Abstract
Lipoxins (LX) are endogenously produced eicosanoids with a spectrum of bioactions that suggest anti-inflammatory, pro-resolution roles for these agents. Mesangial cell (MC) proliferation plays a pivotal role in the pathophysiology of glomerular inflammation and is coupled to sclerosis and tubulointerstitial fibrosis. We have previously reported that LXA4 acts through a specific G-protein-coupled-receptor (GPCR) to modulate MC proliferation in response to the proinflammatory mediators LTD4 and platelet-derived growth factor (PDGF). Further investigations revealed that these effects were mediated by modulation of receptor tyrosine kinase activity. Here we have explored the underlying mechanisms and report inhibition of growth factor (PDGF; epithelial growth factor) activation of Akt/PKB by LXA4. LXA4 (10 nmol/L) modulates PDGF-induced (10 ng/ml, 24 hours) decrements in the levels of cyclin kinase inhibitors p21Cip1 and p27Kip1. PDGF-induced increases in CDK2-cyclin E complex formation are also inhibited by LXA4. The potential of LXA4 as an anti-inflammatory therapeutic is compromised by its degradation; this has been circumvented by synthesis of stable analogs. We report that 15-(R/S)-methyl-LXA4 and 16-phenoxy-LXA4 mimic the native compound with respect to modulation of cell proliferation and PDGF-induced changes in cell cycle proteins. In vivo, MC proliferation in response to PDGF is associated with TGFβ1 production and the subsequent development of renal fibrosis. Here we demonstrate that prolonged (24 to 48 hours) exposure to PDGF is associated with autocrine TGFβ1 production, which is significantly reduced by LXA4. In aggregate these data demonstrate that LX inhibit PDGF stimulated proliferation via modulation of the PI-3-kinase pathway preventing mitogen-elicited G1-S phase progression and suggest the therapeutic potential of LX as anti-fibrotic agents.
Lipoxins (LX) are endogenously produced eicosanoids with potent anti-inflammatory bioactions.1 LX are typically generated by transcellular metabolism initiated by the sequential actions of 15- and 5-, or 5- and 12-lipoxygenases on arachidonic acid, depending on the cellular context.2 In a cytokine-primed milieu, aspirin-induced acetylation of COX-2 shifts its activity from an endoperoxide to a lipoxygenase, thereby promoting synthesis of 15-epi-LX [aspirin-triggered lipoxins (ATL)].1 It has been proposed that some of the desirable effects of aspirin independent of its anti-thrombotic actions may be attributed to the biosynthesis of anti-inflammatory lipid mediators such as ATLs.2 It is increasingly appreciated that there are two distinct phases of lipid mediator production in inflammation, an initial phase characterized by the production of proinflammatory mediators essential to effective host defense, which is superseded by the production of anti-inflammatory mediators such as LX during the resolution phase.3 LXA4 and LXB4 are the principal LX formed in mammals. In particular, LXA4 has been shown to exert potent anti-inflammatory actions, modulating leukocyte trafficking and phagocytic clearance of apoptotic cells.1,3 LX biosynthesis has been demonstrated in many inflammatory conditions such as the glomerulonephritides (GN),4 human pleural disease,3 and in experimental models where the potential for cell-cell interactions is significant.5 LXA4 may exert its anti-inflammatory effects through signals generated by binding to a high-affinity, G-protein-coupled LXA4 receptor (ALXR).2 LX are rapidly metabolized, the major routes of degradation being dehydrogenation at C-15 and possibly ω-oxidation at C-20.2 To circumvent such metabolic inactivation, stable synthetic analogs have been developed that are modified at C-15, C-16, and/or C-20.6 These compounds retain the biological activity of native LXs and have been shown to bind with higher affinity to the ALXR, resulting in greater potency.2,6 The enhanced stability and improved efficacy of these analogs following local and systemic administration in models of inflammation and ischemia-reperfusion suggests significant therapeutic potential.4
Mesangial cells (MC) are modified smooth muscle cells that play a pivotal role in renal physiology by regulating circulation and glomerular structural integrity. Mesangial cell proliferation and or matrix accumulation characterizes many forms of GN and other progressive renal diseases including diabetic nephropathy. MC proliferation can be induced by several mitogens, including platelet-derived growth factor (PDGF) isoforms,7 epidermal growth factor (EGF)8 and eicosanoids such as the cysteinyl leukotriene D4.9 PDGF has been widely implicated in the etiology of GN, triggering MC proliferation, migration, contraction, and synthesis of other cytokines (eg, TGFβ1 and IL-1). In this context, blockade of PDGF bioactions with anti-PDGF antibody10 aptamers,11 soluble receptors,12 or inhibitors of receptor activation13 have been proposed as therapeutic strategies in proliferative GN. Such blockade of PDGF activity inhibits mesangioproliferative changes, scarring and interstitial fibrosis.14
The mitogenic actions of PDGF in MC are mediated via the PDGFR, a member of the receptor tyrosine kinase (RTK) family. Predominant among the PDGF isoforms that are mitogenic for MC is PDGF B which acts via the PDGFRβ.7 Activation of the intrinsic tyrosine kinase activity of the receptor facilitates recruitment of several SH2 domain-containing molecules and associated proteins including the p85 subunit of PI-3-kinase, RasGAP and PLCγ1.15 Our previous investigations have indicated that LXA4 inhibits MC proliferation in response to mitogens such as LTD49 and PDGF.16 These potential anti-inflammatory, pro-resolution bioactions of LX involve complex cross-talk between distinct GPCR and receptor tyrosine kinases.16 We have shown that LX modulate PI-3-kinase activation9 and recruitment of the p85 subunit of PI-3-kinase to the activated PDGFR.16
Here we have investigated the mechanisms underlying LX inhibition of MC proliferation and whether stable synthetic LX analogs can mimic the effects of the native compound in this regard. We report that 15-(R/S)-methyl-LXA4 and 16- phenoxy-LXA4 significantly inhibit PDGF and EGF-stimulated MC proliferation. We demonstrate that LXA4 modulates PDGF-induced decrements in the levels of p21Cip1 and p27Kip1 and promotes nuclear retention of these CKI. Importantly, LXA4 significantly modulated autocrine production of TGFβ1 by PDGF-stimulated MC. MC proliferation in response to PDGF and EGF was coupled to activation of the Ser/Thr kinase, Akt/PKB (Akt), a downstream target of PI-3-kinase. Interestingly, inhibition of PI-3-kinase with LY294002 mimicked the effects of LXA4 with respect to nuclear retention of p27Kip1 in PDGF-stimulated MC. Our data suggest that LXs modulate PDGF-induced proliferation by attenuation of Akt activation and prevention of G1-S progression.
Materials and Methods
Materials
LXA4 and LTD4 were obtained from Biomol (Plymouth Meeting, PA). Human recombinant PDGF-BB and EGF were purchased from Upstate Biotechnology (Milton Keynes, UK). TGFβ1 and anti-TGFβ1 polyclonal antibody were from BD Biosciences (Oxford, UK). AG1296 and AG1478 were acquired from Calbiochem (Nottingham, UK). Transfer membranes were from Millipore (Bedford, MA). All other reagents were purchased from Sigma (Poole, Dorset, UK) unless otherwise stated. Stable synthetic LX analogs were a generous gift from Dr. Nicos Petasis, University of Southern California, Los Angeles, CA.
Cell Culture
Human kidneys were obtained from excess nephrectomy specimens according to the Mater Misericordiae University Hospital ethical guidelines. Renal glomeruli were isolated by differential sieving and mesangial cells were obtained and cultured in RPMI 1640 supplemented with 10% fetal calf serum (FCS), penicillin (100 U/ml), and streptomycin (100 μg/ml) (all purchased from Gibco BRL, Paisley, Scotland). Isolated cells retained the phenotypic characteristics of mesangial cells, including stellate morphology, stained positive for vimentin and α-smooth muscle actin expression, and were negative for factor VIII and cytokeratin excluding endothelial and epithelial cell contamination, respectively.17 The CHOK1 cell line stably expressing ALXR and a control cell line were established and cultured as previously described.16
Cell Proliferation Assay
For analysis of proliferation, primary cultures of mesangial cells and CHOK1 cells were grown to approximately 70% confluence on 24-well plates before serum restriction in 0.2% FCS RPMI-1640 for 48 hours (MC) or serum deprivation in 0% FCS Ham’s F12 medium for 24 hours (CHOK1 cells). After this period, cells were stimulated with various agents in triplicate wells for indicated times (44 hours for MC or 20 hours for CHOK1 cells) as detailed in Figure legends. Proliferation of cells was measured by determining [3H]-thymidine incorporation as follows: 1 μCi [3H]-thymidine (90 to 120 Ci/mmol; NEN, Cambridge, UK) was added to each well and incubated for 4 hours. Cells were washed twice in DPBS, solubilized in 0.2% sodium dodecyl sulfate (SDS) and counts per minute (cpm) were measured in 10 ml of scintillant (σ-Fluor). Data provided are from 4 to 5 independent experiments, as indicated.
Western Blot Analysis
Mesangial cells were serum restricted in 0.2% FCS RPMI 1640 for 48 hours and exposed to various agents for indicated times. Lysates were harvested in RIPA lysis buffer (20 mmol/L Tris-HCl, pH 7.4, 50 mmol/L NaCl, 5 mmol/L ethylene diaminetetraacetic acid, 1% NP-40, 0.5% Na-deoxycholate, 0.1% SDS, 5 mmol/L NaF, 1 mmol/L phenylmethylsulfonyl fluoride, 1 mmol/L Na3VO4, 1 μmol/L leupeptin, 0.3 μmol/L aprotinin). The lysates were clarified by centrifugation at 12,000 × g for 10 minutes and protein concentration in the supernatant was measured by Bradford protein assay. For Western blot analysis, 30 μg of MC protein extract was loaded onto each lane, separated under reducing conditions on an SDS-polyacrylamide gel electrophoresis (PAGE) gel, and transferred to polyvinylidene difluoride (PVDF) membranes (Millipore) by electroblotting. To reduce non-specific antibody binding, the membranes were first blocked with 5% nonfat dried milk for 1 hour at room temperature. This was followed by an overnight incubation at 4°C with antibodies to either cyclin A (Upstate Biotech, Lake Placid, NY), cyclin E (BD Biosciences), CDK2, p21Cip1 or p27Kip1 (all Santa Cruz Biotechnology, Heidelberg, Germany). Controls included omitting the primary antibody and/or replacing the primary antibody with rabbit or mouse serum. Membranes were incubated with a horseradish-peroxidase-conjugated secondary antibody for 1 hour at room temperature and were visualized by chemiluminescence. To check for equal loading, membranes were either stained with Ponceau-S staining solution or were stripped and reprobed for β-actin (Sigma). For analysis of Akt phosphorylation, membranes were blocked with 5% bovine serum albumin (BSA) and probed with antibodies to either phospho-Akt (Ser473) or Akt protein (both New England Biosciences, Hertfordshire, UK).
Analysis of Cyclin E-CDK2 Complex Formation
MC lysate (300 μg) from each condition was precleared with Protein-G agarose beads (Santa Cruz) for 1 hour at 4°C before incubation with antibody to CDK2 (Santa Cruz, 1:500 dilution) overnight at 4°C with constant rocking. Protein-G agarose beads (10 μl) were then added to each immunoprecipitation and incubated for 2 hours at 4°C with constant rocking. Immunocomplexes were washed three times in fresh RIPA lysis buffer, denatured in 5X reducing sample buffer and boiled for 5 minutes to elute protein off beads. Samples were electrophoresed on SDS-PAGE gels, transferred to PVDF membranes, and probed for either cyclin E or CDK2 (loading control).
Quantitation of TGFβ1 Production
Quiescent MC were treated as indicated. At 24-, 48-, and 72-hour time points, medium was removed and assayed for TGFβ1 release by ELISA (R&D systems, Abingdon, UK) as per manufacturer’s protocol. TGFβ1 produced was expressed as picogram TGFβ1 per microgram of cellular protein.
Cellular Fractionation
Using differential centrifugation, nuclear and cytosolic fractions from stimulated cells were harvested using a Nuclear Extract kit (Activemotif, Rixensart, Belgium). Briefly, cell lysates were fractionated at 14,000 × g for 30 seconds. Protein concentration in the pellet and soluble fractions was measured by Bradford protein assay. Cytosolic fractions were concentrated using 10,000 MW cut-off filters (Millipore) to maximize protein yield. Nuclear-cytoplasmic translocation of p27Kip1 was assayed by immunoblotting of cytosolic lysates and densitometric analysis.
Immunocytochemistry
MC were cultured in 4-well chamber slides (Nalge Nunc, Naperville, IL), rendered quiescent and stimulated as indicated. After stimulation, cells were washed with PBS, fixed with 2% paraformaldehyde (10 minutes) and permeabilized with 0.1% Triton X-100 (15 minutes). Fixed cells were then treated with blocking solution (5% BSA) for 1 hour. Localization of p27Kip1 was determined using anti-p27Kip1 polyclonal antibody (1:500). After washes, samples were treated with Oregon Green-conjugated anti-rabbit IgG (1:200). For DNA staining, samples were incubated with 4′,6-diamidino-2-phenylindole, dihydrochloride (DAPI) dye (1 μg/ml, 10 minutes). Cells were viewed and recorded in phase contrast (10× and 40× magnification) and corresponding fields using an Axiovert 200 fluorescent microscope (Carl Zeiss, Jena, Germany).
Results
15-(R/S)-LXA4 and 16-Phenoxy-LXA4 Inhibit Mitogen-Induced Proliferation of MCs
Serum, PDGF, and EGF induced maximal DNA synthesis in primary human MC by 48 hours poststimulation. The mitogenic response of MCs to PDGF-BB and EGF was dose-dependent, with an apparent maximal concentration of 100 ng/ml and 250 ng/ml and an EC50 of 8 ng/ml and 44 ng/ml, respectively. In subsequent experiments, PDGF-BB and EGF were applied at 10 ng/ml and 50 ng/ml concentrations, respectively, which consistently induced a minimum of a 2.5-fold increase in MC DNA synthesis. Incubation of MCs with increasing concentrations of LXA4 did not induce MC DNA synthesis at any concentration tested (10−12 M to 10−8 M). Preincubation of quiescent MC with LXA4 (1 nmol/L) significantly inhibited mitogenesis induced by PDGF (10 ng/ml), as assessed by [3H]-thymidine uptake (Figure 1A). (The effect of LXA4 was found to be dose-dependent [10 pM to 1 μmol/L, data not shown] with an EC50 of approximately 10 nmol/L LXA4, in contrast to other LX responses,9 the dose:response was not bell-shaped). This effect was replicated by 16-phenoxy-LXA4 (10 pM), a stable synthetic analog of LXA4, and 15-(R/S)-LXA4 (10 pM), a stable synthetic analog of ATL (Figure 1A). These data indicate that stable LX analog can act as mimetics of endogenous and aspirin-triggered LXA4 in the context of counter regulation of mitogenic signals. AG1296, the tyrophostin inhibitor of PDGF receptor tyrosine kinase activity was included as a negative control in these experiments.18
Figure 1.
Inhibition of PDGF and EGF-stimulated MC DNA synthesis by lipoxins. A and B: Subconfluent human MCs were serum restricted in 0.2% FCS medium for 48 hours before stimulation with LXA4 (1 nmol/L), 15-(R/S)-LXA4 (10 pM), 16-phenoxy-LXA4 (10 pM), AG1296 (10 μmol/L) (A), AG1478 (100 nmol/L) (B), for 60 minutes before stimulation with PDGF-BB (10 ng/ml) (A) and EGF (50 ng/ml) (B). Cell proliferation was measured by [3H]-thymidine incorporation after 48 hours. Vehicle (EtOH <0.1%) and 10% FCS were used as negative and positive controls, respectively. Results are expressed as fold/basal cell proliferation. Data represent the mean ± SD of four (A) and two (B) independent experiments performed in triplicate. ** P < 0.01 vs. vehicle, * P < 0.05 vs. PDGF alone (unpaired Student’s t-test). C: Inhibition of serum-stimulated CHOK1-ALXR proliferation by LXA4 and stable synthetic analogs, effect not seen in vector control cell line. CHO K1 cells (± ALXR) were preincubated with LXA4 (1 nmol/L), 15-(R/S)-LXA4 (10 pM), 16-phenoxy-LXA4 (10 pM), for 60 minutes before stimulation with serum-containing media. Cell proliferation was measured by [3H]-thymidine incorporation after 48 hours. Results are expressed as percentage cell proliferation. Data are mean ± SEM of four independent experiments performed in triplicate. * P < 0.05 vs. 10% serum alone (unpaired Student’s t-test).
To investigate whether the effect was specific to PDGF-induced proliferation we stimulated cells with LX analogs before treatment with both EGF and serum. As Figure 1B depicts, this anti-proliferative effect was replicated on EGF (50 ng/ml)-stimulated MC proliferation (n = 2). The reduction in DNA synthesis by LX on EGF-treated MC was less than the reduction seen in PDGF-stimulated MC (25% reduction vs. 48% reduction, respectively). Analagous to data with AG1296, the inhibitor of EGF receptor tyrosine kinase activity, AG1478, inhibited EGF-stimulated MC proliferation.
The antiproliferative effect of LXA4 analogs was also observed in serum-stimulated CHO K1 cells stably expressing the lipoxin A4 receptor (ALXR) but not in control cells (Figure 1C). Preincubation of quiescent CHOK1 ± ALXR with LXA4 (1 nmol/L, 60′) significantly (P < 0.05, unpaired Student’s t-test) inhibited mitogenesis (approximately 20% reduction) induced by serum, an effect replicated by 15-(R/S)-methyl-LXA4 (10 pM) and 16-phenoxy-LXA4 (10 pM). Further investigation of the antiproliferative effects of LX using an assay based on reduction of a tetrazolium component (MTT) to an insoluble formazan product indicate that LXA4 inhibition of serum-stimulated proliferation does not involve promotion of apoptosis (data not shown).
LXA4 Modulates Mitogen-Induced Activation of Akt
We have previously observed a reduction in phosphorylation of the PDGFR in MC by LXA4.16 This, in conjunction with data showing LXA4-induced inhibition of LTD4-stimulated PI-3-kinase activation9 and p85 subunit recruitment to the PDGFR,16 suggested that the PI3-kinase pathway may be a locus of the inhibitory effect of LXA4 in MC. As indicated in Figure 2, both PDGF (10 ng/ml) (A) and EGF (50 ng/ml) (B) stimulated activation of Akt via phosphorylation at Ser473. Both PDGF- and EGF-stimulated activation was inhibited by preincubation of MC with LXA4 (10 nmol/L, 30 minutes and 60 minutes) and by a specific inhibitor of PI-3 K activity, LY294002 (3 μmol/L). The LXA4-dependent inhibition was not mimicked by two other GPCR-coupled agonists, LTD4 (10 nmol/L) (A) and 5-HT (10 μmol/L) (B) which have been previously shown to be involved in cross-talk with PDGF and EGF RTK, respectively, in mesangial cells.9,19 Expression of the receptors for both of these GPCR agonists has been previously demonstrated in MC.16,20 The observation that LXA4 inhibition of PDGF and EGF-stimulated proliferation is coupled to modulation of Akt phosphorylation is noteworthy given that expression of dominant-negative Akt in rat mesangial cells inhibits mitogenic responses to PDGF.21
Figure 2.
Akt phosphorylation is modulated by LXA4. Subconfluent cultures of MC were made quiescent in 0.2% FCS medium for 48 hours. MC were then incubated with LXA4 (10 nmol/L, 30′, 60′), LY29002 (3 μmol/L, 30′), LTD4 (10 nmol/L, 30′) (A), 5-HT (10 μmol/L, 30′) (B) before stimulation with PDGF-BB (10 ng/ml, 5′) (A) or EGF (50 ng/ml, 5′) (B). Forty microns of protein were electrophoresed and transferred onto PVDF membrane and immunoblotted for P-Akt (Ser473) or Akt. Results are depicted graphically and represent the mean ± SD for three independent experiments. Values given are fold/basal relative to vehicle-treated cells. * P < 0.001 relative to vehicle, # P < 0.05 relative to PDGF-stimulated cells.
PDGF Decreases CKI p21Cip1 and p27Kip1 Levels: Modulation by LXA4
The levels of G1 phase CKI p21Cip1 and p27Kip1 in quiescent and proliferating MC were determined by western blot analysis (Figure 3A). Quiescent MCs were stimulated with LXA4 (10 nmol/L, 60 minutes) or vehicle before addition of PDGF-BB (10 ng/ml, 24 hours). Detectable levels of p21Cip1 and p27 Kip1 were expressed in quiescent MCs and no change in their levels was observed over a 24-hour period in vehicle or LXA4-treated MC (data not shown). However, incubation with PDGF caused a decrease in detectable levels of both p21Cip1 and p27Kip1 at 24 hours poststimulation (Figure 3A). These decreases were significantly abrogated by preincubation with LXA4 (10 nmol/L, 60 minutes) or by co-stimulation with TGFβ1. TGFβ1 has previously been shown to act in an anti-mitotic manner in PDGF-stimulated MC, acting on G1-phase cell cycle proteins.22 In MC, TGFβ1-treated cells showed no effect on levels of p21Cip1 but showed an increase in levels of p27Kip1 at 24 and 48 hours (data not shown), which is in agreement with previous data.23 Consistent with our data showing inhibition of PDGF-stimulated MC proliferation by the stable synthetic LX analogs (Figure 1), we found that 15-(R/S)-LXA4 and 16-phenoxy-LXA4 replicated the effect of LXA4 on cell cycle protein expression. As Figure 3B depicts, the PDGF-induced reduction in levels of p21Cip1 and p27Kip1 at 24 hours was inhibited by preincubations with 15-(R/S)-LXA4 (10 pM) or 16-phenoxy-LXA4 (10 pM).
Figure 3.
LX regulate PDGF-induced reduction in protein levels of G1 phase. CKI p21Cip1 and p27Kip1 in human MC. A: Subconfluent cultures of human MC were serum restricted in 0.2% FCS medium for 48 hours. Quiescent MC were then stimulated with LXA4 (10 nmol/L, 60′) or vehicle as indicated, before addition of PDGF-BB (10 ng/ml). PDGF-treated MC were co-incubated with TGFβ1 (10 ng/ml) to act as a negative control. MCs were harvested and protein was extracted at 6- and 24-hour intervals poststimulation. Thirty microns protein per lane was resolved by SDS-PAGE and levels of p21Cip1 and p27Kip1 were determined by Western blot. β-actin protein was examined as a loading control. Results are depicted graphically and represent the mean ± SD of three independent experiments. Values given are fold/basal relative to vehicle-treated cells. * P < 0.01 relative to vehicle, # P < 0.05 relative to PDGF-stimulated cells. B: LXA4 stable analogs mimic effect of native LXA4 on PDGF-induced reduction in p21Cip1 and p27Kip1 protein levels. Quiescent MCs were preincubated with LXA4 (10 nmol/L), 15-(R/S)-methyl-LXA4 (10 pM), 16-phenoxy-LXA4 (10 pM) for 60 minutes before treatment with PDGF-BB (10 ng/ml) for 24 hours. Results are depicted graphically and represent the mean ± SD of two independent experiments. Values given are fold/basal relative to vehicle-treated cells.
PDGF Stimulates G1-S Phase Transition: Cyclin E-CDK2 Complex Formation Is Attenuated by LXA4
The levels of G1 associated proteins, CDK2 and cyclin E were determined by Western blot (Figure 4). PDGF stimulation resulted in increased levels of cyclin E at 24 hours and CDK2 at 6 and 24 hours (A). These increases were significantly abrogated by pre-incubation with LXA4. Vehicle-treated MC or MC stimulated with LXA4 (10 nmol/L) alone at 6 and 24 hours showed no alteration in protein levels of either cyclin E or CDK2 (data not shown). Additionally, cyclin A levels were increased by PDGF stimulation at 24 hours (data not shown). However, levels of cyclin A were not attenuated by LXA4 or TGFβ1.
Figure 4.
LXA4 regulates PDGF-induced protein levels of G1 phase CDK2 and cyclin E and CDK2-cyclin E complex formation in human MC. A: Subconfluent cultures of human MC were serum-restricted in 0.2% FCS medium for 48 hours. Quiescent MC were then stimulated with LXA4 (10 nmol/L, 60′) or vehicle as indicated, before addition of PDGF-BB (10 ng/ml). PDGF-treated MC were co-incubated with TGFβ1 (10 ng/ml) to act as a negative control. MCs were harvested and protein was extracted at 6- and 24-hour intervals after stimulation. Thirty microns of protein per lane were size-fractionated by SDS-PAGE and protein expression of cyclin E and CDK2 was determined by Western blot. Expression of β-actin protein was examined as a loading control. Results are depicted graphically and represent the mean ± SD for three independent experiments. Values given are fold/basal relative to vehicle-treated cells. * P < 0.01 relative to vehicle, # P < 0.05 relative to PDGF-stimulated cells. B: The protein from the above conditions was immunoprecipitated with polyclonal antibody to CDK2. The immunoprecipitated lysate was divided in half, separated on an SDS-PAGE, and a Western blot analysis was performed with an antibody to CDK2 and cyclin E. Results are depicted graphically and represent the mean ± SD of two independent experiments. Values given are fold/basal relative to vehicle-treated cells.
Due to the observation that LXA4 significantly inhibited PDGF-stimulated levels of CDK2 and cyclin E, we examined the effect of LXA4 on mitogen-induced cyclin E association with CDK2 to form an active complex, an important step for progression of cells from G1 into S phase.24 As depicted in Figure 4B, PDGF induced association of cyclin E and CDK2 at 24 hours. Preincubation of PDGF-stimulated MCs with LXA4 (10 nmol/L, 60 minutes) inhibits the formation of this complex. To act as a control in this experiment, PDGF-stimulated MCs co-stimulated with TGFβ1 (10 ng/ml) showed a modulation of assembly of cyclin E-CDK2 complexes at 24 hours, as previously reported.23 Incubation of unstimulated control cells with LXA4 or TGFβ1 had no effect on cyclin E-CDK2 association (data not shown).
PDGF-Stimulated Production of TGFβ1 Is Blocked by LXA4
It has previously been shown that PDGF stimulates production of TGFβ1 from MC.25 Consistent with this, an ELISA for levels of secreted TGFβ1 on samples of cell-free medium from PDGF-stimulated MC indicated a significant (P < 0.001) increase in autocrine production of TGFβ1 in a time-dependent manner (24 to 48 hours, Figure 5A; data are expressed as picograms of TGFβ1 produced per microgram of protein present and are consistent with values reported by others25). TGFβ1 release reached a peak of 2.3 ± 0.2 pg/μg (mean ± SEM) at 48 hours poststimulation with PDGF (10 ng/ml). LXA4 (10 nmol/L) reduced the level of autocrine production of TGFβ1 at 24 and 48 hours to basal levels. Extending the timecourse to 72 hours, we also observed a significant decrease (84% reduction) in PDGF-stimulated TGFβ1 production (data not shown).
Figure 5.
PDGF-stimulated TGFβ1 production is attenuated by LXA4. A: Quantitative analysis of TGFβ1 ELISA performed on supernatants of cells at indicated time points. Results are expressed as picograms per microns of MC protein. Data are mean ± SD of seven independent experiments (*P < 0.001 vs. vehicle, # P < 0.05 vs. PDGF alone, unpaired Student’s t-test). B: Subconfluent cultures of human MC were serum restricted in 0.2% FCS medium for 48 hours. Quiescent MC were then stimulated with LXA4 (10 nmol/L, 60′) or vehicle as indicated, before addition of PDGF-BB (10 ng/ml). MCs were harvested and protein was extracted at 6-, 24-, and 48-hour intervals after stimulation. Thirty microns of protein per lane was size-fractionated by SDS-PAGE and protein expression of p27Kip1 was determined by western blot. Expression of β-actin protein was examined as a quantity loading control. C: Western blot analysis at 48 hours poststimulation with above conditions. MCs were stimulated with vehicle, LXA4 (10 nmol/L), PDGF-BB (10 ng/ml) for 48 hours, or pretreated with LXA4 (10 nmol/L) or TGFβ1 monoclonal antibody (1 nmol/L) for 60 minutes before stimulation with PDGF-BB for 48 hours. Results of B and C are depicted graphically and represent the mean ± SD of three independent experiments. Values given are fold/basal relative to vehicle-treated cells. * P < 0.01 relative to vehicle, ** P < 0.05 relative to vehicle, # P < 0.05 relative to PDGF-stimulated cells (24 hours), ## P < 0.05 relative to PDGF-stimulated cells (48 hours).
TGFβ1 has previously been shown to promote MC cell cycle arrest via up-regulation of p27Kip1.24 Consistent with enhanced TGFβ1 production from PDGF-treated cells (Figure 5A), we observed a reversal of the initial diminution in p27Kip1 levels at 48 hours poststimulation with PDGF (Figure 5B). In agreement with our hypothesis that this effect may be mediated through PDGF-stimulated autocrine production of TGFβ1 we observe that this effect is blocked with an inhibitory TGFβ1 antibody (Figure 5C).26
Akt Mediates the Subcellular Localization of p27Kip1: Modulation by LXA4
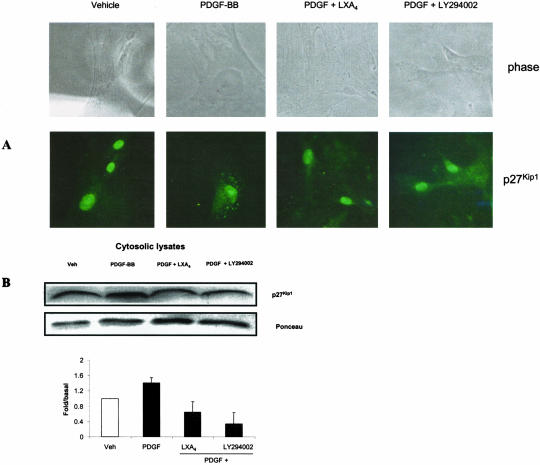
We have investigated the subcellular location of the CKI p27Kip1 in growth factor-stimulated cells. Cells were either processed for indirect immunofluorescence or fractionated to enrich for nuclear and cytoplasmic proteins. Figure 6A shows a representative example of immunofluorescence. In quiescent (vehicle-treated) cells, p27Kip1 localized to the nucleus (90% of cells, counted in two separate fields, 10× magnification, 50 colonies/field, two independent observers). Nuclear staining was confirmed by staining with DAPI stain (data not shown). On stimulation with PDGF an increase in the levels of cytoplasmic p27Kip1 was observed, this effect was inhibited by preincubation with LXA4. PI-3-kinase and subsequent Akt phosphorylation has been proposed to regulate stability of p21Cip1 and p27Kip1 by promoting nuclear to cytoplasmic translocation and subsequent degradation.27–29 We observe that in MC exposed to the PI 3-kinase inhibitor LY294002, PDGF did not promote cytoplasmic (6A) or cytosolic translocation of p27Kip1 (6B). Our observations are consistent with a role for p27Kip1 export from the nucleus in regulating proliferation.
Figure 6.
PDGF increases cytosolic levels of p27Kip1. Influence of LXA4 and LY294002. A: Subcellular localization of p27Kip1 detected by immunocytochemistry. Quiescent MCs on coverslips were incubated with LXA4 (10 nmol/L), LY294002 (3 μmol/L) or vehicle for 30 minutes before stimulation with PDGF (10 ng/ml). At 24 hours, fixed cells were stained with polyclonal antibody against p27Kip1 (green) or phase contrast. B: Cytosolic transition of p27Kip1. Quiescent MCs were incubated with LXA4 (10 nmol/L), LY294002 (3 μmol/L) or vehicle for 30 minutes before stimulation with PDGF (10 ng/ml). After 24 hours, cytosolic and nuclear fractions were extracted. Twenty-five microns of protein per lane were size-fractionated by SDS-PAGE and cytoplasmic levels of p27Kip1 were determined by Western blot. Results are depicted graphically and represent the mean ± SD of three independent experiments. Values given are fold/basal relative to vehicle-treated cells. * P < 0.05 relative to vehicle, # P < 0.05 relative to PDGF-stimulated cells.
Discussion
MC proliferation plays a key role in glomerular inflammation. Proliferative responses to a variety of stimuli are associated with matrix accumulation and the development of glomerulosclerosis, which may lead to interstitial fibrosis, the final common pathway of progressive renal disease.30 In this context it is noteworthy that modulation of MC proliferation is considered an attractive target for therapeutic intervention. PDGF-BB, -AB and -DD, ligands for the PDGFRβ, evoke proliferation of MC.12 A key role for these agents produced either locally or systemically has been demonstrated in renal inflammation.14 Several therapeutic strategies have been proposed to limit PDGF-induced renal damage, including inhibition of either PDGF ligand activity with anti-PDGF antibody,10 or PDGF receptor activation by the tyrosine kinase inhibitor STI 571.13
LX have been proposed as important, endogenously produced pro-resolution agents in host defense and inflammation.1 In the current study we have investigated the mechanisms underlying LX inhibition of mesangial cell proliferation in response to various agents including PDGF and EGF. We report that, similar to the endogenously produced LXA4, the stable synthetic analog 16-phenoxy-LXA4 and the stable analog of ATL, 15-(R/S)-methyl-LXA4, can modulate MC proliferation. Evidence that these effects are mediated through the previously described ALXR are provided by the observation that serum-stimulated proliferation of CHOK1 cells stably expressing the ALXR was attenuated by both 16-phenoxy-LXA4 and 15-(R/S)-methyl-LXA4.
We have investigated the effects of LX on levels of cell cycle proteins. The modulation of proliferation by LX might reflect arrest at G0-G1 or G1-S phase transitions of the cell cycle. Under appropriate stimuli, cells exit quiescence (G0) and enter the cycle at early G1 phase. In late G1 phase, cells pass through the “restriction point” marking the commitment point after which they are no longer sensitive to extracellular signals. Traversing the G1-S phase is coupled to DNA synthesis, followed by entry into G2 and finally mitosis occurs in M phase.31 Progression through the cycle is dependent on the complex interaction of multiple regulatory proteins. The mechanisms of regulation of the G1 phase cell cycle involve the interaction of cyclin-CDK complexes with CDK inhibitors (CKI).32 In MC, a role for decreases in the levels of CKI has been demonstrated in response to PDGF.23 These CKI play an important role regulating proliferation. Both p21Cip1 and p27Kip1 contain binding domains for CDK which may interfere with the ability of CDK to form active complexes with cyclins.33 Furthermore, p21Cip1 can also bind to PCNA, an auxiliary protein to DNA polymerase, either directly or in a quaternary complex with PCNA, cyclins, and CDK.34 Here, we demonstrate that LX attenuate PDGF-induced decrements in p21Cip1 and p27Kip1. Furthermore, the decreases in p21Cip1 and p27Kip1 levels that we observe in PDGF-stimulated cells are coupled to increased levels of cyclin E and establishment of cyclin E-CDK2 complexes, indicative of progression through G1-S. LXA4 inhibited this effect, an observation consistent with maintaining levels of CKIs. It is noteworthy that Shankland and colleagues35 have proposed that the resolution of glomerular inflammation may be associated with a return of depleted p27Kip1 levels. We have previously reported that LX directly attenuated PDGF and EGF receptor activation.16 The underlying mechanisms are not clear and may involve altered phosphatase activity or endocytic trafficking of the RTKs in response to LX.
We have investigated signaling events downstream of receptor activation and report LX modulation of Akt activation in response to PDGF and EGF. Akt is a target of PI-3-kinase where binding of D3-phosphorylated inositides to the PH domain of Akt stimulates phosphorylation of the protein.36 The precise role of Akt in regulating proliferation is unclear; however, recent data suggest that Akt-mediated phosphorylation of p27Kip1 may facilitate its translocation from the nucleus to the cytoplasm where the phosphorylated protein is a target for proteasomal degradation.27–29 Using an analogous mechanism to that proposed for p27Kip1, Akt can also promote p21Cip1 degradation via MDM2 phosphorylation of p53.37 Translocation of CKIs from the nucleus removes repression of cyclin E-CDK2 activity and the cell cycle proceeds through G1-S. There is also evidence for the importance of a trimeric complex with CDK in regulating ubiquitination of p27Kip1. The elevated levels of the cyclin E-CDK2 complex observed in proliferating MC may promote proteasomal degradation of CKIs.38 Consistent with a role for p27Kip1 export from the nucleus in regulating proliferation are our observations using immunostaining. Under basal (quiescent) conditions p27Kip1 is observed as a discrete focus in the nucleus. However, on stimulation with PDGF this becomes cytoplasmic. These data are corroborated by immunoblotting of cytosolic fractions showing increased levels of p27Kip1. In addition to the proposed role of Akt in promoting degradation of p27Kip1 in MC, it may also exert an effect on transcription through phosphorylation-dependent inhibition of the forkhead family of transcription factors.39 Controversy exists over whether p21Cip1 and p27Kip1 follow similar patterns of activation and down-regulation in MC and in models of glomerular disease.40 We observe similar down-regulation patterns of both CKI in response to PDGF and maintenance of levels of both CKI in response to preincubations with LX. We propose a model where PDGF stimulation of human mesangial cells is coupled to PI-3-kinase and Akt activation, resulting in phosphorylation of p27Kip1 and its translocation from the nucleus, removing the repression on cyclin E-CDK2 activity (see Figure 7). In cells treated with LX, inhibition of PDGF receptor phosphorylation is coupled to an attenuation of downstream PI-3-kinase activity and subsequent Akt phosphorylation. In this scenario, p27Kip1 remains within the nucleus where it exerts its inhibitory effects on the activity of cyclin E-CDK2 complexes. After 48-hour stimulation with PDGF we observed a recovery of p27Kip1 to basal levels. We propose that this may be due to autocrine production of TGFβ1. TGFβ1 is known to prevent PDGF induced decreases in p27Kip1 levels.22 Recent evidence demonstrates that the growth-arrest and hypertrophic effects of TGFβ1 are dissociated.41 This may be important in the context of LX, suggesting that mimicry of TGFβ1 with respect to maintenance of p21Cip1 and p27Kip1 levels does not implicate LX as profibrotic indeed, LX significantly attenuated PDGF induced TGFβ1 production.
Figure 7.
Proposed mechanism of LX modulation of PDGF-stimulated MC. proliferation. PDGF stimulation of PDGFRβ in human mesangial cells is coupled to PI-3-kinase and Akt activation, resulting in phosphorylation of p27Kip1 and its translocation from the nucleus, removing the repression on cyclin E-CDK2 activity. In cells treated with LX, inhibition of PDGF receptor phosphorylation is coupled to an attenuation of subsequent Akt phosphorylation and downstream events.
The results presented here are of interest in the context of the bioactions of non-steroidal anti-inflammatory drugs (NSAIDs).42 Whereas the nonselective NSAID aspirin can provoke the generation of 15-epi-LX, thereby acting to promote the resolution of inflammation,43,44 the action of COX-2-specific inhibitors in impeding 15(R)-HETE production may compromise the pro-resolution potential of endogenously produced eicosanoids. Our data elaborate on the potential of LX and ATLs as agents that are not only anti-inflammatory and pro-resolution but may also act as anti-fibrotic agents.
Acknowledgments
We thank Kate Coleman for technical assistance and Dr. John Crean for helpful discussions.
Footnotes
Address reprint requests to Catherine Godson, Department of Medicine and Therapeutics, Conway Institute of Biomolecular and Biomedical Research, University College Dublin, Belfield, Dublin 4, Ireland. E-mail: catherine.godson@ucd.ie.
Supported by the Health Research Board, Ireland, Enterprise Ireland, The Wellcome Trust, and The Government of Ireland Program for Research in Third Level Institutions.
References
- McMahon BM, Mitchell S, Brady HR, Godson C. Lipoxins: revelations on resolution. Trends Pharmacol Sci. 2001;22:391–395. doi: 10.1016/s0165-6147(00)01771-5. [DOI] [PubMed] [Google Scholar]
- Serhan CN. Lipoxins and novel aspirin-triggered 15-epi-lipoxins[ATL]: a jungle of cell-cell interactions or a therapeutic opportunity? Prostaglandins. 1997;53:107–137. doi: 10.1016/s0090-6980(97)00001-4. [DOI] [PubMed] [Google Scholar]
- Levy BD, Clish CB, Schmidt B, Gronert K, Serhan CN. Lipid mediator class switching during acute inflammation: signals in resolution. Nat Med. 2001;2:612–619. doi: 10.1038/89759. [DOI] [PubMed] [Google Scholar]
- Papayianni A, Serhan CN, Phillips ML, Rennke HG, Brady HR. Transcellular biosynthesis of lipoxin A4 during adhesion of platelets and neutrophils in experimental immune complex glomerulonephritis. Kidney Int. 1995:1295–1302. doi: 10.1038/ki.1995.184. [DOI] [PubMed] [Google Scholar]
- Mayadas TN, Mendrick DL, Brady HR, Tang T, Papayianni A, Assmann KJ, Wagner DD, Hynes RO, Cotran RS. Acute passive anti-glomerular basement membrane nephritis in P-selectin-deficient mice. Kidney Int. 1996;49:1342–1349. doi: 10.1038/ki.1996.190. [DOI] [PubMed] [Google Scholar]
- Clish CB, O’Brien JA, Gronert K, Stahl GL, Petasis NA, Serhan CN. Local and systemic delivery of an aspirin-triggered lipoxin prevents neutrophil recruitment in vivo. Proc Natl Acad Sci USA. 1999;96:8247–8252. doi: 10.1073/pnas.96.14.8247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Abboud HE. Growth factors in glomerulonephritis. Kidney Int. 1993;43:252–267. doi: 10.1038/ki.1993.39. [DOI] [PubMed] [Google Scholar]
- Silver BJ, Jaffer FE, Abboud HE. Platelet-derived growth factor synthesis in mesangial cells: induction by multiple growth factors. Proc Natl Acad Sci. 1989;86:1056–1060. doi: 10.1073/pnas.86.3.1056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McMahon B, Stenson C, McPhillips F, Fanning A, Brady HR, Godson C. Lipoxin A4 antagonises the mitogenic effect of LTD4 on human renal mesangial cells: differential activation of MAP kinases through distinct receptors. J Biol Chem. 2000;275:27566–27575. doi: 10.1074/jbc.M001015200. [DOI] [PubMed] [Google Scholar]
- Johnson RJ, Raines EW, Floege J, Yoshimura A, Pritzl P, Alpers C, Ross R. Inhibition of mesangial cell proliferation and matrix expansion in glomerulonephritis in the rat by antibody to platelet-derived growth factor. J Exp Med. 1992;175:1413–1416. doi: 10.1084/jem.175.5.1413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Floege J, Ostendorf T, Janssen U, Burg M, Radeke HH, Vargeese C, Gill SC, Green LS, Janjic N. Novel approach to specific growth factor inhibition in vivo: antagonism of platelet-derived growth factor in glomerulonephritis byaptamers. Am J Pathol. 1999;154:169–179. doi: 10.1016/S0002-9440(10)65263-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ostendorf T, van Roeyen CR, Peterson JD, Kunter U, Eitner F, Hamad AJ, Chan G, Jia XC, Macaluso J, Gazit-Bornstein G, Keyt BA, Lichenstein HS, LaRochelle WJ, Floege J. A fully human monoclonal antibody (CR002) identifies PDGF-D as a novel mediator of mesangioproliferative glomerulonephritis. J Am Soc Nephrol. 2003;14:2237–2247. doi: 10.1097/01.asn.0000083393.00959.02. [DOI] [PubMed] [Google Scholar]
- Gilbert RE, Kelly DJ, McKay T, Chadban S, Hill PA, Cooper ME, Atkins RC, Nikolic-Paterson DJ. PDGF signal transduction inhibition ameliorates experimental mesangial proliferative glomerulonephritis. Kidney Int. 2001;59:1324–1332. doi: 10.1046/j.1523-1755.2001.0590041324.x. [DOI] [PubMed] [Google Scholar]
- Floege J, Johnson RJ. Multiple roles for platelet-derived growth factor in renal disease. Minor Electrolyte Metab. 1995;21:271–282. [PubMed] [Google Scholar]
- Heldin H, Ostmann A, Ronnstrand L. Signal transduction via platelet-derived growth factor receptors. Biochim Biophysica Acta. 1998;1378:F79–F113. doi: 10.1016/s0304-419x(98)00015-8. [DOI] [PubMed] [Google Scholar]
- McMahon B, Mitchell D, Shattock R, Martin F, Brady HR, Godson C. Lipoxin, leukotriene, and PDGF receptors cross-talk to regulate mesangial cell proliferation. EMBO J. 2002;16:1817–1819. doi: 10.1096/fj.02-0416fje. [DOI] [PubMed] [Google Scholar]
- Mene P. Mesangial cell cultures. J Nephrol. 2001;14:198–203. [PubMed] [Google Scholar]
- Goppelt-Struebe M, Fickel S, Reiser CO. The platelet-derived-growth-factor receptor, not the epidermal-growth-factor receptor, is used by lysophosphatidic acid to activate p42/44 mitogen-activated protein kinase and to induce prostaglandin G/H synthase-2 in mesangial cells. Biochem J. 2000;345:217–224. [PMC free article] [PubMed] [Google Scholar]
- Grewal JS, Luttrell LM, Raymond JR. G protein-coupled receptors desensitize and downregulate EGF receptors in renal mesangial cells. J Biol Chem. 2001;276:27335–27344. doi: 10.1074/jbc.M103578200. [DOI] [PubMed] [Google Scholar]
- Nebigil CG, Garnovskaya MN, Spurney RF, Raymond JR. Identification of a rat glomerular mesangial cell mitogenic 5-HT2A receptor. Am J Physiol. 1995;268:F122–F127. doi: 10.1152/ajprenal.1995.268.1.F122. [DOI] [PubMed] [Google Scholar]
- Ghosh-Choudhury G, Zhang JH, Ghosh-Choudhury N, Abboud HE. Ceramide blocks PDGF-induced DNA synthesis in mesangial cells via inhibition of Akt kinase in the absence of apoptosis. Biochim Biophys Res Commun. 2001;286:1183–1190. doi: 10.1006/bbrc.2001.5483. [DOI] [PubMed] [Google Scholar]
- Schoecklmann HO, Rupprecht HD, Zauner I, Sterzel RB. TGF 1-induced cell cycle arrest in renal mesangial cells involves inhibition of cyclin E-cdk 2 activation and retinoblastoma protein phosphorylation. Kidney Int. 1997;51:1228–1236. doi: 10.1038/ki.1997.168. [DOI] [PubMed] [Google Scholar]
- Shankland SJ, Pippin J, Flanagan M, Coats SR, Nangaku M, Gordon KL, Roberts JM, Couser WG, Johnson RJ. Mesangial cell proliferation mediated by PDGF and bFGF is determined by levels of the cyclin kinase inhibitor p27 Kip1. Kidney Int. 1997;51:1088–1099. doi: 10.1038/ki.1997.151. [DOI] [PubMed] [Google Scholar]
- Sherr CJ. G1 phase progression: cycling on cue. Cell. 1994;79:551–555. doi: 10.1016/0092-8674(94)90540-1. [DOI] [PubMed] [Google Scholar]
- Yamabe H, Osawa H, Kaizuka M, Tsunoda S, Shirato K, Tateyama F, Okumura K. Platelet-derived growth factor, basic fibroblast growth factor, and interferon γ increase type IV collagen production in human fetal mesangial cells via a transforming growth factor-β-dependent mechanism. Nephrol Dial Transpl. 2000;15:872–876. doi: 10.1093/ndt/15.6.872. [DOI] [PubMed] [Google Scholar]
- Murphy M, Godson C, Cannon S, Kato S, Mackenzie HS, Martin F, Brady HR. Suppression subtractive hybridization identifies high glucose levels as a stimulus for expression of connective tissue growth factor and other genes in human mesangial cells. J Biol Chem. 1999;274:5830–5834. doi: 10.1074/jbc.274.9.5830. [DOI] [PubMed] [Google Scholar]
- Shin I, Yakes FM, Rojo F, Shin NY, Bakin AV, Baselga J, Arteaga CL. PKB/Akt mediates cell-cycle progression by phosphorylation of p27 Kip1 at threonine 157 and modulation of its cellular localization. Nat Med. 2002;8:1145–1152. doi: 10.1038/nm759. [DOI] [PubMed] [Google Scholar]
- Liang J, Zubovitz J, Petrocelli T, Kotchetkov R, Connor MK, Han K, Lee JH, Ciarallo S, Catzavelos C, Beniston R, Franssen E, Slingerland JM. PKB/Akt phosphorylates p27, impairs nuclear import of p27 and opposes p27-mediated G1 arrest. Nat Med. 2002;8:1153–1160. doi: 10.1038/nm761. [DOI] [PubMed] [Google Scholar]
- Viglietto G, Motti ML, Bruni P, Melillo RM, D’Alessio A, Califano D, Vinci F, Chiappetta G, Tsichlis P, Bellacosa A, Fusco A, Santoro M. Cytoplasmic relocalization and inhibition of the cyclin-dependent kinase inhibitor p27 (Kip1) by PKB/Akt-mediated phosphorylation in breast cancer. Nat Med. 2002;8:1161–1165. doi: 10.1038/nm762. [DOI] [PubMed] [Google Scholar]
- Presig PA, Franch HA. Renal epithelial hyperplasia and hypertrophy. Semin Nephrol. 1995;15:327–340. [PubMed] [Google Scholar]
- Sherr CJ. Mammalian G1 cyclins. Cell. 1993;73:1059–1065. doi: 10.1016/0092-8674(93)90636-5. [DOI] [PubMed] [Google Scholar]
- Morgan DO. principles of CDK regulation. Nature. 1995;374:131–134. doi: 10.1038/374131a0. [DOI] [PubMed] [Google Scholar]
- Sherr CJ, Roberts JM. Inhibitors of mammalian G1 cyclin-dependent kinases. Genes Dev. 1995;9:1149–1163. doi: 10.1101/gad.9.10.1149. [DOI] [PubMed] [Google Scholar]
- Perlman H, Bradley K, Liu H, Cole S, Shamiyeh E, Smith RC, Walsh K, Fiore S, Koch AE, Firestein GS, Haines KG, III, Pope RM. IL-6 and matrix metalloproteinase-1 are regulated by the cyclin-dependent kinase inhibitor p21 in synovial fibroblasts. J Immunol. 2003;170:838–845. doi: 10.4049/jimmunol.170.2.838. [DOI] [PubMed] [Google Scholar]
- Shankland SJ, Hugo C, Coats SR, Nangaku M, Pichler RH, Gordon KL, Pippin J, Roberts JM, Couser WG, Johnson RJ. Changes in cell cycle protein expression during experimental mesangial proliferative glomerulonephritis. Kidney Int. 1996;50:1230–1239. doi: 10.1038/ki.1996.432. [DOI] [PubMed] [Google Scholar]
- Franke TF, Kaplan DR, Cantley LC, Toker A. Direct regulation of the Akt proto-oncogene product by phosphatidylinositol-3, 4-bisphosphate. Science. 1997;275:665–668. doi: 10.1126/science.275.5300.665. [DOI] [PubMed] [Google Scholar]
- Zhou BP, Liao Y, Xia W, Zou Y, Spohn B, Hung MC. HER-2/neu induces p53 ubiquitination via Akt-mediated MDM2 phosphorylation. Nat Cell Biol. 2001;3:973–982. doi: 10.1038/ncb1101-973. [DOI] [PubMed] [Google Scholar]
- Montagnoli A, Fiore F, Eytan E, Carrano AC, Draetta GF, Hershko A, Pagano M. Ubiquitination of p27 is regulated by Cdk-dependent phosphorylation and trimeric complex formation. Genes Dev. 1999;13:1181–1189. doi: 10.1101/gad.13.9.1181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghosh Choudhury G, Lenin M, Calhaun C, Zhang JH, Abboud HE. PDGF inactivates forkhead family transcription factor by activation of Akt in glomerular mesangial cells. Cell Signal. 2002;15:161–70. doi: 10.1016/s0898-6568(02)00057-8. [DOI] [PubMed] [Google Scholar]
- Shankland SJ. Cell-cycle control and renal disease. Kidney Int. 1997;52:294–308. doi: 10.1038/ki.1997.335. [DOI] [PubMed] [Google Scholar]
- Monkawa T, Hiromura K, Wolf W, Shankland SJ. The hypertrophic effect of transforming growth factor b is reduced in the absence of cyclin-dependent kinase-inhibitors p21 and p27. J Am Soc Nephrol. 2002;13:1172–1178. doi: 10.1097/01.asn.0000013162.29833.45. [DOI] [PubMed] [Google Scholar]
- Colville-Nash PR, Gilroy DW. Potential adverse effects of cyclooxygenase-2 inhibition: evidence from animal models of inflammation. BioDrugs. 2001;15:1–9. doi: 10.2165/00063030-200115010-00001. [DOI] [PubMed] [Google Scholar]
- Mitchell S, Thomas G, Harvey K, Cottell D, Reville K, Berlasconi G, Petasis NA, Erwig L, Rees AJ, Savill J, Brady HR, Godson C. Lipoxins, aspirin-triggered epi-lipoxins, lipoxin stable analogues, and the resolution of inflammation: stimulation of macrophage phagocytosis of apoptotic neutrophils in vivo. J Am Soc Nephrol. 2002;13:2497–2507. doi: 10.1097/01.asn.0000032417.73640.72. [DOI] [PubMed] [Google Scholar]
- Serhan CN. Lipoxins and aspirin-triggered 15-epi-lipoxin biosynthesis: an update and role in anti-inflammation and pro-resolution. Prostaglandins Other Lipid Mediat. 2002;68–69:433–455. doi: 10.1016/s0090-6980(02)00047-3. [DOI] [PubMed] [Google Scholar]