Abstract
To gain further insight into the mode of action of S-locus receptor kinase (SRK), a receptor-like kinase involved in the self-incompatibility response in Brassica, different recombinant SRK proteins have been expressed in a membranous environment using the insect cell/baculovirus system. Recombinant SRK proteins exhibited properties close to those of the endogenous stigmatic SRK protein and were found to autophosphorylate on serine and threonine residues in insect cell microsomes. Autophosphorylation was constitutive because it did not require the presence of pollen or stigma extracts in the phosphorylation buffer. Phosphorylation was shown to occur in trans, suggesting the existence of constitutive homooligomers of membrane-anchored recombinant SRK. To investigate the physiological relevance of these results, we have examined the oligomeric status of SRK in planta in cross-linking experiments and by velocity sedimentation on sucrose gradients. Our data strongly suggest that SRK is associated both with other SRK molecules and other stigma proteins in nonpollinated flowers. These findings may have important implications for our understanding of self-pollen signaling.
Keywords: baculovirus, oligomerization, sporophytic self-incompatibility
In the Brassica family, the cell–cell interaction that leads to the rejection of self-pollen at the stigmatic surface [the self-incompatibility (SI) reaction] is controlled genetically by the multiallelic S-locus (1). When the pollen parent and the stigma share common S haplotypes, pollen germination or pollen tube growth is inhibited, thereby preventing self-fertilization. Several genes have been localized at the S locus but only two, the S-locus glycoprotein (SLG) and the S-locus receptor kinase (SRK) genes, exhibit the polymorphic nature expected for genes involved in the SI response (2, 3). SLG encodes a secreted glycoprotein that accumulates in the cell wall of papillae (4), whereas SRK encodes a plasmalemma-anchored glycoprotein (5, 6). SRK is structurally analogous to animal receptor kinases and belongs to the plant receptor-like kinase (RLK) family (7). Although its membrane topology has not been experimentally defined, DNA sequence analysis predicts that SRK consists of three domains: an extracellular glycosylated N-terminal domain (the S-domain) that shares extensive homology with SLG, a membrane-spanning domain, and a cytoplasmic domain (3). The cytoplasmic domain has been shown to have serine/threonine kinase activity when expressed in bacteria (8). In the S3 haplotype, SRK has been shown to encode, in addition to the integral membrane SRK protein, a soluble truncated form corresponding to the S-domain of SRK, the eSRK protein (9).
Although there is some controversy as to whether SLG is required for the SI response (10, 11), there is strong evidence that SRK is necessary. For example, SRK was shown to be mutated in two Brassica lines that exhibited a self-compatible phenotype (12, 13). Moreover, Stahl and coworkers (14) recently have shown that the SI phenotype was altered in transgenic Brassica napus plants expressing a kinase-defective SRK that seems to act as a dominant negative mutant. Their results strongly indicate that SRK is a key component of the SI reaction.
By analogy with animal receptor kinases, which transduce signals after interaction with peptidic extracellular ligands, it has been proposed that SRK initiates the SI reaction after the interaction of its S-domain with an as-yet-unknown self-pollen-borne ligand. Although candidates have been proposed for the male component of the SI response, there is not yet any molecular information concerning this molecule (15–17). The recognition by SRK of its putative ligand is thought to induce an activation of the kinase domain, leading to the recruitment of cytoplasmic targets that mediate the SI reaction. Two types of Brassica proteins have been shown to interact with the cytoplasmic domain of SRK: two thioredoxins and an arm repeat-containing protein (ARC1) (18, 19). The kinase interaction domains of the kinase-associated protein phosphatases of Arabidopsis thaliana and maize also were shown to interact with the kinase domain of SRK (20). ARC1 is perhaps the most interesting of these proteins because it is expressed specifically in Brassica stigmas and it interacts only with the autophosphorylated form of the SRK cytoplasmic domain.
Very little is known about the behavior of SRK in the plasma membrane, and particularly whether it can form homo- or hetero-oligomers. This is a crucial question because of the important role that oligomerization has been shown to play in the activation of receptor kinases in animals (21, 22). Here we show that recombinant integral membrane SRK protein expressed in the insect cell/baculovirus system is able to autophosphorylate. Autophosphorylation does not require the presence of a ligand and occurs at least partially by transphosphorylation mechanisms, indicating that recombinant SRK oligomerizes in the membrane. Moreover, we report biochemical evidence, based on cross-linking experiments and velocity sedimentation on sucrose gradients, that oligomeric complexes containing SRK occur naturally in planta at the stigmatic surface of mature unpollinated Brassica flowers. Current models, based on the structure of S locus genes, propose that self-pollen signaling is mediated by a SRK receptor complex. Our results provide physical evidence for the existence of an SRK receptor complex in planta.
Materials and Methods
Viral and Plasmid DNA Constructs.
Sequences encoding epitope tags were inserted into the reading frame of SRK3 (5) at a ClaI site 13 bp upstream of the termination codon so that the expressed proteins consisted of the reconstructed SRK polypeptide directly followed by either a hexahistidine sequence or the hemagglutinin (HA) epitope (YPYDVPDYA; ref. 23). The kinase-negative form of SRK was created by converting Lys-553 to an arginine codon by using site-specific mutagenesis PCR. SRK constructs were cloned into pBacPAK (CLONTECH) downstream of the polyhedrin promoter and cotransfected with Bsu30I-linearized BacPAK6 baculovius DNA into Spodoptera frugiperda Sf21 insect cells (CLONTECH).
Expression of Recombinant SRK and Protein Extraction.
Amplified recombinant baculoviruses were used to infect exponentially growing cells at a multiplicity of infection of either 10 for single infections or five of each virus for coinfections as described (24).
To extract proteins under denaturing conditions, washed insect cell pellets were resuspended at 1.5 × 106 cells per 100 μl in SDS/PAGE loading buffer [62.5 mM Tris, pH 6.8/10% (vol/vol) glycerol/3% (mass/vol) SDS/2.5% (mass/vol) DTT] and boiled for 5 min. Homogenates were cleared at 18,300 g for 20 min at 4°C, supernatants were acetone-precipitated, and protein concentration was determined (25).
To extract proteins under native conditions, washed cell pellets were solubilized in lysis buffer [50 mM Tris, pH 8/150 mM NaCl/1% (mass/vol) Triton X-100/10% (mass/vol) glycerol], to which 1 mM PMSF, 10 μg/ml leupeptine, 25 mM benzamidine, and 30 μg/ml aprotinine were added. Lysates were incubated for 30 min at 4°C and cleared at 10,000 g for 10 min at 4°C. To test whether recombinant SRK was membrane-anchored, extraction was performed under native conditions in the presence or absence of 1% (mass/vol) Triton X-100, and cleared lysates were centrifuged at 100,000 g for 1 h at 2°C. Proteins contained in the resulting supernatants were analyzed by immunoblotting. Immunocytofluorescence detection and determination of the topology of recombinant SRK are published as supplementary material on the PNAS website, www.pnas.org.
Phosphorylation in Vitro of Microsomal Membrane Preparations.
Microsomal membranes, prepared as described (26), were resuspended at a final protein concentration of 1.6 mg/ml in phosphorylation buffer containing 20 mM Hepes (pH 7.4), 0.1% (mass/vol) Triton X-100, 10 mM MgCl2, 2 mM MnCl2, 0.4 μCi/μl [γ-32P]ATP (Amersham Pharmacia, specific activity: 3,000 Ci/mmol, final concentration of radiolabeled ATP: 0.133 μM), phosphatase inhibitors (10 mM NaF, 0.1 mM Na3VO4, and 1 μM okadaic acid), and protease inhibitors (10 μg/ml leupeptine, 10 μg/ml aprotinine, and 1 mM PMSF) and incubated for 30 min at room temperature. The reaction was stopped by adding EDTA to a final concentration of 60 mM, and proteins were denatured by boiling for 5 min in SDS/PAGE loading buffer.
To specifically recover hexahistidine-tagged recombinant proteins after phosphorylation in vitro, denatured samples were desalted on handmade columns containing 300 μl of Bio-Gel P-6DG (Bio-Rad) equilibrated in chaotropic buffer (8 M urea/100 mM NaH2PO4/10 mM Tris, pH 8.0). Desalted samples then were diluted in 1 ml of chaotropic buffer and incubated at room temperature for 1 h with 50 μl of nickel chelated by nitrilotetra-acetic acid (Ni-NTA) agarose beads (Qiagen, Courtaboeuf, France). Bound hexahistidine-tagged proteins were recovered according to the procedure of the manufacturer.
Phosphoamino Acid and Phosphopeptide Analysis.
Radiolabeled proteins were separated as above and then electroblotted onto a poly(vinylidene difluoride) membrane. After autoradiography, radioactive bands of interest were excised for phosphoamino acid analysis as described (27). For phosphopeptide analysis, microsomes were radiolabeled as above, and proteins (200 μg) were separated by SDS/PAGE and stained with Coomassie blue. Phosphopeptide analysis was performed as described (28). Briefly, the gel band corresponding to SRK was excised and digested with 10 μg of trypsin (sequencing grade; Promega). Tryptic peptides then were separated by HPLC, and phosphopeptides were sequenced by Edman degradation using a Procise 492 Protein Sequencer (Applied Biosystems).
Cross-Linking Analysis of SRK in Planta.
Stigmas of unpollinated flowers of the S3 haplotype were ground in liquid nitrogen in a 1.5-ml tube and resuspended (4 μl per stigma) in solubilization buffer [25 mM Hepes, pH 7.4/1% (mass/vol) Triton X-100/10% (vol/vol) glycerol/5 mM β-mercaptoethanol/1 mM EDTA/5 mM EGTA/2 mM benzamidine/1 mM PMSF/5 μg/ml leupeptine/5 μg/ml aprotinine]. Homogenates were centrifuged at 12,000 g for 10 min at 2°C, and supernatants were recovered. Oligomeric complexes were cross-linked by adding glutaraldehyde to a final concentration of 80 mM. The cross-linking reaction was performed at room temperature for 1 min and quenched by adding glycine at pH 9.0 to a final concentration of 285 mM. Samples were denatured by addition of SDS/PAGE loading buffer and boiled for 5 min.
Velocity Sedimentation on Sucrose Gradients.
Proteins were extracted from 50 stigmas of the S3 haplotype as described above but in octyl-glucoside buffer (20 mM Hepes, pH 7.4/100 mM NaCl/35 mM octyl-glucoside). Molecular mass markers [3 μg/ml BSA (66 kDa), 2.5 μg/ml alcohol dehydrogenase (150 kDa), 1.5 μg/ml β-amylase (200 kDa), Sigma] were diluted in 50 μl of solubilization buffer and added to the stigma protein extract. The resulting mixture (200 μl) was layered at the top of a 4.0-ml gradient of 7.5–30% (mass/vol) sucrose in solubilization buffer, and gradients were centrifuged and analyzed as described (21). Gradient fractions were immunoblotted with mAb 85–36-71 and anti-SLG3N-ter antibodies (29) that detect either SRK3 and its soluble S-domain eSRK3 or SLG3, respectively. To test the effect of a strong denaturing detergent on the oligomeric status of SRK3, SDS at the final concentration of 0.5% (mass/vol) was added in the supernatant containing stigma proteins, in the molecular mass markers mix, and in the sucrose gradient.
Protein Immunoblotting.
Proteins were electrophoresed through a 7.5% SDS/PAGE gel, electroblotted, and immunodetected as described (5, 29).
Results
Expression of Recombinant SRK Proteins.
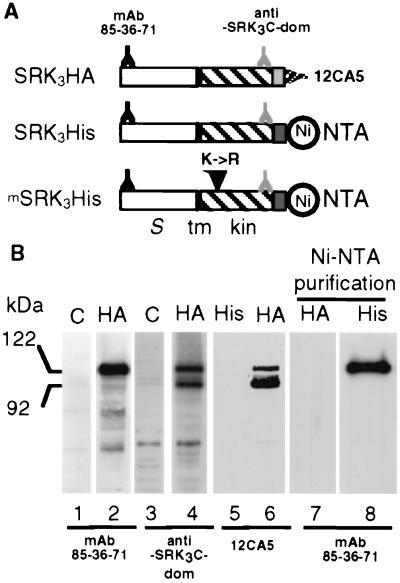
Three different forms of SRK3 were expressed in insect cells (Fig. 1A). These consisted of SRK3 bearing either the HA epitope (SRK3HA) or the hexahistidine tag (SRK3His), and of a kinase-defective form of SRK3His (mSRK3His). Fig. 1B shows that infection of cells with a baculovirus containing the SRK3HA construct resulted in the accumulation of a 122-kDa protein, which was recognized by two different anti-SRK3 antibodies (lanes 2 and 4) and the anti-HA antibody (lane 6). Both the anti-SRK3C-dom and anti-HA antibodies also detected a second major protein of 92 kDa (lanes 4 and 6), which probably corresponded to a truncated product of SRK lacking the N terminus. We confirmed that the 122-kDa protein corresponded to SRK by MS analysis, and we found by deglycosylation experiments that the molecular mass of the glycan side chains was similar to that observed for SRK from stigmas (data not shown). Fig. 1B (lanes 7 and 8) shows that Ni-NTA agarose beads allowed the specific recovery of SRK3His (and mSRK3His, data not shown). Immunocytological analysis indicated that only a small proportion of recombinant SRK was addressed to the plasmalemma, the majority being retained in vesicles under the cell surface (see Fig. 6, which is provided as supplementary material on the PNAS web site, www.pnas.org). Treatment of microsome fractions with trypsin in the presence of different concentrations of Triton X-100 indicated that the recombinant SRK spanned the membrane and was orientated with its S domain in the lumen of the microsomal vesicles (see Fig. 6). Biochemical experiments therefore were carried out on microsomal preparations.
Figure 1.
Expression of recombinant SRK3 proteins in insect cells. (A) Schematic representation of the three recombinant SRK proteins, SRK3HA, SRK3His, and mSRK3His. The position of the different epitopes and tags are shown. The N and C termini of the recombinant proteins are to the left and right, respectively. The white rectangle represents the S-domain (S), the black vertical bar the membrane-spanning (tm) domain, the hatched rectangle indicates the cytoplasmic domain (kin) and the light and dark gray stippled rectangles indicate the HA epitope and hexahistidine tag, respectively. Epitopes recognized by different antibodies and the binding site for Ni-NTA are indicated. The substitution of Lys-553 with an arginine (K->R) in the mSRK3His construct is indicated by a vertical arrowhead. (B) Immunoblotting of SRK recombinant proteins. Proteins extracted from Sf21 cells infected by the parental baculovirus (C) (lanes 1 and 3) or from Sf21 cells infected with baculovirus driving the expression of SRK3HA (HA) (lanes 2, 4, 6, and 7) or SRK3His (His) (lanes 5 and 8) were separated by SDS/PAGE and electroblotted. In some cases, electrophoresis was preceded by a purification with Ni-NTA agarose beads as indicated.
In Vitro Phosphorylation Assays on Microsomal Membranes.
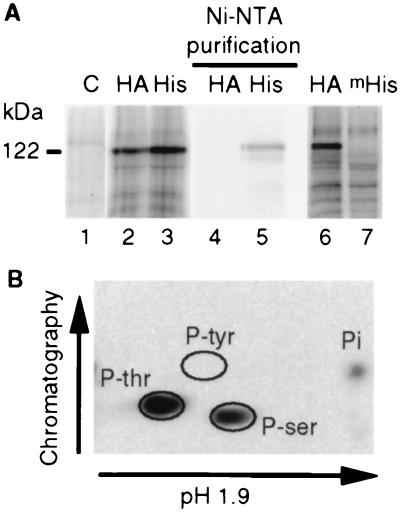
The cytoplasmic domain of SRK has been shown to have serine/threonine kinase activity when expressed in Escherichia coli (8, 30). To determine whether SRK exhibited the same catalytic property in a membranous environment, an in vitro phosphorylation assay was performed on microsomes containing SRK. Phosphorylated proteins were analyzed by SDS/PAGE and detected by autoradiography. Microsomes prepared from noninfected Sf21 cells were used as a negative control. A major phosphorylated band of 122 kDa was labeled in microsomes from cells expressing SRK3HA and SRK3His but was absent from the control (Fig. 2A). To verify that this product corresponded to SRK, we purified SRK3His with Ni-NTA-agarose beads after radiolabeling of microsomes. As shown in Fig. 2A (lanes 4 and 5), the 122-kDa band was specifically recovered from microsomes containing SRK3His but not from SRK3HA-containing microsomes used as a negative control. To demonstrate that the phosphorylation of SRK did not result from an endogenous insect cell kinase, we constructed a kinase-defective form of SRK3His in which the invariant lysine of subdomain II (Lys-553), which is thought to be involved in the interaction with ATP (31), was replaced by an arginine. We characterized the phosphorylation status of this mutated protein (mSRK3His) in radiolabeled microsomes. Our results indicated that mSRK3His was not phosphorylated in contrast with SRK3HA used as a positive control (Fig. 2A, lanes 6 and 7). Taken together these results indicated that SRK was able to autophosphorylate in the membranous environment of insect cell microsomes. The nature of the phosphorylated residues was determined. As shown in Fig. 2B, only phosphorylated serine and threonine residues were detected, demonstrating a constitutive serine/threonine kinase activity.
Figure 2.
Recombinant SRK autophosphorylates on serine and threonine residues in a membranous environment. (A) Microsomes from uninfected Sf21 cells (C) or from Sf21 cells expressing SRK3HA (HA), SRK3His (His) or kinase defective mSRK3His (mHis) were radiolabeled with [γ-32P]ATP. In some cases, proteins (lanes 4 and 5) were purified on Ni-NTA agarose beads after radiolabeling. Proteins were separated by SDS/PAGE and detected by autoradiography. (B) Radiolabeled SRK3His was purified on Ni-NTA agarose beads and hydrolyzed. Free amino acids were separated in two dimensions by chromatography and electrophoresis as indicated, and radiolabeled amino acids were detected by autoradiography. Phosphoamino acids (P-ser, P-thr, and P-tyr for phosphoserine, phosphothreonine, and phosphotyrosine, respectively) were positioned by staining nonradiolabeled phosphoamino acids, added before separation, with nihydrin. Pi indicates inorganic phosphate.
To investigate whether the kinase activity of SRK may be up-regulated by pollen components (containing the putative S-ligand) or as a result of dimerization induced by bivalent antibodies, microsomes containing recombinant SRK were incubated in the presence of various concentrations of pollen proteins or the anti-SRK3N-ter antibody. Neither treatment resulted in an increase in the phosphorylation level of SRK (data not shown). These results suggest that either (i) SRK possesses a constitutive basal kinase activity that is not up-regulated by ligand interaction and/or induced dimerization, or (ii) that SRK may require a cofactor, which is absent in our assay, for its complete activation.
To define more precisely the position of the phosphorylated residues, we attempted to sequence tryptic peptide fragments of radiolabeled recombinant SRK. Only one radioactive peptide (914 cps/min) gave an unambiguous sequence: SPYELDPSSSR. This peptide corresponded to the region from Ser-792 to Arg-802 within the C-terminal region of the SRK3 kinase domain. We were unable to determine which of the four serine residues were phosphorylated but a search for consensus phosphorylation sequences with the prosite database revealed that Ser-792 and Ser-799 reside in typical consensus phosphorylation sites for casein kinase II and protein kinase C, respectively. These data suggest that SRK may possess a kinase activity with a specificity similar to casein kinase II and/or protein kinase C.
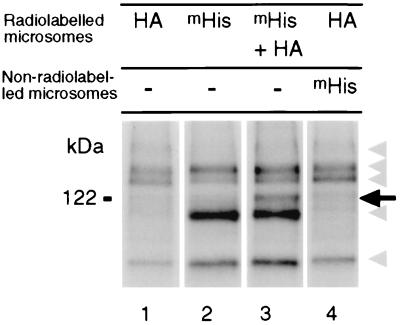
To determine whether SRK autophosphorylation involved an intramolecular or an intermolecular mechanism, we performed in vitro phosphorylation assays on microsomes that contained both the functional SRK3HA and the kinase-defective mSRK3His. These microsomes were prepared from Sf21 cells coinfected with the two appropriate recombinant baculoviruses. After radiolabeling, mSRK3His protein was recovered by affinity binding to Ni-NTA agarose beads. If autophosphorylation had occurred by an intermolecular mechanism, the mSRK3His protein should have been phosphorylated by SRK3HA. In contrast, if autophosphorylation only occurred by an intramolecular mechanism purified mSRK3His should not be radiolabeled. Fig. 3 shows that several different phosphoproteins were recovered, but for the sake of simplicity, we will focus on the integral mSRK3His of 122 kDa. The 122-kDa phosphoprotein was not recovered if SRK3HA or mSRK3His were expressed alone (Fig. 3, lanes 1 and 2) as expected, because SRK3HA does not bind the Ni-NTA agarose beads and mSRK3His is not able to autophosphorylate. However, when mSRK3His was coexpressed with SRK3HA, it became phosphorylated as shown by the appearance of a new radiolabeled protein at 122 kDa (Fig. 3, lane 3). One possibility that we considered was that autophosphorylated SRK3HA might bind to the Ni-NTA agarose beads in the presence of mSRK3His, by virtue of an indirect interaction with the bound mSRK3His protein. To rule out this possibility, we added nonradiolabeled mSRK3His just before the purification step and showed that SRK3HA was not recovered under these conditions (Fig. 3, lane 4). Together, these data strongly suggested that autophosphorylation of membrane-anchored recombinant SRK involved an intermolecular mechanism and transient or stable oligomerization.
Figure 3.
Intermolecular phosphorylation of recombinant SRK proteins. Microsomes from Sf21 cells expressing either SRK3HA (HA) or kinase-defective mSRK3His (mHis) or from Sf21 cells coexpressing mSRK3His and SRK3HA (mHis + HA) were radiolabeled, and proteins carrying the hexahistidine tag were purified on Ni-NTA agarose beads. For the sample shown in lane 4, extracts of unlabeled microsomes from cells expressing mSRK3His were added before purification on Ni-NTA agarose beads. Proteins eluted from Ni-NTA agarose beads were separated by SDS/PAGE and analyzed by autoradiography. Truncated recombinant SRK3 and contaminating polypeptides are indicated by gray arrowheads, and phosphorylated SRK is indicated by an arrow.
Oligomeric Status of SRK in Planta.
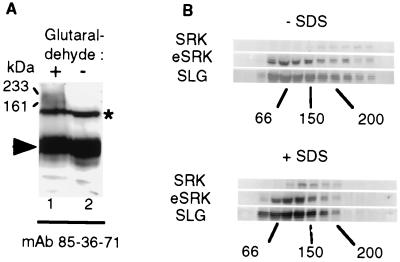
To test whether oligomeric complexes of SRK also exist in planta, proteins from unpollinated stigmas were solubilized in the presence of Triton X-100 and cross-linked by addition of glutaraldehyde. Proteins were analyzed by immunoblotting with mAb 85–36-71. Fig. 4A shows that comparison of glutaraldehyde-treated with untreated extracts revealed two additional products with apparent molecular masses of 233 and 161 kDa. The 233-kDa product had a molecular mass that was very similar to that expected for an SRK dimer (240 kDa). This result indicated that SRK may form complexes either with other SRK molecules and/or with other proteins in unpollinated stigmas.
Figure 4.
Identification of SRK oligomeric complexes in stigma extract. (A) Proteins were extracted from stigmas expressing the S3 haplotype in a Triton X-100-containing buffer and either treated (+) or not (−) with the cross-linking reagent glutaraldehyde. Proteins then were separated by SDS/PAGE and immunoblotted with mAb 85–36-71. SRK3-containing complexes of 161 and 233 kDa are indicated by arrows. Monomeric SRK3 and eSRK3 are indicated with an asterisk and an arrowhead, respectively. (B) Velocity sedimentation on sucrose gradients. Proteins were extracted in buffer containing octyl-glucoside, and stigma extracts were either supplemented with SDS at the concentration of 0.5% (mass/vol) (+SDS) or not (−SDS). The presence of SRK3 (SRK), eSRK3 (eSRK), and SLG3 (SLG) in the different fractions was examined by immunoblotting with mAb 85–36-71 and anti-SLG3 antibodies. The distribution of molecular mass markers (66, 150, and 200 kDa) is indicated at the bottom of each panel.
To verify this result, we performed velocity sedimentation on sucrose gradients and examined the oligomeric status of SRK in the presence or absence of 0.5% SDS, which destabilizes weak oligomeric interactions. Fig. 4B indicates that the majority of SRK protein was in fractions of a molecular mass greater than 200 kDa in the absence of SDS. In contrast, in the presence of SDS, SRK was predominantly concentrated in fractions corresponding to 66–150 kDa. The change in the distribution of SRK in the presence of SDS suggests that SRK was mainly oligomeric under native conditions and that SRK oligomers were dissociated by SDS. Interestingly, the distribution of the soluble proteins eSRK and SLG was not greatly affected in the presence of SDS, suggesting that these proteins are mainly monomeric. However, in the absence of SDS, some of the eSRK and SLG protein was detected in fractions corresponding to high molecular masses, indicating that a small proportion of each protein may be included in oligomeric complexes. As SLG and eSRK are principally monomeric, in contrast to SRK, we conclude that the 160- and 240-kDa products detected after cross-linking experiments corresponded to an SRK heteromer (for example, an SRK monomer associated with one molecule of SLG or eSRK) and an SRK homodimer, respectively. Apart from these molecular complexes based on noncovalent interactions, the existence of disulphide-bonded SLG homodimers has been reported (32). According to our velocity sedimentation experiments, such covalent SLG complexes do not seem to implicate the major fraction of SLG molecules as the peak of SLG was found to correspond to that of the 67-kDa BSA molecular mass marker, in the presence or absence of SDS. We have confirmed that a disulphide-linked SLG complex is present in stigmas of the S3 haplotype by Western blotting of stigmatic extracts separated under nonreducing conditions (data not shown). If stigma proteins are extracted in the absence of DTT a high molecular mass form (approximately 110 kDa) of SLG that could correspond to a disulphide-bonded homodimer is detected. However, this 110-kDa complex is considerably less abundant than the monomeric form of SLG in the S3 haplotype.
Discussion
It is difficult to investigate the molecular mechanism by which SRK mediates the SI response in planta because of the low level of expression of SRK in Brassica. Moreover, attempts to overexpress SRK in Brassica have encountered problems because of cosuppression effects (14, 33). To overcome this obstacle and to gain more insight into how SRK functions at the molecular level, we expressed recombinant SRK in the insect cell/baculovirus system. The recombinant SRK proteins closely resembled SRK extracted from stigmas. They had a similar molecular mass, were solubilized in the presence of Triton X-100, were N-glycosylated, and were membrane-localized. However, although SRK was correctly inserted into the membrane, with the C terminus extending into the cytoplasm, the majority of the recombinant SRK was not targeted to the plasmalemma of the insect cells. This finding contrasts with results obtained for animal receptor kinases, such as epidermal growth factor receptor or insulin receptor, which have been shown to be correctly addressed to the plasmalemma when expressed in insect cells (34, 35). Nevertheless, it is worth noting that plant K+ channels, which are plasma membrane proteins, also were retained mainly in the internal cellular membranes when expressed in the insect cell/baculovirus system (36, 37).
The observation that recombinant protein spanned the membrane reinforces the similarities between SRK and animal receptor kinases and supports models in which SRK transduces a signal from the extracellular space to the cytoplasm.
The kinase domains of SRK (8, 30) and several other RLKs (38–41) have been shown to possess serine/threonine kinase activity when expressed as soluble polypeptides in E. coli. Currently, the only exception among the plant RLK family is PRK1, a pollen-specific RLK from Petunia inflata, which has been shown to autophosphorylate on serine and tyrosine residues (42). In the case of Arabidopsis protein kinase 1, which exhibits serine/threonine/tyrosine kinase activity, it has been proposed that the dual specificity may depend on the presence of consensus sequences from both serine/threonine kinases (subdomains VIb and VIII) and tyrosine kinases (subdomain IX) (43). The catalytic domain of SRK possesses consensus motifs of serine/threonine kinases (subdomains VI and VIII) in addition to sequences closely related to tyrosine kinases (subdomain IX). Therefore, it was particularly interesting to determine the kinase-specific activity of the integral SRK in a membranous environment. Under our experimental conditions, SRK was shown to autophosphorylate on serine and threonine but no tyrosine phosphorylation was detected.
Autophosphorylation of SRK in insect cell microsomes was shown to involve, at least in part, an intermolecular mechanism indicating that recombinant SRK associates in constitutive oligomeric complexes. We cannot exclude the possibility that such complexes form as a result of the high concentration of recombinant SRK in the microsomal environment. However, data from cross-linking experiments and velocity sedimentation on sucrose gradients strongly suggest that SRK oligomers occur naturally in planta. The Arabidopsis RLK CLAVATA1 (CLV1) recently has been shown to be part of a complex including kinase-associated protein phosphatases, a Rho-related protein, and possibly more than one molecule of CLV1 (44), suggesting that signaling complexes may be a common feature of receptors of the RLK family.
In animals, two main mechanisms of receptor kinase activation have been described depending on the kinase specificity of the receptor considered (22). Thus, tyrosine kinase receptors interact with their ligands to form homodimers or heterodimers with closely related proteins. This dimerization step allows transphosphorylation of their kinase domains by bringing them close together and activating the receptor. In contrast, signal transduction by serine/threonine kinase receptors is mediated by two structurally dissimilar receptor types, the type I and type II receptors. For example, in transforming growth factor β signal transduction, the type II receptor forms constitutive, autophoshorylated dimers that interact with the ligand and the type I receptor to form a heterooligomer (21, 45). In this complex, the type I receptor is activated, after its phosphorylation by the type II receptor, and transduces the signal (45). In our study, we have shown that recombinant SRK autophosphorylates constitutively and exists as dimers in nonpollinated stigmas. The fact that a recombinant kinase-defective SRK expressed in transgenic Brassica plants acts as a dominant negative mutant further suggests that homooligomerization of fully functional SRK is necessary for the SI reaction (14). However, the detection of SRK dimers in nonpollinated stigmas indicates that the activation of SRK during the SI response requires an additional step after the formation of SRK homooligomers.
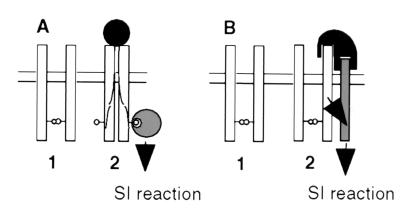
On the basis of our results, we propose that activation of SRK is not coincidental with the formation of SRK oligomers but instead results from the modification of a pre-existing SRK oligomeric complex. Two models are proposed (Fig. 5). In the first model, SRK/ligand interaction would lead to conformational changes in the kinase domain allowing the recruitment of a specific substrate(s) (Fig. 5A). This idea is supported by the fact conformational modification of the kinase domain of SRK910 from B. napus seems to be required for its interaction with thioredoxin THL1 (18). In this model, SRK would mediate both haplotype-specific recognition of the pollen ligand and the recruitment of downstream substrates. Indeed, one (or more) phosphoserine(s) present in the C terminus of recombinant SRK could act as docking sites for the recruitment of cytoplasmic targets. In the second model, SRK would function in a manner analogous to transforming growth factor β receptor type II. The role of SRK would be to recruit (and perhaps activate) a second membrane-anchored coreceptor that could interact with the SRK/pollen ligand complex (Fig. 5B). This model assumes that the two steps of the SI response, i.e., haplotype-specific recognition and signal transduction, are performed by two different receptors. To test these models, it will be necessary to characterize the constituents of SRK complexes in stigmas both before and after pollination with self-incompatible and cross-compatible pollen.
Figure 5.
Two models for the molecular mechanism of signal transduction via SRK in the SI response. (A) This model proposes that SRK (open rectangle) spontaneously associates as a dimer in the plasmalemma of stigmatic papillar cells before pollination. Interaction with a self-pollen borne ligand (filled circle) induces a conformational change of SRK, which allows the recruitment of cytoplasmic targets (large hatched circles) that mediate the SI response. In the example shown here, the cytoplasmic targets recognize phosphorylated residues (small circles) on the SRK protein. (B) In the second model, SRK is also present as a constitutively formed dimer but the activation of SI response by pollen-ligand (in black) requires both binding to SRK in an allele-specific manner, and then binding to a second as-yet-unknown coreceptor (hatched rectangle), which does not need to exhibit any allelic specificity. Signal transduction then may involve interaction of the second receptor with cytosolic targets analogous to those described for serine/threonine receptor kinases in animals.
Studies of self-compatible mutants and transgenic Brassica plants in which the SRK kinase activity is defective have indicated that SRK is necessary for the SI response. If SRK has the same constitutive kinase activity in planta as we have observed in insect cells, we might expect the SI response to be constitutively activated and the stigma to reject both self- and cross-pollen. But as the stigma permits cross-pollination, we suggest that the SRK signal transduction pathway may be inhibited both in the absence of pollination and after cross-pollination, for example by proteins that inhibit SRK kinase activity in stigmas. Interestingly, in animals the FK 506 binding protein (FKBP12) has been shown to inhibit the signaling activity of the transforming growth factor β receptor (TβR) type I/TβR type II complex, which can form even in the absence of ligand (46). In the case of SRK, proteins that interact with the SRK kinase domain, such as thioredoxins, ARC1, and kinase-associated protein phosphatases (18–20), could be involved either in transducing the signal downstream of SRK or in the inhibition of signal transduction in the absence of self-pollen. The identification of a 161-kDa SRK complex in unpollinated stigmas suggests that SRK may be associated with other proteins before pollination, and one possibility is that the formation of such complexes has a negative regulatory effect on SRK. The role of these complexes in the regulation of SRK needs to be precisely defined in the future by investigating their occurrence in cross- and self-pollinated stigmas. The approaches developed in this study, in particular the baculovirus/insect cell system, which allows the coexpression of two different proteins, and velocity sedimentation, open possibilities to characterize physical associations between SRK and other putative components (SLG, eSRK, cytosolic proteins, etc.) of the SRK receptor complex.
Supplementary Material
Acknowledgments
We are very grateful to Christophe Grangeasse, Marguerite-Marie Boutillon, Marie-Claire Ronzière (Institut de Biologie et de Chimie des Protéines de Lyon), and Jérôme Garin (Centre de l'Energie Atomique, Grenoble) for their help. We also thank Anne-Marie Thierry and Richard Blanc for their technical assistance. T.G. is a member of the Centre National de la Recherche Scientifique.
Abbreviations
- Ni-NTA
nickel chelated by nitrilotetra-acetic acid
- RLK
receptor-like kinase
- Sf21
cell line derived from Spodoptera frugiperda
- SI
self-incompatibility
- SLG
S-locus glycoprotein
- SRK
S-locus receptor kinase
- HA
hemagglutinin
Footnotes
Article published online before print: Proc. Natl. Acad. Sci. USA, 10.1073/pnas.070025097.
Article and publication date are at www.pnas.org/cgi/doi/10.1073/pnas.070025097
References
- 1.De Nettancourt D. Incompatibility in Angiosperms. New York: Springer; 1977. [Google Scholar]
- 2.Nasrallah J B, Kao T-H, Goldberg M L, Nasrallah M E. Nature (London) 1985;318:617–618. [Google Scholar]
- 3.Stein J C, Howlett B, Boyes D C, Nasrallah M E, Nasrallah J B. Proc Natl Acad Sci USA. 1991;88:8816–8820. doi: 10.1073/pnas.88.19.8816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kandasamy M K, Paolillo D J, Faraday C D, Nasrallah J B, Nasrallah M E. Dev Biol. 1989;134:462–472. doi: 10.1016/0012-1606(89)90119-x. [DOI] [PubMed] [Google Scholar]
- 5.Delorme V, Giranton J L, Hatzfeld Y, Friry A, Heizmann P, Ariza M J, Dumas C, Gaude T, Cock J M. Plant J. 1995;7:429–440. doi: 10.1046/j.1365-313x.1995.7030429.x. [DOI] [PubMed] [Google Scholar]
- 6.Stein J C, Dixit R, Nasrallah M E, Nasrallah J B. Plant Cell. 1996;8:429–445. doi: 10.1105/tpc.8.3.429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Braun D M, Walker J C. Trends Biochem Sci. 1996;21:70–73. [PubMed] [Google Scholar]
- 8.Goring D, Rothstein S J. Plant Cell. 1992;4:1273–1281. doi: 10.1105/tpc.4.10.1273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Giranton J L, Ariza M J, Dumas C, Cock J M, Gaude T. Plant J. 1995;8:827–834. doi: 10.1046/j.1365-313x.1995.8060827.x. [DOI] [PubMed] [Google Scholar]
- 10.Nasrallah M E, Kandasamy M K, Nasrallah J B. Plant J. 1992;2:497–506. [Google Scholar]
- 11.Gaude T, Rougier M, Heizmann P, Ockendon D J, Dumas C. Plant Mol Biol. 1995;27:1003–1014. doi: 10.1007/BF00037027. [DOI] [PubMed] [Google Scholar]
- 12.Goring D R, Glavin T L, Schafer U, Rothstein S J. Plant Cell. 1993;5:531–539. doi: 10.1105/tpc.5.5.531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Nasrallah J B, Rundle S J, Nasrallah M E. Plant J. 1994;5:373–384. [Google Scholar]
- 14.Stahl R J, Arnoldo M, Glavin T L, Goring D R, Rothstein S J. Plant Cell. 1998;10:209–218. doi: 10.1105/tpc.10.2.209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Boyes D C, Nasrallah J B. Plant Cell. 1995;7:1283–1294. doi: 10.1105/tpc.7.8.1283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pastuglia M, Ruffio-Chable V, Delorme V, Gaude T, Dumas C, Cock J M. Plant Cell. 1997;9:2065–2076. doi: 10.1105/tpc.9.11.2065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Stephenson A G, Doughty J, Dixon S, Elleman C, Hiscock S, Dickinson H G. Plant J. 1997;12:1351–1359. [Google Scholar]
- 18.Bower M S, Matias D D, Fernandes-Carvalho E, Mazzurco M, Gu T, Rothstein S J, Goring D R. Plant Cell. 1996;8:1641–1650. doi: 10.1105/tpc.8.9.1641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gu T, Mazzurco M, Sulaman W, Matias D D, Goring D R. Proc Natl Acad Sci USA. 1998;95:382–387. doi: 10.1073/pnas.95.1.382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Braun D M, Stone J M, Walker J C. Plant J. 1997;12:83–95. doi: 10.1046/j.1365-313x.1997.12010083.x. [DOI] [PubMed] [Google Scholar]
- 21.Gilboa L, Wells R G, Lodish H F, Henis Y I. J Cell Biol. 1998;140:767–777. doi: 10.1083/jcb.140.4.767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Heldin C H. Cell. 1995;80:213–223. doi: 10.1016/0092-8674(95)90404-2. [DOI] [PubMed] [Google Scholar]
- 23.Jou W M, Verhoeyen M, Devos R, Saman E, Fang R, Huylebroeck D, Fiers W, Threlfall G, Barber C, Carey N, Emtage S. Cell. 1980;19:683–696. doi: 10.1016/s0092-8674(80)80045-6. [DOI] [PubMed] [Google Scholar]
- 24.O'Reilly D R, Miller L K, Luckow V A. Baculovirus Expression Vectors: A Laboratory Manual. Oxford, U.K.: Oxford Univ. Press; 1994. [Google Scholar]
- 25.Lowry O, Rosenbrough N, Farr A, Randall R. J Biol Chem. 1951;193:265–275. [PubMed] [Google Scholar]
- 26.Scheel A A, Pelham H R. Biochemistry. 1996;35:10203–10209. doi: 10.1021/bi960807x. [DOI] [PubMed] [Google Scholar]
- 27.Duclos B, Grangeasse C, Vaganay E, Riberty M, Cozzone A J. J Mol Biol. 1996;259:891–895. doi: 10.1006/jmbi.1996.0366. [DOI] [PubMed] [Google Scholar]
- 28.Moritz R L, Eddes J S, Reid G E, Simpson R J. Electrophoresis. 1996;17:907–917. doi: 10.1002/elps.1150170512. [DOI] [PubMed] [Google Scholar]
- 29.Gaude T, Friry A, Heizmann P, Mariac C, Rougier M, Fobis I, Dumas C. Plant Cell. 1993;5:75–86. doi: 10.1105/tpc.5.1.75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Stein J C, Nasrallah J B. Plant Physiol. 1993;101:1103–1106. doi: 10.1104/pp.101.3.1103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hanks S K, Quinn A M, Hunter T. Science. 1988;241:42–52. doi: 10.1126/science.3291115. [DOI] [PubMed] [Google Scholar]
- 32.Doughty J, Dixon S, Hiscock S J, Willis A C, Parkin I A, Dickinson H G. Plant Cell. 1998;10:1333–1347. doi: 10.1105/tpc.10.8.1333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Conner J A, Tantikanjana T, Stein J C, Kandasamy M K, Nasrallah J B, Nasrallah M E. Plant J. 1997;11:809–823. [Google Scholar]
- 34.Greenfield C, Patel G, Clark S, Jones N, Waterfield M D. EMBO J. 1988;7:139–146. doi: 10.1002/j.1460-2075.1988.tb02793.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Paul J I, Tavare J, Denton R M, Steiner D F. J Biol Chem. 1990;265:13074–13083. [PubMed] [Google Scholar]
- 36.Gaymard F, Cerutti M, Horeau C, Lemaillet G, Urbach S, Ravallec M, Devauchelle G, Sentenac H, Thibaud J B. J Biol Chem. 1996;271:22863–22870. doi: 10.1074/jbc.271.37.22863. [DOI] [PubMed] [Google Scholar]
- 37.Zimmermann S, Talke I, Ehrhardt T, Nast G, Muller-Rober B. Plant Physiol. 1998;116:879–890. doi: 10.1104/pp.116.3.879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Chang C, Schaller G E, Patterson S E, Kwok S F, Meyerowitz E M, Bleeker A B. Plant Cell. 1992;4:1263–1271. doi: 10.1105/tpc.4.10.1263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Horn M A, Walker J C. Biochim Biophys Acta. 1994;1208:65–74. doi: 10.1016/0167-4838(94)90160-0. [DOI] [PubMed] [Google Scholar]
- 40.Schulze-Muth P, Irmler S, Schroder G, Schroder J. J Biol Chem. 1996;271:26684–26689. doi: 10.1074/jbc.271.43.26684. [DOI] [PubMed] [Google Scholar]
- 41.Williams R W, Wilson J M, Meyerowitz E M. Proc Natl Acad Sci USA. 1997;94:10467–10472. doi: 10.1073/pnas.94.19.10467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Mu J H, Lee H S, Kao T H. Plant Cell. 1994;6:709–721. doi: 10.1105/tpc.6.5.709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hirayama T, Oka A. Plant Mol Biol. 1992;20:653–662. doi: 10.1007/BF00046450. [DOI] [PubMed] [Google Scholar]
- 44.Trotochaud A E, Hao T, Wu G, Yang Z, Clark S E. Plant Cell. 1999;11:393–406. doi: 10.1105/tpc.11.3.393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Wrana J L, Attisano L, Wieser R, Ventura F, Massague J. Nature (London) 1994;370:341–347. doi: 10.1038/370341a0. [DOI] [PubMed] [Google Scholar]
- 46.Chen Y G, Liu F, Massague J. EMBO J. 1997;16:3866–3876. doi: 10.1093/emboj/16.13.3866. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.