Abstract
During B lymphocyte development, antibody genes are assembled by DNA recombination. Successful cell surface expression of IgM promotes developmental progression. However, when antigen receptors bind autoantigen, development is blocked and ongoing antibody gene recombination occurs, which often alters antibody specificity in a process called receptor editing. We demonstrate here a significant role of developmental block and receptor editing in B cell receptor quality control. During development a functional, non-self-reactive receptor undergoes receptor editing if its expression is below a certain threshold. Doubling the receptor gene dose promotes development in the absence of autoantigen, but allows editing when autoantigen is present. Thus, both underexpressed and harmful B cell receptors can undergo correction by receptor editing.
Each newly formed B cell expresses on the plasma membrane a unique Ig-H/L-chain combination, which is tested for self-reactivity. At this developmental stage, surface Ig (sIg) binding to self-antigens leads to developmental block and elimination of autoreactive B cells by cell death, or to alteration of antibody specificity by receptor editing (1–10). When appropriate IgM appears on the surface of developing B cells, further maturation follows, but expression of a nonautoreactive Ig-H/L-chain complex may be insufficient to promote B cell maturation (11). Because B cell maturation is correlated with reduced abundance of recombinase activator gene (RAG) mRNAs (8, 12–14), lack of recombinase down-regulation may promote receptor editing for reasons unrelated to immune tolerance.
In this paper we study the role of sIg in promoting B cell development in mice expressing the 3-83 B cell receptor (BCR), which binds to H-2K molecules from several haplotypes, including H-2b and H-2k, but fails to react to H-2d (15). In a conventional 3-83 transgenic mouse line (3-83Tg) that carries genes encoding the IgM and IgD forms of 3-83 (16), endogenous Ig-L rearrangements are suppressed on an H-2d (i.e., antigen-free) genetic background, but are stimulated when antigen is present (7, 17–19). In contrast, in mice with individually targeted Ig-H and -L gene loci encoding the 3-83 BCR genes placed in the physiological genomic context (3-83KI mice) (20, 21) H-2d individuals are predicted to lack a reactive self-antigen, yet their B cells undergo extensive receptor editing (21). This result was unexpected because Ig expression should be better regulated in the knock-in (KI) mice than in conventional Tg mice. It was originally suggested that in the 3-83KI mice negative selection induced receptor editing, either because a cryptic self-antigen was contributed by the 129 strain of the embryonic stem cell from which the 3-83KI mouse was derived, or because of a speculated affinity of 3-83 for H-2d antigens (21). An alternative hypothesis to account for the extensive receptor editing in 3-83KI B cells is that 3-83 antigen receptor expression in these cells is insufficient to promote developmental progression. We tested these two hypotheses and present evidence in support of the latter.
Materials and Methods
Mice.
The 3-83Tg mice (16) were backcrossed a minimum of 10 times on a B10.D2nSn/J background. The 3-83 knock-in mice (3-83KI) were described previously (20, 21) and were backcrossed a minimum of 6 times on a B10.D2nSn/J background. For hybridoma analysis, Ig κ-deficient mice (JCκD/JCκD) (22) were bred on the C57BL6 or B10.D2 backgrounds and (JCκD/3-83KI)F1 mice were generated. In the bone marrow chimera experiments depicted in Fig. 3, recipient mice were either C57BL6/J (H-2b) or B10.D2nSn/J (H-2d).
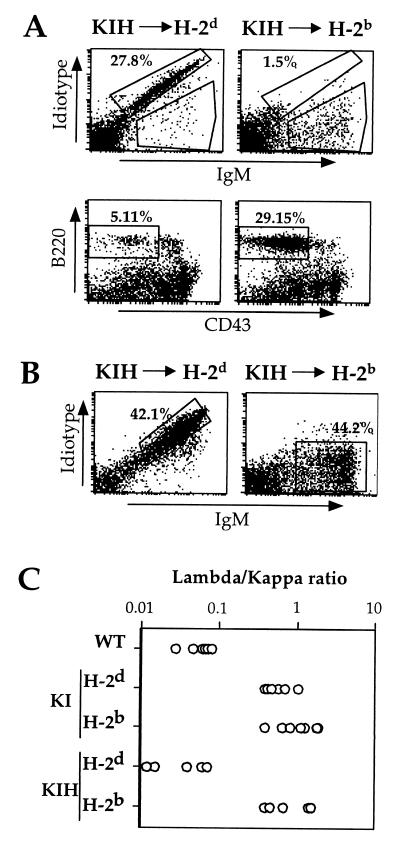
Figure 3.
Efficient antigen-dependent receptor editing in 3-83KIH-reconstituted bone marrow chimeras. Lethally radiated antigen-free B10.D2 (H-2d) or antigen-carrying C57BL6 (H-2b) mice were reconstituted with 3-83KIH bone marrow and analyzed 3 weeks later. (A) Bone marrow B cell expression of 3-83 Id and IgM (Upper). Expression of B220 and CD43 on gated, IgM− cells (Lower); window shows frequencies of small pre-B type cells. (B) 3-83 sIg expression in the spleen of 3-83KIH-reconstituted chimeras in the presence (H-2b) or absence (H-2d) of antigen. Spleen cells were stained with anti-Id and anti-IgM mAbs. The percentage of IgM+ Id− mature B cells was calculated on the lymphocyte-gated population. Data are representative of three independent experiments each containing two individual mice per group. (C) Ratio of the frequencies of λ+ and κ+ B cells in the spleens of the indicated mice as determined by flow cytometry. Each point represents data from an individual mouse.
Flow Cytometry and ELISA.
Cells were incubated on ice with anti-CD16/32 (2.4G2) culture supernatant for 15 min, then stained for 20 min with the indicated antibodies in PBS supplemented with 1% BSA and 0.02% NaN3. The following antibody reagents were used: anti-IgM-FITC (Zymed), anti-λ-FITC (PharMingen), anti-Igκ-phycoerythrin (PE) (PharMingen), anti-B220-PE (RA36B2) (PharMingen), biotin-anti-3-83 idiotype (Id) (54.1) (23), or biotin-anti-IgDa (PharMingen). After two washes, cells were incubated with streptavidin-tri-Color (Caltag, South San Francisco, CA) for 20 min on ice to visualize bound biotinylated antibodies, then washed twice and fixed in a solution of PBS/2% paraformaldehyde. Flow cytometric analysis was done with a FACScan (Becton Dickinson) and the data were analyzed by using the Cell Quest program.
Analysis of 3-83 production was performed as described previously (18).
PCR Assay for κ Locus Rearrangement.
Genomic DNAs were prepared and the status rearrangements of the κ loci were analyzed as described previously (24).
Bone Marrow Chimeric Mice.
B10.D2 and C57BL/6 mice were irradiated with 950 rad and injected 6 h later with 5 × 106 bone marrow cells of indicated origin. Chimeric mice were analyzed 3 weeks later by flow cytometry.
Results
Homozygous 3-83 KI (3-83KIH) Mice Have a Restored 3-83 Id+ Splenic B Cell Compartment.
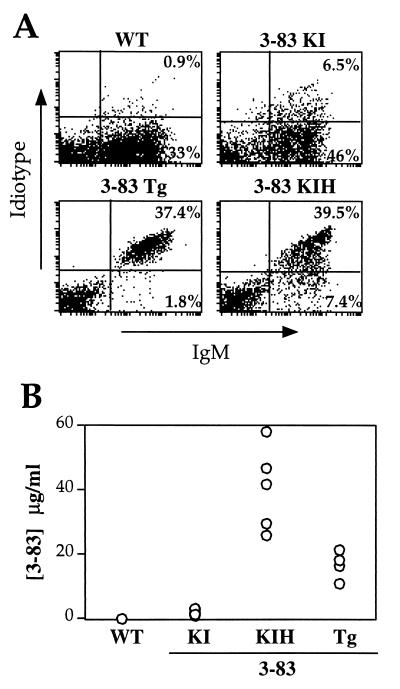
Splenic B cells of conventional Tg (3-83Tg) mice were compared with those of 3-83KI and 3-83KI homozygous (3-83KIH) mice by flow cytometry (Fig. 1A). 3-83 Id+ cells were rare in the 3-83KI mice; instead, 3-83KI splenic B cells were almost entirely IgM+/Id− (Fig. 1A). In contrast, 3-83KIH B cells largely retained high Id expression in proportion to total IgM levels (Fig. 1A), similar to control, 3-83Tg mice. Compared with 3-83KI mice, spleen and bone marrow B cells of 3-83KIH mice expressed a far higher density of 3-83 Id than expected on the basis of their 2-fold increase in gene dose (Fig. 1A; Fig. 2A, second column). Flow cytometry analysis of 3-83KI splenic B cells indicated that >85% retained expression of the IgMa allele linked to the H-chain gene knock-in, but only ≈10% retained 3-83 Id expression (not shown). Correlating with their increased frequency of 3-83 Id+ cells, 3-83KIH mice had significant concentrations (20–60 μg/ml) of serum IgM-3-83 Id, whereas Id levels were low in 3-83KI mice (Fig. 1B).
Figure 1.
Comparison of 3-83 antibody expression on splenic B cells and in sera of conventional (3-83Tg), targeted (3-83KI), and homozygous targeted (3-83KIH) Tg mice. Wild-type (WT) mice were nontransgenic littermates. (A) Surface Id and IgM expression on splenic B cells detected by two-color flow cytometry. (B) 3-83 Id serum levels, as detected by ELISA. 3-83KIH, but not 3-83KI, mice had abundant 3-83 Id IgM in serum. Each symbol represents an individual mouse. Data are representative of at least five mice in each group.
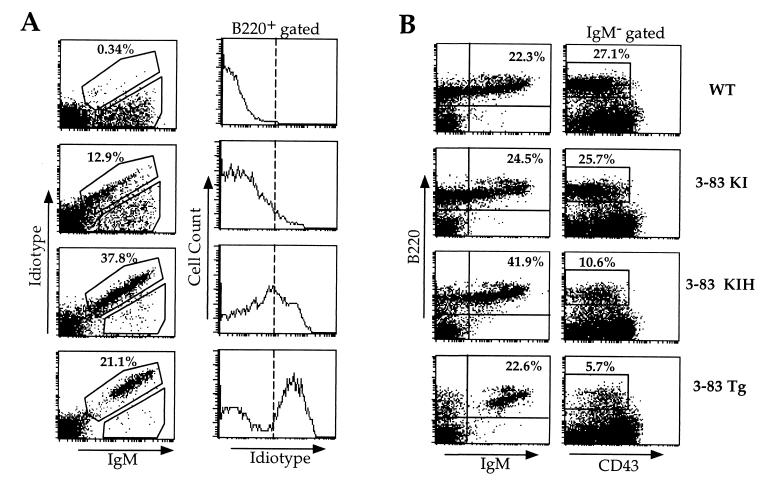
Figure 2.
Restored 3-83 Id expression and B cell maturation in bone marrows of 3-83KIH mice compared with 3-83KI mice. (A) (Left) 3-83 Id/IgM double staining of bone marrow cells. (Right) Id levels on B220-gated bone marrow cells. (B) Enhanced development of sIgM+ cells and reduced pre-B population in 3-83KIH, compared with 3-83KI, bone marrows. (Left) B220/IgM double immunofluorescence. (Right) Pre-B cells enumerated as B220+/IgM−/CD43low cells.
Importantly, the frequency of Id+ B cells was improved when either the H- or the L-chain KI gene was homozygous and the other gene was present in only one copy (Table 3), suggesting that the retention of the transgenic receptor in development was regulated by BCR expression level rather than the number of L-chain loci that needed to be inactivated by editing (see below).
Table 3.
Absolute cell numbers in the spleen of the various 3-83 KI mice
| Mouse type | No. of cells × 10−6
|
||
|---|---|---|---|
| Cells in lymphocyte gate | B220+Id+ | B220+Id− | |
| Wild type | 65 ± 18 | 0.26 ± 0.15 | 30 ± 5 |
| KI/H-2d | 53 ± 15 | 2.4 ± 1.8 | 25 ± 5 |
| KI/H-2b | 47 ± 12 | 0.3 ± 0.2 | 17.2 ± 5 |
| KIH/H-2d | 57 ± 19 | 16 ± 4.6 | 3.8 ± 1 |
| KIH/H-2b | 43 ± 7 | 0.4 ± 0.14 | 22 ± 6 |
| KI (H-locus H)* | 46 ± 3 | 4.2 ± 2 | 17 ± 6 |
| KI (L-locus H)† | 48 ± 7 | 3.6 ± 1.5 | 16 ± 1 |
H-2d mice homozygous for 3-83 H-chain KI gene and hemizygous for the 3-83 κ KI.
H-2d mice homozygous for the 3-83 L-chain KI gene and hemizygous for the 3-83H KI.
3-83KI B Cells Rarely Retain Expression of the Targeted L Chain.
To understand the molecular basis for the loss of transgene-encoded receptor expression in 3-83KI B lymphocytes, we generated hybridomas from 3-83KI spleen cells of either H-2b (antigen-carrying) or H-2d (putative antigen-free) backgrounds and analyzed their L-chain and H-chain proteins. To simplify the analysis, we used cells from mice in which one κ allele was 3-83KI and the other κ locus was germ-line deleted. A random screening of the culture supernatants of 24 such hybridomas with anti-IgM allotype-specific ELISA revealed that all 24 retained expression of the KI-derived IgMa H-chain allele (the nontargeted allele in all cells studied was IgMb). This result supported the IgM allotype analysis of spleen cells and the conclusion of Pelanda et al. (21), that targeted H-chain gene is stably expressed in the vast majority of B cells of 3-83KI mice. For this reason, further analyses to determine the loss of 3-83 Id expression in 3-83KI mice focused on changes in the L-chain loci. Only 4 of 93 H-2d 3-83KI hybridomas and none of 136 H-2b 3-83KI hybridomas secreted antibodies carrying the 3-83 Id (Table 1). An extraordinary percentage of 3-83KI-derived hybridomas expressed λ L chains: 45% or 92%, from H-2d and H-2b mice, respectively (Table 1). Normally, only 6% of mouse B cells express λ. Among hybridomas expressing 3-83 κ chain, none coexpressed λ chain.
Table 1.
Immunoglobulin expression in hybridomas from 3-83KI spleen fusion
| Ig expressed | No. positive hybrids/total tested | |
|---|---|---|
| H-2d | H-2b/d | |
| IgMa | ND | 24/24 |
| 3-83 Id+ | 4/93 | 0/136 |
| κ | 51/93 | 11/136 |
| λ | 42/93 | 125/136 |
L-chain usage and 3-83 secretion were assessed by ELISA. 3-83 idiotype was assessed with mAb 54.1. ND, not determined.
Further evidence for alteration in L-chain gene expression in 3-83KI hybridomas was provided by PCR analysis of κ-locus rearrangement status. Of 49 randomly chosen κ-secreting hybridomas, only 3 (6%) retained the 3-83KI κ gene in an active configuration (Table 2), and these were from hybrids that secreted 3-83 Id. Most κ-secreting hybrids (76%) had detectable new VκJκ rearrangements that displaced or deleted the 3-83KI κ gene (37/49), and an additional 18% of hybrids (6/38) lacked detectable rearrangements, probably because they had deleted the 3-83 κ gene and their active VκJκ genes failed to be amplified with the Vκ primer used. λ-secreting hybridomas were analyzed by PCRs designed to detect recombining sequence (RS) rearrangements (25, 26), which demonstrated that ≈2/3 of the λ-producing hybridomas had deleted their targeted κ allele by V(D)J-mediated RS recombination (Table 2). Taken together, these data, along with earlier studies (21), suggest that the 3-83KI B cells did not efficiently suppress new L-chain gene rearrangements or expression, leading to extensive receptor editing in either the presence or absence of nominal antigen, but in a small subset of H-2d 3-83KI cells the transgene-encoded receptor alone was expressed.
Table 2.
κ-locus rearrangements in 3-83KI spleen hybridomas
| κ-secreting hybridomas
|
γ-secreting hybridomas
|
||||
|---|---|---|---|---|---|
| κ-locus VJ rearrangement | No. positive hybrids/total tested
|
κ-locus RS rearrangement | No. positive hybrids/total tested
|
||
| H-2d | H-2b/d | H-2d | H-2b/d | ||
| 3-83 κ* | 3/38 | 0/11 | Vκ-RS† | 21/42 | 57/125 |
| 3-83 κinv/VJnew‡ | 20/38 | 6/11 | VκJκ-intron-RS† | 7/42 | 20/125 |
| 3-83 κdel/VJnew§ | 9/38 | 2/11 | None detected¶ | 14/42 | 48/125 |
| 3-83 κdel/¶ | 6/38 | 3/11 | |||
Rearrangements of the κ locus were determined by PCR (24). Number of positive hybrids as fraction of total tested is indicated for each group.
Retained 3-83VJκ in active configuration.
See figure 1 of ref. 24 for detailed definition.
Retained 3-83VJκ in inactive, inverted orientation and secondary VκJκ rearrangement was detected.
Secondary VκJκ rearrangement was detected and targeted 3-83VκJκ gene was deleted.
No rearrangement detected.
To determine at which stage of maturation Id+ 3-83KI B cells were blocked, we analyzed by flow cytometry the bone marrow compartments of H-2d 3-83KI mice (Fig. 2A). An IgMlow Idlow population that expressed low levels of B220 was clearly present (Fig. 2A) indicating that at some later point in development Id+ cells were either eliminated or underwent receptor editing. This pattern of receptor expression is most compatible with receptor editing, rather than a cellular selection mechanism. In contrast, 3-83KIH bone marrow B cells expressed high levels of 3-83 Id and largely lacked IgM+/Id− cells. Furthermore, analysis of the frequencies of B cells expressing Ig-κ or Ig-λ indicated that in an H-2d environment 3-83KIH splenic B cells only rarely expressed λ chains, unlike 3-83KI cells (Fig. 3C).
Lack of Evidence for Tolerance-Induced Receptor Editing in H-2d 3-83KI Mice.
Doubling the dosage of 3-83KI genes in H-2d 3-83KIH mice improved the development of 3-83 Id+ cells and suppressed the appearance of Id− cells, compared with 3-83KI mice (Figs. 1 and 2), arguing against the proposition that 3-83-reactive antigens were linked to the 3-83KI loci (21). Other experiments to test for the possible existence of a 3-83-reactive autoantigen on the 3-83KI background, including breeding the 3-83 conventional Tg onto either the targeted L- or H-chain of 3-83KI on the B10.D2 genetic background, failed to manifest any evidence for a tolerogen (data not shown). Furthermore, in 3-83 antibody-binding studies, we were unable to detect 3-83-reactive ligand on the H or L 3-83KI cells (data not shown). In addition, 3-83KIH mice had significant levels of 3-83 Ig in the serum similar to 3-83Tg mice, which should not occur if tolerance was induced, whereas 3-83KI mice lacked serum 3-83 Id (Fig. 1B). Finally, 3-83+ B cells isolated from KIH mice on H-2d background were shown to be functionally competent, as they could be activated upon immunization (data not shown), excluding the possibility that they developed an autoantigen-induced anergic phenotype. Taken together, these data indicate it is unlikely that a self-antigen was responsible for the lack of 3-83 Id+ B cells in the 3-83KI mice.
3-83KIH B Cells Undergo Efficient Antigen-Induced Receptor Editing.
We and others have proposed that, because of the organization of the Ig-L genes, tolerance-induced editing should efficiently rescue and alter the specificity of autoreactive B cells (7, 27), but conventional Tg mice have an inappropriate gene organization to test this idea. Furthermore, no controlled experiment in a targeted Tg system has heretofore been possible, in part because the reactivity of the antibodies whose genes have been tested so far has been to DNA, not to facultative autoantigens (9). The 3-83KIH mice described here provided an opportunity to test the efficacy of receptor editing because B cells generated in the 3-83KIH mice retained Id expression in the absence of antigen (the H-2d background) and could be compared with B cells from 3-83KIH mice expressing antigen (H-2b). Furthermore, because of the potential difficulty in removing two targeted 3-83 κ alleles, this represented a stringent test of the efficiency of receptor editing. To address this issue, we generated chimeras by transferring H-2d 3-83KIH bone marrow cells into lethally irradiated H-2d or H-2b recipients, and analyzed their B cell development by flow cytometry (Fig. 3). In H-2d recipients, Id+ B cells of 3-83KIH mice matured rapidly, resulting in a diminished pre-B cell compartment in the bone marrow (Fig. 3A, Lower Left), and an abundance of Id+ cells in the spleen (Fig. 3B, Left). By contrast, in H-2b recipients, 3-83KIH bone marrow B cells showed a complete down-regulation of the 3-83 receptor and a large population of IgM+ Id− B cells was detected (Fig. 3B, Right). Most significantly, large numbers of Id− B cells appeared in spleens of 3-83KIH-into-H-2b chimeras (Table 3; Fig. 3B, Right), a result that we interpret to indicate that 3-83KIH B cells were able to undergo efficient receptor editing upon encounter with the H-2Kb self-antigen, but in its absence suppressed gene rearrangements. This interpretation was further supported by the reduced κ/λ ratio but similar B cell numbers in 3-83KIH-into-H-2b chimeras compared with 3-83KIH-into-H-2d chimeras (Fig. 3C and Table 3, respectively). These data clearly show that receptor editing is a very efficient process even in the presence of two self-reactive κ alleles. This finding further implies that the robust Id expression in KIH mice on the H-2d background is not the result of poor receptor editing to a putative cryptic autoantigen.
Discussion
To understand the role of sIg in B cell development, we performed an extensive analysis of 3-83KI mice, in which receptor editing of 3-83 Id+ immature cells allowed the production of IgM+ Id− B cells. We show that the generation of B cells with these altered specificities occurs in the bone marrow, but is stimulated by inefficient sIg-mediated maturation, rather than a self-tolerance mechanism. Mice homozygous for 3-83KI chains expressed an increased level of the 3-83 BCR that promoted feedback suppression of new Ig gene rearrangements, hence preventing receptor editing and resulting in a normal development of 3-83+ B cells. Importantly, in the presence of reactive autoantigen KIH B cells underwent extensive and efficient receptor editing. Thus, receptor editing can play an important salvage role in removing and replacing both underexpressed and autoreactive receptors. Because interaction with antigens also causes a reduction in sIg levels, it raises the intriguing possibly that receptor editing in immature B cells is initiated by any process that leads to low BCR levels at the cell surface, including BCR underexpression or receptor down-modulation secondary to sIg engagement. Also consistent with this idea is the observation that in 3-83KI mice bred to signaling mutants that result in increased sIg levels, receptor editing is blocked (V.K., unpublished results).
Analysis of B cell tumor lines and B cells of Ig-Tg mice has indicated that multiple rearrangements can occur on individual Ig-κ loci and that these might be induced spontaneously (13, 28, 29), because initial VJ joins were out of frame (30), because of an inability to pair with H chain (31), or because of immune tolerance (9, 10, 21, 32, 33). In addition, the inability of certain Ig-κ transgenes to mediate feedback suppression of Ig gene rearrangements has sometimes been ascribed to insufficient gene expression (34, 35). However, because Ig-H- and Ig-L-chain genes rearrange and assemble randomly, it has not been possible to prove that secondary L-chain gene rearrangements are induced by inappropriate levels of Ig expression, rather than immune tolerance. Indeed, even high sIg expression is insufficient to inhibit V(D)J recombinase expression in certain models (13, 29); however in these models the possible influence of immune tolerance-induced receptor editing or tumor-related signaling defects was not ruled out. The unique difference between 3-83KI and 3-83KIH immature B cells studied here is in their levels of BCR expression, which are, respectively, insufficient or sufficient for complete maturation of 3-83 Id+ B cells. This observation strongly suggests that not only is a functional antigen receptor required but also a defined level of receptor expression is necessary to promote B cell maturation and recombinase down-regulation.
It is not yet clear whether the mere assembly of a BCR is enough to induce maturation and recombinase down-regulation or whether external ligand triggering of the receptor is required. There is increasing evidence that the CD5+ B cell subset may be positively selected in some way by self-ligands (36–38), but there is no direct evidence for this in other B cell systems (11). Our results suggest that, should such a ligand-induced positive selection signal exist, it must induce a much weaker signal than that induced by the reactive autoantigen H-2Kb, which is known to have low affinity for the 3-83 BCR (18). The simplest model to explain the data is that during development the unligated BCR produces a basal positive signal that informs the B cell to stop rearrangement and to mature further. That this signal is density-dependent is consistent with the concept that random collisions between sIg complexes and their associated signal transducers allow enzymatic trans-modifications, such as phosphorylations, whose signals are further propagated to, and perceived by, the nucleus. In one study in which a truncated sIg lacking variable regions was expressed as a transgene, splenic B cells developed, suggesting that specific antigen is not required (39).
It is very surprising that, individually, each 3-83KI chain is able to pair with other H or L chains to give rise to mature B cells, but in combination with each other at the same expression levels, they fail to sustain B cell development (21). This observation may indicate that not all H- and L-chain associations are comparable, and that some combinations may be fitter in chain pairing, rate of assembly, or stability. Experimental measurements of H/L association are consistent with this possibility (40). On the other hand, it is unclear whether such selectivity is a general phenomenon because the 3-83KI genes may be underexpressed relative to their normal and conventional transgenic counterparts owing to the truncation of their 5′ sequences in the targeting constructs, such that two gene copies are now required to compensate for this artificially lowered expression. In any event, L chains must be tested at the immature B cell stage by pairing with the H chain already present. According to the data presented here, this checkpoint requires a minimal threshold of basal signaling or expression and occurs at a developmental stage at which self-reactivity is also monitored.
Acknowledgments
This paper is dedicated to the memory of Dr. Eugenia Spanopolou. We thank Drs. P. Marrack and J. C. Cambier for their support. We also thank Norman Klinman, Amanda Gavin, and Ann Feeny for critically reading the manuscript and Denis Huszar for providing κ-deficient mice. This study was supported by Grants RO1AI33608 and PO1AI22295 from the National Institutes of Health.
Abbreviations
- sIg
surface Ig
- BCR
B cell receptor
- Tg
transgenic
- KI
knock-in
- KIH
homozygous KI
- Id
idiotype
- RS
recombining sequence
Footnotes
Article published online before print: Proc. Natl. Acad. Sci. USA, 10.1073/pnas.130182597.
Article and publication date are at www.pnas.org/cgi/doi/10.1073/pnas.130182597
References
- 1.Klinman N R. Immunity. 1996;5:189–195. doi: 10.1016/s1074-7613(00)80314-3. [DOI] [PubMed] [Google Scholar]
- 2.Radic M Z, Zouali M. Immunity. 1996;5:505–511. doi: 10.1016/s1074-7613(00)80266-6. [DOI] [PubMed] [Google Scholar]
- 3.Nemazee D, Buerki K. Proc Natl Acad Sci USA. 1989;86:8039–8043. doi: 10.1073/pnas.86.20.8039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chen C, Nagy Z, Radic M Z, Hardy R R, Huszar D, Camper S A, Weigert M. Nature (London) 1995;373:252–255. doi: 10.1038/373252a0. [DOI] [PubMed] [Google Scholar]
- 5.Spanopoulou E, Roman C A, Corcoran L M, Schlissel M S, Silver D P, Nemazee D, Nussenzweig M C, Shinton S A, Hardy R R, Baltimore D. Genes Dev. 1994;8:1030–1042. doi: 10.1101/gad.8.9.1030. [DOI] [PubMed] [Google Scholar]
- 6.Xu H, Li H, Suri-Payer E, Hardy R R, Weigert M. J Exp Med. 1998;188:1247–1254. doi: 10.1084/jem.188.7.1247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Tiegs S L, Russell D M, Nemazee D. J Exp Med. 1993;177:1009–1020. doi: 10.1084/jem.177.4.1009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Melamed D, Benschop R J, Cambier J C, Nemazee D. Cell. 1998;92:173–182. doi: 10.1016/s0092-8674(00)80912-5. [DOI] [PubMed] [Google Scholar]
- 9.Chen C, Prak E L, Weigert M. Immunity. 1997;6:97–105. doi: 10.1016/s1074-7613(00)80673-1. [DOI] [PubMed] [Google Scholar]
- 10.Prak E L, Trounstine M, Huszar D, Weigert M. J Exp Med. 1994;180:1805–1815. doi: 10.1084/jem.180.5.1805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Pillai S. Immunity. 1999;10:493–502. doi: 10.1016/s1074-7613(00)80049-7. [DOI] [PubMed] [Google Scholar]
- 12.Li Y S, Hayakawa K, Hardy R R. J Exp Med. 1993;178:951–960. doi: 10.1084/jem.178.3.951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ma A, Fisher P, Dildrop R, Oltz E, Rathbun G, Achacoso P, Stall A, Alt F W. EMBO J. 1992;11:2727–2734. doi: 10.1002/j.1460-2075.1992.tb05338.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Melamed D, Kench J A, Grabstein K, Rolink A, Nemazee D. J Immunol. 1997;159:1233–1239. [PubMed] [Google Scholar]
- 15.Ozato K, Mayer N, Sachs D H. J Immunol. 1980;124:533–540. [PubMed] [Google Scholar]
- 16.Russell D M, Dembic Z, Morahan G, Miller J F, Bürki K, Nemazee D. Nature (London) 1991;354:308–311. doi: 10.1038/354308a0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hertz M, Nemazee D. Immunity. 1997;6:429–436. doi: 10.1016/s1074-7613(00)80286-1. [DOI] [PubMed] [Google Scholar]
- 18.Lang J, Jackson M, Teyton L, Brunmark A, Kane K, Nemazee D. J Exp Med. 1996;184:1685–1697. doi: 10.1084/jem.184.5.1685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Melamed D, Nemazee D. Proc Natl Acad Sci USA. 1997;94:9267–9272. doi: 10.1073/pnas.94.17.9267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pelanda R, Schaal S, Torres R M, Rajewsky K. Immunity. 1996;5:229–239. doi: 10.1016/s1074-7613(00)80318-0. [DOI] [PubMed] [Google Scholar]
- 21.Pelanda R, Schwers S, Sonoda E, Torres R M, Nemazee D, Rajewsky K. Immunity. 1997;7:765–775. doi: 10.1016/s1074-7613(00)80395-7. [DOI] [PubMed] [Google Scholar]
- 22.Chen J, Trounstine M, Kurahara C, Young F, Kuo C C, Xu Y, Loring J F, Alt F W, Huszar D. EMBO J. 1993;12:821–830. doi: 10.1002/j.1460-2075.1993.tb05722.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Nemazee D A, Bürki K. Nature (London) 1989;337:562–566. doi: 10.1038/337562a0. [DOI] [PubMed] [Google Scholar]
- 24.Retter M W, Nemazee D. J Exp Med. 1998;188:1231–1238. doi: 10.1084/jem.188.7.1231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Durdik J, Moore M W, Selsing E. Nature (London) 1984;307:749–752. doi: 10.1038/307749a0. [DOI] [PubMed] [Google Scholar]
- 26.Selsing E, Daitch L E. In: Immunoglobulin Genes. Honjo T, Alt F, editors. Vol. 2. San Diego: Academic; 1995. pp. 194–203. [Google Scholar]
- 27.Gay D, Saunders T, Camper S, Weigert M. J Exp Med. 1993;177:999–1008. doi: 10.1084/jem.177.4.999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Huber C, Klobeck H G, Zachau H G. Eur J Immunol. 1992;22:1561–1565. doi: 10.1002/eji.1830220632. [DOI] [PubMed] [Google Scholar]
- 29.Rolink A, Grawunder U, Haasner D, Strasser A, Melchers F. J Exp Med. 1993;178:1263–1270. doi: 10.1084/jem.178.4.1263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Feddersen R M, Martin D J, Van Ness B G. J Immunol. 1990;145:745–750. [PubMed] [Google Scholar]
- 31.Kwan S P, Max E E, Seidman J G, Leder P, Scharff M D. Cell. 1981;26:57–66. doi: 10.1016/0092-8674(81)90033-7. [DOI] [PubMed] [Google Scholar]
- 32.Prak E L, Weigert M. J Exp Med. 1995;182:541–548. doi: 10.1084/jem.182.2.541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Radic M Z, Erikson J, Litwin S, Weigert M. J Exp Med. 1993;177:1165–1173. doi: 10.1084/jem.177.4.1165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Rusconi S, Kohler G. Nature (London) 1985;314:330–334. doi: 10.1038/314330a0. [DOI] [PubMed] [Google Scholar]
- 35.Storb U. Annu Rev Immunol. 1987;5:151–174. doi: 10.1146/annurev.iy.05.040187.001055. [DOI] [PubMed] [Google Scholar]
- 36.Gu H, Tarlinton D, Muller W, Rajewsky K, Forster I. J Exp Med. 1991;173:1357–1371. doi: 10.1084/jem.173.6.1357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hayakawa K, Asano M, Shinton S A, Gui M, Allman D, Stewart C L, Silver J, Hardy R R. Science. 1999;285:113–116. doi: 10.1126/science.285.5424.113. [DOI] [PubMed] [Google Scholar]
- 38.Ye J, McCray S K, Clarke S H. EMBO J. 1996;15:1524–1533. [PMC free article] [PubMed] [Google Scholar]
- 39.Shaffer A L, Schlissel M S. J Immunol. 1997;159:1265–1275. [PubMed] [Google Scholar]
- 40.Hamel P A, Isenman D E, Klein M H, Luedtke R, Dorrington K J. Mol Immunol. 1984;21:277–283. doi: 10.1016/0161-5890(84)90098-1. [DOI] [PubMed] [Google Scholar]