Abstract
Aims: To investigate the immunohistochemical expression of a panel of biologically relevant markers in patients with non-small cell lung cancer using fresh frozen specimens and to test their prognostic relevance for identification of patients at risk.
Methods: Seventy nine tumour infiltrated lung cancer specimens and 66 adjacent histologically tumour free tissues were analysed; 11 postmortem specimens from patients who did not suffer from a malignant disease served as a control group. Cryostat sections were stained with monoclonal antibodies against epidermal growth factor receptor (EGFR), c-erbB-2, c-erbB-3, CD82, Ki-67, p120, p53, bcl-2, and CD31.
Results: At least one of the tested markers was raised above the defined cut off point in 75 of the tumours. In 55, three to six factors were increased. EGFR was raised in 32, c-erbB-2 in 29, c-erbB-3 in 46, p53 in 29, bcl-2 in 26, Ki-67 in 36, p120 in 46, and CD31 in 29. None of the tested parameters was significant in univariate survival analysis. In a second step, three variables were combined (c-erbB3, p53, and microvessel density), and cases with increased expression of two or three parameters proved to have a significantly lower survival probability than those expressing none or only one factor. In the tumour free group only 10 showed raised marker expression.
Conclusion: Characterisation of tumour cells in surgical specimens with immunohistological markers could help identify those patients at risk for early cancer death who could possibly profit from adjuvant treatment after curative tumour resection.
Keywords: lung cancer; immunohistochemistry; p53, bcl-2; epidermal growth factor receptor; c-erbB-2; c-erbB 3; Ki-67; p120; microvessel density
Immunohistochemical analysis of the gene expression pattern in non-small cell lung cancer (NSCLC) can yield important insights into the biological course of the disease. These markers can provide information on the proliferative activity,1 metastatic potential,2,3 response to chemotherapy,4,5 and the patient’s prognosis.6 Moreover, identification of tumour specific markers can help develop new diagnostic and potentially therapeutic strategies.7 Among other methods, immunohistochemical analysis is widely used to analyse these markers, because it is technically easy to perform with standard laboratory equipment and therefore a valuable tool for routine diagnostic application.
Growth factor receptors (epidermal growth factor receptor (EGFR or c-erbB-1), c-erbB-2, and c-erbB-3), metastasis inhibiting parameters (CD82), proliferation associated markers (Ki-67 and p120), and apoptosis regulating markers (p53 and bcl-2) are of special interest. EGFR expression is increased in many lung carcinomas (32–81%),8–13 particularly squamous cell carcinomas (SCCs) rather than non-squamous cell carcinomas.13 However, most studies failed to demonstrate prognostic significance.10,12,14,15 Others suggested that EGFR expression is a useful indicator of nodal metastasis,10,13 pathological stage,12 and even survival.9,16 Approximately 20–50% of NSCLC cases show overexpression of the c-erbB-2 protein product, especially in adenocarcinomas (ACs). High expression is linked to shortened survival,9,17 and is associated with intrinsic multiple drug resistance.18 Other authors found no impact on prognosis.9,14 Only a few studies investigated the influence of c-erbB-3 protein expression in NSCLC, and high expression was associated with shorter survival in advanced NSCLC.19 CD82 is a member of the transmembrane 4 superfamily of cell membrane glycoproteins, and previous studies have shown that CD82 expression is associated with a significantly more favourable prognosis, especially for AC. It has been speculated that the KAI1/CD82 protein may have metastasis inhibiting activity.3 p53 overexpression has frequently been identified as a prognostic factor,4,6,17,20–23 although these results have been disputed.9,24–27 Downstream in the apoptotic pathway bcl-2 inhibits apoptosis. The expression of bcl-2 was reported to be a prognostic factor inversely correlated with p53.26 Others reported that bcl-2 positive tumours showed a prolonged time to recurrence, but no difference in survival.9,28 Ki-67 is a marker of proliferative activity and can provide prognostic information29 on the risk of recurrence and cancer death1,17 in NSCLC. The p120 nucleolar protein is another proliferation associated antigen detected predominantly in SCCs.30 Its influence on survival remains unclear. Neoangiogenesis is essential for tumour growth and metastatic spread and represents an important prognostic indicator in NSCLC.25,31–33 Therefore, more vascularised tumours, defined by microvessel density (MVD), predicted metastatic disease and showed reduced survival.31,32
“Neoangiogenesis is essential for tumour growth and metastatic spread and represents an important prognostic indicator in non-small cell lung cancer”
However, most of the reports described above were based on paraffin wax embedded material and because of a lack of standardisation in preparation, fixation, staining procedure, and analysis, the immunohistological results were contradictory. Our present study investigated the expression of a panel of relevant biological markers in 79 patients with lung cancer to find out whether cryostat sections can provide more reliable results than paraffin wax embedded material. A second aim of our study was to test different marker combinations to identify patients at risk.
PATIENTS AND METHODS
Patients
In total, 79 tumour infiltrated lung cancer specimens and 66 adjacent histologically tumour free tissues were analysed. Freshly resected tissues were collected for routine diagnosis. One portion of the sample was fixed in 10% formalin and routinely processed for paraffin wax embedding. All surgical tumour specimens were classified histopathologically according to the World Health Organisation classification.34 The second portion was used for immunohistochemical analysis. The samples were immediately snap frozen in liquid nitrogen and stored at −80°C until sectioning.
All 79 patients showed histological evidence of tumour cell infiltration. Adjacent histologically tumour free tissues from patients in the same group were analysed separately. Routine postmortem lung tissue probes from 11 patients who did not suffer from a malignant disease served as a control group for immunohistochemical analysis. In the 79 patients with tumours the histological diagnosis was as follows: AC in 37, SCC in 37, three patients with large cell carcinomas, and two with carcinoids. According to UICC recommendations, 52 patients were classified as stage I, 16 as stage II, nine as stage III, and two as stage IV. All patients were treated surgically between 1994 and 2001. Patients in advanced clinical stages were also treated with chemotherapy and/or radiotherapy. Median observation time was 24 (range, 1–109) months and the 50% survival probability was reached at 85 months; 26 patients died. At the time of surgery, the median age of the patients was 61 (range, 37–79) years. The male to female ratio was 3.4 : 1 (61 and 18)
Immunohistochemical staining
Frozen lung tissue specimens were cut at 3–5 μm on a cryostat, fixed, and incubated with monoclonal antibodies and with alkaline phosphatase complex (D651; Dako, Glostrup, Denmark), as described previously.35,36 All antibodies were diluted with phosphate buffered saline containing 1% bovine serum albumin. Monoclonal antibodies to the following molecules were used: cytokeratin (A45-B/B3; Micromet, Planegg-Martinsreid, Germany), CD82 (clone 50F11; Pharmingen, San Diego, California, USA), EGFR (clone EGFR.1; Pharmingen), c-erbB-2 (clone 3B5, Pharmingen), c-erbB-3 (clone RTJ.1; Pharmingen), p53 (PAb 1801, clone 1801; Pharmingen), bcl-2 (clone 100, Ab-1; Calbiochem, Oncogene Research, Cambridge, Massachusetts, USA), Ki-67 (Dakopatts, Glostrup, Denmark), p120 (Ab-1, Clone FB-1; Calbiochem), CD3 (clone SK7; Becton Dickinson, San Jose, California, USA), and CD19 (clone HIB19; Becton Dickinson).
Immunohistochemical evaluation
Slides were evaluated by two of the authors (WH and SD) using a semiquantitative method on a Zeiss AXIOSKOP 2 light microscope. The percentage of immunopositive cells in representative areas of the sections was assessed. The intensity of immunostaining was divided into four categories, namely: negative (−), low (+), moderate (++), and high (+++). Based on the results achieved in the control group, the cut off levels were defined as follows (mean value plus 2SD): c-erbB-2, > 15.7%; c-erbB-3, > 5.5%; bcl-2, > 12.9%; and CD3, > 16%. For some stainings there were very few positive cells in the control group, so that the cut off value was defined by means of the adjacent tumour free specimens from patients with tumours (mean value plus 2SD): EGFR, > 0.3%; p53, > 1.3%, Ki-67, > 2.7%; CD19, > 3.9%; p120, > 11.4%. Only cases with moderate (++) or high (+++) staining intensity and a percentage of immunopositive cells above the cut off point were scored as “positive”. CD82 was positive in all tumour cells and in normal epithelium of tumour and non-tumour specimens. Consequently, no cut points could be defined for this marker, and it was excluded from further statistical evaluation.
Microvessel density
Microvessel counting was performed in accordance with previous reports,31,32 with slight modifications. Endothelial cells were stained with an anti-CD31 (clone WM59; Serotec, Kidlington, Oxford, UK) monoclonal antibody. The three most intense regions were identified under low magnification (×100); using a higher magnification, positive cells were counted in each of the three areas. The median number of CD31 positive cells in these areas was then defined as the cut off point (15%).
Statistical analysis
The Pearson χ2 test was used to evaluate differences between the groups. Significance was determined using 95% confidence intervals. The log rank test and Cox proportional hazards model were applied to examine the relation between cancer specific survival and the immunohistochemical markers. Cancer specific survival was defined as the time between surgery and death or last follow up. All statistical procedures were performed with SPSS, version 7.5, statistical software (SPSS Inc, Chicago, Illinois, USA).
RESULTS
The tumour specimens from 75 of the patients with NSCLC revealed immunohistochemical alterations for at least one of the markers tested (EGFR, c-erbB-2, c-erbB-3, bcl-2, p53, bcl-2, Ki-67, or p120). One or two parameters were raised in 20, and in 29 three or four and in 26 five or six factors were altered (table 1; fig 1). Interestingly, only 10 of the 66 adjacent tumour free specimens from patients with tumours showed increased expression of the markers tested. In detail, one marker was raised in six cases and two markers in two cases. Alterations in three and four markers were found once. Tables 1 and 2 show the results in detail.
Table 1.
Percentage of immunopositive cells in the tumour specimens and the tumour free specimens
| Tumour | Tumour free specimens | |||
| Parameters | No. positive (%) cases | Median % positive cells (range)* | No. positive (%) cases | Median % positive cells (range)* |
| Number | 79 | 66 | ||
| EGFR | 32 (41%) | 3 (0–80) | 1 (2%) | 0 (0–1) |
| c-erbB-2 | 29 (37%) | 10 (0–80) | 3 (5%) | 1 (0–30) |
| c-erbB-3 | 46 (58%) | 20 (0– 80) | 3 (5%) | 0 (0–10) |
| bcl-2 | 26 (33%) | 10 (0–70) | 3 (5%) | 3 (0–20) |
| p53 | 29 (37%) | 0.1 (0– 50) | 1 (2%) | 0 (0–5) |
| Ki-67 | 36 (46%) | 1 (0–20) | 1 (2%) | 0 (0–10) |
| p120 | 46 (58%) | 10 (0–50) | 5 (8%) | 0.1 (0–20) |
| MVD | 29 (37%) | 20 (0.1–50) | ND | |
Also listed are the number and percentage of cases in which the immunopositive cells were raised above the defined cut off limits.
*Percentage of immunopositive cells/specimen.
EGFR, epidermal growth factor receptor; MVD, microvessel density; ND, not defined.
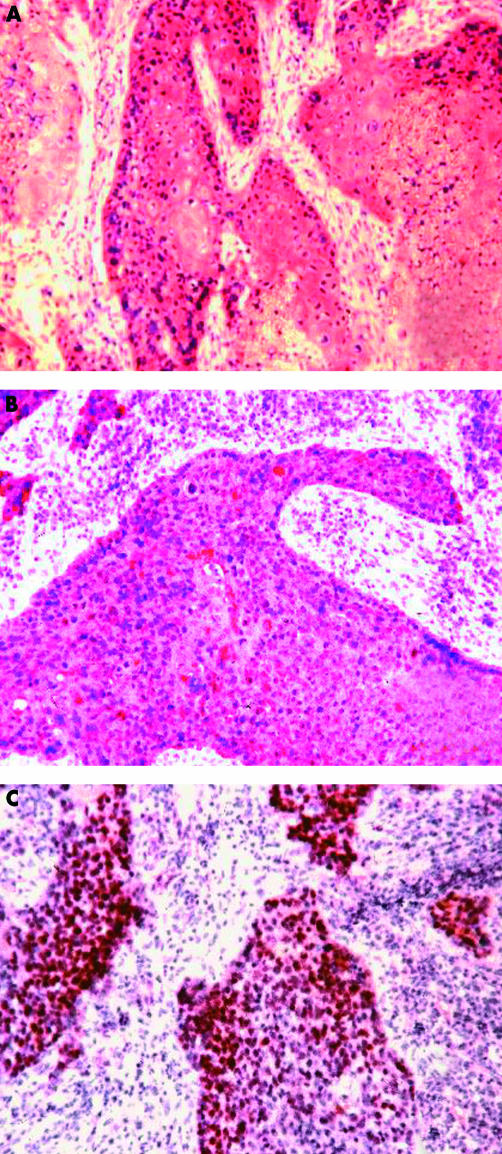
Figure 1.
Positive immunostaining of cryostat sections for (A) epidermal growth factor receptor, (B) c-erbB-3, and (C) p53.
Table 2.
Frequency of clinical parameters and raised immunohistochemical markers in NSCLC tumour specimens
| Parameters | Total | ||||||||
| Total | 79 | EGFR | cerbB2 | cerbB3 | bcl-2 | p53 | Ki-67 | p120 | MVD |
| Clinical parameters | |||||||||
| AC | 37 | 11 | 16 | 17 | 16 | 13 | 13 | 21 | 11 |
| SCC | 37 | 19 | 12 | 27 | 7 | 15 | 24 | 23 | 17 |
| LCC | 3 | 1 | 1 | 2 | 2 | 1 | 0 | 1 | 1 |
| Carcinoid | 2 | 1 | 0 | 0 | 1 | 0 | 0 | 1 | 0 |
| Death | 26 | 9 | 10 | 18 | 7 | 10 | 11 | 14 | 10 |
| IHC | |||||||||
| EGFR | 32 | – | 10 | 22 | 9 | 9 | 18 | 19 | 10 |
| c-erbB-2 | 29 | 10 | – | 21 | 8 | 12 | 15 | 22 | 5 |
| c-erbB-3 | 46 | 22 | 21 | – | 12 | 18 | 28 | 31 | 18 |
| bcl-2 | 26 | 9 | 8 | 12 | – | 8 | 12 | 13 | 11 |
| p53 | 29 | 9 | 12 | 18 | 8 | – | 18 | 22 | 12 |
| Ki-67 | 36 | 18 | 15 | 28 | 12 | 18 | – | 25 | 13 |
| p120 | 46 | 19 | 22 | 31 | 13 | 22 | 25 | – | 19 |
| MVD | 29 | 10 | 5 | 18 | 11 | 12 | 13 | 19 | – |
| CD3 | 7 | 2 | 2 | 5 | 4 | 4 | 3 | 3 | 2 |
| CD19 | 39 | 14 | 16 | 26 | 17 | 17 | 21 | 21 | 5 |
The bold numbers indicate a significant correlation between the two parameters (Pearson χ2 test, p < 0.05).
AC, adenocarcinoma; EGFR, epidermal growth factor; IHC, immunohistochemistry; LCC, large cell carcinoma; NSCLC, non-small cell lung cancer; MVD, microvessel density; SCC, small cell carcinoma.
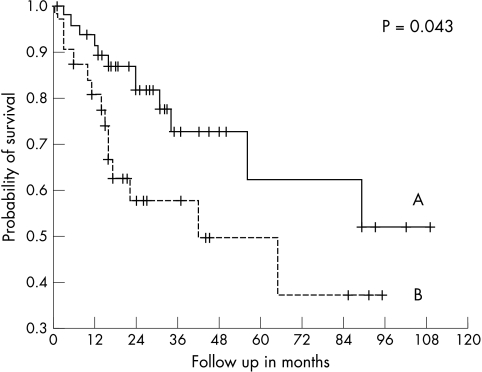
None of the tested parameters (EGFR, c-erbB-2, c-erbB-3, ki-67, p120, p53, bcl-2, and MVD) revealed a significant difference in univariate survival analysis. In a second step, the variables c-erbB-3, p53, and MVD were combined. Cases with increased expression of two or three parameters had a significantly lower survival probability in univariate analysis than those expressing none or only one or two factors (log rank: p = 0.043) (fig 2). A subanalysis revealed that this survival difference was predominantly seen in AC (p = 0.011), whereas in SCC these parameters were not able to differentiate prognostic subtypes.
Figure 2.
Survival of the 79 patients with non-small cell lung cancer categorised according to immunohistochemical marker expression (c-erbB-3, p53, and microvessel density). Line A, no markers or only one marker raised (n = 8); line B, two or three markers raised (n = 31); the difference was significant (p = 0.043).
The application of previously recommended cut off values revealed that for p53 a reduction to 0.1%26 and for c-erbB-319 an increase to 50% rendered prognostic significance to both markers (p53, p = 0.02; c-erbB-3, p = 0.04).
DISCUSSION
Seventy five of the 79 NSCLC tumour specimens revealed increased expression of at least one of the tested markers (EGFR, c-erbB-2, c-erbB-3, bcl-2, p53, Ki-67, p120, and MVD). In 55 of the cases three to six factors were raised. Using the predefined cut off points these data show that immunohistochemistry is a highly sensitive and specific means of identifying malignant tumour infiltration. Only 10 of the 66 adjacent microscopically tumour free specimens showed immunohistochemical alterations, and in most of these cases only single parameters were raised. However, more sophisticated methods, such as the polymerase chain reaction and fluorescence in situ hybridisation analysis, can more frequently detect molecular alterations in adjacent tumour free tissues and in tumour specimens with low infiltrations,37,38 so that immunohistochemistry is of little use in screening for premalignant lesions.
“In c-erbB-3 positive cases, Ki-67 was seen more frequently than in negative cases, indicating a more aggressive tumour type”
Cryostat sections were chosen to guarantee optimal staining by eliminating technical bias, including antigen damage as a result of uncontrolled duration of fixation or from heating during paraffin wax embedding.39–41 Another advantage of using frozen specimens is that it facilitates subsequent molecular examination. We calculated cut off values in a control group or using microscopically tumour free tissue—the biologically most relevant definition of “positivity”. Many other studies defined the cut off artificially or as the median value of the tumour group.42
CD82 expression was similar in tumour and non-tumour tissues. Almost all tumours were clearly positive with this monoclonal antibody. In contrast, a previous report3,43 analysing CD82 expression on cryostat sections from 200 patients found clearly positive expression in only 32.5% of the patients, and this correlated with a more favourable prognosis. Like others, we found EGFR expression more frequently in SCC,13 although we could not demonstrate a significant correlation between EGFR expression and prognosis,12,44 in accordance with several other reports.10,12,15,27 More interestingly, 29 of the patients had increased c-erbB-2 (Her2neu) expression, which correlated with increased p120 expression and reduced MVD. C-erbB-3 positivity was found in 46 of the tumour specimens, and correlated with the SCC histological subtype. In c-erbB-3 positive cases, Ki-67 was seen more frequently than in negative cases, indicating a more aggressive tumour type. Using paraffin wax embedded material Yi et al found that high c-erbB-3 protein expression was associated with shorter survival in advanced NSCLC.19 In their study, 18.5% of the tumours showed a very high c-erbB-3 positivity (score 3), with the highest percentage seen for SCC (29%). Similarly, in our study, only patients with high expression of c-erbB-3 showed a significantly lower survival probability (p = 0.04).
The apoptosis regulating protein p53 was raised in 29 of the tumour samples but only once in the adjacent tumour free group. In the p53 positive tumour cases, the numbers of proliferating cells (Ki-67 and p120 positive) were significantly increased. Previous immunocytochemical studies using various monoclonal antibodies and either paraffin wax embedded or fresh frozen material found that between 17% and 69% of NSCLC tumour samples had increased expression of p53. Consequently, it is not surprising that reports on the prognostic role of p53 have been inconsistent.4,6,17,21,23–27,35 In our cohort, p53 expression alone did not have an influence on prognosis when applying the predefined cut off value. However, p53 combined with c-erbB-3, and MVD was able to define a subgroup of patients with NSCLC who had a poor prognostic outcome. These results are similar to those of Harpole et al,17 who showed that p53 and c-erbB-2 expression were independent predictors of early recurrence and cancer death in multivariate analysis. Furthermore, when tumours with single p53 positive cells were included (cut off, 0.1%) p53 gained significance in our analysis.
The expression of the proliferation associated marker Ki-67 was raised in 36 of the tumour specimens and only once in the adjacent tumour free group. However, in our cohort with limited follow up data, Ki-67 did not reach prognostic significance. Its value for predicting recurrence and cancer death has already been proved,1,17,29 although in a large multivariate analysis by Harpole et al Ki-67 lost its significance because it was significantly associated with p53 status.17 Similarly, we found a clear correlation between Ki-67 and p53 in our cohort. The p120 nucleolar protein is a proliferation associated antigen and is initially expressed in the mid-G1 phase of the cell cycle and increases upon entry into the S phase. We found increased numbers of p120 positive cells in 46 of the tumour specimens and in only five of the adjacent tumour free samples. The major histological subtypes were equally distributed (AC, 21; SCC, 23) and no correlation with high c-erbB-2 and p53 expression was seen. In a previous smaller study (n = 37) the labelling index was 67% in SCCs, 35% in ACs, and 30% in large cell carcinomas.30 The prognostic role of p120 as a single factor in NSCLC has yet to be confirmed.
As reported previously, the single markers had no prognostic significance. However, the combination of various factors resulted in prognostic significance, reflecting the fact that the malignant phenotype is heterogeneous, different between individuals, and is the result of a multistep process.6,17,45 Schneider et al investigated c-Ki-ras mutation, together with p53 and c-erbB-2 expression, and found that the “multiple molecular marker parameter” improved the estimation of prognosis compared with single marker testing.45 Harpole et al combined clinical parameters with c-erbB-2 and p53 expression and vascular invasion.17 They proposed a multivariate model for risk assessment. A third group tested molecular markers on 244 patients6 and discovered that in addition to well known clinical parameters, K-ras codon 12 mutation, p53 expression, and the absence of H-ras p21 expression were prognostically relevant. They also proposed a pathological molecular substaging system. In their studies, the combination of immunohistochemistry, molecular methods, and clinical parameters resulted in an improvement in the predictive value. The usefulness of such promising staging systems in daily routine is limited by the fact that different techniques need to be used. However, our study used a single method that is in widespread use; immunohistochemical typing of tumours can be performed in most laboratories with standard equipment.
“By combining only a few key markers a more aggressive tumour phenotype can be defined”
Tumour associated alterations are seen using immunohistochemistry in nearly all patients suffering from NSCLC, and by combining only a few key markers a more aggressive tumour phenotype can be defined. The characterisation of tumour cells in surgical specimens by means of immunohistological markers could help identify those patients at risk for early cancer death, who could possibly profit from adjuvant treatment after curative tumour resection.
Take home messages.
Immunohistochemistry for p53, c-erbB-3, and microvessel density (as measured by anti-CD31) was able to define a subgroup of patients with non-small cell lung cancer who had a poor prognostic outcome
Thus, the characterisation of tumour cells in surgical specimens with immunohistological markers could help identify those patients at risk for early cancer death who could possibly profit from adjuvant treatment after curative tumour resection
Acknowledgments
Thanks to S Schnabl for her secretarial help. This study was supported by the Association for Cancer Research, Innsbruck (Verein für Tumorforschung, Innsbruck).
Abbreviations
AC, adenocarcinoma
EGFR, epidermal growth factor
NSCLC, non-small cell lung cancer
MVD, microvessel density
SCC, small cell carcinoma
REFERENCES
- 1.Pence JP, Kerns BM, Dodge RK, et al. Prognostic significance of the proliferation index in surgically resected non-small-cell lung cancer. Arch Surg 1993;128:1382–90. [DOI] [PubMed] [Google Scholar]
- 2.Miyoshi T, Kondo K, Hino N, et al. The expression of the CD44 variant exon 6 is associated with lymph node metastasis in non-small cell lung cancer. Clin Cancer Res 1997;3:1289–97. [PubMed] [Google Scholar]
- 3.Higashiyama M, Kodama K, Yokouchi H, et al. KAI1/CD82 expression in nonsmall cell lung carcinoma is a novel, favorable prognostic factor: an immunohistochemical analysis. Cancer 1998;83:466–74. [DOI] [PubMed] [Google Scholar]
- 4.Rusch V, Klimstra D, Venkatraman E, et al. Aberrant p53 expression predicts clinical resistance to cisplatin-based chemotherapy in locally advanced non-small cell lung cancer. Cancer Res 1995;55:5038–43. [PubMed] [Google Scholar]
- 5.Perdomo JA, Naomoto Y, Haisa M, et al. In vivo influence of p53 status on proliferation and chemoradiosensitivity in non-small-cell lung cancer. J Cancer Res Clin Oncol 1998:124:10–18. [DOI] [PubMed] [Google Scholar]
- 6.Kwiatkowski DJ, Harpole DH, Jr, Godleski J, et al. Molecular pathologic substaging in 244 stage I non-small-cell lung cancer patients: clinical implications. J Clin Oncol 1998:16:2468–77. [DOI] [PubMed] [Google Scholar]
- 7.Bunn PA. Novel targeted agents for the treatment of lung cancer. ASCO Educational Book, Spring 2002, 683–92.
- 8.Kurie JM, Shin HJ, Lee JS, et al. Increased epidermal growth factor receptor expression in metaplastic bronchial epithelium. Clin Cancer Res 1996;2:1787–93. [PubMed] [Google Scholar]
- 9.Pastorino U, Andreola S, Tagliabue E, et al. Immunocytochemical markers in stage I lung cancer: relevance to prognosis. J Clin Oncol 1997;15:2858–65. [DOI] [PubMed] [Google Scholar]
- 10.Fujino S, Enokibori T, Tezuka N, et al. A comparison of epidermal growth factor receptor levels and other prognostic parameters in non-small cell lung cancer. Eur J Cancer 1996;32A:2070–4. [DOI] [PubMed] [Google Scholar]
- 11.Brabender J, Danenberg KD, Metzger R, et al. Epidermal growth factor receptor and HER2-neu mRNA expression in non-small cell lung cancer is correlated with survival. Clin Cancer Res 2001;7:1850–5. [PubMed] [Google Scholar]
- 12.Rusch V, Klimstra D, Venkatraman E, et al. Overexpression of the EGFR and its ligand, TGFalpha, is frequent in resectable non-small cell lung cancer but does not predict tumour progression. Clin Cancer Res 1997;3:515–22. [PubMed] [Google Scholar]
- 13.Fontanini G, Vignati S, Bigini D, et al. Epidermal growth factor receptor (EGFr) expression in non-small cell lung carcinomas correlates with metastatic involvement of hilar and mediastinal lymph nodes in the squamous subtype. Eur J Cancer 1995;31A:178–83. [DOI] [PubMed] [Google Scholar]
- 14.Pfeiffer P, Clausen PP, Andersen K, et al. Lack of prognostic significance of epidermal growth factor receptor and the oncoprotein p185HER-2 in patients with systemically untreated non-small-cell lung cancer: an immunohistochemical study on cryosections. Br J Cancer 1996;74:86–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gerdes AM, Nielsen O, Mohr U, et al. Correlation between molecular genetic analyses and immunohistochemical evaluation of the epidermal growth factor receptor and p185HER2. Anticancer Res 1998;18(4A):2529–34. [PubMed] [Google Scholar]
- 16.Volm M, Rittgen W, Drings P. Prognostic value of ERBB-1, VEGF, cyclin A, FOS, JUN and MYC in patients with squamous cell carcinomas. Br J Cancer 1998;77:663–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Harpole DH, Jr, Herndon JE, Wolfe WG, et al. A prognostic model of recurrence and death in stage I non-small cell lung cancer utilizing presentation, histopathology, and oncogene expression. Cancer Res 1995;55:51–6. [PubMed] [Google Scholar]
- 18.Tsai CM, Chang KT, Wu LH, et al. Correlation between intrinsic chemoresistance and HER-2/neu gene expression, p53 gene mutations, and cell proliferation characteristics in non-small cell lung cancer lines. Cancer Res 1996;56:206–9. [PubMed] [Google Scholar]
- 19.Yi ES, Harclerode D, Gondo M, et al. High c-erbB-3 protein expression is associated with shorter survival in advanced non-small cell lung carcinomas. Mod Pathol 1997;10:142–8. [PubMed] [Google Scholar]
- 20.Kawasaki M, Nakanishi Y, Kuwano K, et al. The utility of p53 immunostaining of transbronchial biopsy specimens of lung cancer: p53 overexpression predicts poor prognosis and chemoresistance in advanced non-small cell lung cancer. Clin Cancer Res 1997;3:1195–200. [PubMed] [Google Scholar]
- 21.Harpole DH, Marks JR, Richards WG, et al. Localized adenocarcinoma of the lung: oncogene expression of erbB-2 and p53 in 150 patients. Clin Cancer Res 1995;1:659–64. [PubMed] [Google Scholar]
- 22.Ohsaki Y, Toyoshima E, Fujiuchi S, et al. bcl-2 and p53 protein expression in non-small cell lung cancers: correlation with survival time. Clin Cancer Res 1996;2:915–20. [PubMed] [Google Scholar]
- 23.Nishio M, Koshikawa T, Kuroishi T, et al. Prognostic significance of abnormal p53 accumulation in primary, resected non-small cell lung cancer. J Clin Oncol 1996;14:497–502. [DOI] [PubMed] [Google Scholar]
- 24.Vega FJ, Iniesta P, Caldes T, et al. p53 exon 5 mutations as a prognostic indicator of shortened survival in non-small-cell lung cancer. Br J Cancer 1997;76:44–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Apolinario RM, van der Valk P, de Jong JS, et al. Prognostic value of the expression of p53, bcl-2, and bax oncoproteins, and neovascularization in patients with radically resected non-small-cell lung cancer. J Clin Oncol 1997;15:2456–66. [DOI] [PubMed] [Google Scholar]
- 26.Fontanini G, Vignati S, Bigini D, et al. bcl -2 protein: a prognostic factor inversely correlated to p53 in non-small cell lung cancer. Br J Cancer 1995;71:1003–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Greatens TM, Niehans GA, Rubins JB, et al. Do molecular markers predict survival in non-small-cell lung cancer? Am J Respir Crit Care Med 1998;157:1093–7. [DOI] [PubMed] [Google Scholar]
- 28.Fleming MV, Guinee DG, Jr, Chu WS, et al. Bcl-2 immunohistochemistry in a surgical series of non-small cell lung cancer patients. Hum Pathol 1998;29:60–4. [DOI] [PubMed] [Google Scholar]
- 29.Pence JC, Kerns BJ, Dodge RK, et al. Prognostic significance of the proliferation index in surgically resected non-small-cell lung cancer. Arch Surg 1993;128:1382–90. [DOI] [PubMed] [Google Scholar]
- 30.Uchiyama B, Saijo Y, Kumano N, et al. Expression of nucleolar protein p120 in human lung cancer: difference in histological types as a marker for proliferation. Clin Cancer Res 1997;3:1873–7. [PubMed] [Google Scholar]
- 31.Fontanini G, Lucchi M, Vignati S, et al. Angiogenesis as a prognostic indicator of survival in non-small-cell lung carcinoma: a prospective study. J Natl Cancer Inst 1997;89:881–6. [DOI] [PubMed] [Google Scholar]
- 32.Fontanini G, Bigini D, Vignati S, et al. Microvessel count predicts metastatic disease and survival in non-small cell lung cancer. J Pathol 1995;177:57–63. [DOI] [PubMed] [Google Scholar]
- 33.Ushijima C, Tsukamoto S, Yamazaki K, et al. High vascularity in the peripheral region of non-small cell lung cancer tissue is associated with tumor progression. Lung Cancer 2001;34:233–41. [DOI] [PubMed] [Google Scholar]
- 34.Brambilla E, Travis WD, Colby TV, et al. The new World Health Organization classification of lung tumours. Eur Respir J 2001;18:1059–68. [DOI] [PubMed] [Google Scholar]
- 35.Hilbe W, Gächter A, Duba HC, et al. Comparison of ACISTM (automated cellular imaging system) and manual microscopy for immunohistochemically stained sections of lung cancer specimens applying p53, ki-67 and p120. Oncol Rep 2003;10:15–20. [PubMed] [Google Scholar]
- 36.Cordell JL, Falini B, Erber WN, et al. Immunoenzymatic labeling of monoclonal antibodies using immune complexes of alkaline phosphatase and monoclonal anti-alkaline phosphatase (APAAP-complexes). J Histochem Cytochem 1984;32:219–29. [DOI] [PubMed] [Google Scholar]
- 37.Niklinski J, Niklinska W, Chyczewski L, et al. Molecular genetic abnormalities in premalignant lung lesions: biological and clinical implications. Eur J Cancer Prev 2001;10:213–26. [DOI] [PubMed] [Google Scholar]
- 38.Zojer N, Dekan G, Ackermann J, et al. Aneuploidy of chromosome 7 can be detected in invasive lung cancer and associated premalignant lesions of the lung by fluorescence in situ hybridisation. Lung Cancer 2000;28:225–35. [DOI] [PubMed] [Google Scholar]
- 39.Jacobs TW, Prioleau JE, Stillman IE, et al. Loss of tumor marker-immunostaining on stored paraffin slides of breast cancer. J Natl Cancer Inst 1996;88:1054–9. [DOI] [PubMed] [Google Scholar]
- 40.Binks S, Clelland CA, Ronan J, et al. p53 gene product expression in resected non-small cell carcinoma of the lung, with studies of concurrent cytological preparations and microwave antigen retrieval. J Clin Pathol 1997;50:320–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kerns BJ, Jordan PA, Moore MB, et al. p53 overexpression in formalin-fixed, paraffin-embedded tissue detected by immunohistochemistry. J Histochem Cytochem 1992;40:1047–51. [DOI] [PubMed] [Google Scholar]
- 42.Altman DG, Lausen B, Sauerbrei W, et al. Danger of using “optimal” cutpoints in the evaluation of prognostic factors. J Natl Cancer Inst 1994;86:829–35. [DOI] [PubMed] [Google Scholar]
- 43.Adachi M, Taki T, Ieki Y, et al. Correlation of KAI1/CD82 gene expression with good prognosis in patients with non-small cell lung cancer. Cancer Res 1996;56:1751–55. [PubMed] [Google Scholar]
- 44.Rusch V, Baselga J, Cordon-Cardo C, et al. Differential expression of the epidermal growth factor receptor and its ligands in primary non-small cell lung cancers and adjacent benign lung. Cancer Res 1993;53:2379–85. [PubMed] [Google Scholar]
- 45.Schneider PM, Praeuer HW, Stoeltzing O, et al. Multiple molecular marker testing (p53, C-Ki-ras, c-erbB-2) improves estimation of prognosis in potentially curative resected non-small cell lung cancer. Br J Cancer 2000;83:473–9. [DOI] [PMC free article] [PubMed] [Google Scholar]