Abstract
Tenascin-C (TN-C) is an extracellular matrix molecule that is expressed during wound healing in various tissues. Although not detectable in the normal adult heart, it is expressed under pathological conditions. Previously, using a rat model, we found that TN-C was expressed during the acute stage after myocardial infarction and that α-smooth muscle actin (α-SMA)-positive myofibroblasts appeared in TN-C-positive areas. In the present study, we examined whether TN-C controls the dynamics of myofibroblast recruitment and wound healing after electrical injury to the myocardium of TN-C knockout (TNKO) mice compared with wild-type (WT) mice. In TNKO mice, myocardial repair seemed to proceed normally, but the appearance of myofibroblasts was delayed. With cultured cardiac fibroblasts, TN-C significantly accelerated cell migration, α-SMA expression, and collagen gel contraction but did not affect proliferation. Using recombinant fragments of murine TN-C, the functional domain responsible for promoting migration of cardiac fibroblasts was mapped to the conserved fibronectin type III (FNIII)-like repeats and the fibrinogen (Fbg)-like domain. Furthermore, alternatively spliced FNIII and Fbg-like domains proved responsible for the up-regulation of α-SMA expression. These results indicate that TN-C promotes recruitment of myofibroblasts in the early stages of myocardial repair by stimulating cell migration and differentiation.
Tenascin-C (TN-C), an extracellular matrix molecule expressed at high levels during embryonic development and cancer invasive fronts, as well as in response to injury, is known to influence various cell activities.1–4 Each subunit of a hexameric glycoprotein consists of TA (tenascin assembly domain), epidermal growth factor (EGF)-like repeats, fibronectin type III (FN III)-like repeats, and a C-terminal fibrinogen (Fbg)-related domain. Alternative splicing results in several different forms of TN-C, containing variable numbers of FN III repeats. Accumulating results of in vitro studies point to each domain having specific functions, for example in the regulation of cell adhesion, migration, or growth.1–4
In the heart, TN-C is expressed at very early stages of embryonic development,5 is not detected in normal adult myocardium, but is re-expressed in various pathological conditions.6–13 After myocardial infarction, TN-C appears during the acute stages, at the interface between infarcts and intact myocardium.7,8 We previously reported that TN-C may loosen the linkage between cardiomyocytes and connective tissue and thus helps with tissue remodeling at the edges of residual myocardium.8 Furthermore, we found α-smooth muscle actin (α-SMA)-positive myofibroblasts in TN-C-positive areas and that deposition of TN-C precedes their recruitment.8
Myofibroblasts are specialized fibroblasts that share characteristics with smooth muscle cells expressing α-SMA. They play an important role in wound healing by synthesizing collagens and exerting strong contraction forces to minimize wound areas.14–17 It is thought that residential interstitial fibroblasts at the edges of injured tissue differentiate into myofibroblasts and migrate into damaged areas. In the present study, we investigated whether TN-C contributed to myocardial tissue repair, with particular attention to recruitment of myofibroblasts. For this purpose TN-C knockout (TNKO) and wild-type (WT) mice were compared with regard to the healing processes after electrical injury to the myocardium. Furthermore, the effect of TN-C on cell proliferation, migration, and differentiation of cardiac fibroblasts into myofibroblasts was examined in vitro. Functional domains were also determined using recombinant fragments of murine TN-C.
Materials and Methods
TN-C Knockout Mice
The originally produced TN-C knockout mouse18 was backcrossed with BALB/c inbred mice.
Myocardial Injury Model
TNKO and WT mice, 8 weeks old, were handled according to guidelines approved by the Mie University Animal Experiment and Care Committee. Animals were anesthetized by an intraperitoneal injection of 50 mg/kg of sodium pentobarbital and the left diaphragm was exposed through a midline incision in the abdomen. The ventricular wall was injured through the diaphragm by an electric pulse current (160 μA, 0.5 second) using an electric coagulator (OPERER II-F; J. Morita, Tokyo, Japan) that was delivered with a probe of 2 mm in diameter. This procedure caused a transmural injury at the postinferior wall of the left ventricle of ∼2 to 3 mm in diameter. Mice were sacrificed 1, 2, 3, and 5 days after injury. The hearts were removed and fixed in a 4% paraformaldehyde solution at 4°C for 16 hours, embedded in paraffin, and cut into 4-μm-thick sections. To examine cell proliferation, some mice received an intraperitoneal administration of 20 mg/kg of 8-bromodeoxiuridine (BrdU) 30 minutes before sacrifice.
Immunohistochemistry
Production and characterization of an anti-TN-C polyclonal rabbit antibody and mouse monoclonal antibody clones 4F10TT (IBL, Gunma, Japan) and 4C8MS (IBL), as well as the immunostaining procedures with tissue sections, were previously described.8,9,19 4C8MS specifically recognizes the alternative splicing sites, whereas 4F10TT reacts with constitutive sites of TN-C molecules. Sections on slides were incubated with either rabbit polyclonal antibodies (1 μg/ml), 4F10TT (1 μg/ml), or 4C8MS (5 μg/ml), overnight at 4°C, and subsequently with peroxidase-conjugated anti-mouse or anti-rabbit IgG Fab′ (1:500; MBL, Nagoya, Japan) for 1 hour. After washing, diaminobenzidine/H2O2 solution was used to demonstrate antibody binding. The sections were then lightly counterstained with methylgreen or hematoxylin to facilitate orientation. Incorporated BrdU in tissue sections was immunostained according to Yoshimura and colleagues.20 The labeled cells excluding inflammatory cells in the injured areas near the border zone were counted in three different fields of ×40 objective, and the average of each sample (n = 5 for each) was calculated. Myofibroblasts were labeled by a direct immunoperoxidase method with anti-α-SMA antibody (EPOS; Dako Japan, Kyoto, Japan) and the α-SMA-positive cells in the injured areas were also counted. Double immunohistochemistry for TN-C and α-SMA was performed as previously described.8
Purification of TN-C and Its Recombinant Fragments
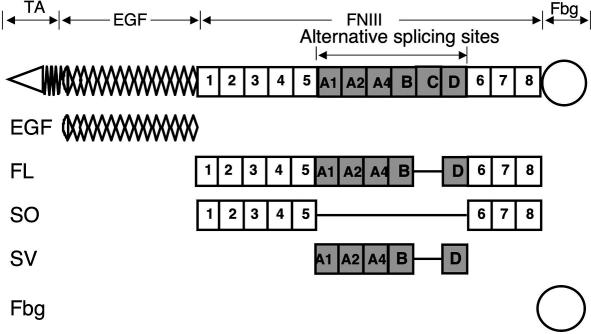
TN-C was purified from conditioned medium of the U-251MG human glioma cell line.21 Recombinant fragments of TN-C (Figure 1): FNIII repeats including the alternative splicing site (FL), FNIII repeats of the alternative splicing site (SV), FNIII repeats without the site (SO), the EGF-like domain, and the fibrinogen (Fbg)-like domain, were obtained from conditioned media of CHO K-1 cells permanently transfected with cDNAs encoding the respective domains, and purified.19
Figure 1.
Diagram of mouse TN-C and its recombinant fragments. FL: FNIII repeats including both conserved (1 to 5, 6 to 9) and alternatively spliced repeats (A1, A2, A4, B, D). SV: Alternatively spliced FNIII repeats. SO: Conserved FNIII repeats. EGF: the EGF-like domain. Fbg: fibrinogen-like domain.
Cell Cultures
Primary cultures of cardiac fibroblasts were obtained from cardiac ventricles of either TNKO or WT mice. Five mice were used for each preparation. The mice were sacrificed by cervical dislocation and the hearts were quickly removed under sterile conditions. Ventricular tissue was excised, thoroughly minced, and digested with 0.1% collagenase (class II; Worthington, Freehold, NJ) in Dulbecco’s phosphate-buffered saline. The isolated cells were collected by centrifugation, resuspended in Iscov’s modified Dulbecco’s medium (IMDM; Sigma, St. Louis, MO) supplemented with 10% fetal bovine serum (FBS; Life Technologies, Inc., Grand Island, NY), and plated on a 75-cm2 Primaria culture flask (Becton Dickinson Labware, Franklin Lakes, NJ). After the cardiac fibroblasts had attached to the bottom surface of the flask, nonadherent cells (primarily myocytes, leukocytes, and endothelial cells) could be washed away. This approach enabled us to obtain virtually pure cultures of fibroblasts. The cells were grown in IMDM with 10% FBS at 37°C in a humidified incubator gassed with 5% CO2. Experiments were performed using cells at the second passage. The effects of intact TN-C and recombinant fragments of TN-C on fibroblasts were examined using the cells from TN-C-null mice to exclude the influence of intrinsically synthesized TN-C.
BrdU Incorporation
Cells (1 × 105) of cardiac fibroblasts from TNKO mice were plated on each well of Falcon four-chamber culture slides (Becton Dickinson Labware), grown in IMDM/10% FBS for 12 hours, washed with serum-free medium, and then incubated in medium with 0.1% FBS for 24 hours. Thereafter, TN-C was added to a final concentration of 1 to 10 μg/ml. After incubation for 12 hours, the cells were labeled with BrdU (10 μg/ml) for 2 hours, fixed with 100% ethanol at −20°C for 30 minutes, and treated with 1 N HCl solution at room temperature for 20 minutes. Labeled nuclei were detected with monoclonal anti-BrdU antibody (Dako Japan) and peroxidase-conjugated goat anti-mouse IgG (MBL). The BrdU-positive nuclei and total number of nuclei (more than 500) were counted and percentage values were determined.
Migration Assays
We first compared the migration of cardiac fibroblasts from TNKO and WT mice, by a transwell migration assay using cell culture inserts (8-μm pore size, Becton Dickinson Labware). Cells (5 × 104 of either type) in 0.5% bovine serum albumin/serum-free IMDM were plated into the inner chamber. The medium, containing 5% FBS as a chemoattractant, was poured into the outer chamber (Falcon 24-well plate, Becton Dickinson Labware). To examine the effect of TN-C on cell migration, intact TN-C (0 to 10 μg/ml) or one of the recombinant fragments (10 μg/ml) was added to medium of the upper chamber of TN-C-null cells. The cells were allowed to migrate to the lower membrane surface for 8 hours. The cells remaining on the upper surface were then wiped off and the inserts were fixed with 100% ethanol and stained with 0.1% crystal violet (Sigma) in 10% ethanol. Stained cells on 1 mm2 of the lower membrane surface were counted under a ×10 objective lens.
Expression of α-SMA
Cardiac fibroblasts from either TNKO or WT mice were plated on Falcon four-chamber culture slides (5 × 104 for each well) in IMDM/10% FBS for 3 hours, washed with serum-free medium, then incubated in medium with 0.1% FBS for 24 hours. Thereafter, intact TN-C (5 to 20 μg/ml) or one of the recombinant fragments (10 μg/ml) was added to TN-C-null fibroblasts. After incubation for 24 hours, the cells were fixed with 4% paraformaldehyde and labeled with antibodies against α-SMA (Sigma) for 1.5 hours, then exposed to secondary antibodies (fluorescein isothiocyanate-conjugated anti-mouse IgG, MBL) and rhodamine-phalloidin (Molecular Probes, Eugene, OR) for 1 hour. Cells were examined with a ×20 objective lens by epifluorescence microscopy (BX-50; Olympus, Tokyo, Japan). Polyclonal monospecific antibodies against either TN-C or calsequestrin were affinity-purified with antigen-conjugated columns. Calsequestrin is protein of the sarcoplasmic reticulum, and anti-calsequestrin antibody was used as a control IgG. Purified TN-C (10 μg/ml) was incubated with either antibodies (10 μg/ml) at room temperature for 30 minutes, and then added to the culture medium.
Collagen Gel Contraction Assay
Cellular collagen gel contraction assays were performed in Falcon 24-well plates. A collagen solution in IMDM with 0.1% FBS was prepared from porcine collagen I according to the manufacturer’s instructions (Nitta Gelatin, Osaka, Japan) and combined with cardiac fibroblasts of either TNKO or WT mice at 2 × 105/ml. Five hundred μl of the collagen/cell mixture (final collagen concentration, 1.0 mg/ml) was plated into each well and allowed to polymerize at 37°C. After incubation for 24 hours, 0.5 ml of IMDM containing 0.1% FBS with or without TN-C (10 μg/ml) was added to each well, and the gels were carefully detached from the dishes. The mean size of the gel (n = 3 for each sample) was taken at each time point with the aid of a flatbed scanner (Epson, Tokyo, Japan). To estimate the contractility of the cells, the size of the gel was subtracted from the starting area and expressed as a percentage. This experiment was repeated three times.
Results
Wound Healing and Expression of TN-C in Injured Mouse Myocardium
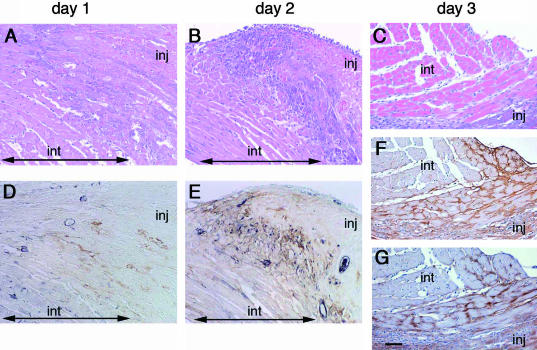
In WT mice, coagulation necrosis of cardiomyocytes and edema around lesions were apparent 1 day after injury (Figure 2A), becoming more pronounced on day 2 (Figure 2B). On day 3, granulation tissues began to be formed in the border zone and eventually replaced necrotic masses (Figure 2C). TN-C began to be expressed within 1 day after injury at the border between intact myocardial tissues and necrotic areas (Figure 2D) and was gradually deposited in developing granulation tissue (Figure 2, E and F). Using the monoclonal antibody 4C8MS, it was confirmed that the deposited molecules contained the large splice variants (Figure 2G). In TNKO mice, myocardial healing appeared to proceed normally and there was no obvious difference from WT mice on routine histological analyses (Figure 3).
Figure 2.
Tissue healing and TN-C expression after electrical injury of the myocardium at day 1 (A, D), day 2 (B, E), and day 3 (C, F, G). A–C: H&E staining; D, E: double immunolabeling for α-SMA (blue) and TN-C (brown). F and G: Immunolabeling with the antibody clone 4F10TT reacting with constitutive sites of TN-C (F), and 4C8MS that specifically recognizes an alternative spliced repeat of TN-C (G). On day 1, TN-C deposition is clearly detectable in the interstitial spaces of border zone myocardium, but only vascular walls are positive for α-SMA (D). E: On day 2, myofibroblasts are apparent in TN-C-positive areas. G: Note staining for large splice variants of TN-C. int, intact area; inj, injured area. Scale bar, 50 μm.
Figure 3.
Comparison of the histopathology of myocardial repair in a WT (A) and TN-C knockout mouse (B) 5 days after electric injury. H&E staining. Scale bar, 50 μm.
Recruitment of Myofibroblasts in Granulation Tissue of the Heart
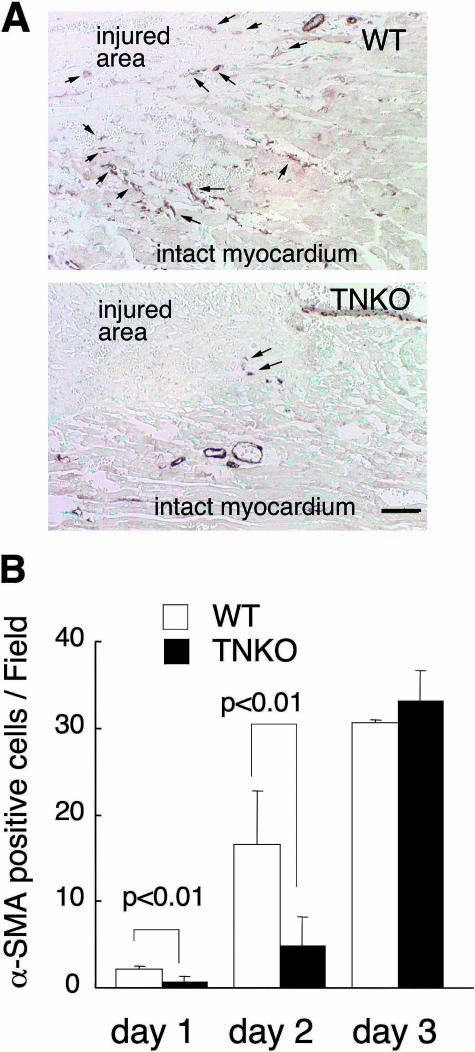
Immunostaining of α-SMA of WT mice showed that on day 1, only vascular walls were positive and few myofibroblasts were present, although TN-C deposition was clearly detectable (Figure 2D). On day 2, myofibroblasts emerged in TN-C-positive areas of the interstitium of the border zone (Figure 2E), and increased in number on day 3, moving into necrotic areas. The number of myofibroblasts per optic field in WT mice on days 1, 2, and 3 were 2.2 ± 0.3, 16.6 ± 6.2, and 30.6 ± 0.3, respectively (Figure 4). In TNKO mice, they were 0.7 ± 0.6, 4.9 ± 3.3, and 33.2 ± 3.5. On days 1 and 2, myofibroblasts in WT mice were significantly more numerous than in TNKO mice (P < 0.01), but the difference disappeared on day 3 (Figure 4).
Figure 4.
Myofibroblasts in the border zones of necrotic cardiac tissue. A: Myofibroblasts labeled with anti-α-SMA antibody. On day 2, α-SMA-positive cells are evident (arrows) in the myocardial interstitium near the necrotic area in a WT mouse. Fewer cells are apparent in a TN-C knockout (TNKO) mouse. B: α-SMA-positive cells were counted in three fields of view under a ×40 objective and the average of each sample was calculated. On days 1 and 2, the myofibroblasts in WT mice were more frequent than in TNKO mice (P < 0.01), but there was no difference on day 3. The data are averages and SDs of results from five animals. Scale bar, 50 μm.
Cell Proliferation in Granulation Tissue of the Heart
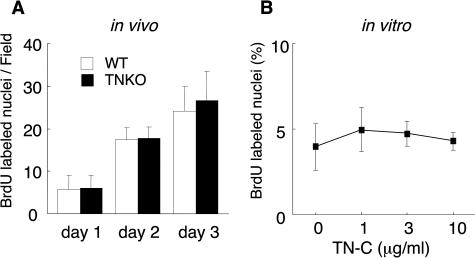
Since it is well known that TN-C enhances proliferation of some cell types, we examined DNA synthesis in granulation tissue by BrdU incorporation assay. The percentages of BrdU-labeled nuclei in WT mice on days 1, 2, and 3 were 5.7 ± 3.4%, 17.5 ± 2.7%, and 24.2 ± 5.9%, respectively, whereas in TNKO mice they were 6.1 ± 2.9%, 17.7 ± 2.8%, and 26.6 ± 7.0%, with no significant differences between the two animal groups (Figure 5A).
Figure 5.
Effect of TN-C on proliferation of cardiac fibroblasts in vivo and in vitro. A: BrdU-labeled cells in the injured areas of WT and TNKO mice. BrdU-positive nuclei were counted in three fields of view of a ×40 objective and the average of each sample was calculated. The data are expressed as averages and SDs of results from five animals. Note the lack of significant differences between WT and TNKO. B: Cells isolated from TNKO mice were plated and grown on culture glass slides. After serum starvation for 24 hours, TN-C was added. Cells labeled with BrdU were visualized by immunocytochemistry and BrdU-positive and total number of nuclei (more than 500) were counted and percentage values generated. The data are averages and SDs from three independent experiments. Note the lack of any increase with TN-C treatment.
Effect of TN-C on Proliferation, Migration, and Differentiation of Cultured Cardiac Fibroblasts in Vitro
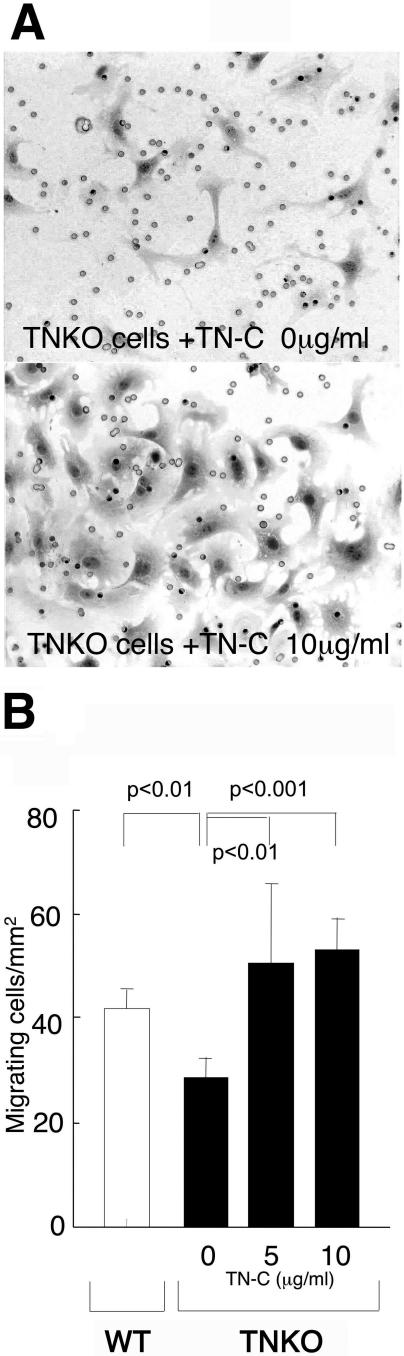
To confirm the in vivo results, we first investigated the effect of TN-C on cell proliferation of cultured cardiac fibroblasts from TNKO mice by BrdU incorporation assay. Addition of TN-C in various concentrations to the fibroblasts was without significant effect (Figure 5B). In transwell migration assays, the number of TN-C-null fibroblasts that migrated through insert membranes was significantly lower than those from the WT mice (28.8 ± 3.7 cells/mm2 versus 42.0 ± 3.6 cells/mm2, P < 0.01). Addition of TN-C to TN-C-null cells at the concentration of 5 and 10 μg/ml, significantly increased the number of migrated cells to 50.8 ± 12.3 cells/mm2 and 53.1 ± 5.2 cells/mm2, respectively (P < 0.01 and P < 0.001, Figure 6).
Figure 6.
Effects of TN-C on migration of cardiac fibroblasts in a transmigration assay. Cells isolated from cardiac ventricles of either TNKO or WT mice were plated on the culture inserts, treated with TN-C (5 or 10 μg/ml) or without TN-C, and allowed to migrate for 8 hours. A: Cells migrating through the membrane were stained with 0.1% crystal violet. B: Migration of TN-C-null cells was significantly lower than that of WT-cells, and addition of TN-C significantly enhanced cell migration in a dose-dependent manner. Cells were counted in three fields of view of 1 mm2 in each insert, and the data are averages and SDs of results from three independent experiments.
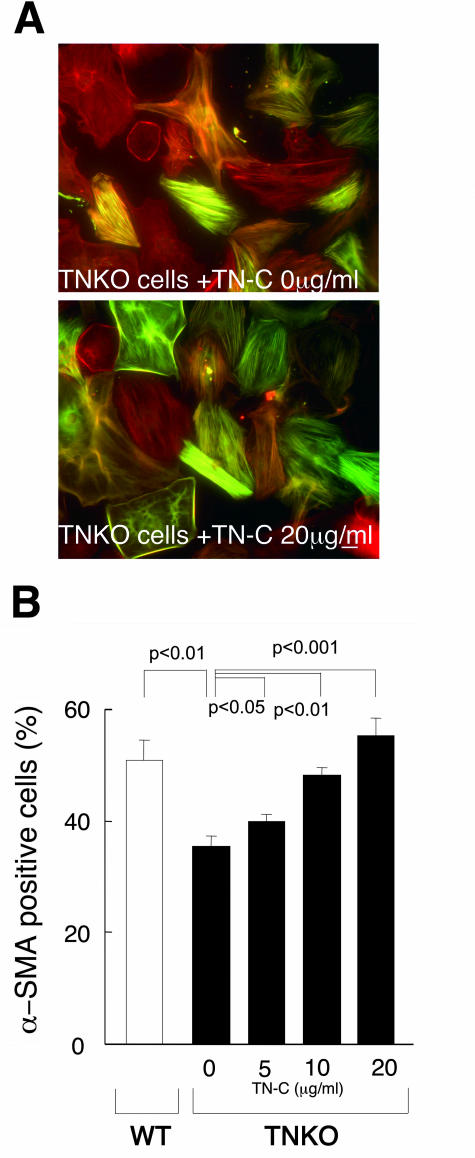
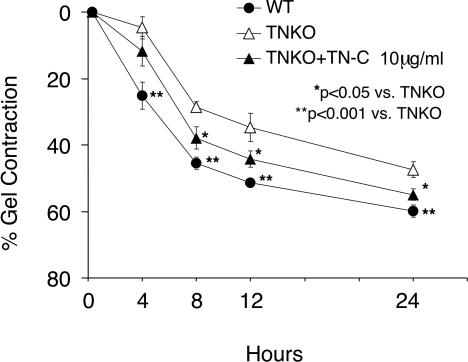
Differentiation to myofibroblasts was assessed by expression of α-SMA (Figure 7) and collagen gel contraction assay (Figure 8). Cardiac fibroblasts in culture from TNKO mice showed well developed stress fibers and some cells expressed α-SMA (Figure 7A). However, the percentage of α-SMA-positive cells was significantly lower than that of WT cardiac fibroblasts (35.4 ± 2.03% versus 50.8 ± 3.7%, P < 0.01). Addition of TN-C significantly increased the α-SMA-positive ratio of the TN-C-null fibroblasts in a dose-dependent manner (Figure 7B). To confirm this result, we tested whether addition of an antibody against TN-C blocked this effect and found that treatment with the TN-C antibody (95 ± 5%) restored the α-SMA-positive cells to the levels in fibroblasts that were cultured without TN-C (100 ± 5%), while addition of control IgG had no effect (117 ± 8%). Both TN-C-null and WT cells contracted collagen gels, but contraction by TN-C-null cells was significantly weaker than those by WT cells. Addition of TN-C significantly increased the gel contraction of TN-C-null fibroblasts (Figure 8).
Figure 7.
Effects of TN-C on α-SMA expression in cardiac fibroblasts. Cells isolated from cardiac ventricles of either TNKO or WT mice were plated and grown on the culture glass slides. After 24-hour serum starvation, TN-C (0 or 20 μg/ml) was added to TN-C-null cells and incubated for another 24 hours. A: α-SMA expression was detected by indirect immunofluorescent staining with fluorescein isothiocyanate-conjugated secondary antibody, and all cells were counterstained with rhodamine-phalloidin. The α-SMA-positive ratio of TN-C-null cells was significantly lower than that of WT cells. B: Addition of TN-C significantly up-regulated α-SMA expression in a dose-dependent manner. The α-SMA-positive and total cells (more than 200) were counted to allow generation of percentage values. The data are averages and SDs of results from three independent experiments.
Figure 8.
Effects of TN-C on collagen gel contraction by cardiac fibroblasts. Cardiac fibroblasts of either TNKO or WT mice were combined with collagen gel mixture and allowed to polymerize. After 24 hours medium with or without TN-C (10 μg/ml) was added, then the gels were detached from the dish. Percentages of the contraction were measured at 4, 8, 12, and 24 hours. Contraction by WT cells (closed circle) was significantly stronger than that by TN-C-null cells (open triangle) at each time point. Addition of TN-C (closed triangle) significantly increased the gel contraction of TNKO fibroblasts at 8, 12, and 24 hours. The data are expressed as averages and SDs of triplicate samples.
Functional Domains of TN-C on Migration and α-SMA Expression of Cardiac Fibroblasts
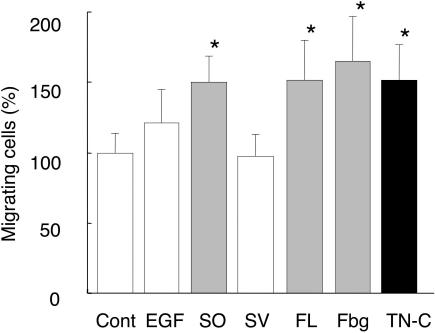
Having shown that TN-C promoted migration and α-SMA expression, we tried to determine the responsible functional domains of TN-C, using the following recombinant fragments: FNIII repeats including alternative splicing sites (FL), conserved FNIII repeats (SO), alternative splice sites (SV), the EGF-like domain, and the fibrinogen (Fbg)-like domain (Figure 1). On addition to fibroblast cultures, FL and SO increased the numbers of migrating cells to 151 ± 28% and 151 ± 18%, respectively, of the control value. The Fbg-like domain also caused a 165 ± 32% elevation, while addition of neither EGF nor SV fragments had significant effects (Figure 9). Therefore, the conserved FNIII repeats and the Fbg-like domain may have the ability to promote migration of fibroblasts.
Figure 9.
Determination of functional domains promoting cell migration using recombinant fragments of TN-C. TN-C-null cardiac fibroblasts were plated on culture inserts and treated with TN-C (10 μg/ml) or TN-C fragments (10 μg/ml), and allowed to migrate for 8 hours. The cells were counted in three fields of view of 1 mm2 in each insert, and the data are averages and SDs of results from six independent experiments, relative to the control without TN-C. *P < 0.01.
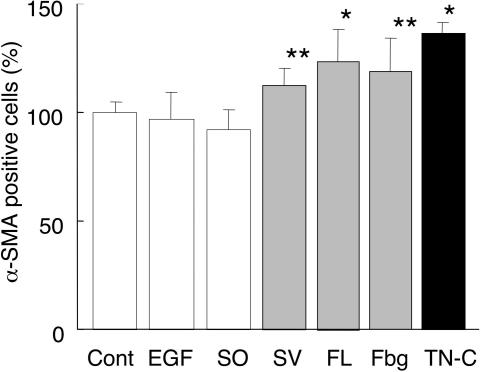
FL also augmented the proportion of α-SMA-positive fibroblasts (123 ± 15%) as compared with the controls (Figure 10). Although SO did not exert any apparent influence, SV also caused a significant increase, along with the Fbg-like domain (118 ± 16%). Addition of EGF was without effect. Therefore, alternatively spliced FNIII repeats and the Fbg-like domain may thus induce differentiation from fibroblasts to myofibroblasts.
Figure 10.
Determination of functional domains up-regulating α-SMA expression using recombinant fragments of TN-C. TN-C-null cardiac fibroblasts were plated and grown on the culture glass slides. After 24-hour serum starvation, they were treated with TN-C (10 μg/ml) or TN-C fragments (10 μg/ml), and incubated for 24 hours. α-SMA expression was detected by immunofluorescence. α-SMA-positive cells and total number of cells (more than 200) were counted and the percentage values were generated. The data are averages and SDs of results from six independent experiments, relative to the control without TN-C. *P < 0.01, **P < 0.05.
Discussion
TN-C has been proposed to play significant roles in wound healing in various tissues because of its dramatic increase after injury.22 Although TN-C knockout mice were initially reported to be phenotypically normal,18 several reports have documented abnormal tissue restoration after injury to the cornea,23 skin,24 or after habu venom-induced glomerulonephritis.25,26 Using different models, others have reported no significant differences in the wound healing on morphological analysis,27,28 but deposition of fibronectin was found to be reduced in the granulation tissue of skin wounds in TN-C-null mice.27 In vitro, TN-C promotes epithelial cell migration and proliferation,19 which could be partly responsible for abnormal repair of epithelial tissue in TNKO mice.
Because of the limited ability for regeneration of cardiomyocytes, myocardial wound healing mostly depends on interstitial fibroblasts, which form and contract granulation and scar tissue via multiple cell activities.29 Especially, myofibroblasts, specialized fibroblasts, are key players in the myocardial repair.30–32 In the present study, we found that their appearance in injured myocardium was clearly delayed in TN-C knockout mice, indicating that TN-C promotes recruitment of myofibroblasts.
On tissue injury, primarily in response to mechanical stress, interstitial fibroblasts evolve into proto-myofibroblasts with a contractile microfilamentous apparatus, then, with various stimuli, into differentiated myofibroblasts characterized by expression of α-SMA. These then migrate into the injured areas, generate contraction forces, synthesize, organize, and degrade collagen and other ECMs, and finally results in a shortening of collagen matrix with corresponding wound closure.14,15,33,34
A close relationship between TN-C and myofibroblasts in cancer stroma or normal tissue has been reported.35–37 In cancer stroma, myofibroblasts have often been identified as the source of TN-C deposits38 and it is supposed that they may provide a proinvasive signal to cancer cells mediated with TN-C.39 By careful sequential observation in a rat model after myocardial infarction, we have found that, initially α-SMA-negative interstitial cells in the border zones express TN-C, and then myofibroblasts appear in the TN-C-positive areas.8 Similar results were observed in mouse myocardial wound healing in this study. It seems likely that interstitial cells synthesize TN-C, which, in an autocrine and paracrine manner, then causes interstitial cells to change their phenotype to differentiated myofibroblasts and promotes migration into damaged areas.
Our in vitro findings support this possibility. Cardiac fibroblasts from TNKO mice showed lower cell migration and α-SMA expression than WT cells that synthesize TN-C in culture. Addition of TN-C to TN-C-null cells recovered both cell migration and α-SMA expression. Expression of α-SMA is a critical step for myofibroblast differentiation and functionally important for force generation.40,41 Although gel contraction could be regulated by various factors including differentiation of myofibroblasts, cell-matrix adhesion, and maturation of cytoskeletal contractile apparatus,34,42–44 the increment of contraction by addition of TN-C in our experiment should partly reflect the up-regulation of α-SMA expression. Although TN-C is well known to stimulate migration of various types of cells, it has also been suggested to modulate differentiation of cells.45–47
Functional domains responsible for differentiation into myofibroblasts were mapped to alternative spliced FNIII repeats, but not the conserved repeats, and the Fbg-like domain of TN-C. In contrast, the responsible domain for promoting migration of cardiac fibroblasts was mapped to the conserved FNIII repeats and the Fbg-like domain. Several receptors might bind to the respective domains of TN-C and transmit multiple signals that could trigger various cellular functions.
Interestingly, using the same recombinant fragments and the same assays, our previous study demonstrated that the alternative spliced FNIII domains promoted migration of breast cancer cells, but the conserved repeats and Fbg domain did not.19 The conserved repeats of FNIII 3 and 6 have similarly been demonstrated to be involved in glioma migration.48 It has also been reported that the Fbg-like domain, but not the entire FNIII repeats, can mediate migration of aortic smooth muscle cells on a TN-C substrate.49 Conversely, a recombinant fragment of alternatively spliced FNIII A to D, but not the conserved FN III repeats or the Fbg domain, was found to enhance migration of endothelial cells.50 Therefore, cell-type specificity is apparent. A number of molecules, such as members of the integrin family, annexin II, EGF receptor, cell adhesion molecules (CAMs), syndecan, and phosphacan/receptor-type protein tyrosine phosphatase ζ/β, have been reported as receptors for TN-C.3 Moreover, TN-C may also regulate cell behavior by direct or indirect modulation of other ECM proteins. For example, it has been reported that TN-C may inhibit fibronectin initiating signaling by interfering fibronectin-syndecan 4 binding.44,51,52 Therefore, functional activities provided by TN-C could occur through different mechanisms, possibly using separate receptors depending on the cell.
In this study, we demonstrated that TN-C plays critical roles in stimulating myofibroblasts in response to injury. However, after 3 days there was no longer any difference in myofibroblasts in TN-C-null and control mice, and myocardial healing did not show distinct morphological differences so that a compensatory mechanism must exist. Indeed, TN-C is not the only factor that controls behavior of myofibroblasts. For example, the transition between proto-myofibroblasts and differentiated myofibroblasts is known to be stimulated by many factors, such as various cytokines, growth factors, endothelin 1, angiotensin II, and newly synthesized extracellular matrix proteins.16 It is well established that the combined action of the splice variant of cellular fibronectin containing ED-A and transforming growth factor-β is important in this regard.53,54 In cancer tissues, TN-C is often co-expressed with fibronectin containing ED-A,37,55 and also can enhance the functions of transforming growth factor-β in cancer cells.56 Therefore, TN-C might control behavior of myofibroblasts in a complex way, collaborating and interfering with many other factors.
Nevertheless, it is clear that TN-C is a major factor for recruitment of myofibroblasts in early stages of myocardial tissue repair. Although this would increase contraction forces to prevent ventricular dilatation, TN-C also has been reported to inhibit cell contraction by suppressing focal adhesion kinase and Rho A activity in fibrinogen-fibronectin matrix.43,44 Furthermore, TN-C may loosen adhesion of cardiomyocytes8 and stimulate MMP expression in some types of cells.56 Although these effects, as well as integrin shedding, should be beneficial for cells to modify cell-ECM interaction during tissue remodeling,57 they also have potential to cause slippage of myocytes resulting in ventricular dilatation. Therefore, TN-C could be a key molecule in controlling the balance of beneficial and undesirable cellular responses in cardiac remodeling.
Acknowledgments
We thank M. Hara and M. Namikata for their technical assistance.
Footnotes
Address reprint requests to Dr. Kyoko Imanaka-Yoshida, Department of Pathology and Biology, Mie University Graduate School of Medicine, 2-174 Edobashi, Tsu, Mie 514-8507, Japan. E-mail: imanaka@doc.medic.mie-u.ac.jp.
Supported by the Ministry of Education, Culture, Sports, Science, and Technology of Japan (grant-in-aid for scientific research no. 15590735 to K.I.-Y); and the Ministry of Health, Labor, and Welfare of Japan (grant for intractable disease to K.I.-Y. and M.H.).
References
- Chiquet-Ehrismann R, Chiquet M. Tenascins: regulation and putative functions during pathological stress. J Pathol. 2003;200:488–499. doi: 10.1002/path.1415. [DOI] [PubMed] [Google Scholar]
- Chiquet-Ehrismann R, Tucker RP. Connective tissues: signalling by tenascins. Int J Biochem Cell Biol. 2004;36:1085–1089. doi: 10.1016/j.biocel.2004.01.007. [DOI] [PubMed] [Google Scholar]
- Jones FS, Jones PL. The tenascin family of ECM glycoproteins: structure, function, and regulation during embryonic development and tissue remodeling. Dev Dyn. 2000;218:235–259. doi: 10.1002/(SICI)1097-0177(200006)218:2<235::AID-DVDY2>3.0.CO;2-G. [DOI] [PubMed] [Google Scholar]
- Jones PL, Jones FS. Tenascin-C in development and disease: gene regulation and cell function. Matrix Biol. 2000;19:581–596. doi: 10.1016/s0945-053x(00)00106-2. [DOI] [PubMed] [Google Scholar]
- Imanaka-Yoshida K, Matsumoto K, Hara M, Sakakura T, Yoshida T. The dynamic expression of tenascin-C and tenascin-X during early heart development. Differentiation. 2003;71:291–298. doi: 10.1046/j.1432-0436.2003.7104506.x. [DOI] [PubMed] [Google Scholar]
- Tamura A, Kusachi S, Nogami K, Yamanishi A, Kajikawa Y, Hirohata S, Tsuji T. Tenascin expression in endomyocardial biopsy specimens in patients with dilated cardiomyopathy: distribution along margins of fibrotic lesions. Heart. 1996;75:291–294. doi: 10.1136/hrt.75.3.291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Willems IE, Arends JW, Daemen MJ. Tenascin and fibronectin expression in healing human myocardial scars. J Pathol. 1996;179:321–325. doi: 10.1002/(SICI)1096-9896(199607)179:3<321::AID-PATH555>3.0.CO;2-8. [DOI] [PubMed] [Google Scholar]
- Imanaka-Yoshida K, Hiroe M, Nishikawa T, Ishiyama S, Shimojo T, Ohta Y, Sakakura T, Yoshida T. Tenascin-C modulates adhesion of cardiomyocytes to extracellular matrix during tissue remodeling after myocardial infarction. Lab Invest. 2001;81:1015–1024. doi: 10.1038/labinvest.3780313. [DOI] [PubMed] [Google Scholar]
- Imanaka-Yoshida K, Hiroe M, Yasutomi Y, Toyozaki T, Tsuchiya T, Noda N, Maki S, Nishikawa T, Sakakura T, Yoshida T. Tenascin-C is a useful marker for disease activity in myocarditis. J Pathol. 2002;197:388–394. doi: 10.1002/path.1131. [DOI] [PubMed] [Google Scholar]
- Imanaka-Yoshida K, Hiroe M, Yoshida T. Interaction between cell and extracellular matrix in heart disease: multiple roles of tenascin-C in tissue remodeling. Histol Histopathol. 2004;19:517–525. doi: 10.14670/HH-19.517. [DOI] [PubMed] [Google Scholar]
- Sato M, Toyozaki T, Odaka K, Uehara T, Arano Y, Hasegawa H, Yoshida K, Imanaka-Yoshida K, Yoshida T, Hiroe M, Tadokoro H, Irie T, Tanada S, Komuro I. Detection of experimental autoimmune myocarditis in rats by 111In monoclonal antibody specific for tenascin-C. Circulation. 2002;106:1397–1402. doi: 10.1161/01.cir.0000027823.07104.86. [DOI] [PubMed] [Google Scholar]
- Frangogiannis N, Shimoni S, Chang SM, Ren G, Dewald O, Gersch C, Espada R, Mahesh R, Entman ML, Zoghbi WA. Active interstitial remodeling: an important process in the hibernating human myocardium. J Am Coll Cardiol. 2002;39:1468–1474. doi: 10.1016/s0735-1097(02)01792-8. [DOI] [PubMed] [Google Scholar]
- Morimoto S, Imanaka-Yoshida K, Hiramitsu S, Kato S, Ohtsuki M, Uemura A, Kato Y, Nishikawa T, Toyozaki T, Hishida H, Yoshida T, Hiroe M. The diagnostic utility of tenascin-C for evaluation of activity of human acute myocarditis. J Pathol. 2005;205:460–467. doi: 10.1002/path.1730. [DOI] [PubMed] [Google Scholar]
- Gabbiani G. Evolution and clinical implications of the myofibroblast concept. Cardiovasc Res. 1998;38:545–548. doi: 10.1016/s0008-6363(98)00065-0. [DOI] [PubMed] [Google Scholar]
- Gabbiani G. The myofibroblast in wound healing and fibrocontractive diseases. J Pathol. 2003;200:500–503. doi: 10.1002/path.1427. [DOI] [PubMed] [Google Scholar]
- Serini G, Gabbiani G. Mechanisms of myofibroblast activity and phenotypic modulation. Exp Cell Res. 1999;250:273–283. doi: 10.1006/excr.1999.4543. [DOI] [PubMed] [Google Scholar]
- Powell DW. Myofibroblasts: paracrine cells important in health and disease. Trans Am Clin Climatol Assoc. 2000;111:271–292. [PMC free article] [PubMed] [Google Scholar]
- Saga Y, Yagi T, Ikawa Y, Sakakura T, Aizawa S. Mice develop normally without tenascin. Gene Dev. 1992;6:1821–1831. doi: 10.1101/gad.6.10.1821. [DOI] [PubMed] [Google Scholar]
- Tsunoda T, Inada H, Kalembeyi I, Imanaka-Yoshida K, Sakakibara M, Okada R, Katsuta K, Sakakura T, Majima Y, Yoshida T. Involvement of large tenascin-C splice variants in breast cancer progression. Am J Pathol. 2003;162:1857–1867. doi: 10.1016/S0002-9440(10)64320-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshimura E, Majima A, Sakakura Y, Sakakura T, Yoshida T. Expression of tenascin-C and the integrin alpha 9 subunit in regeneration of rat nasal mucosa after chemical injury: involvement in migration and proliferation of epithelial cells. Histochem Cell Biol. 1999;111:259–264. doi: 10.1007/s004180050356. [DOI] [PubMed] [Google Scholar]
- Yoshida T, Yoshimura E, Numata H, Sakakura Y, Sakakura T. Involvement of tenascin-C in proliferation and migration of laryngeal carcinoma cells. Virchows Arch. 1999;435:496–500. doi: 10.1007/s004280050433. [DOI] [PubMed] [Google Scholar]
- Mackie EJ, Halfter W, Liverani D. Induction of tenascin in healing wounds. J Cell Biol. 1988;107:2757–2767. doi: 10.1083/jcb.107.6.2757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsuda A, Yoshiki A, Tagawa Y, Matsuda H, Kusakabe M. Corneal wound healing in tenascin knockout mouse. Invest Ophthalmol Vis Sci. 1999;40:1071–1080. [PubMed] [Google Scholar]
- Koyama Y, Kusubata M, Yoshiki A, Hiraiwa N, Ohashi T, Irie S, Kusakabe M. Effect of tenascin-C deficiency on chemically induced dermatitis in the mouse. J Invest Dermatol. 1998;111:930–935. doi: 10.1046/j.1523-1747.1998.00401.x. [DOI] [PubMed] [Google Scholar]
- Nakao N, Hiraiwa N, Yoshiki A, Ike F, Kusakabe M. Tenascin-C promotes healing of Habu-snake venom-induced glomerulonephritis: studies in knockout congenic mice and in culture. Am J Pathol. 1998;152:1237–1245. [PMC free article] [PubMed] [Google Scholar]
- Matsumoto K, Hiraiwa N, Yoshiki A, Ohnishi M, Kusakabe M. PDGF receptor-a deficiency in glomerular mesangial cells of tenascin-C knockout mice. Biochem Biophys Res Commun. 2002;290:1220–1227. doi: 10.1006/bbrc.2001.6316. [DOI] [PubMed] [Google Scholar]
- Forsberg E, Hirsch E, Frohlich L, Meyer M, Ekblom P, Aszodi A, Werner S, Fassler R. Skin wounds and severed nerves heal normally in mice lacking tenascin-C. Proc Natl Acad Sci USA. 1996;93:6594–6599. doi: 10.1073/pnas.93.13.6594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iglesia DD, Gala PH, Qiu T, Stepp MA. Integrin expression during epithelial migration and restratification in the tenascin-C-deficient mouse cornea. J Histochem Cytochem. 2000;48:363–376. doi: 10.1177/002215540004800306. [DOI] [PubMed] [Google Scholar]
- Camelliti P, Borg TK, Kohl P. Structural and functional characterisation of cardiac fibroblasts. Cardiovasc Res. 2005;65:40–51. doi: 10.1016/j.cardiores.2004.08.020. [DOI] [PubMed] [Google Scholar]
- Cleutjens JPM, Blankesteijn WM, Daemen MJAP, Smits JFM. The infarcted myocardium: simply dead tissue, or a lively target for therapeutic interventions. Cardiovasc Res. 1999;44:232–241. doi: 10.1016/s0008-6363(99)00212-6. [DOI] [PubMed] [Google Scholar]
- Heeneman S, Cleutjens JP, Faber BC, Creemers EE, van Suylen RJ, Lutgens E, Cleutjens KB, Daemen MJ. The dynamic extracellular matrix: intervention strategies during heart failure and atherosclerosis. J Pathol. 2003;200:516–525. doi: 10.1002/path.1395. [DOI] [PubMed] [Google Scholar]
- Virag JI, Murry CE. Myofibroblast and endothelial cell proliferation during murine myocardial infarct repair. Am J Pathol. 2003;163:2433–2440. doi: 10.1016/S0002-9440(10)63598-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tomasek JJ, Gabbiani G, Hinz B, Chaponnier C, Brown RA. Myofibroblasts and mechano-regulation of connective tissue remodelling. Nat Rev Mol Cell Biol. 2002;3:349–363. doi: 10.1038/nrm809. [DOI] [PubMed] [Google Scholar]
- Grinnell F. Fibroblast biology in three-dimensional collagen matrices. Trends Cell Biol. 2003;13:264–269. doi: 10.1016/s0962-8924(03)00057-6. [DOI] [PubMed] [Google Scholar]
- Ramadori G, Schwogler S, Veit T, Rieder H, Chiquet-Ehrismann R, Mackie EJ, Meyer zum Buschenfelde KH. Tenascin gene expression in rat liver and in rat liver cells. In vivo and in vitro studies. Virchows Arch B Cell Pathol Incl Mol Pathol. 1991;60:145–153. doi: 10.1007/BF02899540. [DOI] [PubMed] [Google Scholar]
- Kalembeyi I, Yoshida T, Iriyama K, Sakakura T. Analysis of tenascin mRNA expression in the murine mammary gland from embryogenesis to carcinogenesis: an in situ hybridization study. Int J Dev Biol. 1997;41:569–573. [PubMed] [Google Scholar]
- Yoshida T, Matsumoto E, Hanamura N, Kalembeyi I, Katsuta K, Ishihara A, Sakakura T. Co-expression of tenascin and fibronectin in epithelial and stromal cells of benign lesions and ductal carcinomas in the human breast. J Pathol. 1997;182:421–428. doi: 10.1002/(SICI)1096-9896(199708)182:4<421::AID-PATH886>3.0.CO;2-U. [DOI] [PubMed] [Google Scholar]
- Hanamura N, Yoshida T, Matsumoto E, Kawarada Y, Sakakura T. Expression of fibronectin and tenascin-C mRNA by myofibroblasts, vascular cells and epithelial cells in human colon adenomas and carcinomas. Int J Cancer. 1997;73:10–15. doi: 10.1002/(sici)1097-0215(19970926)73:1<10::aid-ijc2>3.0.co;2-4. [DOI] [PubMed] [Google Scholar]
- De Wever O, Nguyen QD, Van Hoorde L, Bracke M, Bruyneel E, Gespach C, Mareel M. Tenascin-C and SF/HGF produced by myofibroblasts in vitro provide convergent pro-invasive signals to human colon cancer cells through RhoA and Rac. FASEB J. 2004;18:1016–1018. doi: 10.1096/fj.03-1110fje. [DOI] [PubMed] [Google Scholar]
- Hinz B, Dugina V, Ballestrem C, Wehrle-Haller B, Chaponnier C. {Alpha}-smooth muscle actin is crucial for focal adhesion maturation in myofibroblasts. Mol Biol Cell. 2003;14:2508–2519. doi: 10.1091/mbc.E02-11-0729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hinz B, Celetta G, Tomasek JJ, Gabbiani G, Chaponnier C. Alpha-smooth muscle actin expression upregulates fibroblast contractile activity. Mol Biol Cell. 2001;12:2730–2741. doi: 10.1091/mbc.12.9.2730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanekar S, Borg TK, Terracio L, Carver W. Modulation of heart fibroblast migration and collagen gel contraction by IGF-I. Cell Adhes Commun. 2000;7:513–523. doi: 10.3109/15419060009040308. [DOI] [PubMed] [Google Scholar]
- Midwood KS, Schwarzbauer JE. Tenascin-C modulates matrix contraction via focal adhesion kinase- and Rho-mediated signaling pathways. Mol Biol Cell. 2002;13:3601–3613. doi: 10.1091/mbc.E02-05-0292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Midwood KS, Valenick LV, Hsia HC, Schwarzbauer JE. Coregulation of fibronectin signaling and matrix contraction by tenascin-C and syndecan-4. Mol Biol Cell. 2004;15:5670–5677. doi: 10.1091/mbc.E04-08-0759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wirl G, Hermann M, Ekblom P, Fassler R. Mammary epithelial cell differentiation in vitro is regulated by an interplay of EGF action and tenascin-C downregulation. J Cell Sci. 1995;108:2445–2456. doi: 10.1242/jcs.108.6.2445. [DOI] [PubMed] [Google Scholar]
- Dandachi N, Hauser-Kronberger C, More E, Wiesener B, Hacker GW, Dietze O, Wirl G. Co-expression of tenascin-C and vimentin in human breast cancer cells indicates phenotypic transdifferentiation during tumour progression: correlation with histopathological parameters, hormone receptors, and oncoproteins. J Pathol. 2001;193:181–189. doi: 10.1002/1096-9896(2000)9999:9999<::AID-PATH752>3.0.CO;2-V. [DOI] [PubMed] [Google Scholar]
- Garcion E, Halilagic A, Faissner A, French-Constant C. Generation of an environmental niche for neural stem cell development by the extracellular matrix molecule tenascin C. Development. 2004;131:3423–3432. doi: 10.1242/dev.01202. [DOI] [PubMed] [Google Scholar]
- Phillips GR, Krushel LA, Crossin KL. Domains of tenascin involved in glioma migration. J Cell Sci. 1998;111:1095–1104. doi: 10.1242/jcs.111.8.1095. [DOI] [PubMed] [Google Scholar]
- LaFleur DW, Chiang J, Fagin JA, Schwartz SM, Shah PK, Wallner K, Forrester JS, Sharifi BG. Aortic smooth muscle cells interact with tenascin-C through its fibrinogen-like domain. J Biol Chem. 1997;272:32798–32803. doi: 10.1074/jbc.272.52.32798. [DOI] [PubMed] [Google Scholar]
- Chung CY, Murphy-Ullrich JE, Erickson HP. Mitogenesis, cell migration, and loss of focal adhesions induced by tenascin-C interacting with its cell surface receptor, annexin II. Mol Biol Cell. 1996;7:883–892. doi: 10.1091/mbc.7.6.883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang W, Chiquet-Ehrismann R, Moyano JV, Garcia-Pardo A, Orend G. Interference of tenascin-C with syndecan-4 binding to fibronectin blocks cell adhesion and stimulates tumor cell proliferation. Cancer Res. 2001;61:8586–8594. [PubMed] [Google Scholar]
- Orend G, Huang W, Olayioye MA, Hynes NE, Chiquet-Ehrismann R. Tenascin-C blocks cell-cycle progression of anchorage-dependent fibroblasts on fibronectin through inhibition of syndecan-4. Oncogene. 2003;22:3917–3926. doi: 10.1038/sj.onc.1206618. [DOI] [PubMed] [Google Scholar]
- Serini G, Bochaton-Piallat M-L, Ropraz P, Geinoz A, Borsi L, Zardi L, Gabbiani G. The fibronectin domain ED-A is crucial for myofibroblastic phenotype induction by transforming growth factor-beta 1. J Cell Biol. 1998;142:873–881. doi: 10.1083/jcb.142.3.873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dugina V, Fontao L, Chaponnier C, Vasiliev J, Gabbiani G. Focal adhesion features during myofibroblastic differentiation are controlled by intracellular and extracellular factors. J Cell Sci. 2001;114:3285–3296. doi: 10.1242/jcs.114.18.3285. [DOI] [PubMed] [Google Scholar]
- Matsumoto E, Yoshida T, Kawarada Y, Sakakura T. Expression of fibronectin isoforms in human breast tissue: production of extra domain A+/extra domain B+ by cancer cells and extra domain A+ by stromal cells. Jpn J Cancer Res. 1999;90:320–325. doi: 10.1111/j.1349-7006.1999.tb00750.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalembeyi I, Inada H, Nishiura R, Imanaka-Yoshida K, Sakakura T, Yoshida T. Tenascin-C upregulates matrix metalloproteinase-9 in breast cancer cells: direct and synergistic effects with transforming growth factor. Int J Cancer. 2003;105:53–60. doi: 10.1002/ijc.11037. [DOI] [PubMed] [Google Scholar]
- Goldsmith EC, Carver W, McFadden A, Goldsmith JG, Price RL, Sussman M, Lorell BH, Cooper G, Borg TK. Integrin shedding as a mechanism of cellular adaptation during cardiac growth. Am J Physiol. 2003;284:H2227–H2234. doi: 10.1152/ajpheart.00920.2002. [DOI] [PubMed] [Google Scholar]