Abstract
Reduction in the functional mass of β-cells is a common denominator in most forms of diabetes. Since the replicative potential of β-cells is limited, the search for factors that trigger islet neogenesis becomes imperative. Here we tested the hypothesis that regenerating factors for the pancreas are either secreted by or present within the pancreatic milieu itself. For this purpose, we intraperitoneally injected pancreatic cell culture supernatant (PCCS), from normal pancreas, into streptozotocin(STZ)-induced diabetic mice for 15 consecutive days. The PCCS-treated mice showed sustained reversal in 77.77% of experimental diabetic mice as evidenced by restoration of normoglycemia, increase in serum insulin levels and occurrence of neo islets in histopathological studies during a two month follow up, as opposed to the control diabetic mice which remained hyperglycemic throughout. In order to examine the potential of PCCS to bring about the regeneration of islets, we treated intra-islet precursor cells with PCCS in vitro, which led to the neogenesis of islets as evidenced by dithiozone and insulin immunostaining. These findings substantiate our hypothesis and make the search for regenerative factors converge towards the pancreas and its immediate surroundings. Such regenerative approaches, in combination with other therapeutic strategies to promote islet neogenesis may, in future, provide a cure and/or better means for the control and management of diabetes.
Keywords: regeneration, pancreas, pancreatic cell culture supernatant, diabetes, nesidioblastosis
Introduction
Diabetes mellitus results from β-cell dysfunction and decrement in insulin levels. It is generally believed that the number of functionally intact β-cells in the islets of Langerhans is critical to the progression and outcome of diabetes mellitus and also for its possible reversal [1-5].
It is known that the pancreas has a 'sensing' mechanism which responds to deficit in the islet/pancreatic cell mass and regenerates so as to maintain normal β-cell/islet cell mass [6-8]. Evidence indicates that islet stem cells or progenitor cells exist in the adult pancreas and are located in or near pancreatic ducts, and can differentiate into endocrine cells in response to stimuli [9-12]. Work by Guz et al. suggests that there are islet progenitor cells elsewhere in the pancreas [13, 14], and studies by Banerjee et al. have demonstrated that STZ-induced diabetic mice retain their pool of intra-islet precursor cells and can give rise to mature functional islets [10].
Strategies which can trigger replication and/or growth in islet mass in a diabetic pancreas are desirable for the effective treatment of diabetes [4]. In such a scenario, any agents which can increase the level of regeneration, by triggering nesidioblastosis in the otherwise dormant precursor cells, would be able to retard the progression of diabetes.
Many inductive substances have been shown to stimulate pancreatic regeneration and the restoration of islet cell mass in vivo. Recently it was demonstrated that the administration of tungstate to neonatal STZ-treated mice brought about regeneration of the pancreas [15]. Various natural products like gastrointestinal neuropeptides, colecystokinin, terminalia catappa linn fruits, vinca rosea extracts and alkaloid extract of ephedrae herba are effective in regeneration of the pancreas and enhancement of β-cell growth. They are, hence, candidates for regenerative therapy in the treatment of diabetes [16-19].
It has been previously shown in our lab that partial pancreatectomy in diabetic mice led to regeneration of the pancreas with rapid improvement in glycemic status and maintenance of normoglycemia throughout a 90-day follow up [20].
In a model of 90% pancreatectomy in rats and mice, treatment with poly ADP synthase inhibitors was shown to result in exocrine and endocrine cell regeneration [21, 22]. Rosenberg and Vinik have shown that partial duct obstruction of hamster pancreas leads to the induction of islet cell regeneration and neo islet formation in diabetic mice [23, 24]. The trophic effects observed in this model are mediated by the paracrine and/or autocrine mechanism. It has also been shown that tissue extract from cellophane-wrapped pancreas contains protein(s) exhibiting trophic effects on pancreatic tissue including islet cell regeneration [25-28]. Based on these lines, Hardikar et al. from this lab reported in 1999 a reversal of diabetes following serial injections of cytosolic extract from regenerating pancreas [29].
Studies carried out by us and other groups have prompted us to hypothesize that the factor(s) leading to the regeneration of pancreas and restoration of islet mass are either secreted by or are present within the pancreatic milieu itself [11, 20, 29]. For verification of this hypothesis, we studied the effects of serial intraperitoneal injections of PCCS and pancreatic cell cytosolic extract (CE) from normal pancreas, along with appropriate controls, on the hyperglycemic status of STZ-diabetic mice in vivo and the potential of PCCS to trigger neo islet formation from intra-islet precursor cell monolayer in vitro, and our findings confirm this hypothesis.
Materials and Methods
Animals
Male Swiss albino mice (6-8 weeks) were rendered diabetic by administering an intraperitoneal injection of 100 mg/kg body weight of streptozotocin (STZ; Sigma Chemical Co., St. Louis, USA). STZ was reconstituted in chilled sodium citrate buffer (pH 4.5) just before injection. STZ-treated animals were considered as frankly diabetic after they consistently exhibited random plasma glucose levels of more than 200 mg/dl for a minimum period of two weeks. Diabetic mice were randomly divided into six groups viz. non-diabetic control mice, sham-treated diabetic control mice, diabetic mice treated with heat-inactivated PCCS, diabetic mice treated with CE of pancreatic cell culture and diabetic mice treated with consecutive injections of PCCS.
All animals were obtained from an inbred population of swiss albino mice maintained at the animal house of the National Centre for Cell Science, Pune, India and supplied with food and water ad libitum throughout the period of the study. All procedures were carried out according to specified regulations of the Central Animal Ethical Committee. The animals used were cared for in accordance with the principles of the "guide for care and use of laboratory animals" prepared by the National Academy of Sciences (NIH publication 86-23 revised 1985).
Preparation of PCCS
Islets were isolated by an islet isolation protocol designed in our lab by Shewade et al. (30). Briefly, pancreata were removed aseptically from sacrificed mice, minced and digested using dissociation medium containing Dulbecco’s modified essential medium (DMEM) supplemented with 1mg/ml Collagenase type V (Sigma, Chemical Co., St. Louis, USA). The reaction was stopped by the addition of RPMI 1640 with hepes, glucose and bicarbonate and 10% fetal bovine serum (GIBCOTM, Grand Island, NY, USA). The suspension was centrifuged and the resulting pellet was seeded in 25 cm2 tissue culture flasks containing 5ml RPMI 1640 with H,G and B and 10% FBS per pancreas. These flasks were then incubated at 37°C in an atmosphere comprising of 5% CO2 for 48 hrs, after which the culture was centrifuged at 2,000 rpm for 5 minutes at room temperature. The supernatant was collected and centrifuged at 12,000 rpm for 30 minutes at 4°C to remove any cells or cell debris in suspension. The resulting supernatant was collected, distributed into aliquots and stored thereafter at -80°C. The total protein content in the above-mentioned supernatant was estimated using a BCA protein detection kit. (Pierce Biotechnology, Rockford, IL, USA).
Heat inactivation of PCCS was carried out by boiling it at 100°C in a water bath for 10 minutes and then immediately cooling it on ice. This procedure was repeated 3 times.
Preparation of the cytosolic extract
After collecting the supernatant following the procedure described above, the cell pellet was used for the preparation of the CE. The pellet was homogenized in a lysis buffer containing 20mM Tris, 1mM EDTA, 0.1% triton X-100 and 1mM PMSF for inhibition of proteases. After incubating on ice for 30 minutes the pellet was sonicated for 5 minutes (10 pulses) and supernatant was collected after spinning at 4°C, 30 minutes and 12,000rpm. This extract was then dialyzed against 20mM Tris using a 3Kd cut-off dialysis membrane for 4 hours at 4°C, after which the CE was aliquoted, quantified and stored at -80°C.
Treatment
Diabetic mice were divided into groups each comprising of 10 mice. Nondiabetic control mice were left untreated while sham-treated control diabetic mice were injected with RPMI 1640 with H, G and B +10% FBS. Heat-inactivated PCCS was injected into another set of control mice. One set of diabetic mice was injected with CE of cell pellet (10 μg of total protein per day) while a set of experimental mice was treated with 200 μl PCCS (10μg of total protein per day).
Plasma glucose and serum insulin estimation
Plasma glucose was monitored every six days from the administration of injections using Accu-Check glucose sensor (Roche Diagnostics Co., Germany). Blood for serum was collected immediately prior to termination of the experiment (after a 60-day follow-up period) by retro orbital bleeding. Serum was collected after centrifugation at 12,000 rpm at 4°C for 30 minutes and then stored at -80°C. Serum insulin levels were measured using the Insulin ELISA kit (Biosource, Europe SA, Nevilles, Belgium).
Histopathological procedures
Pancreas from experimental and control mice were removed after termination of the experiment (60 days after completion of the injection regime) and fixed in 4% buffered paraformaldehyde at room temperature. Sections 7μm thick were taken on a rotary microtome. (Reichert-Jung 2030 Biocut, Cambridge Instruments, Heidelberg, Germany) and stained with hematoxylin and eosin.
Intraperitoneal glucose tolerance test
60 days after completion of the injection course, control and experimental animals were fasted overnight and intraperitoneally administered a glucose dose of 2 g/kg body weight. Blood glucose was measured at 0, 30, 60 and 90 minutes from the administration of glucose.
Preparation and treatment of intra-islet precursor cell monolayer
The intra-islet precursor cell isolation protocol was followed as standardized in our lab by Banerjee et al. [10], with some modifications. After the formation of monolayer, the medium was changed to a serum free medium (SFM) which consisted of DMEM: Ham's F12K (1:1), β mercaptoethanol (1%), bovine serum albumin (BSA) (0.1%). Nicotinamide, keratinocyte growth factor (KGF) and insulin transferrin selenium (ITS) were not added to the SFM. The intra-islet precursor cell monolayer was treated with 50μl of PCCS (2.961 μg of total protein), in experimental wells. Control wells 1 and 2 were left untreated or treated with RPMI 1640 with H, G and B +10 % FBS respectively. Media was replenished every four days. These monolayer wells were kept under constant observation for collecting the newly formed cellular aggregates which budded off and separated from the cell monolayer on maturation.
Dithizone staining
To evaluate the total number and assess the specificity of the newly generated islets, these islets were collected after budding off, and stained with insulin-specific stain, dithiozone (DTZ) (Sigma, St. Louis, MO, USA), and scored using an inverted microscope (Olympus, Tokyo, Japan) as described [10].
Insulin immunostaining
To ascertain the insulin immunoreactive status of the newly generated islets, they were collected from control and experimental wells, washed with 1x PBS and immunostained for insulin as described [10]. Briefly, they were fixed with 4% paraformaldehyde and permeabilized using 0.2% Triton X-100. Samples were blocked using 2% BSA and primary staining for insulin was performed using anti-insulin goat polyclonal antisera (Santa Cruz Biotechnology, Santa Cruz, CA, USA. dilution 1:1000 μl). After washing, samples were incubated in fluorescein isothiocyanate (FITC)-tagged rabbit anti goat polyclonal IgG (Santa Cruz Biotechnology, Santa Cruz, CA, USA dilution 1:1500) secondary antibody. The samples were then mounted on slides in mounting media containing anti fade. These were then observed under confocal laser microscope (Carl Zeiss Meditec AG, Jena, Germany) using an argon air-cooled laser of wavelength 488 nm (LASOS, Lasertechnik GmbH, Jena, Germany).
Statistics
All blood glucose and serum insulin values are reported as mean ± SD. Data were analyzed by the one-way ANOVA method using the statistical software SIGMA STAT, version 2.03 from SPSS Inc.
Results
It can be clearly seen that diabetic mice treated with PCCS gradually attain normoglycemia after 7 days of treatment, while the sham-treated mice and the heat-inactivated PCCS-treated control diabetic mice showed sustained hyperglycemia. After 15 days of treatment, the PCCS-treated mice retained normoglycemia, the CE-treated mice showed an increase in glucose levels, while the sham-treated diabetic controls and the control diabetic mice treated with heat-inactivated PCCS showed sustained hyperglycemia. The nondiabetic controls exhibited consistent normoglycemia throughout the course of the experiment (Table 1).
Table 1. Random plasma glucose levels of mice in experimental and control groups.
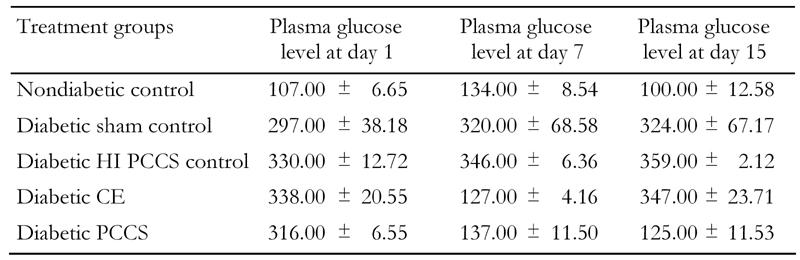
Data represents average random plasma glucose levels in control and experimental diabetic animals take during continuation of injection course. Values expressen in mg/dl ± SD with n = 10 in each group; p ≤ 0.001.
A PCCS regime on varied degrees of hyperglycemic mice showed gradual reduction in random blood glucose levels in all sets of experimental animals after 15 days of serial injections (Table 2). None of the reversed mice showed any symptoms of hypoglycemia. The sham-treated diabetic control mice did not show any lowering of random blood glucose levels, nor did nondiabetic control mice injected with PCCS show any symptoms of hypoglycemia (data not included). Table 3 indicates the serum insulin levels of experimental and control mice. It can be observed that diabetic mice after treatment with PCCS exhibited serum insulin levels in the same range as the nondiabetic animals (15-25 μIU/ml) while the serum insulin levels in sham-treated diabetic control mice were considerably lower than those of the normal range.
Table 2. Effects of PCCS on variable degrees of hyperglycemia.
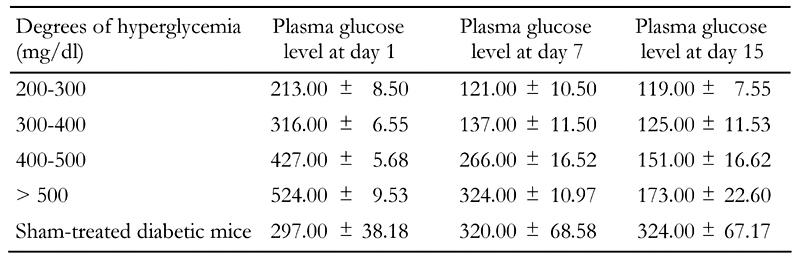
Data represents random plasma glucose levels in mice with variable degrees of hyperglycemia treated with PCCS and sham-treated diabetic mice. Values indicate random mean plasma glucose levels expressed in mg/dl ± SD with n = 10 in each group; p ≤ 0.001.
Table 3. Serum insulin levels of PCCS-treated diabetic and control mice.

Serum was collected from mice at the end of the 60-day follow-up. The normal mice and PCCS-treated reversed mice survived, while the sham-treated diabetic mice showed 50% mortality during the follow-up. Data represents average serum insulin levels expressed in µIU/ml ± SD with n = 8 in each group; p ≤ 0.001.
Intraperitoneal glucose clearance of experimental and control diabetic mice showed that PCCS-treated diabetic mice showed a reduction in blood glucose levels after the initial overshoot, and in the course of 90 minutes regained normoglycemia, thereby exhibiting normal glucose clearance, as did the nondiabetic control mice. The sham-treated diabetic control mice did not show a reduction in blood glucose levels even after 90 minutes, which indicates an abnormal glucose clearance (Figure 1). Histopathological sectioning of pancreas from the non diabetic, diabetic and PCCS-treated mice showed a newly generated islet, close to a duct with the islet ductal connection intact (Figure 2).
Figure 1. Glucose clearance in experimental and control mice.
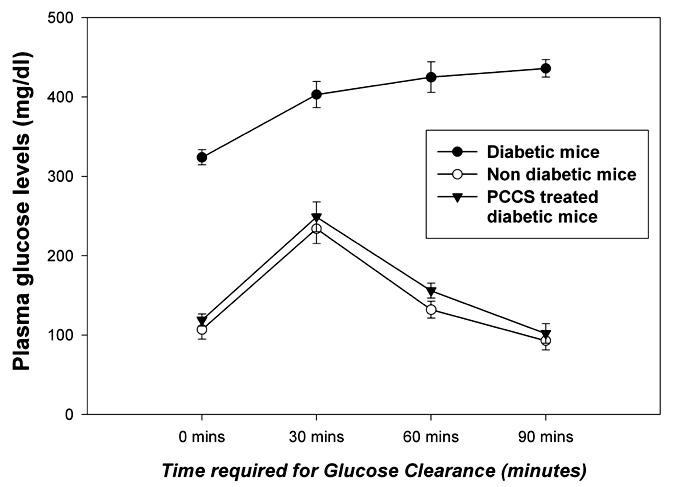
The figure illustrates the intraperitoneal glucose tolerance test of PCCS-treated diabetic and control diabetic mice at time points from 0 minutes (fasting blood glucose) to 90 minutes. The data represents plasma glucose levels expressed in mg/dl ± SD with n = 10 in each group; p ≤ 0.001.
Figure 2.
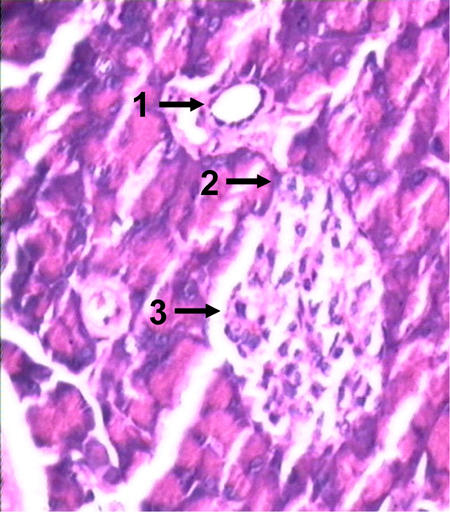
Hematoxylin- and eosin-stained histopathological sections of pancreas of PCCS-treated diabetic mice on attaining normoglycemia, showing the development of neo islets from the pancreatic duct. 1: duct from which the islet sprouted. 2: connection between the duct and the islet. 3: newly formed islet.
Intra-islet precursor cells on treatment with PCCS showed formation of neo islets in vitro. The control wells did not show any formation of similar cell aggregates or a budding of mature islets as was observed in PCCS-treated wells. These cellular aggregates stained positive for dithizone (Figure 3A) and also stained positive for insulin (Figure 3B).
Figure 3.
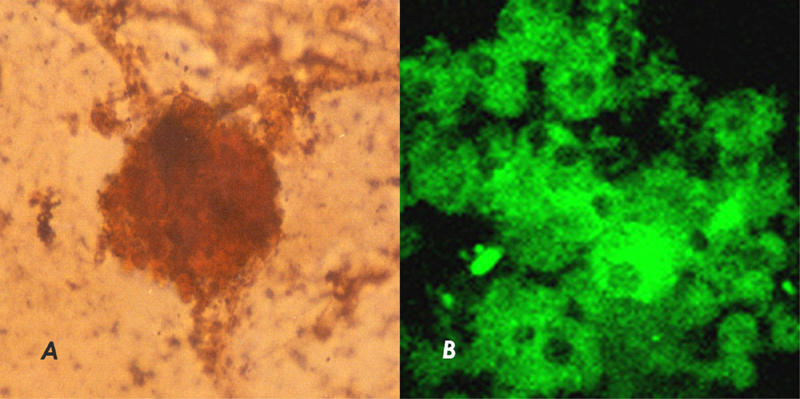
A: New islet generated from intra-islet precursor cells on treatment with PCCS. Islets were stained with DTZ. B: New islets generated from intra-islet precursor cells on treatment with PCCS stained for insulin immunoreactivity.
Discussion
In the present study we have demonstrated that serial injections of PCCS in STZ-diabetic mice led to a restoration and retention of normoglycemia due to the induction of nesidioblastosis.
PCCS was administered serially for 15 days, and a gradual lowering of random blood glucose levels was observed during the course, after which the injections were discontinued. PCCS contain islet cells and hence may have some insulin content. To avoid the influence of this exogenous insulin content on random blood glucose levels in PCCS-treated mice, all experimental and control mice were rested (not given injections) for a period of 24 hours before estimation of random blood glucose levels. Though it is known that insulin therapy causes reversal in cases of hypoleptinemia, diabetic retinopathy, neuropathic and gastrointestinal complications related to diabetes mellitus in adolescents and reversal in abnormalities of ocular fluorophotometry, it is also known that insulin alone is incapable of bringing about a reversal or regeneration of a consistent or transient nature in diabetic animal models [31-34]. PCCS treatment in diabetic mice brought about a gradual reduction in random blood glucose levels as opposed to sham-treated or heat-inactivated PCCS-treated controls. The sham treatment consisted of RPMI 1640 with H, G and B +10% FBS. It was observed that none of the sham-treated mice regained normoglycemia, thus excluding the possibility of any of the components of the media or FBS having caused reversal or regeneration. It can also be noted that heat-inactivated PCCS did not cause reversal or regeneration either. It was shown earlier in our lab that CE from regenerating pancreas induces normoglycemia and a reversal of experimental diabetes in mice, and our current findings concur with this [29]. The ability of CE derived from the cell pellet to induce transient normoglycemia indicates further that the nesidioblastotic factor is produced by the pancreatic cells, while the supernatant from the same culture induces a stable reversal of diabetes (Table 1). It would, therefore, be logical to conclude that the regenerating factors are secreted by the cells of the pancreas.
It is known from previous studies that regeneration of the diabetic pancreas is induced by some amount of injury to the pancreas in the form of pancreatectomy, duct ligation, chemical toxins and viruses [8, 11, 35, 36]. Based on our results, it can be further speculated that the pancreas itself may be a storehouse of regenerating and inhibitory factors [20]. In vivo pancreatic injury induced by pancreatectomy could be a reason for the release of the regenerating factors which results in the reversal of experimental diabetes [6, 7, 20, 37, 38]. This is perhaps the first report demonstrating in vitro pancreatic injury being harnessed for in vivo nesidioblastosis leading to a stable reversal of diabetes. The injury that triggers the production and/or secretion of the regenerating factors into the supernatant may be implicated in the culture process. In the present study, pancreatic injury induced in vitro during the process of primary culture may trigger production and/or secretion of the nesidioblastotic factors into the cell culture supernatant, which on being injected into experimental diabetic mice triggers nesidioblastosis and thereby a reversal of diabetes.
Our results confirm the nesidioblastotic potential of PCCS and necessitate the identification and characterization of these regenerating factor.
On treating diabetic mice showing varied degrees of hyperglycemia with PCCS it was observed that PCCS effectuates a reversal of diabetes at varied stages of disease progression, as can be deduced from Table 2. Treatment of PCCS in nondiabetic controls did not induce hypoglycemia (data not shown). Also, no mortality was observed in any of the reversed mice due to hypoglycemia, which is indicative of good glycemic control. The regenerative potential of PCCS is further documented by the elevated serum insulin levels in PCCS-treated mice, which are comparable to nondiabetic mice, after a 60 day follow up as seen from Table 3. On the other hand, the serum insulin levels of sham-treated diabetic mice were considerably lower than the experimental and the nondiabetic control mice, as seen in Table 3. These elevated levels of insulin correspond with a lowering in random blood glucose levels. The PCCS-treated diabetic mice also showed normal glucose clearance, while the sham-treated diabetic mice showed abnormal glucose clearance, as can be observed in Figure 1, ensuring sustained normoglycemia. These results suggest that mice showed reversal due to insulin production by new β-cells/islets.
Histopathological studies of PCCS-treated mice show evidence of nesidioblastosis, as can be seen by the intact ductal connection between the islet and associated duct in the pancreas of PCCS-treated mice (Figure 2), as compared to diabetic controls where no such connection between islets and ducts was observed (data not shown). The newly generated islets may have formed from the ductal precursor cells. Studies by other groups have demonstrated a reversal of diabetic mice on formation of new islets from ductal cells [10, 39]. These studies, along with studies related to pancreatectomy in mice, and our present results, strengthen our conclusion that PCCS has the capacity to bring about nesidioblastosis in vivo. This suggests that the factors causing regeneration are present within the pancreas or in the pancreatic milieu.
To further test the regenerative potential of PCCS, we treated the intra-islet precursor cell monolayer with PCCS, along with appropriate controls in vitro. Intra-islet precursor cells were chosen as a suitable model system because it has been recently shown that new β-cells differentiate from intra-islet precursors in pancreatic islets of mice in which diabetes is induced by injecting a high dose of the β-cell toxin STZ. [10, 40].
It could be observed that the intra-islet precursor cell monolayer, on treatment with PCCS in vitro, lead to the formation of cellular aggregates, which budded off upon maturation, to the scale of 32.85 ± 13.984 islets per 9.06 cm2 (where p = 0.05), while no islets or islet-like clusters were found in the control wells. Since no known neo islet inducing/nesidioblastotic factors like KGF or nicotinamide were extrinsically added to the SFM, it can be safely stated that the islets formed in the PCCS-treated experimental wells were due to the treatment of the intra-islet precursor monolayer with PCCS alone. This observation is further supported by the fact that no neo islets were formed in the control wells containing monolayer with SFM alone or in the wells sham-treated with RPMI 1640 with H, G and B +10% FBS. Furthermore, cell aggregation could not be observed in the control wells, which confirms that the aggregation and maturation of the islets is brought about by PCCS. The identity of these cellular aggregates was further confirmed by the use of the DTZ staining (Figure 3A) and the insulin immunostaining method (Figure 3B). These results indicate the cellular aggregates to be newly formed islets in vitro. This brings into focus the potential of PCCS to bring about the formation of new islets from intra-islet precursor cells in vitro.
Our present studies demonstrate that PCCS carries the potential to induce islet neogenesis in vivo as well as in vitro and thus proves our hypothesis that the regenerating factors for the pancreas lie within the pancreas and are capable of bringing about nesidioblastosis in vivo as well as in vitro. These results help in narrowing down the search for regenerating factors to the pancreatic milieu in mice. The pancreatic cell culture supernatant contains cells of exocrine and endocrine nature and is thereby a milieu of secretions of these cells, which may act independently of each other or in synergism. These aspects of mechanism are the subject of an independent investigation.
Acknowledgments
This work was supported by a grant from the Department of Biotechnology, Government of India. We also like to thank the Director of the NCCS for the use of the departmental facility and Dr. Avinash Pradhan and Ashwini Atre for technical assistance. We declare that there is no conflict of interest that would prejudice the impartiality of this research work.
References
- 1.Zalzman M, Gupta S, Giri RK, Berkovich I, Sappal BS, Karnieli O, Zern MA, Fleischer N, Efrat S. Reversal of hyperglycemia in mice by using human expandable insulin-producing cells differentiated from fetal liver progenitor cells. Proc Natl Acad Sci U S A. 2003;100:7253–7258. doi: 10.1073/pnas.1136854100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Risbud MV, Bhonde RR. Models of pancreatic regeneration in diabetes. Diabetes Res Clin Pract. 2002;58(3):155–165. doi: 10.1016/s0168-8227(02)00103-1. [DOI] [PubMed] [Google Scholar]
- 3.Petrovsky N, Silva D, Schatz DA. Prospects for the prevention and reversal of type 1 diabetes mellitus. Drugs. 2002;62(18):2617–2635. doi: 10.2165/00003495-200262180-00005. [DOI] [PubMed] [Google Scholar]
- 4.Okamoto H. The reg gene family and reg proteins: with special attention to the regeneration of pancreatic beta-cells. J Hepatobiliary Pancreat Surg. 1999;6:254–262. doi: 10.1007/s005340050115. [DOI] [PubMed] [Google Scholar]
- 5.Yamaoka T. Regeneration therapy for diabetes mellitus. Expert Opin Biol Ther. 2003;3:425–433. doi: 10.1517/14712598.3.3.425. [DOI] [PubMed] [Google Scholar]
- 6.Brockenbrough JS, Weir GC, Bonner-Weir S. Discordance of exocrine and endocrine growth after 90% pancreatectomy in rats. Diabetes. 1988;37:232–236. doi: 10.2337/diab.37.2.232. [DOI] [PubMed] [Google Scholar]
- 7.Parekh D, Townsend CM Jr, Rajaraman S, Ishizuka J, Thompson JC. Pancreatic regeneration after partial pancreatectomy. Am J Surg. 1991;161:84–89. doi: 10.1016/0002-9610(91)90365-k. [DOI] [PubMed] [Google Scholar]
- 8.Wang RN, Kloppel G, Bouwens L. Duct to islet cell differentiation and islet growth in the pancreas of duct ligated adult rats. Diabetologia. 1995;38:1405–1411. doi: 10.1007/BF00400600. [DOI] [PubMed] [Google Scholar]
- 9.Paris M, Tourrel-Cuzin C, Plachot C, Ktorza A. Review: pancreatic beta-cell neogenesis revisited. Exp Diabesity Res. 2004;5(2):111–121. doi: 10.1080/15438600490455079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Banerjee M, Bhonde RR. Regeneration of pancreatic beta-cells from intra-islet precursor cells in an experimental model of diabetes. JOP. 2003;4(4):137–145. [PubMed] [Google Scholar]
- 11.Hardikar AA. Generating new pancreas from old. Trends Endocrinol Metab. 2004;15:198–203. doi: 10.1016/j.tem.2004.05.001. [DOI] [PubMed] [Google Scholar]
- 12.Bell DS, Grizzle WE, Dunlap NE. Nesidioblastosis causing reversal of insulin-dependent diabetes and development of hyperinsulinemic hypoglycemia. Diabetes Care. 1995;18(10):1379–1380. doi: 10.2337/diacare.18.10.1379. [DOI] [PubMed] [Google Scholar]
- 13.Bonner-Weir S, Taneja M, Weir GC, Tatarkiewicz K, Song KH, Sharma A, O'Neil JJ. In vitro cultivation of human islets from expanded ductal tissue. Proc Natl Acad Sci U S A. 2000;97(14):7999–8004. doi: 10.1073/pnas.97.14.7999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Katdare MR, Bhonde RR, Parab PB. Analysis of morphological and functional maturation of neoislets generated in vitro from pancreatic ductal cells and their suitability for islet banking and transplantation. J Endocrinol. 2004;182(1):105–112. doi: 10.1677/joe.0.1820105. [DOI] [PubMed] [Google Scholar]
- 15.Fernandez-Alvarez J, Barbera A, Nadal B, Barcello-Batllori S, Piquer S, Claret M, Guinovart JJ, Gomis R. Stable and functional regeneration of pancreatic beta-cell population in nSTZ-rats treated with tungstate. Diabetologia. 2004;47:470–477. doi: 10.1007/s00125-004-1332-8. [DOI] [PubMed] [Google Scholar]
- 16.Morisset J. Intervention of GI neuropeptides in pancreatic growth and regeneration: comparison with cholecystokinin. J Physiol Pharmacol. 2003;54:127–141. [PubMed] [Google Scholar]
- 17.Nagappa AN, Thakurdesai PA, Venkat Rao N, Singh J. Antidiabetic activity of terminalia catappa linn fruits. J Ethnopharmacol. 2003;88:45–50. doi: 10.1016/s0378-8741(03)00208-3. [DOI] [PubMed] [Google Scholar]
- 18.Ghosh S, Suryawanshi SA. Effect of Vinca rosea extract in treatment of alloxan diabetes in male albino rats. Indian J Exp Biol. 2001;39:748–759. [PubMed] [Google Scholar]
- 19.Xiu LM, Miura AB, Yamamoto K, Kobayashi T, Song QH, Kitamura H, Cyong JC. Pancreatic islet regeneration by epheridine in mice with streptozotocin-induced diabetes. Am J Chin Med. 2001;29:493–500. doi: 10.1142/S0192415X01000514. [DOI] [PubMed] [Google Scholar]
- 20.Hardikar AA, Karandikar MS and Bhonde RR. Effect of partial pancreatectomy on diabetic status in BALB/c mice. J Endocrinol. 1999;162:189–195. doi: 10.1677/joe.0.1620189. [DOI] [PubMed] [Google Scholar]
- 21.Terazono K, Yamamoto H, Takasawa S, Shiga K, Yonemura Y, Tochino Y, Okamoto H. A novel gene activated in regenerating islets. J Biol Chem. 1988;263:2111–2114. [PubMed] [Google Scholar]
- 22.Akiyama T, Takasawa S, Nata K, Kobayashi S, Abe M, Shervani NJ, Ikeda T, Nakagawa K, Unno M, Matsuno S et al. Activation of reg gene, a gene for insulin producing beta-cell regeneration: poly (ADP-ribose) polymerase binds reg promoter and regulates transcription by autopoly (ADP-ribosyl)ation. Proc Natl Acad Sci U S A. 2001;98:43–53. doi: 10.1073/pnas.240458597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Rosenberg L, Brown RA, Duguid WP. A new approach to the induction of duct epithelial cells hyperplasia and nesidioblastosis by cellophane wrapping of the hamster pancreas. J Surg Res. 1983;35:63–72. doi: 10.1016/0022-4804(83)90127-0. [DOI] [PubMed] [Google Scholar]
- 24.Rosenberg L, Vinik AI. Induction of endocrine cell differentiation: a new approach to management of diabetes. J Lab Clin Med. 1989;114:75–83. [PubMed] [Google Scholar]
- 25.Clas D, Rosenberg L, Duguid WP, Malenshenko E. Islet cell differentiation and proliferation induced by a pancreatic cytosol extract. Surgical Forum. 1988;39:164–166. [Google Scholar]
- 26.Rosenberg L, Duguid WP, Healy M, Clas D, Vinik AI. Reversal of diabetes by induction of islet cell neogenesis. Transplant Proc. 1992;24:1027–1028. [PubMed] [Google Scholar]
- 27.Raefeloff R, Pittenger GL, Barlow SW, Qin XF, Yan B, Rosenberg L, Duguid WP, Vinik AI. Cloning and sequencing of the pancreatic islet neogenesis associated protein (INGAP) gene and its expression in islet neogenesis in hamsters. J Clin Invest. 1997;99:2100–2109. doi: 10.1172/JCI119383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Rafaeloff R, Qin XF, Barlow SW, Rosenberg L, Vinik AI. Identification of deferentially expressed genes induced in pancreatic islet neogenesis. FEBS Lett. 1996;378:219–223. doi: 10.1016/0014-5793(95)01457-8. [DOI] [PubMed] [Google Scholar]
- 29.Hardikar AA and Bhonde R. Modulating experimental diabetes by treatment with cytosolic extract from the regenerating pancreas. Diabetes Res Clin Pract. 1999;46:203–211. doi: 10.1016/s0168-8227(99)00098-4. [DOI] [PubMed] [Google Scholar]
- 30.Shewade Y, Umrani M, Bhonde RR. Large scale isolation of islets by tissue culture of adult mouse pancreas. Transplant Proc. 1999;31:1721–1723. doi: 10.1016/s0041-1345(99)00077-9. [DOI] [PubMed] [Google Scholar]
- 31.Havel PJ, Uriu-Hare JY, Liu T, Stanhope KL, Stern JS, Keen CL, Ahren B. Marked and rapid decreases of circulating leptin in streptozotocin diabetic rats: reversal by insulin. Am J Physiol. 1998;274:1482–1491. doi: 10.1152/ajpregu.1998.274.5.R1482. [DOI] [PubMed] [Google Scholar]
- 32.White MC, Kohner EM, Pickup JC, Keen H. Reversal of diabetic retinopathy by continuous subcutaneous insulin infusion: a case report. Br J Ophthalmol. 1981;65(5):307–311. doi: 10.1136/bjo.65.5.307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.White NH, Waltman SR, Krupin T, Santiago JV. Reversal of neuropathic and gastrointestinal complications related to diabetes mellitus in adolescents with improved metabolic control. J Pediatr. 1981;99(1):41–45. doi: 10.1016/s0022-3476(81)80954-7. [DOI] [PubMed] [Google Scholar]
- 34.White NH, Waltman SR, Krupin T, Santiago JV. Reversal of abnormalities in ocular fluorophotometry in insulin-dependent diabetes after five to nine months of improved metabolic control. Diabetes. 1982;31(1):80–85. doi: 10.2337/diab.31.1.80. [DOI] [PubMed] [Google Scholar]
- 35.Bonner-Weir S, Baxter LA, Schuppin GT, Smith FE. A second pathway for regeneration of adult exocrine and endocrine pancreas. A possible recapitulation of embryonic development. Diabetes. 1993;42:1715–1720. doi: 10.2337/diab.42.12.1715. [DOI] [PubMed] [Google Scholar]
- 36.Fernandes A, King LC, Guz Y, Stein R, Wright CV, Teitelman G. Differentiation of new insulin-producing cells is induced by injury in adult pancreatic islets. Endocrinology. 1997;138:1750–1762. doi: 10.1210/endo.138.4.5049. [DOI] [PubMed] [Google Scholar]
- 37.Hayashi KY, Tamaki H, Handa K, Takahashi T, Kakita A, Yamashina S. Differentiation and proliferation of endocrine cells in the regenerating rat pancreas after 90% pancreatectomy. Arch Histol Cytol. 2003;66(2):163–174. doi: 10.1679/aohc.66.163. [DOI] [PubMed] [Google Scholar]
- 38.Jetton TL, Liu YQ, Trotman WE, Nevin PW, Sun XJ, Leahy JL. Enhanced expression of insulin receptor substrate-2 and activation of protein kinase B/Akt in regenerating pancreatic duct epithelium of 60%-partial pancreatectomy rats. Diabetologia. 2001;44(11):2056–2065. doi: 10.1007/s001250100011. [DOI] [PubMed] [Google Scholar]
- 39.Kodama S, Kuhtreiber W, Fujimura S, Dale EA, Faustman DL. Islet regeneration during the reversal of autoimmune diabetes in NOD mice. Science. 2003;302(5648):1223–1227. doi: 10.1126/science.1088949. [DOI] [PubMed] [Google Scholar]
- 40.Guz Y, Torres A and Teitelman G. Detrimental effect of protracted hyperglycemia on beta-cell neogenesis in a murine model of diabetes. Diabetologia. 2002;45:1689–1696. doi: 10.1007/s00125-002-0970-y. [DOI] [PubMed] [Google Scholar]


