Abstract
The diagnosis of paediatric solid tumours is often based on small tissue needle biopsies in which many different entities demonstrate a “small round cell tumour” phenotype and in which there may be insufficient tissue to allow the interpretation of diagnostic architectural features, which may be present in larger specimens. Therefore, the extensive use of a panel of immunohistochemical markers is part of the routine handling and investigation of such biopsies to reach a definite diagnosis. However, in some cases the morphological and routine immunohistochemical findings may be insufficient for a precise diagnosis or they may be difficult to interpret in the given clinical context. Although many paediatric tumours exhibit characteristic chromosomal translocations with resultant specific fusion transcripts, these require molecular methods for their detection, usually on fresh tissue samples, which may not always be available. As more immunohistochemical markers become available, more precise diagnosis on such small biopsies may be possible. This review examines the use of the immunohistochemical markers, MyoD1 and myogenin, in the diagnosis of paediatric rhabdomyosarcoma, including its subtypes.
Keywords: alveolar, embryonal, MyoD1, myogenin, rhabdomyosarcoma
Rhabdomyosarcoma (RMS) is the most common paediatric solid tumour.1 The diagnosis is almost always on the basis of a small tissue biopsy supplemented by immunohistochemical confirmation for the definitive diagnosis. Although many historical classification schemes have been reported,2 the most widely used is the modified World Health Organisation classification,3 which describes two main morphological subtypes, embryonal RMS (ERMS) and alveolar RMS (ARMS). The third major type, pleomorphic or anaplastic rhabdomyosarcoma, is essentially a tumour that affects adults; in children, ERMS cases may demonstrate pronounced cellular pleomorphism but their classification remains ERMS. The diagnosis of RMS and its subtypes is important because ARMS is reported to have a worse prognosis, with a greater frequency of disseminated metastases.1 Overall, multimodal treatment has increased survival in RMS from 25% in 1970 to more than 70% in recent studies, with particular improvements in those patients with residual tumour after the initial treatment.4
“The diagnosis of rhabdomyosarcoma and its subtypes is important because alveolar rhabdomyosarcoma is reported to have a worse prognosis, with a greater frequency of disseminated metastases”
The diagnosis of most paediatric solid tumours requires an extensive panel of immunohistochemical markers because many entities exhibit a non-characteristic “small round cell tumour” phenotype. The main differential diagnoses in this age group are lymphoma, neuroblastoma, primitive neuroectodermal tumour, and RMS, most of which can be readily distinguished on the basis of their simple immunohistochemical profile. In some cases, however, the material submitted and/or the immunohistochemical profile may not be diagnostic, and further investigations are required. This review examines the use of the immunohistochemical markers MyoD1 and myogenin in the diagnosis of paediatric RMS.
SUBTYPES OF PAEDIATRIC RHABDOMYOSARCOMA
RMS may be classified as embryonal, including the pathologically distinct botryoid subtype, spindle cell, and alveolar, including the solid variant.1 Most adult cases are of the pleomorphic type, occurring in skeletal muscles, whereas in the paediatric age group, RMS mainly occurs in the head and neck region (especially perorbital), the urogenital tract, the biliary tract, and the trunk or limb. The classification of RMS was initially based purely on the morphological and cytological appearances of the tumour. ERMS refers to a tumour composed of primitive mesenchymal cells with varying stages of morphological skeletal muscle differentiation (strap cells, striated cytoplasm, etc) in a loose, myxoid, or cellular collagenous stroma. There may be nuclear pleomorphism but nucleoli are inconspicuous. The botryoid subtype simply refers to an ERMS in a subepithelial location, which exhibits a layer of densely cellular neoplastic cells just beneath the epithelium (cambium layer). ARMS classically demonstrates an architectural pattern with thin fibrous septae lined by tumour cells, some of which are centrally dissociated or discohesive. However, it has become apparent that this architectural pattern may not always be present (solid variant ARMS) and the morphological diagnosis of ARMS therefore relies on the cytological characteristics of round tumour cells with hyperchromatic nuclei, coarse chromatin, and prominent nucleoli. Cytological features of skeletal muscle differentiation may be absent, particularly in small biopsy specimens.
The distinction of ARMS from ERMS is important because ARMS carries a worse prognosis and requires a modified therapeutic regimen.1 Definitive diagnosis of ARMS may require molecular and/or cytogenetic investigation because most cases exhibit characteristic translocations enabling subtype diagnosis even on the basis of small tissue samples. Two characteristic gene fusion products have been described in ARMS: the fusion of PAX3 to FKHR5–7 and PAX7 to FKHR,8 corresponding to the t(2;13)9–19 and t(1;13) translocations, respectively.10,20,21
MOLECULAR DIAGNOSIS OF ALVEOLAR RHABDOMYOSARCOMA
Because most cases of ARMS express the PAX3–FKHR or PAX7–FKHR gene fusions, resulting from the t(2;13) or t(1;13) translocations, respectively, molecular methods have been increasingly used in their diagnosis, both on fresh and paraffin wax embedded tissue. In a study of 171 childhood cases of RMS, including 78 cases of ARMS, the reverse transcriptase polymerase chain reaction (RT-PCR) was able to identify either the PAX3–FKHR or the PAX7–FKHR fusion transcripts in 55% and 22% of patients with ARMS, respectively. Importantly, no case of ERMS expressed either transcript.22 In a further study of 91 cases of primary RMS, the PAX3–FKHR or PAX7–FKHR translocations were present in more than 80% of ARMS cases, with PAX3–FKHR expression appearing to be an adverse prognostic factor.23 Several other studies have confirmed the high specificity for the diagnosis of ARMS because no cases of ERMS exhibit these fusion products.24–26 Furthermore, molecular and morphological review of RMS cases suggests that, in some cases, the detection of translocations may clarify the histopathological diagnosis in cases where morphological features are equivocal.27 Although such cytogenetic abnormalities are often detected, in some ARMS cases the PAX3–FKHR or PAX7–FKHR fusion gene products can be detected by molecular methods, but the translocation cannot be identified cytogenetically,28,29 and rarely other variant translocations may be present.30 Nevertheless, in some ARMS cases these gene fusion products cannot be detected, for example congenital ARMS.31
“Two characteristic gene fusion products have been described in alveolar rhabdomyosarcomas: the fusion of PAX3 to FKHR and PAX7 to FKHR, corresponding to the t(2;13) and t(1;13) translocations, respectively”
Patients with ERMS do have characteristic translocations, hyperdiploid or hypertetraploid karyotypes have been reported,32 and many ERMS cases show breakpoints at the 1p11–q11 region or loss of heterozygosity at 11p15.28 Although fusion products are not present, ERMS cells have been reported to express increased amounts of wild-type PAX3 or PAX7 compared with non-RMS myoblasts.33
IMPLICATIONS FOR PROGNOSIS
In patients with localised ARMS, the presence or absence of the PAX3–FKHR or PAX7–FKHR fusion products does not appear to be associated with differences in outcome. However, in patients with metastatic ARMS, survival is significantly worse for those expressing the PAX3–FKHR translocation.22,23
MOLECULAR MECHANISM OF ARMS FUSION PRODUCT FUNCTION
The t(2;13)(q35;q14) and t(1;13)(p36;q14) translocations rearrange PAX3 and PAX7, which are members of the paired box transcription factor family. These are juxtaposed with FKHR, a member of the fork head transcription factor family. The fusion genes thus produced encode chimaeric proteins containing the PAX3/PAX7 DNA binding and the FKHR transcriptional activation domains, allowing transcriptional activation with a higher potency than the wild-type PAX proteins.34,35 In addition to their altered functional behaviour, such fusion products are also overexpressed in ARMS because of the amplification of PAX7–FKHR and an increase in the transcription rate of PAX3–FKHR.36,37 This aberrant gene expression presumably contributes to the malignant behaviour of ARMS by affecting cellular growth, apoptosis, and differentiation.38,39 The PAX3–FKHR and PAX7–FKHR translocations differ with regard to the presence of reciprocal translocation products and amplification, further suggesting differences between the mechanisms of these translocation events.40
RELATION BETWEEN PAX AND MORPHOLOGY
Although PAX3–FKHR is only found in ARMS, RMS cell lines transfected with the PAX3–FKHR translocation and grown as tumour xenografts in immunodeficient mice show faster growth, more invasion, and have a more pleomorphic appearance, although the characteristic alveolar architecture is not apparent.41 Furthermore, the proportion of tumour cells staining with Ki67 or in terminal deoxynucleotidyl transferase mediated dUTP nick end labelling based assays is greater in tumours expressing PAX3–FKHR.42 Clearly, such fusion gene products have important effects on biological behaviour, but may be insufficient in isolation to transform cells to an ARMS phenotype.
MYOGENIC REGULATORY PROTEINS: MYOD1 AND MYOGENIN
The myogenic nuclear regulatory proteins are a group of DNA binding proteins, which act as transcription factors and stimulate myogenesis. Transfection into multipotential mesodermal cells stimulates myogenic differentiation,43–45 and a variety of differentiated cell types can be converted to skeletal muscle after transfection with MyoD1.46 MyoD induces differentiation by activating muscle specific genes and is important in the switch from cellular proliferation to differentiation. Loss of this normal control could theoretically lead to the formation of RMS tumours, which have lost control of cell proliferation.47 Northern blot analyses demonstrate expression of both MyoD1 and myogenin in RMS cell lines, which act as lineage markers and differentiation markers, respectively.48 Fetal myoblasts express both MyoD and myogenin in culture, whereas adult myoblasts are negative.49 That these genes are important for normal skeletal muscle differentiation can be demonstrated by reports that myogenin knockout mice show severe skeletal muscle defects, differing in different regions, despite expressing normal amounts of MyoD, with committed cells unable to form muscle sheets without the presence of myogenin. Therefore, myogenin and MyoD appear to have different roles in myogenesis rather than there simply being differences in expression.50,51
“MyoD induces differentiation by activating muscle specific genes and is important in the switch from cellular proliferation to differentiation”
Traditionally, immunohistochemistry to detect myoid differentiation has been based around expression of the intermediate filament desmin, the contractile protein actin, or the oxygen transport molecule, myoglobin. In one early study, histological examination of 65 RMS samples reported that cross striations were seen on light microscopy in about 25–30% of cases; however, immunohistochemical staining for myoglobin was present in only 30% of ERMS and 70% of ARMS cases. Overall, 64% of ERMS and 80% of ARMS cases showed either positive immunostaining or ultrastructural features of skeletal muscle differentiation.52 These traditional immunomarkers require considerable differentiation along the myogenic pathway before cellular expression occurs. In contrast, the analysis of the expression of the myogenic nuclear regulatory proteins, MyoD1 and myogenin, should allow the identification of primitive tumours that are relatively undifferentiated. However, because they are markers of skeletal muscle differentiation rather than RMS, these molecules may also be expressed in many other tumours demonstrating skeletal muscle differentiation, such as rhabdomyomatous Wilms’s tumour, and will also be expressed in regenerating muscle fibres entrapped within any infiltrating tumour.
Anti-MyoD1 antibody (Novocastra, Newcastle, UK) recognises an epitope near the C-terminus of the MyoD1 protein. MyoD1 immunostaining is positive in almost all RMS cases, and is localised to the nuclei (cytoplasmic staining may occur but is less specific).53 Antimyogenin (Dako, Ely, Cambridgeshire, UK) recognises an epitope in amino acid region 138–158 of the myogenin molecule. Myogenin staining is positive in most cases of RMS (see below) and is localised to the nuclei. Antigen (epitope) retrieval techniques are required for both antibodies and only true nuclear expression should be considered positive.
MYOGENIN AND MYOD IMMUNOHISTOCHEMISTRY IN RHABDOMYOSARCOMA
Several studies have examined the use of immunohistochemical staining with MyoD and myogenin in the diagnosis of RMS. Of 150 formalin fixed, paraffin wax embedded archival samples, including 32 RMS cases and several other tumours, myogenin was expressed in all RMS samples but in no other soft tissue tumours, including nodular fasciitis, malignant fibrous histiocytoma, malignant peripheral nerve sheath tumour, inflammatory myofibroblastic tumour, myofibrosarcoma, leiomyoma, leiomyosarcoma, and alveolar soft part sarcoma. However, focal nuclear myogenin staining was seen in occasional desmoid, infantile myofibromatosis, and infantile fibrosarcoma specimens, but this may have represented entrapped, regenerating non-neoplastic skeletal muscle. Similarly, all RMS samples showed nuclear immunoreactivity for MyoD1 but background cytoplasmic staining with this antibody makes interpretation more difficult.54,55 In another report, 119 paediatric tumours were studied including 48 cases of ARMS, 20 cases of ERMS, and a range of other entities. Nuclear myogenin positivity was present in all RMS samples but none of the other paediatric tumour types.56 Cui et al reported nuclear expression of MyoD1 and myogenin in about 80% of RMS cases, with MyoD1 generally expressed in the small, primitive tumour cells, whereas the larger cells showing morphological evidence of skeletal muscle differentiation were negative.57 Positive nuclear expression of myogenin was stronger than for MyoD1 in cases with differentiated tumour cells, but was less prominent in cases in which small, primitive tumour cells predominated. In that study, no other soft tissue tumours exhibited positive nuclear staining for either MyoD1 or myogenin.57 Overall, these studies suggest that almost all RMS samples show positive nuclear staining with antibodies to MyoD and/or myogenin, with non-RMS paediatric tumours being consistently negative.58
IMMUNOCYTOCHEMICAL DIFFERENTIATION OF EMBRYONAL FROM ALVEOLAR SUBTYPES
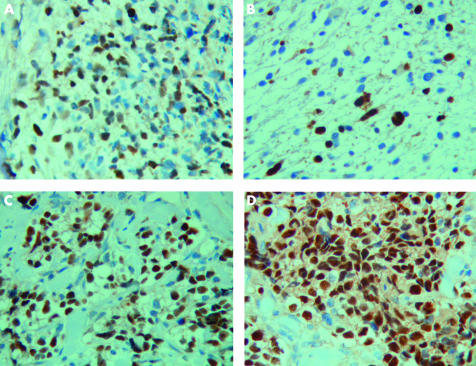
Although essentially all RMS samples show some degree of immunostaining with antibodies to MyoD and myogenin, the expression patterns differ between ERMS and ARMS. In one study examining 68 RMS cases, there was extensive nuclear myogenin staining in most cases of ARMS (expression in > 75% of tumour cells), whereas in ERMS, although all cases showed some tumour cells with positive nuclear expression, immunopositivity was less uniform, and in many cases < 25% of tumour cells were positive.56 Similarly, Cessna and colleagues55 examined 32 ARMS cases and reported that there was strong nuclear staining for myogenin, which was most pronounced in the tumour cells lining fibrous septae, highlighting the alveolar architecture. Again, ERMS showed consistent positivity but with a much more variable and focal staining pattern. In another series, all nine ARMS cases stained strongly positively for myogenin, whereas in the 15 ERMS, staining was weak, patchy, and a larger proportion of tumour cells were negative for myogenin (fig 1).59
Figure 1.
Photomicrographs showing embryonal rhabdomyosarcoma (A,B) and alveolar rhabdomyosarcoma (C,D) immunostained with antibodies to MyoD1 (A,C) and myogenin (B,D). Note that both tumours demonstrate nuclear positivity but staining is much more widespread and intense with myogenin in rhabdomyosarcomas of the alveolar subtype (original magnification, ×400).
Solid variant alveolar rhabdomyosarcoma
In the only small study examining 15 cases of ERMS, classic alveolar ARMS, and solid variant ARMS, sections were immunostained for both myogenin and MyoD1 and semiquantitative scoring of each section was carried out for percentage tumour cells stained and staining intensity. In all cases, the tumour cells stained with both antibodies, but in the ARMS groups staining for myogenin was much stronger and most (> 90%) of the tumour cells stained. However, there was no difference in staining extent or intensity between the classic and solid ARMS variants, suggesting that myogenin immunohistochemistry is useful even in patients with atypical morphological findings on needle biopsy.60
RELATION BETWEEN IMMUNOSTAINING AND MOLECULAR ALTERATIONS
Few studies have directly examined the inter-relation between the presence of the fusion gene products and immunostaining patterns. However, in one small series, in six of seven ARMS cases with strong nuclear immunostaining for myogenin the presence of PAX3–FKHR or PAX7–FKHR was demonstrated by RT-PCR. Furthermore, there was one tumour, which was initially diagnosed as ERMS on morphological grounds, but which stained strongly for myogenin and was retrospectively found to be positive for the PAX3–FKHR transcript. In this study, western blotting for myogenin was also carried out and there was good correlation between the extent of immunohistochemical staining and western blot findings. ARMS cases were found to express three times more myogenin than ERMS. It is therefore possible that ERMS results from an early block in myogenesis, before the expression of myogenin, whereas ARMS originates from cells later in the myogenic pathway.59 Further evidence for the inter-relation is provided by a study in which PAX3–FKHR was introduced into cell lines and gene expression changes analysed by means of cDNA microarrays. Expression of the PAX3–FKHR product stimulated myogenic differentiation, including the induction of MyoD and myogenin expression.61
“It is possible that embryonal rhabdomyosarcoma results from an early block in myogenesis, before the expression of myogenin, whereas alveolar rhabdomyosarcoma originates from cells later in the myogenic pathway”
RT-PCR FOR THE DETECTION OF METASTATIC DISEASE
The detection of minimal residual disease or micrometastases in RMS may be difficult. PCR based detection of MyoD1 may be of value in both ARMS and ERMS, in which characteristic fusion products are absent. MyoD1 mRNA may be detected in tissue specimens using RT-PCR. In one series of 35 cases of RMS, the MyoD1 transcript was detected in almost all RMS cases, whereas no expression was found in non-RMS samples.62 However, in a similar study, the detection of MyoD1 mRNA was not specific for RMS, being amplified in some other childhood tumours.63 The detection rate of metastatic disease is significantly higher with RT-PCR than by morphological means. RT-PCR is positive in all patients with morphological evidence of metastatic disease and also in some in whom metastases were identified by RT-PCR alone. Therefore, such methodology may be particularly useful for the detection of minimal bone marrow involvement in children with RMS, although the clinical relevance of such micrometastases remains uncertain.64
Take home messages.
Rhabdomyosarcoma is the most common malignant soft tissue tumour of childhood
Rhabdomyosarcoma may be categorised into embryonal (including botryoid), alveolar (including solid variant), and spindle cell subtypes according to morphology and immunohistochemical findings
Prognosis and treatment are different for the alveolar subtype, which shows characteristic chromosomal translocations and gene fusion products involving chromosome 13 (2:13 PAX3–FKHR and 1:13 PAX7–FKHR)
On small biopsy specimens, morphology may be difficult to determine and immunohistochemistry plays an important role
Essentially all paediatric rhabdomyosarcomas show nuclear expression of myogenin and MyoD1 (myogenic nuclear regulatory proteins), whereas almost no other paediatric tumours demonstrate positive immunostaining
Rhabdomyosarcomas of the alveolar subtype, alveolar or solid variants, demonstrate widespread and strong myogenin expression compared with those of the embryonal subtype
SUMMARY
The immunohistochemical staining of paediatric rhabdomyosarcomas with antibodies to MyoD and myogenin provides sensitive and specific diagnostic information. Almost all cases demonstrate nuclear expression of both products, but myogenin immunostaining is usually more clinically useful because it is more consistent and is associated with less non-specific staining. Furthermore, widespread and intense immunostaining for myogenin in RMS is significantly associated with tumours of the alveolar subtype, both alveolar and solid variants. Further studies are required to investigate the precise relation between the immunohistochermical expression of myogenin, the presence of PAX3–FKHR or PAX7–FKHR gene fusion products, and prognosis.
Abbreviations
ARMS, alveolar rhabdomyosarcoma
ERMS, embryonal rhabdomyosarcoma
RMS, rhabdomyosarcoma
RT-PCR, reverse transcriptase polymerase chain reaction
REFERENCES
- 1.O’Shea PA. Myogenic tumors of soft tissue. In: Coffin CM, Dehner LP, ed. Pediatric soft tissue tumors. Baltimore: Williams & Wilkins, 1997:214–53.
- 2.Parham DM. Pathologic classification of rhabdomyosarcomas and correlations with molecular studies. Mod Pathol 2001;14:506–14. [DOI] [PubMed] [Google Scholar]
- 3.Weiss SW, Goldblum JR. General considerations. In: Weiss SW, Goldblum JR, eds. Enzinger and Weiss’s soft tissue tumors. St Louis: Mosby, 2001:1–19.
- 4.Pappo AS, Shapiro DN, Crist WM, et al. Biology and therapy of pediatric rhabdomyosarcoma. J Clin Oncol 1995;13:2123–39. [DOI] [PubMed] [Google Scholar]
- 5.Galili N, Davis RJ, Fredericks WJ, et al. Fusion of a fork head domain gene to PAX3 in the solid tumour alveolar rhabdomyosarcoma. Nat Genet 1993;5:230–5. [DOI] [PubMed] [Google Scholar]
- 6.Shapiro DN, Sublett JE, Li B, et al. Fusion of PAX3 to a member of the forkhead family of transcription factors in human alveolar rhabdomyosarcoma. Cancer Res 1993;53:5108–12. [PubMed] [Google Scholar]
- 7.Barr FG, Galili N, Holick J, et al. Rearrangement of the PAX3 paired box gene in the paediatric solid tumour alveolar rhabdomyosarcoma. Nat Genet 1993;3:113–17. [DOI] [PubMed] [Google Scholar]
- 8.Davis RJ, D’Cruz CM, Lovell MA, et al. Fusion of PAX7 to FKHR by the variant t(1;13)(p36;q14) translocation in alveolar rhabdomyosarcoma. Cancer Res 1994;54:2869–72. [PubMed] [Google Scholar]
- 9.Douglass EC, Shapiro DN, Valentine M, et al. Alveolar rhabdomyosarcoma with the t(2;13): cytogenetic findings and clinicopathologic correlations. Med Pediatr Oncol 1993;21:83–7. [DOI] [PubMed] [Google Scholar]
- 10.Whang-Peng J, Knutsen T, Theil K, et al. Cytogenetic studies in subgroups of rhabdomyosarcoma. Genes Chromosomes Cancer 1992;5:299–310. [DOI] [PubMed] [Google Scholar]
- 11.Barr FG, Holick J, Nycum L, et al. Localization of the t(2;13) breakpoint of alveolar rhabdomyosarcoma on a physical map of chromosome 2. Genomics 1992;13:1150–6. [DOI] [PubMed] [Google Scholar]
- 12.Shapiro DN, Valentine MB, Sublett JE, et al. Chromosomal sublocalization of the 2;13 translocation breakpoint in alveolar rhabdomyosarcoma. Genes Chromosomes Cancer 1992;4:241–9. [DOI] [PubMed] [Google Scholar]
- 13.Dal Cin P, Brock P, Aly MS, et al. A variant (2;13) translocation in rhabdomyosarcoma. Cancer Genet Cytogenet 1991;55:191–5. [DOI] [PubMed] [Google Scholar]
- 14.Mitchell CD, Ventris JA, Warr TJ, et al. Molecular definition in a somatic cell hybrid of a specific 2:13 translocation breakpoint in childhood rhabdomyosarcoma. Oncogene 1991;6:89–92. [PubMed] [Google Scholar]
- 15.Nojima T, Abe S, Yamaguchi H, et al. A case of alveolar rhabdomyosarcoma with a chromosomal translocation, t(2;13)(q37;q14). Virchows Arch A Pathol Anat Histopathol 1990;417:357–9. [DOI] [PubMed] [Google Scholar]
- 16.Rowe D, Gerrard M, Gibbons B, et al. Two further cases of t(2;13) in alveolar rhabdomyosarcoma indicating a review of the published chromosome breakpoints. Br J Cancer 1987;56:379–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lizard-Nacol S, Mugneret F, Volk C, et al. Translocation (2;13)(q37;q14) in alveolar rhabdomyosarcoma: a new case. Cancer Genet Cytogenet 1987;25:373–4. [DOI] [PubMed] [Google Scholar]
- 18.Lai JL, Savary JB, Deminatti M, et al. Translocation (2;13)(q37;q14) in rhabdomyosarcoma: a new case. Cancer Genet Cytogenet 1987;25:371–2. [DOI] [PubMed] [Google Scholar]
- 19.Douglass EC, Valentine M, Etcubanas E, et al. A specific chromosomal abnormality in rhabdomyosarcoma. Cytogenet Cell Genet 1987;45:148–55. [DOI] [PubMed] [Google Scholar]
- 20.Douglass EC, Rowe ST, Valentine M, et al. Variant translocations of chromosome 13 in alveolar rhabdomyosarcoma. Genes Chromosomes Cancer 1991;3:480–2. [DOI] [PubMed] [Google Scholar]
- 21.Biegel JA, Meek RS, Parmiter AH, et al. Chromosomal translocation t(1;13)(p36;q14) in a case of rhabdomyosarcoma. Genes Chromosomes Cancer 1991;3:483–4. [DOI] [PubMed] [Google Scholar]
- 22.Sorensen PH, Lynch JC, Qualman SJ, et al. PAX3–FKHR and PAX7–FKHR gene fusions are prognostic indicators in alveolar rhabdomyosarcoma: a report from the children’s oncology group. J Clin Oncol 2002;20:2672–9. [DOI] [PubMed] [Google Scholar]
- 23.Anderson J, Gordon T, McManus A, et al. Detection of the PAX3–FKHR fusion gene in paediatric rhabdomyosarcoma: a reproducible predictor of outcome? Br J Cancer 2001;85:831–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Edwards RH, Chatten J, Xiong QB, et al. Detection of gene fusions in rhabdomyosarcoma by reverse transcriptase-polymerase chain reaction assay of archival samples. Diagn Mol Pathol 1997;6:91–7. [DOI] [PubMed] [Google Scholar]
- 25.Reichmuth C, Markus MA, Hillemanns M, et al. The diagnostic potential of the chromosome translocation t(2;13) in rhabdomyosarcoma: a PCR study of fresh-frozen and paraffin-embedded tumour samples. J Pathol 1996;180:50–7. [DOI] [PubMed] [Google Scholar]
- 26.Arden KC, Anderson MJ, Finckenstein FG, et al. Detection of the t(2;13) chromosomal translocation in alveolar rhabdomyosarcoma using the reverse transcriptase-polymerase chain reaction. Genes Chromosomes Cancer 1996;16:254–60. [DOI] [PubMed] [Google Scholar]
- 27.Anderson J, Renshaw J, McManus A, et al. Amplification of the t(2;13) and t(1;13) translocations of alveolar rhabdomyosarcoma in small formalin-fixed biopsies using a modified reverse transcriptase polymerase chain reaction. Am J Pathol 1997;150:477–82. [PMC free article] [PubMed] [Google Scholar]
- 28.Gordon T, McManus A, Anderson J, et al. Cytogenetic abnormalities in 42 rhabdomyosarcoma: a United Kingdom cancer cytogenetics group study. Med Pediatr Oncol 2001;36:259–67. [DOI] [PubMed] [Google Scholar]
- 29.Frascella E, Toffolatti L, Rosolen A. Normal and rearranged PAX3 expression in human rhabdomyosarcoma. Cancer Genet Cytogenet 1998;102:104–9. [DOI] [PubMed] [Google Scholar]
- 30.Kenet G, Sharon N, Rosner E, et al. Chromosomal translocation (1:13) in a case of alveolar rhabdomyosarcoma. J Pediatr Hematol Oncol 1998;20:86–7. [DOI] [PubMed] [Google Scholar]
- 31.Grundy R, Anderson J, Gaze M, et al. Congenital alveolar rhabdomyosarcoma: clinical and molecular distinction from alveolar rhabdomyosarcoma in older children. Cancer 2001;91:606–12. [DOI] [PubMed] [Google Scholar]
- 32.Kullendorff CM, Donner M, Mertens F, et al. Chromosomal aberrations in a consecutive series of childhood rhabdomyosarcoma. Med Pediatr Oncol 1998;30:156–9. [DOI] [PubMed] [Google Scholar]
- 33.Bernasconi M, Remppis A, Fredericks WJ, et al. Induction of apoptosis in rhabdomyosarcoma cells through down-regulation of PAX proteins. Proc Natl Acad Sci U S A 1996;93:13164–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Fredericks WJ, Galili N, Mukhopadhyay S, et al. The PAX3–FKHR fusion protein created by the t(2;13) translocation in alveolar rhabdomyosarcomas is a more potent transcriptional activator than PAX3. Mol Cell Biol 1995;15:1522–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Bennicelli JL, Edwards RH, Barr FG. Mechanism for transcriptional gain of function resulting from chromosomal translocation in alveolar rhabdomyosarcoma. Proc Natl Acad Sci U S A 1996;93:5455–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Davis RJ, Barr FG. Fusion genes resulting from alternative chromosomal translocations are overexpressed by gene-specific mechanisms in alveolar rhabdomyosarcoma. Proc Natl Acad Sci U S A 1997;94:8047–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Barr FG, Nauta LE, Davis RJ, et al. In vivo amplification of the PAX3–FKHR and PAX7–FKHR fusion genes in alveolar rhabdomyosarcoma. Hum Mol Genet 1996;5:15–21. [DOI] [PubMed] [Google Scholar]
- 38.Sublett JE, Jeon IS, Shapiro DN. The alveolar rhabdomyosarcoma PAX3/FKHR fusion protein is a transcriptional activator. Oncogene 1995;11:545–52. [PubMed] [Google Scholar]
- 39.Barr FG. Gene fusions involving PAX and FOX family members in alveolar rhabdomyosarcoma. Oncogene 2001;20:5736–46. [DOI] [PubMed] [Google Scholar]
- 40.Fitzgerald JC, Scherr AM, Barr FG. Structural analysis of PAX7 rearrangements in alveolar rhabdomyosarcoma. Cancer Genet Cytogenet 2000;117:37–40. [DOI] [PubMed] [Google Scholar]
- 41.Anderson J, Ramsay A, Gould S, et al. PAX3–FKHR induces morphological change and enhances cellular proliferation and invasion in rhabdomyosarcoma. Am J Pathol 2001;159:1089–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Collins MH, Zhao H, Womer RB, et al. Proliferative and apoptotic differences between alveolar rhabdomyosarcoma subtypes: a comparative study of tumors containing PAX3–FKHR or PAX7–FKHR gene fusions. Med Pediatr Oncol 2001;37:83–9. [DOI] [PubMed] [Google Scholar]
- 43.Wright WE, Sassoon DA, Lin VK. Myogenin, a factor regulating myogenesis, has a domain homologous to MyoD. Cell 1989;56:607–17. [DOI] [PubMed] [Google Scholar]
- 44.Aurade F, Pinset C, Chafey P, et al. Myf5, MyoD, myogenin and MRF4 myogenic derivatives of the embryonic mesenchymal cell line C3H10T1/2 exhibit the same adult muscle phenotype. Differentiation 1994;55:185–92. [DOI] [PubMed] [Google Scholar]
- 45.Weintraub H, Davis R, Tapscott S, et al. The MyoD gene family: nodal point during specification of muscle cell lineage. Science 1991;151:761–6. [DOI] [PubMed] [Google Scholar]
- 46.Faerman A, Pearson-White S, Emerson C, et al. Ectopic expression of MyoD1 in mice causes prenatal lethalities. Dev Dyn 1993;196:165–73. [DOI] [PubMed] [Google Scholar]
- 47.Tintignac LA, Leibovitch MP, Leibovitch SA. [New insight into MyoD regulation: involvement in rhabdomyosarcoma pathway?] Bull Cancer 2001;88:545–8. [PubMed] [Google Scholar]
- 48.Hosoi H, Sugimoto T, Hayashi Y, et al. Differential expression of myogenic regulatory genes, MyoD1 and myogenin, in human rhabdomyosarcoma sublines. Int J Cancer 1992;50:977–83. [DOI] [PubMed] [Google Scholar]
- 49.Yablonka-Reuveni Z, Paterson BM. MyoD and myogenin expression patterns in cultures of fetal and adult chicken myoblasts. J Histochem Cytochem 2001;49:455–62. [DOI] [PubMed] [Google Scholar]
- 50.Myer A, Olson EN, Klein WH. MyoD cannot compensate for the absence of myogenin during skeletal muscle differentiation in murine embryonic stem cells. Dev Biol 2001;229:340–50. [DOI] [PubMed] [Google Scholar]
- 51.Nabeshima Y, Hanaoka K, Hayasaka M, et al. Myogenin gene disruption results in perinatal lethality because of severe muscle defect. Nature 1993;364:532–5. [DOI] [PubMed] [Google Scholar]
- 52.Kahn HJ, Yeger H, Kassim O, et al. Immunohistochemical and electron microscopic assessment of childhood rhabdomyosarcoma. Increased frequency of diagnosis over routine histologic methods. Cancer 1983;51:1897–903. [DOI] [PubMed] [Google Scholar]
- 53.Engel ME, Mouton SC, Emms M. Paediatric rhabdomyosarcoma: MyoD1 demonstration in routinely processed tissue sections using wet heat pretreatment (pressure cooking) for antigen retrieval. J Clin Pathol 1997;50:37–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Cessna M, Coffin C, Perkins S, et al. Myogenin (MYOG) and MyoD1 (MD1) expression in rhabdomyosarcoma (RMS) and spindle cell mimics: a study of 135 cases. Lab Invest 2001;81:36. [DOI] [PubMed] [Google Scholar]
- 55.Cessna MH, Zhou H, Perkins SL, et al. Are myogenin and myoD1 expression specific for rhabdomyosarcoma? A study of 150 cases, with emphasis on spindle cell mimics. Am J Surg Pathol 2001;25:1150–7. [DOI] [PubMed] [Google Scholar]
- 56.Kumar S, Perlman E, Harris CA, et al. Myogenin is a specific marker for rhabdomyosarcoma: an immunohistochemical study in paraffin-embedded tissues. Mod Pathol 2000;13:988–93. [DOI] [PubMed] [Google Scholar]
- 57.Cui S, Hano H, Harada T, et al. Evaluation of new monoclonal anti-MyoD1 and anti-myogenin antibodies for the diagnosis of rhabdomyosarcoma. Pathol Int 1999;49:62–8. [DOI] [PubMed] [Google Scholar]
- 58.Folpe AL. MyoD1 and myogenin expression in human neoplasia: a review and update. Adv Anat Pathol 2002;9:198–203. [DOI] [PubMed] [Google Scholar]
- 59.Dias P, Chen B, Dilday B, et al. Strong immunostaining for myogenin in rhabdomyosarcoma is significantly associated with tumors of the alveolar subclass. Am J Pathol 2000;156:399–408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Williams S, Sebire NJ, Ramsay A, et al. Immunohistochemical expression of myogenin and MyoD1 in embryonal, classic alveolar and solid-variant paediatric alveolar rhabdomyosarcoma [abstract]. J Pathol 2002;198:25A. [Google Scholar]
- 61.Khan J, Bittner ML, Saal LH, et al. cDNA microarrays detect activation of a myogenic transcription program by the PAX3–FKHR fusion oncogene. Proc Natl Acad Sci U S A 1999;96:13264–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Frascella E, Rosolen A. Detection of the MyoD1 transcript in rhabdomyosarcoma cell lines and tumor samples by reverse transcription polymerase chain reaction. Am J Pathol 1998;152:577–83. [PMC free article] [PubMed] [Google Scholar]
- 63.Gattenloehner S, Dockhorn-Dworniczak B, Leuschner I, et al. A comparison of MyoD1 and fetal acetylcholine receptor expression in childhood tumors and normal tissues: implications for the molecular diagnosis of minimal disease in rhabdomyosarcomas. J Mol Diagn 1999;1:23–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Athale UH, Shurtleff SA, Jenkins JJ, et al. Use of reverse transcriptase polymerase chain reaction for diagnosis and staging of alveolar rhabdomyosarcoma, Ewing sarcoma family of tumors, and desmoplastic small round cell tumor. J Pediatr Hematol Oncol 2001;23:99–104. [DOI] [PubMed] [Google Scholar]