Abstract
Soluble low-affinity receptors for IgG are known to inhibit immune complex (IC)-mediated inflammation, and expression by leukocytes is elevated in several inflammatory diseases. Immunoglobulin M (IgM) rheumatoid factors (RF), anti-Fc autoantibodies, are found in autoimmune diseases, such as rheumatoid arthritis (RA), as well as in normal immune responses. This study demonstrated that soluble FcγRIIa inhibits the interaction of rheumatoid factors with ICs. The recombinant soluble low-affinity FcγR, rsFcγRIIa, partially inhibited (30–70%) the rate of precipitation of soluble ICs by RF-positive RA sera. This required the normal interaction of FcγRIIa with Fc as the effect could be abrogated with the Fab fragment of the blocking mAb IV-3. Furthermore, rsFcγRIIa partially inhibited (40%) the binding of a monoclonal IgM RF (RF-AN) to an IC formed by IgG2 antibody binding to an antigen-coated biosensor chip. Since RF-AN has been characterized by crystallography to bind to the CH2/CH3 interface of the IgG-Fc, and leukocyte FcγRIIa binds to a distinct site centred on the lower hinge, this inhibition is uncompetitive. Some inhibition (15%) of staphylococcal protein A binding to IC was also observed. As soluble FcγRIIa disrupts Fc:Fc interactions in IgG-ICs, we propose that this alteration of the IC also reduces the accessibility of Fc portions in the IC, resulting in the partial inhibition of ligands, particularly IgM RF, which bind Fc. We propose that the high concentrations of soluble FcγR found during inflammation can affect the properties of ICs and their interaction with the immune system.
Introduction
FcγRI, FcγRII and FcγRIII are cell surface receptors expressed on various leukocytes specifically binding immunoglobulin (Ig) G. IgG immune complexes (ICs) crosslink these FcγRs and activate leukocytes inducing effector functions, such as respiratory burst, cytokine secretion, antibody-dependent cellular cytotoxicity (ADCC) and phagocytosis.1–6 These cell surface FcγRs have a well-defined role in antibody-induced inflammation and clearance of antigen–antibody complexes.7–11 Recent studies using mice deficient in various FcγRs have demonstrated an important role for FcγRs in the pathology of rheumatoid arthritis (RA). In both methylated bovine serum albumin (BSA)-induced arthritis and collagen-induced arthritis models, mice deficient in the activating receptor FcγRI were resistant to matrix degradation, while mice deficient in FcγRIII had reduced inflammation. Deficiency in the inhibitory mouse receptor FcγRII increased disease susceptibility in arthritis models, with effects in both joint inflammation and destruction.12–17 Furthermore, a transgenic model of RA, in which disease can be induced by transfer of antiglucose-6-phosphate isomerase antibody, shows a dependence on FcγRIII and complement.18
While the biology of human low-affinity FcγRs, i.e. FcγRII and FcγRIII, as cell surface receptors is well known, these receptors also exist in soluble forms, the physiological functions of which are not fully characterized.19–21 Soluble forms of FcγRIIa can result from either the shedding of the transmembrane receptor or expression of alternatively spliced mRNA,22,23 while soluble FcγRIII is shed by proteolysis.24 The production of soluble FcγRs occurs following the activation of neutrophils, natural killer (NK) cells, Langerhans cells and platelets. Consequently elevated levels of soluble receptors are found in various scenarios of cellular immunity or disease states, including RA, where high levels of soluble FcγR occur, but their role in disease in uncertain.25–27 A protective role is possible as soluble FcγRIIa inhibits inflammation in a reverse passive Arthus model.10 Soluble FcγR inhibits inflammation both by blocking immune complexes from binding membrane receptors and by inhibiting Fc:Fc-mediated immune precipitation.28
In RA, an autoimmune disease of the synovial membrane, the synovial fluid of an affected joint is a milieu containing proinflammatory cytokines, monokines, chemokines29–31 and, in most cases, rheumatoid factors (RFs). RFs are IgM, IgG and IgA autoantibodies directed against the Fc region of IgG.32 Although IgM RF is found at low levels in healthy individuals, high-titre IgM RF in RA is an indicator of accelerated disease progression and greater likelihood of systemic features such as vasculitis and other IC-related features.33,34 The role of RFs in RA is not well understood, although they probably contribute to pathology by participating in forming ICs by crosslinking IgG.35 This crosslinking by RFs of ICs bound to NK cell FcRs has been reported to enhance cellular activation.36 This in vitro study examined the interplay between soluble low-affinity FcγR and RFs and found recombinant soluble FcγRIIa (rsFcγRIIa) inhibited the binding of RFs to ICs.
Materials and methods
Preparation of IgG complexes (ICs) and RF
Heat-aggregated IgG (HAGG) was prepared from Sandoglobulin (Sandoz, Novartis Pharmaceuticals Co, East Hanover, NJ), comprising ≥96% IgG and traces of IgA and IgM, using two different methods for use in either an enzyme-linked immunosorbent assay (ELISA) or turbidimetric and biosensor assays. Firstly, for the ELISA, the Sandoglobulin, at 30 mg/ml in phosphate-buffered saline (PBS), was heated for 30 min at 63° and centrifuged for 5 min at 10 000 g and 4°, and the supernatant was incubated on ice for 30 min with 1% (w/v) polyethylene glycol 6000 (PEG 6000, Sigma Chemical Co, St. Louis, MO) in PBS. The precipitated complexes were collected by centrifugation at 10 000 g and 4° for 10 min, dissolved in PBS and centrifuged at 10 000 g and 4° for 10 min, and the supernatant was used to coat ELISA plates. Secondly, for use in the turbidimetric and biosensor assays, the Sandoglobulin, at 30 mg/ml in PBS, was heated for 30 min at 63° and centrifuged for 5 min at 10 000 g and 4°, and the supernatant was fractionated by gel filtration chromatography on Superdex 200 (Amersham Pharmacia Biotech, Melbourne, Australia). The soluble IgG aggregates used in the assays eluted at the void volume of the column, and were thus separated from the monomeric IgG. IgM was purified from sera by euglobin precipitation37 and chromatography on Superdex 200. IgM was purified from two RA patients, BEA and PIK, and from a normal donor, WIN. Sera from patients BEA, BRU, COT, ELL, GRI, HEY, MIE, MOL, PIK, RIT and TOG for use in assays was heat-inactivated by treatment at 56° for 15 min (see Table 1). Heat inactivation (56° for 15 min) had only a small effect on RF activity as the treatment of the whole serum TOG, or purified RF-AN, resulted in retention of > 90% of the HAGG binding activity in a biosensor assay (data not shown).
Table 1.
Inhibition of RF-mediated IC precipitation by rsFcγRIIa
| RA patient | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| BRU | COT | ELL | GRI | HEY | MIE | MOL | PIK | RIT | TOG | |||
| % inhibition | 49 | 40 | 48 | 69 | 38 | 22 | 42 | 52 | 54 | 41 | 27 | |
Values are the per cent inhibition of immune complex precipitation by 10 µl of heat-inactivated RA patient serum at 5 min in the presence of 7·6 µm rsFcγRIIa.
Recombinant soluble FcγRIIa was produced in CHO-K1 cells and purified as described previously.10 IgM RF was labelled with horseradish peroxidase (HRP, Sigma) as described previously.38 HRP-labelled protein A was purchased from Sigma.
Turbidimetric assay for RF precipitation of HAGG
The immune complex precipitating activity of RF was measured using a turbidimetric assay where heat-inactivated RA patient serum (10 µl) and HAGG (100 µg/ml, 0·7 µm) were incubated together in a volume of 400 µl and the increased light scattering at 350 nm was recorded. Subsequent experiments used sera stored in 40% v/v glycerol at − 20°. The use of the Fab fragment of the anti-FcγRIIa mAb IV-3 to block FcγRIIa binding to the Fc has been described previously.28 The inactivated serum (10 µl, stored at 40% v/v glycerol) and HAGG (100 µg/ml, 0·7 µm) were incubated together with 0·7 µm rsFcγRIIa in the presence or absence of 1·4 µm IV-3 Fab, and IC precipitation was measured.
ELISA analysis of inhibition of RF binding by FcγRIIa
The binding activity of HRP-labelled IgM RF was measured in an ELISA using HAGG-coated 96-well plates. ELISA plates (Serocluster U vinyl, Costar, Cambridge, MA) were incubated with 50 µl PEG-fractionated HAGG (25 µg/ml in 50 mm carbonate/bicarbonate pH 9·6) for 16 hr and blocked with 120 µl of 1% BSA (Commonwealth Serum Laboratories, Melbourne, Australia) in PBS for 1 hr at 37°.
Inhibition of IgM RF binding by purified rsFcγRIIA was achieved by incubating HAGG-coated plates with 0·5% BSA in PBS containing rsFcγRII, in the concentration range of 0·1 to 10 µm, for 60 min at 37°. HRP-labelled IgM RF was added for 40 min, before washing with PBS and detection with 2,2′azino-bis(3–ethylbenzthiazoline–6–sulfonic acid) (ABTS) substrate (Boehringer Mannheim, Nunawading, Australia). HRP-labelled IgMs RF-BEA and PIK were used at concentrations of ∼ 1 µg/ml. To test for inhibition of protein A binding to HAGG by rsFcγRII, the same protocol was followed, except HRP-labelled protein A (Sigma) was used at 1 : 12 000.
Biosensor analysis of inhibition of RF binding to HAGG by FcγRIIa
HAGG (50 µg/ml) in 50 mm acetate, pH 5·0, was coupled (1000 RU) to a CM5 biosensor chip (BIAcore, Melbourne, Australia) using the manufacturer's instructions for carbodiimide chemistry and binding recorded for the separate injections of PIK IgM RF (3 µm) and rsFcγRIIa (2 µm). Combining PIK IgM RF (3 µm) and rsFcγRIIa (2 µm) together in a single injection onto the immobilized HAGG was used to measure the effect of rsFcγRIIa on RF binding.
Preparation of human monoclonal IgM RF-AN
The procedure for purification of RF was based on the non-solubility of IgM in water.39 RF was prepared from the tissue culture supernatant derived from the Epstein-Barr virus (EBV)-immortalized RF-AN cell line.40 Cells were grown in RPMI-1640 supplemented with 10% fetal calf serum (FCS) to a final density of 106 cells/ml. The cell supernatant was mixed at 4° with an equal volume of saturated ammonium sulfate and left overnight at 4°. The precipitate was washed twice with cold 50% saturated ammonium sulfate, then dissolved in PBS at 1/40 the original volume of the cell supernatant and dialysed extensively against PBS. The material in the dialysis tube was spun for 1 hr at 10 000 g and the fluid was then dialysed against H2O. The delicate precipitate which was formed under these conditions was collected by centrifugation as above and dissolved finally in 1/200 the original volume of the cell supernatant. The concentration of protein was 300 µg/ml as determined by Bradford, with BSA as standard.
Biosensor analysis of inhibition of RF-AN binding to IgG2:NP by FcγRIIa
Diaminopropane (Sigma) at 11 mm in water was coupled to a CM5 biosensor chip (BIAcore) using the manufacturer's instructions for carbodiimide chemistry. The chip was then reacted with 3-nitro-4-hydroxyphenylacetic acid (NP-OSu, Genosys, Cambridgeshire, UK). Chimeric IgG2 anti-NP with human constant domains (Serotec, Oxford, UK) was injected (12 µg/ml) onto the biosensor (20 µl, 10 µl/min, in 20 mm sodium phosphate, 150 mm NaCl, 2 mm EDTA, 0·05% surfactant p20) over a NP coupled chip and binding recorded. An injection of RF-AN (20 µl, 11 nm) was performed and binding to the IgG2 recorded. The chip was regenerated to remove bound IgG2 and RF. IgG2 was injected as previously and a second injection of buffer alone was carried out to provide the binding response of the IgG2 alone. RF-AN binding to the IgG2 was measured by the increased response of the RF-AN sensogram above that of the IgG2 alone. This measure of RF-AN binding activity was taken at the arbitrary endpoint of 14 min (total run time). Following regeneration of bound protein from the chip, other interactions with bound IgG2 were measured. These were: the injection of rsFcγRIIa (20 µl, 10 µm), a mixture of rsFcγRIIa (10 µm) and RF-AN (11 nm), and a mixture of heat-aggregated IgG (200 µg/ml) and RF-AN (11 nm). Non-specific binding was evaluated by simultaneously passing injected material over a biosensor channel containing bound chimeric IgE anti-NP (Serotec) (12 µg/ml, 20 µl). Small transient non-specific responses on the IgE channel of 11 and 29 RU were recorded during the injection of the RF-AN and rsFcγRIIa samples.
Biosensor analysis of inhibition of recombinant protein A binding to IgG2:NP by FcγRIIa
Chimeric IgG2 anti-NP (Serotec) was injected (12 µg/ml) onto the biosensor (20 µl, 10 µl/min) as previously described and the binding of recombinant staphylococcal protein A (20 µl, 11 nm, from Calbiochem, Castle Hill, Australia) was recorded. The protein A was mixed with rsFcγRIIa or HAGG to determine the inhibition of binding as described above.
Results
Soluble FcγRIIa inhibits IC precipitation mediated by IgM RF-positive sera
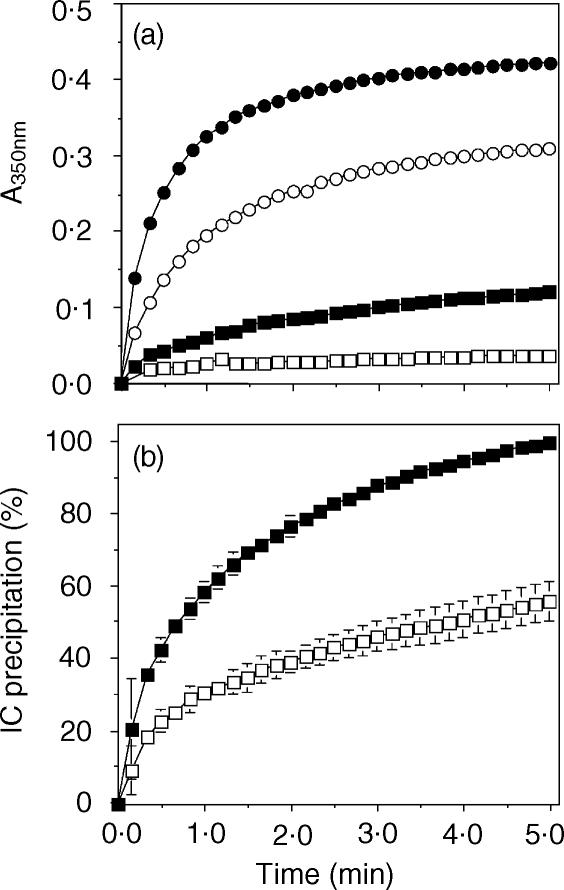
IC precipitation by IgM RF-positive sera from RA patients was demonstrated in a turbidimetric assay. The IC precipitating activity of all the RF-positive RA sera tested (11 patients) was inhibited by rsFcγRIIa (Table 1). The activity of the RF-positive sera affected the extent of inhibition of precipitation by rsFcγRIIa (Fig. 1a). A serum with a low IC precipitating activity, from patient ELL, for which IC precipitation reached a light-scattering value (A350nm) of 0·12 at 5 min, was inhibited 69% in the presence of 7·6 µm rsFcγRII. A serum with high IC precipitating activity, from patient TOG (A350nm = 0·42 at 5 min), was inhibited by 27% in the presence of 7·6 µm rsFcγRII. The inhibited rate of IC precipitation by the serum TOG in the presence of rsFcγRIIa was evident only in the first 30 s of the incubation. Four different patient sera (from BEA, BRU, COT and GRI) of approximately equivalent IC precipitating activity were tested, and the kinetics of the inhibition by rsFcγRIIa were similar. At the assay endpoint, the IC precipitation by each serum was inhibited by 44 ± 5% in the presence of rsFcγRIIa (Fig. 1b).
Figure 1.
rsFcγRIIa inhibits RF-mediated precipitation of immune complexes. (a) HAGG (100 µg/ml, 0·7 µm) was incubated with RF-positive serum ELL (squares) or with RF-positive serum TOG (circles) in the absence (filled symbols) or presence (open symbols) of 7·6 µm rsFcγRIIa and complex formation measured by A350nm. (b) Four different RA-positive sera of equivalent RF activity were incubated with HAGG in the absence (filled squares) or presence (open squares) of 7·6 µm rsFcγRIIa as above. The data are presented as the per cent IC precipitation averaged for the four samples ± SD.
Inhibition of IgM RF-mediated IC precipitation is rsFcγRIIa concentration-dependent
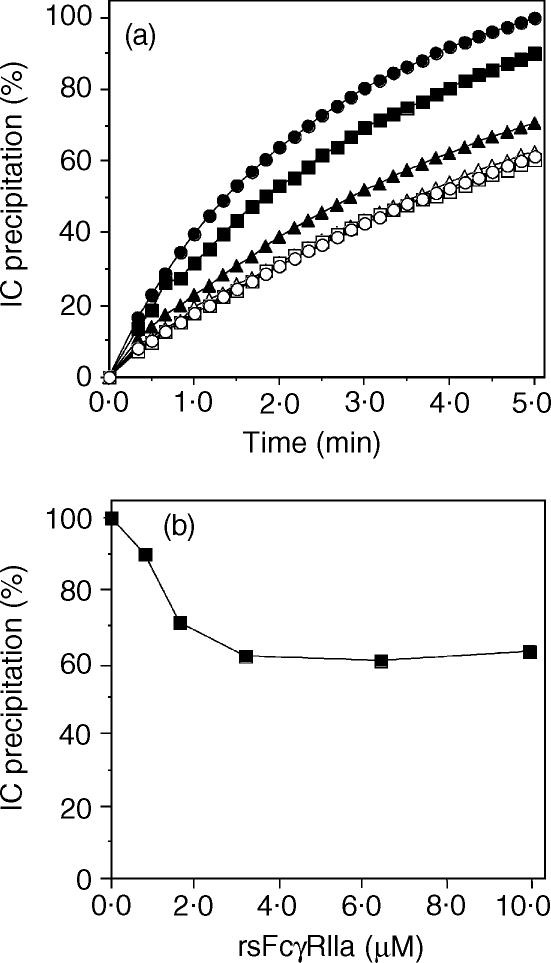
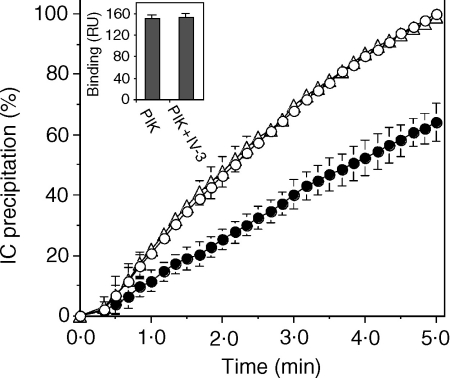
The dependence of the inhibition of RF-mediated IC precipitation on rsFcγRIIa concentration in the range of 0·8–10 µm was examined using the serum from a RA patient, BEA. RsFcγRIIa up to a concentration of 3·3 µm gave a dose-dependent inhibition of HAGG (0·7 µm) precipitation (Fig. 2a). This inhibition was partial, 10% at 0·8 µm, 29% at 1·7 µm and 39% at 3·3 µm rsFcγRIIa, and most pronounced at the early time-points. Inhibition reached a maximum at 3·3 µm, and higher concentrations did not result in any further inhibition of IC precipitation (10 µm rsFcγRIIa resulted in 37% inhibition). In a separate experiment, a greater, but still partial, inhibition of 49% of serum BEA-mediated IC precipitation was observed with 7·6 µm rsFcγRIIa (Table 1). This difference reflects interassay variation (± ∼10%). The partial inhibition is illustrated by plotting the precipitation endpoints against rsFcγRIIa concentration (Fig. 2b). The inhibition by rsFcγRIIa had a IC50 of ∼1 µm, the same concentration as the equilibrium dissociation constant (KD) value for the FcγRII:IgG interaction. This correlation suggests the binding of rsFcγRIIa to IgG is necessary for the observed inhibition of RF activity. The Fab fragment of the mAb IV-3, which blocks the IgG binding site of FcγRIIa, was used to confirm that rsFcγRIIa inhibition of RF activity is mediated by its Fc binding activity. The precipitation of IC by the RA serum PIK in the presence of 0·7 µm rsFcγRIIa at 5 min was inhibited by 36%, i.e. it was 64 ± 6% of that reached in the absence of rsFcγRIIa (Fig. 3). The further addition of the Fab fragment of mAb IV-3 at 1·4 µm abrogated all inhibition, restoring the IC precipitation to 99 ± 3%. Thus the inhibitory effect of rsFcγRIIa on RF-mediated IC precipitation is mediated through its IgG binding activity. IV-3 is highly specific for FcγRIIa and has no effect itself on RF binding activity, as found in a biosensor assay where PIK-RF binding to immobilized HAGG (150 ± 7 RU) was unaffected by the presence of 0·6 µm mAb IV-3 (153 ± 35 RU, Fig. 3 insert).
Figure 2.
rsFcγRIIa inhibition of RF-mediated precipitation is dose dependent. (a) HAGG (100 µg/ml) was incubated with the RF-positive sera (BEA) at 1/40 dilution and complex formation measured in the presence of rsFcγRIIa concentrations of 0 (filled circles), 0·8 (filled squares), 1·7 (filled triangles), 3·3 (open circles), 6·6 (open squares) and 9·9 µm (open triangles). (b) The per cent IC precipitation at the endpoint (5 min) of each assay was plotted against rsFcγRIIa concentration to show that only 40% of the IC precipitation appears sensitive to inhibition by rsFcγRIIa.
Figure 3.
The Fab fragment of mAb IV-3 ablates inhibition by rsFcγRIIa of RF immune precipitation. HAGG (100 µg/ml, 0·7 µm) was incubated with a 1/40 dilution of the RF-positive serum PIK alone (open circles), or in the presence of rsFcγRIIa (0·7 µm, closed circles), or in the presence of both rsFcγRIIa and the IV-3 Fab (1·4 µm, open triangles). Values represent the average of four measurements. Insert: PIK serum at 1/40 dilution was bound to HAGG immobilized on a biosensor chip as described in ‘Materials and methods’ and shown in Fig. 5. In the presence of IV-3 mAb (0·6 µm), no effect on RF binding activity was seen (n = 9).
Soluble FcγRIIa inhibits IgM RF binding to HAGG
RsFcγRIIa may inhibit RF-mediated IC precipitation by inhibiting RF binding to the Fc regions of the IC. This possibility was investigated directly by measuring the binding of HRP-labelled IgM RF to HAGG. Two purified RF IgM fractions, BEA and PIK, were labelled with HRP and the binding to HAGG was measured by ELISA (Fig. 4). Dose-dependent partial inhibition of the binding of the labelled IgM RF to the immobilized IgG by rsFcγRIIa was observed. The binding of RF-BEA and RF-PIK was inhibited by 75% in the presence of 10 µm rsFcγRII. Like the IC precipitation assays, the concentration of rsFcγRIIa near its KD value, ∼1 µm, gave half-maximal inhibition of RF binding. In contrast, the binding of HRP-labelled protein A was only slightly inhibited over this same range of rsFcγRIIa concentrations.
Figure 4.
RsFcγRIIa inhibits the binding of HRP-labelled RF to HAGG. HRP-labelled IgM RF BEA (open circles), PIK (closed circles) or protein A (open triangles) was reacted with HAGG-coated ELISA plates as described in the ‘Materials and methods’ in the presence of the indicated concentrations of rsFcγRII. Normalized binding is expressed as a percentage of maximum binding in the absence of rsFcγRIIa. Values represent the average of duplicate measurements.
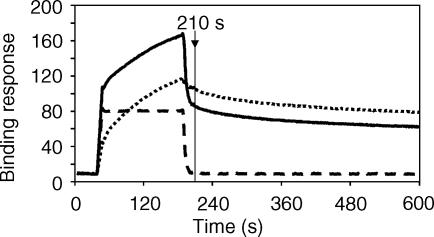
The inhibition of RF binding to IC was also measured using a biosensor assay. HAGG was coupled to a biosensor chip and purified IgM RF (patient PIK, dotted line in Fig. 5) at 500 µg/ml (∼500 nm) bound with 110 RU at an arbitrary endpoint near the commencement of the dissociation phase (dotted line at 210 s, Fig. 5). When the RF was mixed with 2 µm rsFcγRIIa (solid line, Fig. 5) and injected onto the biosensor surface, there was a small but consistent decrease of approximately 20% in RF binding with 85 RU of RF bound at the same time-point (solid line, 210 s). The rsFcγRIIa does not contribute to the binding response during the dissociation of the RF because its rapid off rate results in its complete dissociation from the chip long before the RF (dashed line). Thus, as observed previously, rsFcγRIIa partially inhibits RF binding to HAGG.
Figure 5.
Biosensor analysis of rsFcγRIIa inhibition of the binding of IgM RF to HAGG. HAGG (∼1000 RU) was coupled to a biosensor chip and the purified IgM (3 µm) from the RA patient PIK was injected (at 210 s, 110 RU bound, sensogram indicated by the dotted line). When the IgM RF preparation was combined with 2 µm rsFcγRIIa in a single injection (solid line), a small but consistent approximate 20% decrease in RF binding was seen at the 210-s time-point. An injection of rsFcγRIIa (2 µm) alone showed that this low-affinity receptor rapidly dissociated from the HAGG layer (dashed line).
Soluble FcγRIIa inhibits monoclonal IgM RF-AN binding to IgG2:NP immune complex
The mechanistic interpretation of how rsFcγRIIa inhibits RF binding to immune complexes is made difficult by the multivalent nature of the IgM RF and the IC interaction. For example, it is not possible to assume that all the Fc portions accessible for monovalent rsFcγRIIa binding are also equally accessible for IgM RF binding. None the less, some difficulties can be overcome by using defined monoclonal reagents. RA sera may contain RFs of different epitope specificities, and HAGG formed by partial heat denaturation of IgG may not display some properties of an antigen:antibody IC. Thus the inhibition of binding of the IgM monoclonal RF-AN to an immune complex formed by the binding of recombinant IgG2 to a haptenylated surface was investigated.
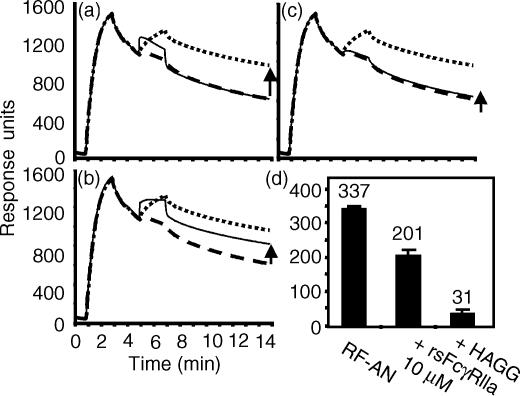
The chimeric IgG2 was reacted with a NP coupled biosensor chip to form an immune complex on the chip (∼1500 RU) and after this injection, as the IgG2 began to dissociate from the NP, RF-AN (20 µl, 11 µm) was injected (Fig. 6a–c, dotted line). At an arbitrary endpoint (14 min total run time), the binding response for this IgG2:RF-AN sensogram was 327 RU above the sensogram for IgG2 binding alone (Fig. 6a–c, dashed line). This measure of RF-AN binding (indicated by the arrow in Fig. 6a) has contributions from the effects of both RF binding and crosslinking of IgG2, thus slowing its dissociation from the NP layer. In contrast, because of its rapid dissociation, the injection of rsFcγRIIa (10 µm, Fig. 6a, thin solid line) over the IgG2 complex resulted in a transient binding response, with the sensogram rapidly returning to the signal produced by IgG2 alone.
Figure 6.
RsFcγRIIa inhibits RF-AN binding to immobilized chimeric IgG2. (a, b, c) Chimeric IgG2 anti-NP with human constant domains (12 µg/ml, 80 nm) was injected onto the biosensor (20 µl, 10 µl/min, in 20 mm sodium phosphate, 150 mm NaCl, 2 mm EDTA, 0·05% surfactant p20) over a NP coupled CM5 biosensor chip and binding recorded as response units. An injection of RF-AN (20 µl, 10 µg/ml, 11 nm) was carried out and binding to the IgG2 recorded (dotted line). The chip was regenerated and IgG2 injected as previously, and a second injection of buffer alone carried out to show any non-specific effects and the dissociation of the IgG2 from the chip (dashed line). The binding response of the RF-AN injection above that of the IgG2 injected alone (marked with an arrow) indicates RF-AN binding activity. Following regeneration, other test injections to show binding to the IgG2 layer were carried out and are shown as thin lines. These are the injection of (a) rsFcγRIIa (20 µl, 10 µm), (b) a mixture of rsFcγRIIa (10 µm) and RF-AN (11 nm) and (c) a mixture of heat-aggregated IgG (200 µg/ml, 1·3 µm) and RF-AN (11 nm). Non-specific binding was evaluated by simultaneously passing injected samples over a biosensor channel containing bound chimeric IgE anti-NP. Transient responses on the IgE layer of 11 and 29 RU were recorded during the injection of the RF-AN and rsFcγRIIa samples. (d) Shows the averaged RF binding activity (i.e. the binding response above IgG2 alone) measured at 14 min in the absence of an inhibitor, in the presence of 10 µm rsFcγRIIa and in the presence of 1·3 µm HAGG (average ± SD, n = 4).
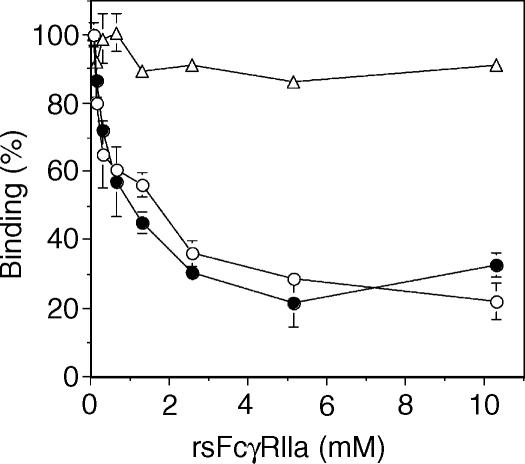
However, when a mixture of rsFcγRIIa (10 µm) and RF-AN (11 µm) was injected over the preformed IgG2 IC, the response (Fig. 6b, thin solid line) was initially greater than that of RF-AN alone (dotted line), as a result of the additional transient rsFcγRIIa binding. Then, by completion of the injection, the sensogram fell below that of the RF-AN alone. Thus the binding of RF-AN to the IgG2:NP complex (indicated by the arrow in Fig. 6b) is, after binding in the presence of rsFcγRIIa, only 185 RU above the sensogram for IgG2 binding alone (dashed line). The data from four independent experiments showed the RF-AN binding response (337 ± 11 RU) was inhibited by 40% to 201 ± 22 RU in the presence of 10 µm rsFcγRIIa (Fig. 6d). In contrast, direct competition with HAGG (1·3 µm in IgG) inhibited RF-AN binding by 91%.
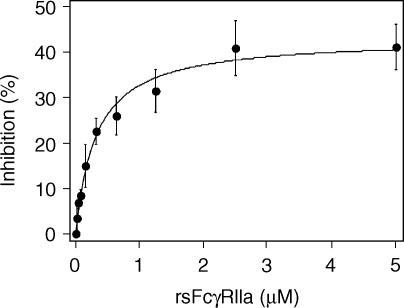
Next, the concentration dependence of the inhibition of RF-AN activity by rsFcγRIIa in this system was tested (Fig. 7). As seen with the polyclonal RF preparations, a rsFcγRIIa concentration-dependent titration of RF inhibition was demonstrated with a half-maximal inhibition reached at ∼0·3 µm rsFcγRIIa. Again, inhibition was partial and approached a maximum of 43%.
Figure 7.
RsFcγRIIa inhibition of RF-AN binding activity is concentration dependent. Chimeric IgG2 was reacted with a NP coupled biosensor chip and rsFcγRIIa was used at the indicated concentrations as an inhibitor of RF-AN binding as described for Figure 6 and in the ‘Materials and methods’.
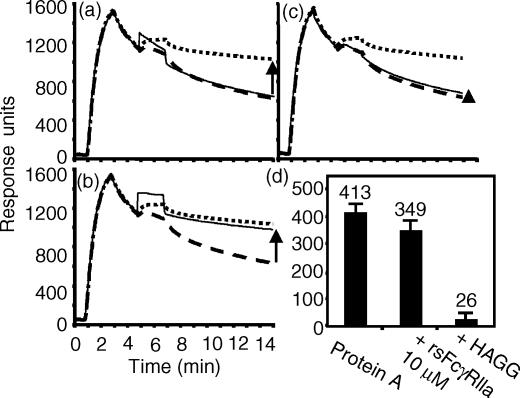
The specificity of the rsFcγRIIa-mediated inhibition of RF binding to the CH2/CH3 interface was tested using recombinant staphylococcal protein A binding to the IgG2:NP complex. The binding of protein A was near maximal at 450 nm and was used at 11 nm, where ∼70% of the maximum binding response was observed (data not shown). The inhibition of protein A binding by rsFcγRIIa was tested using the same experimental protocol as described for Fig. 6, but replacing RF-AN with protein A. Data from a typical experiment are shown in Fig. 8(a–c), with the binding of the rsProtein A being 447 RU above that of the IgG2 alone (indicated by the arrow, Fig. 8a). rsProtein A binding was slightly inhibited (368 RU) in the presence of 10 µm rsFcγRIIa (Fig. 8b, thin solid line) and strongly inhibited (12 RU) in the presence of soluble HAGG (Fig. 8c, thin solid line). Averaged data show the injection of protein A alone gave a binding endpoint of 413 ± 33 (n = 9), while the injection of a mixture containing protein A (11 nm) and rsFcγRIIa (10 µm) gave a protein A binding endpoint that was reduced to 349 ± 32 RU (Fig. 8d). The average inhibition of protein A binding was 15 ± 4% (n = 9). In contrast, direct competition with HAGG (1·3 µm in IgG) inhibited protein A binding by 94%. Thus, while the inhibition of RF binding was more substantial, there was also a small but reproducible inhibition of protein A binding to the Fc regions of ICs by rsFcγRIIa.
Figure 8.
There is little inhibition by rsFcγRIIa of protein A binding to immobilized IgG2. The inhibition of protein A (11 nm) binding to IgG2 by rsFcγRIIa was assessed using the conditions described in Fig. 6. IgG2 was bound to the NP coupled chip (20 µl, 10 ml/min) and ∼4 min later rsProtein A (20 µl, 11 nm) was injected (thick dotted lines) or buffer alone (thick dashed lines) was injected. The binding response of the IgG2 and rsProtein A injection above that of the IgG2 and buffer injection (marked with an arrow) indicates protein A binding activity. Following regeneration, other injections to test binding to the IgG2 layer were carried out and are shown as thin lines. These were the injection of (a) rsFcγRIIa (20 µl, 10 µm), (b) a mixture of rsFcγRIIa (10 µm) and rsProtein A (11 nm), and (c) a mixture of heat-aggregated IgG (200 µg/ml, 1·3 µm) and rsProtein A (11 nm). (d) The average rsProtein A binding activity measured at 14 min in the absence of an inhibitor, in the presence of 10 µm rsFcγRIIa, and in the presence of 1·3 µm HAGG is shown (average ± SD, n = 9).
Discussion
Fc receptors have been shown in various systems2,3,6,8,9 to play pivotal roles in antibody-mediated inflammatory responses, and more recently have demonstrated unexpected importance in a number of arthritis models (reviewed in 16). The interaction of IC with particular FcγRs, in concert with other factors, influences aspects of the disease including joint inflammation, matrix and cartilage destruction, metalloprotease activation and proinflammatory cytokine secretion.12–18,41,42 In contrast, despite the observation of elevated levels in some chronic inflammatory diseases, the function of soluble low-affinity FcγRs in antibody-mediated inflammatory responses is not well elucidated. The potential importance of soluble FcγRs as immune modulators is underscored by the ability of administered rsFcγRIIa to reduce a reverse passive Arthus interaction in the rat.10 The inhibitory activities of soluble FcRs include simple blockade of cellular FcRs and the inhibition of IC formation by disruption of Fc:Fc interactions.28
This study investigated whether a low-affinity soluble FcγR, recombinant soluble FcγRIIa, could modulate binding of RF to the Fc regions of preformed aggregates of normal human IgG, or authentic immune complexes of human IgG2 and NP on a biosensor chip. The results herein showed that a rsFcγRIIa inhibited the binding of polyclonal IgM RF and monoclonal IgM RF-AN43 to IC and consequently also inhibited the precipitation of IC mediated by polyclonal IgM RF. This inhibition operated through the normal binding of rsFcγRIIa to the Fc region of IgG, as the Fab fragment of the blocking anti-FcγRIIa mAb IV-3 abrogated this effect. Furthermore, the half-maximal inhibition of RF-mediated IC precipitation (or RF binding to IgG-IC) was observed at concentrations of rsFcγRIIa approximating the micromolar KD for the interaction of rsFcγRII with IgG. This is consistent with IgG binding by the rsFcγRIIa mediating the inhibition of RF binding as, at the concentration of rsFcγRIIa approximately equal to its µM KD, half of the IgG molecules should be bound by the soluble receptor. Although rsFcγRIIa was always titrated to an excess (10 µm, ∼10 times KD) over the IC (e.g. ∼10 femtomol/mm2 on the biosensor chip in Fig. 7) to which it bound, the inhibition of RF binding to the IC was always only partial. This partial inhibition of RF activity supports the conclusion that inhibition of IgM RF binding by rsFcγRIIa is uncompetitive. The reciprocal experiment, where IgM RF was tested as an inhibitor of rsFcγRIIa binding, was uninformative. The main difficulty is that it is unlikely that all Fc regions in an IC, whether formed by aggregation or antigen binding, will be equal in availability for binding to both monovalent rsFcγRIIa (22 kDa) and pentavalent IgM RF (900 kDa).
The conclusion that the inhibition of the RF by rsFcγRIIa is not brought about through simple competition for binding to the Fc is supported by the location of binding sites within the Fc. The leukocyte FcγRs binding sites and the canonical RF binding site are in distinct regions in the Fc. Mutational analysis of IgG has shown that leukocyte FcγR binding activity resides in the lower hinge and upper CH2 domains of the antibody.1,44–50 Recently, the solved structure of the complex between rsFcγRIII and IgG1 Fc has been elucidated at the atomic level.44 RsFcγRIII and rsFcγRIIa consist of two extracellular domains with 50% amino acid identity and are likely to interact in a similar manner with the Fc.51
RF and protein A bind elsewhere on the Fc at overlapping sites at the CH2/CH3 interface. In fact, bacterial FcRs compete with RF for binding to the Fc.52,53 The X-ray structure of a complex of rheumatoid factor Fab (RF-AN) with an IgG4-Fc has provided precise molecular detail for the interaction of this RF with the interface between the CH2 and CH3 domains of IgG.43,54,55 The RF-AN Fab bound to the CH2/CH3 domain interface orientated at about 45° to the Fc and pointing away from the CH2 domain. The bound RF Fab is thus unlikely to interfere sterically with soluble FcγR binding to the upper CH2 domain. Therefore, the inhibition of RF-AN binding by soluble FcγRIIa probably results from an uncompetitive mechanism, as the separation of the sites for RF-AN and FcγR binding in the Fc precludes simple competition for binding.
Interactions between Fc portions in an IC are disrupted by rsFcγRIIa;28 thus one possible mechanism for the inhibition of RF binding is that disruption of Fc:Fc interactions eliminates some RF binding sites. That is, the Fc:Fc interactions between IgG molecules in an IC may be important in the presentation of clusters of Fc portions for interaction with multivalent effector molecules46 or IgM-RF. An alternative explanation may lie in a comprehensive mutational study of the human IgG1 Fc, which found that a few residues outside the crystallographic Fc:FcγRIII interface (e.g. Lys 414) none the less have a minor and unexplained role in IgG binding to FcγRII and FcγRIII.49
In accord with the interpretation that the soluble FcγR binding in some way reduces the accessibility of some of the Fc portions in the IC, there was a small inhibition of protein A binding to the IC by rsFcγRIIa. That the effect on protein A binding was much less than for RF could be a result of the higher affinity of protein A binding compared with the IgM RF, which requires multiple weak Fab:Fc interactions for binding.
An important issue in evaluating the possible physiological role of soluble FcγRs is whether they reach sufficient concentrations to have an effect at sites of inflammation. For soluble FcγRIIa, the inhibitory activities reported in vitro and in vivo appear to require micromolar concentrations. Such concentrations might appear not to be physiological, considering the reported normal systemic concentration of soluble FcγRIIa is approximately 0·3 nm.56 However, soluble FcγRIII may be a more effective physiological IC modulator, as this has been reported to reach systemic concentrations of 15 µm in healthy individuals and 90 µm in patients with RA.27 In the activated environment of the RA joint, soluble FcγRIII has been measured at concentrations of up to 28 µm.25 The KD for the FcγRIII:IgG interaction (KD∼ 0·1 µm1) is about 10-fold lower than that of FcγRIIa, which suggests this soluble FcR may inhibit IC precipitation at submicromolar concentrations. Thus, at the high concentrations measured in the synovial fluid, this soluble FcγRIII would be expected to be a potent inhibitor of IC formation28 and RF binding to these ICs. The low-affinity soluble FcγRs, in particular sFcγRIII, are therefore potentially significant players in mediating control of IC-initiated inflammation. These activities, controlling IC formation and binding by RF, may significantly affect IC-mediated leukocyte activation, IC clearance and associated pathology.
Acknowledgments
The authors wish to thank Dr Gary Jamieson for help with computing and data analysis and Sandra Esparon for expert technical assistance. The National Health and Medical Research Council provided grant support.
Abbreviations
- ADCC
antibody-dependent cellular cytotoxicity
- HAGG
heat-aggregated IgG
- HRP
horse radish peroxidase
- IC
immune complex
- KD
equilibrium dissociation constant
- RA
rheumatoid arthritis
- RF
rheumatoid factor
- rs
recombinant soluble
References
- 1.Hulett MD, Hogarth PM. Molecular basis of Fc receptor function. Adv Immunol. 1994;57:1–127. doi: 10.1016/s0065-2776(08)60671-9. [DOI] [PubMed] [Google Scholar]
- 2.Sylvestre DL, Ravetch JV. Fc receptors initiate the Arthus reaction: redefining the inflammatory cascade. Science. 1994;265:1095–8. doi: 10.1126/science.8066448. [DOI] [PubMed] [Google Scholar]
- 3.Ravetch JV. Fc receptors: rubor redux. Cell. 1994;78:553–60. doi: 10.1016/0092-8674(94)90521-5. [DOI] [PubMed] [Google Scholar]
- 4.Ravetch JV, Kinet JP. Fc receptors. Annu Rev Immunol. 1991;9:457–92. doi: 10.1146/annurev.iy.09.040191.002325. [DOI] [PubMed] [Google Scholar]
- 5.Bolland S, Ravetch JV. Spontaneous autoimmune disease in Fc (gamma) RIIB-deficient mice results from strain-specific epistasis. Immunity. 2000;13:277–85. doi: 10.1016/s1074-7613(00)00027-3. [DOI] [PubMed] [Google Scholar]
- 6.Ravetch JV. Fc receptors. Curr Opin Immunol. 1997;9:121–5. doi: 10.1016/s0952-7915(97)80168-9. [DOI] [PubMed] [Google Scholar]
- 7.Fossati-Jimack L, Reininger L, Chicheportiche Y, Clynes R, Ravetch JV, Honjo T, Izui S. High pathogenic potential of low-affinity autoantibodies in experimental autoimmune hemolytic anemia. J Exp Med. 1999;190:1689–96. doi: 10.1084/jem.190.11.1689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Clynes R, Dumitru C, Ravetch JV. Uncoupling of immune complex formation and kidney damage in autoimmune glomerulonephritis. Science. 1998;279:1052–4. doi: 10.1126/science.279.5353.1052. [DOI] [PubMed] [Google Scholar]
- 9.Clynes R, Maizes JS, Guinamard R, Ono M, Takai T, Ravetch JV. Modulation of immune complex-induced inflammation in vivo by the coordinate expression of activation and inhibitory Fc receptors. J Exp Med. 1999;189:179–85. doi: 10.1084/jem.189.1.179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ierino FL, Powell MS, McKenzie IF, Hogarth PM. Recombinant soluble human Fc gamma RII: production, characterization, and inhibition of the Arthus reaction. J Exp Med. 1993;178:1617–28. doi: 10.1084/jem.178.5.1617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Samuelsson A, Towers TL, Ravetch JV. Anti-inflammatory activity of IVIG mediated through the inhibitory Fc receptor. Science. 2001;291:484–6. doi: 10.1126/science.291.5503.484. [DOI] [PubMed] [Google Scholar]
- 12.Blom AB, van Lent PL, van Vuuren H, Holthuysen AE, Jacobs C, van de Putte LB, van de Winkel JG, van den Berg WB. Fc gamma R expression on macrophages is related to severity and chronicity of synovial inflammation and cartilage destruction during experimental immune-complex-mediated arthritis (ICA) Arthritis Res. 2000;2:489–503. doi: 10.1186/ar131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kleinau S, Martinsson P, Heyman B. Induction and suppression of collagen-induced arthritis is dependent on distinct fcgamma receptors. J Exp Med. 2000;191:1611–6. doi: 10.1084/jem.191.9.1611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.van Lent PL, van Vuuren AJ, Blom AB, Holthuysen AE, van de Putte LB, van de Winkel JG, van den Berg WB. Role of Fc receptor gamma chain in inflammation and cartilage damage during experimental antigen-induced arthritis. Arthritis Rheum. 2000;43:740–52. doi: 10.1002/1529-0131(200004)43:4<740::AID-ANR4>3.0.CO;2-0. [DOI] [PubMed] [Google Scholar]
- 15.van Lent PL, Nabbe K, Blom AB, Holthuysen AE, Sloetjes A, van de Putte LB, Verbeek S, van den Berg WB. Role of activatory Fc gamma RI and Fc gamma RIII and inhibitory Fc gamma RII in inflammation and cartilage destruction during experimental antigen-induced arthritis. Am J Pathol. 2001;159:2309–20. doi: 10.1016/s0002-9440(10)63081-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Van den Berg WB. Lessons from animal models of arthritis. Curr Rheumatol Rep. 2002;4:232–9. doi: 10.1007/s11926-002-0070-5. [DOI] [PubMed] [Google Scholar]
- 17.Ioan-Facsinay A, de Kimpe SJ, Hellwig SM, et al. FcgammaRI (CD64) contributes substantially to severity of arthritis, hypersensitivity responses, and protection from bacterial infection. Immunity. 2002;16:391–402. doi: 10.1016/s1074-7613(02)00294-7. [DOI] [PubMed] [Google Scholar]
- 18.Ji H, Ohmura K, Mahmood U, et al. Arthritis critically dependent on innate immune system players. Immunity. 2002;16:157–68. doi: 10.1016/s1074-7613(02)00275-3. [DOI] [PubMed] [Google Scholar]
- 19.Galon J, Bouchard C, Fridman WH, Sautes C. Ligands and biological activities of soluble Fc gamma receptors. Immunol Lett. 1995;44:175–81. doi: 10.1016/0165-2478(94)00211-9. [DOI] [PubMed] [Google Scholar]
- 20.Fridman WH, Teillaud JL, Bouchard C, et al. Soluble Fc gamma receptors. J Leukoc Biol. 1993;54:504–12. [PubMed] [Google Scholar]
- 21.Sautes C, Varin N, Hogarth PM, Unkeless JC, Teillaud C, Even J, Lynch A, Fridman WH. Molecular and functional studies of recombinant soluble Fc gamma receptors. Mol Immunol. 1990;27:1201–7. doi: 10.1016/0161-5890(90)90023-s. [DOI] [PubMed] [Google Scholar]
- 22.Astier A, Merle-Beral H, de la Salle H, Moncuit J, Cazenave JP, Fridman WH, Hanau D, Teillaud JL. Soluble Fcgamma receptor, Fc gammaRIIa2, is present in two forms in human serum and is increased in patients with stage C chronic lymphocytic leukemia. Leuk Lymphoma. 1997;26:317–26. doi: 10.3109/10428199709051781. [DOI] [PubMed] [Google Scholar]
- 23.Astier A, de la Salle H, de la Salle C, et al. Human epidermal Langerhans cells secrete a soluble receptor for IgG (Fc gamma RII/CD32) that inhibits the binding of immune complexes to Fc gamma R+ cells. J Immunol. 1994;152:201–12. [PubMed] [Google Scholar]
- 24.Galon J, Moldovan I, Galinha A, Provost-Marloie MA, Kaudewitz H, Roman-Roman S, Fridman WH, Sautes C. Identification of the cleavage site involved in production of plasma soluble Fc gamma receptor type III (CD16) Eur J Immunol. 1998;28:2101–7. doi: 10.1002/(SICI)1521-4141(199807)28:07<2101::AID-IMMU2101>3.0.CO;2-W. [DOI] [PubMed] [Google Scholar]
- 25.Fleit HB, Kobasiuk CD, Daly C, Furie R, Levy PC, Webster RO. A soluble form of Fc gamma RIII is present in human serum and other body fluids and is elevated at sites of inflammation. Blood. 1992;79:2721–8. [PubMed] [Google Scholar]
- 26.de Haas M, Kleijer M, Minchinton RM, von Roos D, dem Borne AE. Soluble Fc gamma RIIIa is present in plasma and is derived from natural killer cells. J Immunol. 1994;152:900–7. [PubMed] [Google Scholar]
- 27.Lamour A, Baron D, Soubrane C, Cartron J, Khayat D, Adler Y, Le Goff P, Youinou P. Anti-Fc gamma receptor III autoantibody is associated with soluble receptor in rheumatoid arthritis serum and synovial fluid. J Autoimmun. 1995;8:249–65. doi: 10.1006/jaut.1995.0019. [DOI] [PubMed] [Google Scholar]
- 28.Gavin AL, Wines BD, Powell MS, Hogarth PM. Recombinant soluble Fc gamma RII inhibits immune complex precipitation. Clin Exp Immunol. 1995;102:620–5. doi: 10.1111/j.1365-2249.1995.tb03862.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Burmester GR, Stuhlmuller B, Keyszer G, Kinne RW. Mononuclear phagocytes and rheumatoid synovitis. Mastermind or workhorse in arthritis? Arthritis Rheum. 1997;40:5–18. doi: 10.1002/art.1780400104. [DOI] [PubMed] [Google Scholar]
- 30.Feldmann M, Maini RN. The role of cytokines in the pathogenesis of rheumatoid arthritis. Rheumatology. 1999;38(Suppl. 2):3–7. [PubMed] [Google Scholar]
- 31.Maini RN, Elliott MJ, Brennan FM, Feldmann M. Beneficial effects of tumour necrosis factor-alpha (TNF-alpha) blockade in rheumatoid arthritis (RA) Clin Exp Immunol. 1995;101:207–12. doi: 10.1111/j.1365-2249.1995.tb08340.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Mannik M. Rheumatoid factors in the pathogenesis of rheumatoid arthritis. J Rheumatol Suppl. 1992;32:46–9. [PubMed] [Google Scholar]
- 33.Visser H, Gelinck LB, Kampfraath AH, Breedveld FC, Hazes JM. Diagnostic and prognostic characteristics of the enzyme linked immunosorbent rheumatoid factor assays in rheumatoid arthritis. Ann Rheum Dis. 1996;55:157–61. doi: 10.1136/ard.55.3.157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tinghe H, Carson DA. Rheumatoid factor. In: Ruddy S, Harris ED, Sledge CB, editors. Kelly's Textbook of Rheumatology. Philadelphia: W.B. Saunders Co.; 2001. pp. 130–60. [Google Scholar]
- 35.Williams RC. Rheumatoid factors: historical perspective, origins and possible role in disease. J Rheumatol Suppl. 1992;32:42–5. [PubMed] [Google Scholar]
- 36.Hendrich C, Kuipers JG, Kolanus W, Hammer M, Schmidt RE. Activation of CD16+ effector cells by rheumatoid factor complex. Role of natural killer cells in rheumatoid arthritis. Arthritis Rheum. 1991;34:423–31. doi: 10.1002/art.1780340407. [DOI] [PubMed] [Google Scholar]
- 37.Johnstone A, Thorpe R. Purification of Immunoglobulins, Constituent Chains and Fragments. Immunochemistry in Practice. Oxford: Blackwell Scientific Publications; 1982. [Google Scholar]
- 38.Catty D, Raykundalia C. Elisa and related enzyme immunoassays. In: Catty D, editor. Antibodies: A Practical Approach. Oxford: IRL Press; 1989. pp. 97–154. [Google Scholar]
- 39.Vollmers HP, Wozniak E, Stepien-Botsch E, Zimmermann U, Muller-Hermelink HK. A rapid method for purification of monoclonal human IgM from mass culture. Hum Antibodies Hybridomas. 1996;7:37–41. [PubMed] [Google Scholar]
- 40.Steinitz M, Izak G, Cohen S, Ehrenfeld M, Flechner I. Continuous production of monoclonal rheumatoid factor by EBV-transformed lymphocytes. Nature. 1980;287:443–5. doi: 10.1038/287443a0. [DOI] [PubMed] [Google Scholar]
- 41.Debets JM, Van der Linden CJ, Dieteren IE, Leeuwenberg JF, Buurman WA. Fc-receptor cross-linking induces rapid secretion of tumor necrosis factor (cachectin) by human peripheral blood monocytes. J Immunol. 1988;141:1197–201. [PubMed] [Google Scholar]
- 42.Abrahams VM, Cambridge G, Lydyard PM, Edwards JC. Induction of tumor necrosis factor alpha production by adhered human monocytes: a key role for Fcgamma receptor type IIIa in rheumatoid arthritis. Arthritis Rheum. 2000;43:608–16. doi: 10.1002/1529-0131(200003)43:3<608::AID-ANR18>3.0.CO;2-G. [DOI] [PubMed] [Google Scholar]
- 43.Corper AL, Sohi MK, Bonagura VR, et al. Structure of human IgM rheumatoid factor Fab bound to its autoantigen IgG Fc reveals a novel topology of antibody–antigen interaction. Nat Struct Biol. 1997;4:374–81. doi: 10.1038/nsb0597-374. [DOI] [PubMed] [Google Scholar]
- 44.Sondermann P, Huber R, Oosthuizen V, Jacob U. The 3.2-A crystal structure of the human IgG1 Fc fragment-Fc gammaRIII complex. Nature. 2000;406:267–73. doi: 10.1038/35018508. [DOI] [PubMed] [Google Scholar]
- 45.Lund J, Winter G, Jones PT, et al. Human Fc gamma RI and Fc gamma RII interact with distinct but overlapping sites on human IgG. J Immunol. 1991;147:2657–62. [PubMed] [Google Scholar]
- 46.Burton DR, Woof JM. Human antibody effector function. Adv Immunol. 1992;51:1–84. doi: 10.1016/s0065-2776(08)60486-1. [DOI] [PubMed] [Google Scholar]
- 47.Ward ES, Ghetie V. The effector functions of immunoglobulins: implications for therapy. Ther Immunol. 1995;2:77–94. [PubMed] [Google Scholar]
- 48.Sarmay G, Lund J, Rozsnyay Z, Gergely J, Jefferis R. Mapping and comparison of the interaction sites on the Fc region of IgG responsible for triggering antibody dependent cellular cytotoxicity (ADCC) through different types of human Fc gamma receptor. Mol Immunol. 1992;29:633–9. doi: 10.1016/0161-5890(92)90200-h. [DOI] [PubMed] [Google Scholar]
- 49.Shields RL, Namenuk AK, Hong K, et al. High resolution mapping of the binding site on human IgG1 for Fcgamma RI, Fcgamma RII, Fcgamma RIII, and FcRn and design of IgG1 variants with improved binding to the Fcgamma R. J Biol Chem. 2001;276:6591–604. doi: 10.1074/jbc.M009483200. [DOI] [PubMed] [Google Scholar]
- 50.Wines BD, Powell MS, Parren PW, Barnes N, Hogarth PM. The IgG Fc contains distinct Fc receptor (FcR) binding sites: the leukocyte receptors Fc gamma RI and Fc gamma RIIa bind to a region in the Fc distinct from that recognized by neonatal FcR and protein A. J Immunol. 2000;164:5313–8. doi: 10.4049/jimmunol.164.10.5313. [DOI] [PubMed] [Google Scholar]
- 51.Sondermann P, Kaiser J, Jacob U. Molecular basis for immune complex recognition: a comparison of Fc- receptor structures. J Mol Biol. 2001;309:737–49. doi: 10.1006/jmbi.2001.4670. [DOI] [PubMed] [Google Scholar]
- 52.Nardella FA, Schroder AK, Svensson ML, Sjoquist J, Barber C, Christensen P. T15 group A streptococcal Fc receptor binds to the same location on IgG as staphylococcal protein A and IgG rheumatoid factors. J Immunol. 1987;138:922–6. [PubMed] [Google Scholar]
- 53.Schroder AK, Nardella FA, Mannik M, Johansson PJ, Christensen P. Identification of the site on IgG Fc for interaction with streptococci of groups A, C and G. Immunology. 1987;62:523–7. [PMC free article] [PubMed] [Google Scholar]
- 54.Sutton BJ, Corper AL, Sohi MK, Jefferis R, Beale D, Taussig MJ. The structure of a human rheumatoid factor bound to IgG Fc. Adv Exp Med Biol. 1998;435:41–50. doi: 10.1007/978-1-4615-5383-0_4. [DOI] [PubMed] [Google Scholar]
- 55.Sutton B, Corper A, Bonagura V, Taussig M. The structure and origin of rheumatoid factors. Immunol Today. 2000;21:177–83. doi: 10.1016/s0167-5699(00)01589-9. [DOI] [PubMed] [Google Scholar]
- 56.Astier A, de la Salle H, Moncuit J, Freund M, Cazenave JP, Fridman WH, Hanau D, Teillaud JL. Detection and quantification of secreted soluble Fc gamma RIIA in human sera by an enzyme-linked immunosorbent assay. J Immunol Meth. 1993;166:1–10. doi: 10.1016/0022-1759(93)90323-y. [DOI] [PubMed] [Google Scholar]