Abstract
Background/aim: The authors investigated the concentrations of 8-isoprostaglandin F2a, a marker of oxidative stress in vivo, and ascorbic acid, a protectant against oxidative damage, in the aqueous humour of patients with exfoliation syndrome (XFS) and cataract and compared the results with those in age matched patients with cataract, but without XFS, to determine whether XFS is associated with increased oxidative stress.
Methods: Aqueous humour was aspirated at the beginning of phacoemulsification cataract surgery from 27 eyes of 27 cataract patients with XFS and 27 eyes of 27 age matched cataract patients without XFS. 8-Isoprostaglandin F2aconcentration in the aqueous was determined with a commercial immunoassay; ascorbic acid concentration was measured with a microplate assay method.
Results:. The mean concentration of 8-isoprostaglandin F2ain the aqueous from patients with XFS (2429 (SD 2940) pg/ml; range 400–10500 pg/ml) was significantly higher than that measured in the aqueous of age matched control patients (529.1 (226.8) pg/ml; range 325–1000 pg/ml); (p = 0.0028). Furthermore, mean ascorbic acid concentration in XFS patients (0.75 (0.39) mM; range 0.28–1.70 mM) was significantly lower than that found in control patients (1.19 (0.47) mM; range 0.53–2.4 mM); (p = 0.0005). There was a reverse correlation between 8-isoprostaglandin F2aand ascorbic acid concentration.
Conclusion: 8-Isoprostaglandin F2awas significantly increased in the aqueous of patients with XFS, and ascorbic acid was decreased, providing evidence of a role for free radical induced oxidative damage in the pathobiology of XFS.
First described by Lindberg in 1917, exfoliation syndrome (XFS) is an age related disease characterised by the synthesis and progressive accumulation of a fibrillar extracellular material in many ocular tissues. It is the most common identifiable condition leading to the development of open angle glaucoma (exfoliative glaucoma, XFG).1,2The pathogenesis and mechanisms underlying the development of XFS and the subsequent progression from XFS to XFG remain unknown.2,3
Ultraviolet radiation, a main cause of free radical formation, has been implicated in various ocular disorders such as pterygium, cataract, and age related macular degeneration. Differences in exposure to ultraviolet radiation have been suggested to be a cause for the variable prevalence of XFS around the world.4 Growth factors5 and metalloproteinases6,7 can be activated by free radicals. Furthermore, growth factors and metalloproteinases have been shown to be involved in the pathobiology of XFS.8,9 In particular, increased concentrations of transforming growth factor beta 1 (TGF-β1), a powerful modulator of extracellular matrix formation,10 have been detected in the aqueous humour of patients with XFS and XFG11,12 and increased TGF-β1 synthesis has been associated with increased levels of reactive oxygen species.13,14
There is a significant link between ultraviolet irradiation and ascorbic acid levels in the aqueous humour.15 Ascorbic acid, a major antioxidant and free radical scavenger in the eye, may be reduced in the aqueous humour of patients with XFS suggesting a role for free radical induced oxidative damage in XFS pathobiology.
The levels of free radical catalysed arachidonic acid products, such as 8-isoprostaglandin F2a (8-IPGF), in urine and other biological fluids are currently employed as markers of oxidative stress in vivo.16,17 These compounds in their esterified form may contribute to free radical catalysed cell membrane injury.18 Furthermore, isoprostanes influence extracellular matrix synthesis and express growth factor activity in cultured rat aortic smooth muscle cells.19 These experimental data support the notion that isoprostanes formed by free radicals in the anterior segment of the eye may have a role in XFS development. We investigated the role of free radicals in XFS by comparing and correlating the aqueous levels of 8-IPGF and ascorbic acid in patients with XFS and cataract and age matched cataract controls.
PATIENTS AND METHODS
Aqueous humour samples were collected at two collaborating centres at the university department of ophthalmology, Aristotle University of Thessaloniki, Greece and at the department of ophthalmology, General Hospital of Patra, Greece. We entered in this prospective study samples from normotensive patients with and without XFS who were undergoing routine cataract surgery. We excluded patients with other ophthalmic conditions (for example, glaucoma, uveitis, progressive retinal disease, etc) and systemic diseases (for example, diabetes, arthritis) that might have influenced the levels of 8-IPGF or ascorbic acid. Excluded were patients who received topical medications and patients who had undergone previous laser or intraocular surgery. The only systemic medications allowed in this study were those used for control of arterial hypertension.
All patients underwent a comprehensive ocular examination before enrolment. A dilated examination was performed the day before surgery to look for exfoliation material. All patients had intraocular pressure lower than 20 mm Hg and did not receive any local medication except mydriatics before surgery. Consent was obtained from all patients. For the purposes of this study only patients exhibiting exfoliation material deposits on the lens and/or pupillary border were deemed to have XFS. Patients exhibiting pigment liberation signs commensurate with the definition of exfoliation suspect20 were excluded from the study to ensure lack of possible contamination of the control group.
Aqueous humour samples were obtained before fashioning of the scleral tunnel. A small amount of aqueous humour (10–40 μl) was aspirated through an ab externo limbal paracentesis site using a 27 gauge needle on a tuberculin syringe, with special care to avoid blood contamination. Immediately after collection, aqueous samples were stored at −70°C until biochemical analysis. Samples that could not be evaluated within 6–9 weeks of storage were not included in this study and were used for the preparation of ascorbic control samples. This investigation included a total of 54 aqueous samples, 27 from cataract patients with XFS (mean age 77.1 (SD 5.9) years) and 27 from age matched cataract patients without XFS (mean age 74.2 (6.3) years).
8-IPGF measurement assay
The aqueous concentration of 8-IPGF was measured with a commercial immunoassay kit (Assay Designs Inc, Ann Arbor, MI, USA). The kit is a competitive enzyme linked immunoassay. An 8-IPGF polyclonal antibody binds either to the 8-IPGF contained in the sample or to alkaline phosphatase covalently bound 8-IPGF. A calibration curve was constructed using a standard 8-IPGF solution provided by the kit. Ten μl of aqueous was diluted with 40 μl of the provided diluent solution and used directly, without previous hydrolysis. The 8-IPGF concentration measurement assay was used according to the instructions of the provider, following the overnight incubation protocol. The lower detection limit of the assay was determined as the concentration of 8-IPGF measured at 2 SD from the zero along the calibration curve and was calculated to be 62 pg/ml. Thus, the detection limit for the diluted aqueous samples was 310 pg/ml. Interassay variation was found to be 6.1% for a concentration of 2.0 pg/ml.
Ascorbic acid measurement assay
Ascorbic acid concentration in the aqueous was assessed with a microplate assay method, in accordance with the technique described by Shichi et al.21 The technique employed has been reported previously.22 Before analysis aqueous humour samples were treated with 6% trichloroacetic acid, incubated at room temperature for 15 minutes and centrifuged for 15 minutes at 13 000 g in order to precipitate protein. Protein was then measured in the sediment using the bicinchonic acid (BCA) assay as described before.23
Ten μl of each supernatant, standard solution, or control was mixed in a microplate well with 5 μl of 1M NaOH and was incubated for 30 minutes at room temperature. Following incubation another 10 μl of each supernatant was placed in a second microplate well. Each sample was then mixed with 200 μl of BCA reagent (Pierce, Rockford, IL, USA). After incubation for 30 minutes at room temperature the absorbance at 562 nm was measured in a microplate photometer. Ascorbic acid (Sigma-Aldrich Chemie Gmbh, Steinheim, Germany) solutions with known concentrations served as standards for the construction of a calibration curve.
For the construction of a control sample we employed a pool of 15 aqueous humour samples, which were left for 4 weeks at 4°C and were then found to have undetectable levels of ascorbic acid. Subsequently, a known amount of ascorbic acid (0.7 mM) was added to this control pool. This pool was then aliquoted and kept at −70°C until measurement concurrently with the study samples. Throughout the study a satisfactory recovery, 96% (6.2%), interassay variation 4.3% and intra-assay variation 7.1% was calculated from this sample.
Statistical calculations were performed with the GraphPad InStat statistical package for Windows. Data are expressed as mean (SD). Normal distribution fitting was checked by the Kolmogorov-Smirnov goodness of fit test. Statistical significance of the differences between XFS and controls was calculated by the two tailed unpaired Student’s t test with Welch correction or by the Mann-Whitney non-parametric test where applicable. Correlations between sets of data were estimated by Pearson or by Spearman non-parametric correlation.
RESULTS
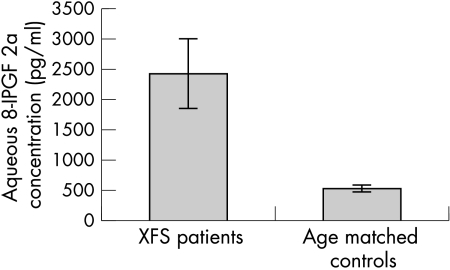
8-IPGF concentration was above the lowest detection limits of the assay (310 pg/ml) in 19 of the 27 samples derived from patients with XFS and in only 11 of the 27 controls. The values of 8-IPGF did pass Kolmogorov-Smirnov normality test only in controls. The mean concentration of 8-IPGF from patients with XFS was 2429 (2940) pg/ml (range 400–10 500 pg/ml). A significantly lower 8-IPGF concentration was measured in the aqueous of age matched control patients (mean 529.1 (226.8) pg/ml; range 325–1000 pg/ml); (p = 0.0028; Mann-Whitney test) (Fig 1).
Figure 1.
8-Isoprostaglandin F2a concentration in the aqueous humour of cataract patients with and without XFS. A significantly higher concentration (p = 0.0054) was found in the XFS samples. Error bars represent standard error of means (SEM).
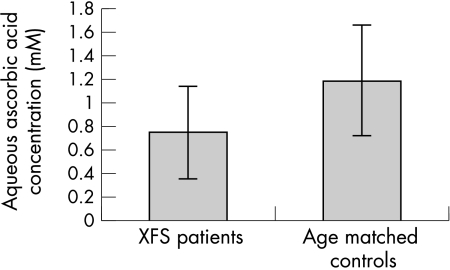
Ascorbic acid was detected in all samples collected. The mean concentration of ascorbic acid in the aqueous humour from patients with XFS (0.75 (0.39) mM; range 0.28–1.70 mM) was significantly lower than that of the age matched control patients (mean 1.19 (0.47) mM; range 0.53–2.4 mM); (p = 0.0005; Student t test); (Fig 2).
Figure 2.
Ascorbic acid concentration in the aqueous humour of cataract patients with and without XFS. A significantly lower concentration (p = 0.0005) was found in the XFS samples. Error bars represent standard deviation (SD).
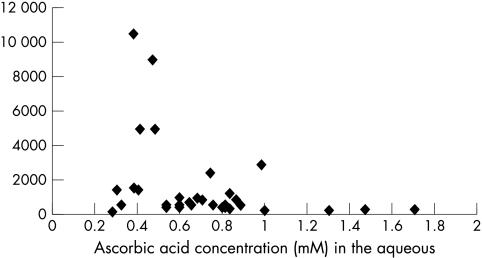
A significant reverse correlation was documented between 8-IPGF and ascorbic levels in all samples with measurable 8-IPGF concentrations (40 samples); (p = 0.0004; Spearman correlation). This is graphically shown in Figure 3. 8-IPGF and ascorbic acid concentration did not correlate with age (p = 0.765 and p = 0.936 respectively; Pearson correlation).
Figure 3.
Correlation between 8-isoprostaglandin F2a and ascorbic acid concentration in all aqueous humour samples with measurable 8-isoprostaglandin F2a values. A significant correlation (Spearman p = 0.0004) was found.
DISCUSSION
In the present study, the mean concentration of 8-IPGF in aqueous from patients with XFS and cataract was found to be approximately five times greater than that measured in the aqueous from age matched control cataract patients. Furthermore, in those patients with XFS mean ascorbic acid concentration was significantly decreased. To the best of our knowledge, this is the first time that the level of 8-IPGF has been assessed in the human aqueous humour.
Isoprostanes are formed by free radical mediated peroxidation of arachidonic acid and serve as biomarkers of lipid peroxidation and potential mediators of free radical injury. These molecules are geometric stereoisomers to cyclo-oxygenase generated prostaglandins with their hydrocarbon chains in a cis position in relation to the pentane ring.24 8-IPGF is produced by free radical attack on arachidonate, which resides on the SN-2 position of cell membrane phospholipids.18 The resulting esterified peroxy-arachidonate is then converted to isoprostane by β cleavage followed by rearrangement and finally cleaved by a phospholipase and released into the surrounding fluid. The same molecule can also be synthesised by prostaglandin endoperoxide synthase-2 in human monocytes.25 8-IPGF circulates in plasma and is excreted in urine and its concentration in these biological fluids has been used as an indicator of oxidative stress status in vivo.16,17,26–28 Among other important biological actions 8-IPGF is a potent vasoconstrictor and a mitogen for fibroblasts and vascular smooth muscle cells.29–31 Thus 8-IPGF may also mediate free radical injury to the surrounding tissues.
The mean value of non-esterified 8-IPGF in the control aqueous samples was approximately three times greater than the reported value in the plasma, while the mean value of IPGF in aqueous from XFS patients was approximately 15 times greater than in plasma, which was reported to be about 140 pg/ml.32,33 Since the concentration of 8-IPGF reflects the oxidative stress status in vivo,17,26,27 the higher 8-IPGF concentration in aqueous may be attributed to higher oxidative stress applied to ocular tissues exposed to ultraviolet radiation.34 It is well documented that aqueous humour acts as an ultraviolet filter protecting the lens and retina against ambient radiation.35 It is for this purpose that aqueous humour contains a series of molecules such as ascorbic acid, urate, l-tyrosine, l-cysteine, and glutathione that protect the eye against free radical damage.36 Ascorbic acid is the most important of these molecules in terms of ocular antioxidant defence.15 The reverse correlation established in the current study between ascorbic acid and 8-IPGF concentrations in the aqueous further supports this notion.
We found significantly lower ascorbate levels in the aqueous humour of patients with XFS compared to age matched controls. The finding further supports our recent report in another series of XFS patients.22 However, it remains to be determined whether the decreased level of ascorbic acid in the aqueous is part of the underlying XFS pathobiology, or merely reflects a secondary phenomenon due to concurrent damage of the ciliary secretory cells or increased oxidation.
Ultraviolet irradiation, a main cause of free radical formation, has been implicated in the past for the variable prevalence of XFS in various ethnic cohorts around the world.4,37 Both decreased ascorbate levels and increased 8-IPGF concentration in the aqueous of XFS patients may be attributed to the influence of ultraviolet irradiation. The interrelation between ascorbic acid and 8-IPGF is complex and a cause and effect relation between these two molecules cannot be proved. Nevertheless, the data presented herein suggest a faulty antioxidant defence system and enhanced oxidative stress in the anterior chamber of patients with XFS. We hypothesise that 8-IPGF is implicated in XFS pathobiology, either by expressing growth factor activity and directly influencing extracellular matrix metabolism,19,30 or, alternatively, by influencing matrix formation indirectly via upregulation of growth factors and downregulation of proteolytic enzymes such as matrix metalloproteinases (MMPs).
8-IPGF has been reported to increase TGF-β1 synthesis in experimental type I diabetes38 and to decrease MMP-2 and MMP-9 activity in vitro.39 Recently, a significantly increased level of TGF-β1 has been measured in the aqueous humour of patients with XFS and has been suggested to play an important part in the pathogenesis of XFS.11,12 Moreover, levels of activated MMP-2 were significantly decreased in aqueous samples from XFS patients,9 suggesting that reduced MMP activity in aqueous humour may promote the abnormal matrix accumulation. There is increasing evidence to suggest that oxidative stress together with anterior segment hypoxia, upregulation of growth factors, particularly TGF-β1, and a MMP/TIMP imbalance constitute major molecular mechanisms involved in the pathogenesis of XFS. These mechanisms ultimately may lead to increased extracellular matrix formation and progressive accumulation of exfoliation material in the anterior segment tissues of patients with XFS.
The hypothesis that oxidative stress is causally involved in XFS development and that a cascade of complex events links 8-IPGF, ascorbate, and TGF-β1 in XFS is supported by the observation that TGF-β1 synthesis is promoted by oxidative stress caused by hydrogen peroxide.13,14 Further evidence is provided by a number of reports which demonstrate that oxidative injury can trigger fibrogenic responses and that oxidative stress resulting in lipid peroxidation is involved in the pathogenesis of fibrotic disorders—for example, hepatic and pulmonary fibrosis.40
It should also be emphasised that 8-IPGF itself is a potent vasoconstrictor and promotes the secretion of endothelin, a peptide with additional mitogenic and vasoconstrictor activities.30,31,41 These actions of 8-IPGF may provide a logical explanation for the microvascular abnormalities documented in the iris vasculature in patients with XFS/XFG resulting in a pronounced hypoperfusion and hypoxia in the anterior chamber of these patients.41–45
In conclusion, our findings indicate that free radical action together with a depleted antioxidant defence system may represent important molecular mechanisms in the development of XFS. Future research is required to improve the understanding of these mechanisms in order to elucidate the pathogenesis of XFS, as a first step towards development of specific treatment strategies in the future.
Acknowledgments
Supported by grant 912-OPT-0091-149 from Pharmacia (New Jersey, USA) to Dr AGP Konstas.
We acknowledge Ms Sophia Kotsinou for excellent technical assistance and the help of Dr Ioannis Tersis with the aqueous samples.
REFERENCES
- 1.Ritch R. Exfoliation syndrome. Curr Opin Ophthalmol 2001;12:124–30. [DOI] [PubMed] [Google Scholar]
- 2.Ritch R, Schlotzer-Schrehardt U. Exfoliation syndrome. Surv Ophthalmol 2001;45:265–315. [DOI] [PubMed] [Google Scholar]
- 3.Konstas AG, Marshall GE, Lee WR. Immunogold localisation of laminin in normal and exfoliative iris. Br J Ophthalmol 1990;74:450–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Taylor HR. Pseudoexfoliation, an environmental disease? Trans Ophthalmol Soc UK 1979;99:302–7. [PubMed] [Google Scholar]
- 5.Barcellos-Hoff MH, Dix TA. Redox-mediated activation of latent transforming growth factor-beta 1. Mol Endocrinol 1996;10:1077–83. [DOI] [PubMed] [Google Scholar]
- 6.Cejkova J, Lojda Z. The apearance of active plasminogen activator of urokinase type (u-PA) in the rabbit anterior eye segment irradiated by UVB rays. A histochemical and biochemical study. Acta Histochem 1995;97:257–62. [DOI] [PubMed] [Google Scholar]
- 7.Cejkova J, Lojda Z. The damaging effect of UV rays below 320nm on the rabbit anterior eye segment II. Enzyme histochemical changes and plasmin activity after prolonged irradiation. Acta Histochem 1995;97:183–262. [DOI] [PubMed] [Google Scholar]
- 8.Koliakos GG, Konstas AG, Triantos A, et al. Increased growth factor activity in the aqueous humour of patients with exfoliation syndrome. Graefes Arch Clin Exp Ophthalmol 2000;238:491–5. [DOI] [PubMed] [Google Scholar]
- 9.Schlotzer-Schrehardt U, Zenkel M, Lommatzsch J, et al. Altered balance between matrix metalloproteinasses and their inhibitors in pseudoexfoliation syndrome and pseudoexfoliation glaucoma. ARVO meeting 2002.
- 10.Border WA, Noble NA. Transforming growth factor beta in tissue fibrosis. N Engl J Med 1994;331:1286–92. [DOI] [PubMed] [Google Scholar]
- 11.Koliakos GG, Schlotzer-Schrehardt U, Konstas AG, et al. Transforming and insulin-like growth factors in the aqueous humour of patients with exfoliation syndrome. Graefes Arch Clin Exp Ophthalmol 2001;239:482–7. [DOI] [PubMed] [Google Scholar]
- 12.Schlötzer-Schrehardt U, Zenkel M, Küchle M, et al. Role of transforming growth factor-β1 and its latent form binding protein in pseudoexfoliation syndrome. Exp Eye Res 2001;73:765–80. [DOI] [PubMed] [Google Scholar]
- 13.Iglesias-De La Cruz MC, Ruiz-Torres P, Alcami J, et al. Hydrogen peroxide increases extracellular matrix mRNA through TGF-beta in human mesangial cells. Kidney Int 2001;59:87–95. [DOI] [PubMed] [Google Scholar]
- 14.Weiss MF, Scivittaro V, Anderson JM. Oxidative stress and increased expression of growth factors in lesions of failed hemodialysis access. Am J Kidney Dis 2001;37:970–80. [DOI] [PubMed] [Google Scholar]
- 15.Ringvold A. The significance of ascorbate in the aqueous humour protection against UV-A and UV-B. Exp Eye Res 1996;62:261–4. [DOI] [PubMed] [Google Scholar]
- 16.Roberts LJ, Morrow JD. Measurement of F(2)-isoprostanes as an index of oxidative stress in vivo. Free Radic Biol Med 2000;28:505–13. [DOI] [PubMed] [Google Scholar]
- 17.Morrow JD. The isoprostanes: their quantification as an index of oxidant stress status in vivo. Drug Metab Rev 2000;32:377–85. [DOI] [PubMed] [Google Scholar]
- 18.Morrow JD, Awad JA, Boss HJ, et al. Non-cyclooxygenase-derived prostanoids (F2-isoprostanes) are formed in situ on phospholipids. Proc Natl Acad Sci USA 1992;89:10721–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Fukunaga M, Makita N, Roberts LJ, et al. Evidence for the existence of F2-isoprostane receptors on rat vascular smooth muscle cells. Am J Physiol 1993;264:C1619–24. [DOI] [PubMed] [Google Scholar]
- 20.Prince AM, Streeten BW, Ritch R, et al. Preclinical diagnosis of pseudoexfoliation syndrome. Arch Ophthalmol 1987;105:1076–82. [DOI] [PubMed] [Google Scholar]
- 21.Shichi H, Page T, Sahouri MJ, et al. Microplate assay of ascorbic acid in aqueous humor with bicinchoninic acid. J Ocul Pharmacol Ther 1997;13:201–6. [DOI] [PubMed] [Google Scholar]
- 22.Koliakos GG, Konstas AG, Schlotzer-Schrehardt U, et al. Ascorbic acid concentration is reduced in the aqueous humor of patients with exfoliation syndrome. Am J Ophthalmol 2002; (in press). [DOI] [PubMed]
- 23.Kuchle M, Ho TS, Nguyen NX, et al. Protein quantification and electrophoresis in aqueous humor of pseudoexfoliation eyes. Invest Ophthalmol Vis Sci 1994;35:748–52. [PubMed] [Google Scholar]
- 24.Roberts LJ, Morrow JD. The isoprostanes: novel markers of lipid peroxidation and potential mediators of oxidant injury. Adv Prostaglandin Thromboxane Leukot Res 1995;23:219–24. [PubMed] [Google Scholar]
- 25.Pratico D, FitzGerald GA. Generation of 8-epiprostaglandin F2alpha by human monocytes. Discriminate production by reactive oxygen species and prostaglandin endoperoxide synthase-2. J Biol Chem 1996;271:8919–24. [DOI] [PubMed] [Google Scholar]
- 26.Vanharanta M, Voutilainen S, Nurmi T, et al. Association between low serum enterolactone and increased plasma F(2)-isoprostanes, a measure of lipid peroxidation. Atherosclerosis 2002;160:465–9. [DOI] [PubMed] [Google Scholar]
- 27.Morrow JD, Roberts LJ. Mass spectrometric quantification of F2-isoprostanes in biological fluids and tissues as measure of oxidant stress. Methods Enzymol 1999;300:3–12. [DOI] [PubMed] [Google Scholar]
- 28.Pratico D, Barry OP, Lawson JA, et al. IPF2alpha-I: an index of lipid peroxidation in humans. Proc Natl Acad Sci USA 1998;95:3449–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hoffman SW, Moore S, Ellis EF. Isoprostanes: free radical-generated prostaglandins with constrictor effects on cerebral arterioles. Stroke 1997;28:844–9. [DOI] [PubMed] [Google Scholar]
- 30.Yura T, Fukunaga M, Khan R, et al. Free-radical-generated F2-isoprostane stimulates cell proliferation and endothelin-1 expression on endothelial cells. Kidney Int 1999;56:471–8. [DOI] [PubMed] [Google Scholar]
- 31.Lahaie I, Hardy P, Hou X, et al. A novel mechanism for vasoconstrictor action of 8-isoprostaglandin F2 alpha on retinal vessels. Am J Physiol 1998;274:R1406–16. [DOI] [PubMed] [Google Scholar]
- 32.Berger TM, Polidori MC, Dabbagh A, et al. Antioxidant activity of vitamin C in iron-overloaded human plasma. J Biol Chem 1997;272:15656–60. [DOI] [PubMed] [Google Scholar]
- 33.Haas JA, Krier JD, Bolterman RJ, et al. Low-dose angiotensin II increases free isoprostane levels in plasma. Hypertension 1999;34:983–6. [DOI] [PubMed] [Google Scholar]
- 34.Rose RC, Richer SP, Bode AM. Ocular oxidants and antioxidant protection. Proc Soc Exp Biol Med 1998;217:397–407. [DOI] [PubMed] [Google Scholar]
- 35.Ringvold A. Quenching of UV-induced fluorescence by ascorbic acid in the aqueous humour. Acta Ophthalmol Scand 1995;73:529–33. [DOI] [PubMed] [Google Scholar]
- 36.Richer SP, Rose RC. Water soluble antioxidants in mammalian aqueous humor: interaction with UV B and hydrogen peroxide. Vis Res 1998;38:2881–8. [DOI] [PubMed] [Google Scholar]
- 37.Forsius H. Exfoliation syndrome in various ethnic populations. Acta Ophthalmol Suppl 1988;184:71–85. [DOI] [PubMed] [Google Scholar]
- 38.Montero A, Munger KA, Khan RZ, et al. F(2)-isoprostanes mediate high glucose-induced TGF-beta synthesis and glomerular proteinuria in experimental type I diabetes. Kidney Int 2000;58:1963–72. [DOI] [PubMed] [Google Scholar]
- 39.Staff AC, Ranheim T, Henriksen T, et al. 8-Iso-prostaglandin f(2alpha) reduces trophoblast invasion and matrix metalloproteinase activity. Hypertension 2000;35:1307–13. [DOI] [PubMed] [Google Scholar]
- 40.Parola M, Robino G. Oxidative stress-related molecules and liver fibrosis. J Hepatol 2001;35:297–306. [DOI] [PubMed] [Google Scholar]
- 41.Kahler J, Ewert A, Weckmuller J, et al. Oxidative stress increases endothelin-1 synthesis in human coronary artery smooth muscle cells. J Cardiovasc Pharmacol 2001;38:49–57. [DOI] [PubMed] [Google Scholar]
- 42.Shimizu T. Changes of iris vessels in capsular glaucoma: three-dimensional and electron microscopic studies. Jpn J Ophthalmol 1985;29:434–52. [PubMed] [Google Scholar]
- 43.Vannas A. Flurescein angiography of the vessels of the iris in pseudoexfoliation of the lens capsule, capsular glaucoma and some other forms of glaucoma. Acta Ophthalmol Suppl 1969;105:9–75. [PubMed] [Google Scholar]
- 44.Konstas AG, Marshall GE, Lee WR. Iris vasculopathy in exfoliation syndrome. An immunocytochemical study. Acta Ophthalmol (Copenh)1991;69:472–83. [DOI] [PubMed] [Google Scholar]
- 45.Konstas AG, Marshall GE, Cameron SA, et al. Morphology of iris vasculopathy in exfoliation glaucoma. Acta Ophthalmol (Copenh)1993;71:751–9. [DOI] [PubMed] [Google Scholar]