell rounding is bacterial strain and cell line dependent.
Although nonapoptotic cell rounding could be induced by all strains of H. pylori tested, we noted that some strains induced this effect more profoundly than others and that some cell lines appeared to be more susceptible. We quantified this by systematically adding doubling dilutions of concentrated broth culture supernatants of four strains of H. pylori to AGS, HeLa, and RK13 cells. We selected microscopy fields for examination in an unbiased manner by starting one viewing field from the lower edge of each well and moving two fields in at a time. We examined 100 cells per well to calculate the percentage of rounded and/or vacuolated cells. In order to compare bacterial strains and cell lines, we used as our major endpoint the dilution at which >80% of total cells counted were vacuolated or rounded. We and other workers have found that this is a harder and more reliable endpoint than that obtained by comparing total numbers of vacuolated cells within an individual dilution because of the difficulties in scoring borderline cases (3, 8). All four bacterial strains caused separation and rounding of epithelial cells, but this effect varied according to the strain of H. pylori and the cell line (Table 2). Fourfold-concentrated and unconcentrated broth supernatant from M99 caused 80% (or more) cell rounding in all three cell lines tested. 60190 (at a fourfold concentration) caused 80% rounding in AGS and RK13 cells, whereas Tx30a and J116 (both at a fourfold concentration) caused 80% rounding only in AGS cells. Overall, M99 caused more rounding than the other strains (Table 2). AGS cells became rounded significantly more easily than HeLa cells (P = 0.004, as determined by the Fisher exact test), while RK13 cells appeared to be intermediate.
TABLE 2.
H. pylori strain and cell line specificity of the abilities of broth culture supernatants (fourfold concentrated) to cause 80% cell rounding
| H. pylori strain | No. of expts in which there was cell rounding with the following cell linesa:
|
|||
|---|---|---|---|---|
| AGS (n = 5) | HeLa (n = 5) | RK13 (n = 3) | Total (n = 13) | |
| SFBBb (negative control) | 0 | 0 | 0 | 0 (0)c |
| M99 | 5 | 4 | 3 | 12 (92)d |
| 60190 | 3 | 0 | 2 | 5 (38)e |
| Tx30a | 4 | 0 | 0 | 4 (31) |
| J116 | 2 | 0 | 0 | 2 (14) |
| Total | 14/20 (70)f | 4/20 (20) | 5/12 (42) | |
Cell rounding was present at various levels throughout the experiments. To quantify differences, we recorded the number of experiments in which >80% cell rounding was induced by fourfold-concentrated bacterial culture supernatants after 24 h of incubation. All statistical data were calculated by using Fisher's exact test (two tailed) and contingency tables.
SFBB, sulfite-free brucella broth.
The numbers in parentheses are percentages.
P < 0.01 for a comparison with any other strain or SFBB.
P = 0.04 for a comparison with SFBB.
P = 0.04 for a comparison with HeLa cells.
Cell rounding is not dependent on VacA.
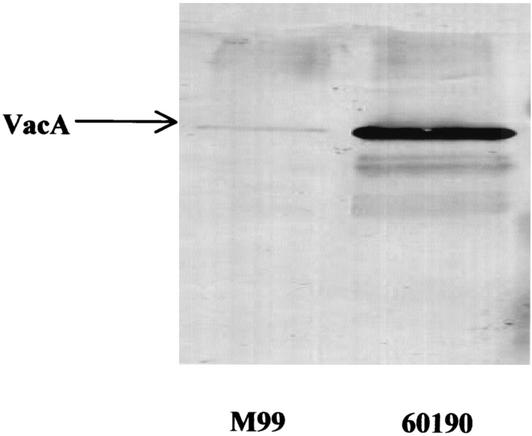
Because the two strains which induced nonapoptotic cell rounding most profoundly (M99 and 60190) had the most toxigenic genotype of vacA (s1/m1) and because other bacterial toxins cause similar effects, we initially hypothesized that the cell-rounding effect was due to VacA. However, two experiments led us to question this. First, although dilution of concentrated broth culture supernatants of 60190 revealed cytoplasmic vacuolation, dilution of M99 supernatants did not, implying that rounding was not just an effect of concentrated VacA. Second, although strain M99 produced more profound rounding than 60190 produced, supernatant immunoblots obtained by using polyclonal anti-VacA antibodies (a gift from T. L. Cover) showed that it produced less VacA (Fig. 3). This could be because the effect was not due to VacA or because it was a separate effect of VacA and M99 VacA was a strong inducer of this effect but did not induce vacuolation.
FIG. 3.
Immunoblot of supernatants from strains M99 and 60190. A representative immunoblot obtained by using polyclonal anti-VacA antibodies shows that the two strains secrete similar-size VacA but that 60190 secretes significantly more than M99 secretes. Supernatants were corrected for the bacterial optical density at 550 nm before concentration and loading of equal volumes onto sodium dodecyl sulfate-polyacrylamide gel electrophoresis gels.
To directly and definitively test the hypothesis that VacA caused cell rounding, we constructed VacA− isogenic insertion mutants of the two strains which induced rounding most strongly, M99 and 60190. To do this, we ligated the kanamycin resistance cassette, aphA, from plasmid pILL600 (15) (a kind gift from A. Labigne) into the unique EcoRI site of vacA in plasmid pCTB8 (8) (a kind gift from T. L. Cover), as previously described (8). We used the resulting plasmid, pCTB8::aphA, to replace vacA in strains M99 and 60190, using natural transformation, homologous recombination, and marker rescue on blood agar plates containing 30 μg of kanamycin per ml (8) to obtain strains M99v1 and 60190v1. To confirm insertion of the cassette within vacA, we PCR amplified genomic DNA from strains M99, M99v1, 60190, and 60190v1 using vacA-specific primers 5′ATGGAAATACAACAAACACAC3′ and 5′ CTCCAGAACCCACACGATT3′ (and PCR conditions previously described [2]) and showed that there was insertion of a 1.4-kb fragment, consistent with the antibiotic cassette, into vacA in M99v1 and 60190v1. We confirmed that supernatants from the VacA mutant strains did not contain VacA or VacA fragments by immunoblotting with anti-VacA antibodies, as described above. To assess the effects on cells, we concentrated supernatants from M99, M99v1, 60190, and 60190v1 grown in identical conditions and added them to cells in doubling dilutions as described above. M99v1 and 60190v1 broth media caused rounding and separation of epithelial cells just like the parent M99 and 60190 strains on all three occasions tested. We further characterized the effects of the mutant strains by staining the actin cytoskeletons in the cells, and we observed changes identical to those observed with the wild-type strains.
We are currently further characterizing the factor in H. pylori supernatant responsible for cell rounding, and initial experiments have shown that it is heat sensitive. We heated broth culture supernatants from strains 60190 and M99 to 80°C in a water bath for 30 min prior to concentration, concentrated them as described above, added them to AGS cells, and observed the effects over 24 h. The heated supernatants did not induce the cell-rounding effect, whereas paired control nonheated supernatants caused profound rounding.
In this study, we demonstrated that H. pylori culture supernatants induce cytoskeletal disruption and cell rounding of epithelial cell lines. The effect precedes, but does not inevitably result in, apoptosis. In general, there are two triggers of apoptosis: cell damage (particularly DNA damage), which leads to altruistic apoptosis, and isolation from other cells or the surrounding matrix, which leads to anoikis (death by homelessness) (12). We demonstrated that there is disruption of the epithelial actin cytoskeleton that leads to separation of epithelial cells and subsequent apoptosis, suggesting that H. pylori-induced anoikis occurs. This is similar to the apoptosis through anoikis induced by toxins from other gram-negative bacteria, such as Clostridium difficile (18), Bacteroides fragilis (9, 13, 31), and enteroaggregative Escherichia coli (21). Although the mechanisms vary, these toxins all cause epithelial cytoskeletal rearrangements and dramatic alterations in cellular morphology.
The effect of H. pylori on the cytoskeleton and particularly the role of bacterial virulence factors are controversial. Segal et al. have described the following two cellular phenotypes induced by H. pylori coculture: a “hummingbird” phenotype, characterized by lamellipodia and filopodia, which is dependent on a functioning bacterial cag-encoded type IV secretory system, and a stress fiber-associated phenotype that is independent of cag and the VacA toxin (29). Palovuori et al. have also observed H. pylori induction of cag- and VacA-independent actin changes, but they described ruffles and lamellipodia-like structures (25). Pai et al. described cytoskeletal changes with reduced stress fibers induced by H. pylori culture supernatants in a rat gastric epithelial cell line and reproduced these changes with purified VacA (24). In our experiments, as expected with culture supernatants and so absence of cell contact with the cag type IV secretion system, we did not observe the hummingbird phenotype. The changes which we observed with reduced stress fibers are most similar to those described by Pai et al. (24), but we show definitively that they are not due to VacA.
Acknowledgments
J.R.B. was supported by fellowships from The Wellcome Trust (entry level) and the Medical Research Council (United Kingdom) (clinical training fellowship). J.C.A. was supported by a senior clinical fellowship from the Medical Research Council (United Kingdom).
Editor: A. D. O'Brien
REFERENCES
- 1.Atherton, J. C. 1998. H. pylori virulence factors. Br. Med. Bull. 54:105-120. [DOI] [PubMed] [Google Scholar]
- 2.Atherton, J. C. 1997. Molecular methods for detecting ulcerogenic strains of H. pylori, p. 133-143. In M. H. Clayton (ed.), Methods in molecular medicine. Helicobacter pylori protocols. Humana Press Inc., Totawa, N.J. [DOI] [PubMed]
- 3.Atherton, J. C., P. Cao, R. M. Peek, Jr., M. K. Tummuru, M. J. Blaser, and T. L. Cover. 1995. Mosaicism in vacuolating cytotoxin alleles of Helicobacter pylori. Association of specific vacA types with cytotoxin production and peptic ulceration. J. Biol. Chem. 270:17771-17777. [DOI] [PubMed] [Google Scholar]
- 4.Atherton, J. C., R. M. Peek, Jr., K. T. Tham, T. L. Cover, and M. J. Blaser. 1997. Clinical and pathological importance of heterogeneity in vacA, the vacuolating cytotoxin gene of Helicobacter pylori. Gastroenterology 112:92-99. [DOI] [PubMed] [Google Scholar]
- 5.Censini, S., C. Lange, Z. Xiang, J. E. Crabtree, P. Ghiara, M. Borodovsky, R. Rappuoli, and A. Covacci. 1996. cag, a pathogenicity island of Helicobacter pylori, encodes type I-specific and disease-associated virulence factors. Proc. Natl. Acad. Sci. USA 93:14648-14653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chen, G., E. M. Sordillo, W. G. Ramey, J. Reidy, P. R. Holt, S. Krajewski, J. C. Reed, M. J. Blaser, and S. F. Moss. 1997. Apoptosis in gastric epithelial cells is induced by Helicobacter pylori and accompanied by increased expression of BAK. Biochem. Biophys. Res. Commun. 239:626-632. [DOI] [PubMed] [Google Scholar]
- 7.Correa, P. 1995. Helicobacter pylori and gastric carcinogenesis. Am. J. Surg. Pathol. 19:S37-S43. [PubMed]
- 8.Cover, T. L., M. K. Tummuru, P. Cao, S. A. Thompson, and M. J. Blaser. 1994. Divergence of genetic sequences for the vacuolating cytotoxin among Helicobacter pylori strains. J. Biol. Chem. 269:10566-10573. [PubMed] [Google Scholar]
- 9.Donelli, G., A. Fabbri, and C. Fiorentini. 1996. Bacteroides fragilis enterotoxin induces cytoskeletal changes and surface blebbing in HT-29 cells. Infect. Immun. 64:113-119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Drazek, E. S., A. Dubois, and R. K. Holmes. 1994. Characterization and presumptive identification of Helicobacter pylori isolates from rhesus monkeys. J. Clin. Microbiol. 32:1799-1804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.El-Omar, E. M., M. Carrington, W. H. Chow, K. E. McColl, J. H. Bream, H. A. Young, J. Herrera, J. Lissowska, C. C. Yuan, N. Rothman, G. Lanyon, M. Martin, J. F. Fraumeni, Jr., and C. S. Rabkin. 2000. Interleukin-1 polymorphisms associated with increased risk of gastric cancer. Nature 404:398-402. [DOI] [PubMed] [Google Scholar]
- 12.Frisch, S. M., and H. Francis. 1994. Disruption of epithelial cell-matrix interactions induces apoptosis. J. Cell Biol. 124:619-626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Koshy, S. S., M. H. Montrose, and C. L. Sears. 1996. Human intestinal epithelial cells swell and demonstrate actin rearrangement in response to the metalloprotease toxin of Bacteroides fragilis. Infect. Immun. 64:5022-5028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kuck, D., B. Kolmerer, C. Iking-Konert, P. H. Krammer, W. Stremmel, and J. Rudi. 2001. Vacuolating cytotoxin of Helicobacter pylori induces apoptosis in the human gastric epithelial cell line AGS. Infect. Immun. 69:5080-5087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Labigne-Roussel, A., P. Courcoux, and L. Tompkins. 1988. Gene disruption and replacement as a feasible approach for mutagenesis of Campylobacter jejuni. J. Bacteriol. 170:1704-1708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Le'Negrate, G., V. Ricci, V. Hofman, B. Mograbi, P. Hofman, and B. Rossi. 2001. Epithelial intestinal cell apoptosis induced by Helicobacter pylori depends on expression of the cag pathogenicity island phenotype. Infect. Immun. 69:5001-5009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Leunk, R. D., P. T. Johnson, B. C. David, W. G. Kraft, and D. R. Morgan. 1988. Cytotoxic activity in broth-culture filtrates of Campylobacter pylori. J. Med. Microbiol. 26:93-99. [DOI] [PubMed] [Google Scholar]
- 18.Mahida, Y. R., S. Makh, S. Hyde, T. Gray, and S. P. Borriello. 1996. Effect of Clostridium difficile toxin A on human intestinal epithelial cells: induction of interleukin 8 production and apoptosis after cell detachment. Gut 38:337-347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Martin, D. F., E. Montgomery, A. S. Dobek, G. A. Patrissi, and D. A. Peura. 1989. Campylobacter pylori, NSAIDS, and smoking: risk factors for peptic ulcer disease. Am. J. Gastroenterol. 84:1268-1272. [PubMed] [Google Scholar]
- 20.Moss, S. F., J. Calam, B. Agarwal, S. Wang, and P. R. Holt. 1996. Induction of gastric epithelial apoptosis by Helicobacter pylori. Gut 38:498-501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Navarro-Garcia, F., C. Sears, C. Eslava, A. Cravioto, and J. P. Nataro. 1999. Cytoskeletal effects induced by Pet, the serine protease enterotoxin of enteroaggregative Escherichia coli. Infect. Immun. 67:2184-2192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nomura, A., G. N. Stemmermann, P. H. Chyou, I. Kato, G. I. Perez-Perez, and M. J. Blaser. 1991. Helicobacter pylori infection and gastric carcinoma among Japanese Americans in Hawaii. N. Engl. J. Med. 325:1132-1136. [DOI] [PubMed] [Google Scholar]
- 23.Nomura, A., G. N. Stemmermann, P. H. Chyou, G. I. Perez-Perez, and M. J. Blaser. 1994. Helicobacter pylori infection and the risk for duodenal and gastric ulceration. Ann. Intern. Med. 120:977-981. [DOI] [PubMed] [Google Scholar]
- 24.Pai, R., T. L. Cover, and A. S. Tarnawski. 1999. Helicobacter pylori vacuolating cytotoxin (VacA) disorganizes the cytoskeletal architecture of gastric epithelial cells. Biochem. Biophys. Res. Commun. 262:245-250. [DOI] [PubMed] [Google Scholar]
- 25.Palovuori, R., A. Perttu, Y. Yan, R. Karttunen, S. Eskelinen, and T. J. Karttunen. 2000. Helicobacter pylori induces formation of stress fibers and membrane ruffles in AGS cells by rac activation. Biochem. Biophys. Res. Commun. 269:247-253. [DOI] [PubMed] [Google Scholar]
- 26.Parsonnet, J., S. Hansen, L. Rodriguez, A. B. Gelb, R. A. Warnke, E. Jellum, N. Orentreich, J. H. Vogelman, and G. D. Friedman. 1994. Helicobacter pylori infection and gastric lymphoma. N. Engl. J. Med. 330:1267-1271. [DOI] [PubMed] [Google Scholar]
- 27.Robinson, G., and T. Gray. 1990. Electron microscopy 2: tissue preparation, sectioning and staining, p. 525-562. In S. A. Bancroft (ed.), Theory and practice of histological techniques, 3rd ed. Churchill Livingstone, London, United Kingdom.
- 28.Rudi, J., D. Kuck, S. Strand, A. von Herbay, S. M. Mariani, P. H. Krammer, P. R. Galle, and W. Stremmel. 1998. Involvement of the CD95 (APO-1/Fas) receptor and ligand system in Helicobacter pylori-induced gastric epithelial apoptosis. J. Clin. Investig. 102:1506-1514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Segal, E. D., J. Cha, J. Lo, S. Falkow, and L. S. Tompkins. 1999. Altered states: involvement of phosphorylated CagA in the induction of host cellular growth changes by Helicobacter pylori. Proc. Natl. Acad. Sci. USA 96:14559-14564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wagner, S., W. Beil, J. Westermann, R. P. Logan, C. T. Bock, C. Trautwein, J. S. Bleck, and M. P. Manns. 1997. Regulation of gastric epithelial cell growth by Helicobacter pylori: evidence for a major role of apoptosis. Gastroenterology 113:1836-1847. [DOI] [PubMed] [Google Scholar]
- 31.Weikel, C. S., F. D. Grieco, J. Reuben, L. L. Myers, and R. B. Sack. 1992. Human colonic epithelial cells, HT29/C1, treated with crude Bacteroides fragilis enterotoxin dramatically alter their morphology. Infect. Immun. 60:321-327. [DOI] [PMC free article] [PubMed] [Google Scholar]