Abstract
In mammalian astrocytes, calcium waves are transmitted between cells via both a gap junction-mediated pathway and an extracellular, P2 receptor-mediated pathway, which link the cells into a syncytium. Calcium waves in astrocytes have also been shown to evoke calcium transients in neurons, and activity in neurons can elicit calcium waves in astrocytes. In this study, we show that in primary human fetal astrocytes, the P2 receptor-mediated and gap junction-mediated pathways are differentially regulated by the cytokine IL-1β. Confocal microscopy of astrocytes loaded with Indo-1 demonstrated that intercellular calcium wave transmission in IL-1β-treated cultures was potentiated compared with controls. However, transmission of calcium waves via the gap junction-mediated pathway was strikingly reduced. The major component of functional gap junctions in human fetal astrocytes was demonstrated to be connexin43 (Cx43), and there was a marked reduction of junctional conductance, loss of dye coupling, loss of Cx43 protein, and down-regulation of Cx43 mRNA expression after IL-1β treatment of cultures. Conversely, transmission of calcium waves via the P2 receptor-mediated pathway was potentiated in IL-1β-treated cultures compared with controls. This potentiation was associated with an increase in the number of cells responsive to UTP, and with a transient increase in expression of the P2Y2 purinoceptor mRNA. Because in inflammatory conditions of the human central nervous system IL-1β is produced both by resident glia and by invading cells of the immune system, our results suggest that inflammatory events may have a significant impact on coordination of astrocytic function and on information processing in the central nervous system.
Intercellular calcium signaling has been shown to occur in a variety of cells in culture and may represent a general mechanism for cell–cell communication (1). Two models have been proposed by which intercellular calcium waves may be transmitted in populations of nonexcitable cells. In one model, the increase in intracellular calcium concentration is transmitted by diffusion of inositol triphosphate (IP3) and/or calcium between adjacent cells via gap junctions composed of connexin subunits (2, 3). In the second, the calcium wave is mediated by autocrine or paracrine activity of secreted tri- and dinucleotide phosphates on P2 receptors, which may further be divided into the metabotropic P2Y receptor family and the ionotropic P2X receptor family (4). In the central nervous system (CNS), intercellular calcium waves have been observed in neurons (5), astrocytes (6, 7), and oligodendrocytes (8), and although these observations were originally made in populations of cells in culture, more recently intercellular calcium waves have also been observed in more integrated systems, for example in astrocytes in brain slice cultures (9), in neurons in cerebral cortical slices (5), and in glial cells in an intact preparation of retina (10). Intercellular calcium signaling in astrocytes has attracted particular attention, because it may represent a form of glial cell excitability that constitutes an information-processing system operating in parallel with neuronal circuits within the CNS (6). Although it has been recognized for some time that calcium signaling occurs between mammalian astrocytes via the gap junction-mediated pathway (11), the contribution of an extracellular, P2 receptor-mediated pathway has only recently been appreciated (12, 13). A number of observations would also suggest that glial and neuronal circuits interact. It has been demonstrated that in cocultures, intercellular calcium waves in astrocytes can elicit calcium transients in neurons (14), whereas in brain slices in culture, an intercellular calcium wave can be generated in astrocytes by stimulation of neurons (9). Taken together, these observations indicate that the mechanisms underlying information processing in the CNS may be substantially more complex and plastic than has previously been appreciated.
Recent work in our laboratories has focused on the modulation of astrocytic function by inflammatory mediators (15). As a result of these studies, we have shown that the cytokine IL-1β is a key activator for human fetal astrocytes, inducing the expression of multiple proinflammatory genes. Exposure to IL-1β also induces a change in cellular morphology from flat and polygonal to stellate, reminiscent of the change in astrocyte morphology that accompanies a reactive astrogliosis, a common response to brain injury, in vivo. In normal healthy brain tissues, constitutive expression of IL-1β is extremely low and restricted to specific neuronal tracts (16). However, in a number of different inflammatory and degenerative conditions of the CNS, increased expression of IL-1β has been extensively documented (17). In the present study, we have tested the hypothesis that IL-1β modulates the mechanisms underlying calcium wave transmission between human fetal astrocytes. Our results indicate that IL-1β differentially regulates calcium wave transmission mediated via the P2 receptor-mediated pathway as opposed to that mediated via gap junctions.
METHODS
Astrocyte Cultures and Cytokines.
Enriched human fetal cortical astrocyte cultures were established from second-trimester abortuses, and culture purity was determined by immunostaining for GFAP (glial fibrillary acidic protein; astrocytes), MAP-2 (microtubule-associated protein-2; neurons) and CD68 (microglia), as described (18). All tissue collection was approved by the Institutional Clinical Review Committee of the Albert Einstein College of Medicine. Recombinant human IL-1β was a gift from the Biological Response Modifiers Program at the National Cancer Institute (Frederick, MD); the dose used (10 ng/ml) was based on studies that we have performed previously on the response of human fetal astrocytes to IL-1 (15). This dose is identical to that used in studies that have examined the IL-1 signaling cascade leading to the activation of NF-κB in several other cell types (19, 20).
Calcium Measurements.
Astrocytes were plated on uncoated glass-bottomed microwells (Mat-Tek, Ashland, MA) at a density of 2.5 × 105 cells/ml. At 7–30 days after plating, confluent control or IL-1β-treated cultures of the same case plated at the same time were loaded with 10 μM Indo-1-AM (Molecular Probes) at 37°C for 45 min, rinsed in PBS (pH 7.4), and the ratio of Indo-1 fluorescence intensity emitted at two wavelengths (390–440 nm and >440 nm) was imaged by using UV laser excitation at 351 nm on a Nikon RCM 8000 real-time confocal microscope. After background and shading correction with a UV large pinhole setting and a Nikon 40× water immersion objective (n.a. 1.15, working distance 0.2 mm), Indo-1 fluorescence ratio images were continuously acquired at 1 Hz before and after application of a mechanical or chemical stimulus. Gentle mechanical stimulation of a single cell in the confocal field (171 × 128 μm) with a glass microelectrode was used to evoke intercellular calcium waves. The efficacy of calcium spread between cells was calculated as the proportion of cells within the field responding with an increase in intracellular calcium during calcium wave transmission. Calcium wave velocity was calculated as the distance between the stimulated and responding cells, divided by the time interval between the half-maximal calcium increases in the stimulated and responding cells. Half-maximal calcium increases were obtained from sigmoidal curves fitted to the ascending phase of each Indo-1 fluorescence ratio increase. In separate experiments, the response of cultures to a panel of P2 receptor agonists was also determined. Suramin (Calbiochem), apyrase, heptanol, 2-methylthioadenosine 5′-triphosphate (2-MeSATP), α,β-methyleneadenosine 5′-triphosphate, pyridoxalphosphate-6-azophenyl-2′,4′-disulfonic acid, tetrasodium, ATP and UTP (all from Sigma-Aldrich) were dissolved in PBS, pH adjusted to 7.4, and used at the concentrations indicated in the text. To ensure that the doses of heptanol and suramin used in these studies were appropriate and specific for their respective pathways, preliminary experiments were performed. Astrocytes were loaded with Fura2 AM, and intracellular calcium levels were measured from ratiometric images (dual excitation at 350 and 380, Em: >450 nm; Metafluor Software, Universal Imaging Corporation, Westchester, PA). Application of 5 μM ATP (approximately EC50 measured in cortical astrocytes: E.S. and S.O.S., unpublished work) evoked a rapid rise in intracellular calcium that slowly recovered during the next minute (data not shown). Subsequent to washes, cells were rinsed with 3 mM heptanol for 2 min, and 5 μM ATP was reapplied; neither response amplitude nor duration was markedly attenuated in the presence of heptanol. Cells were then rinsed for 10 min to remove the heptanol and exposed to 100 μM suramin for 5 min. In the presence of suramin, the response to 5 μM ATP was abolished. Finally, suramin was rinsed from the bath and 5 μM ATP was applied once again, giving rise to an abrupt calcium increase that was similar to responses before treatment and after heptanol. In addition, double whole cell recording studies performed in our laboratories have demonstrated that 100 μM suramin does not appreciably affect junctional coupling, whereas 3 mM heptanol totally uncouples astrocytes (D.C.S. and E.S., unpublished observations).
Northern Blot Analysis.
At indicated times, total RNA was extracted from confluent astrocyte cultures by using Trizol (Life Technologies) and Northern blot analysis for connexin43 (Cx43) was performed as described (15). Rat Cx43 cDNA was kindly supplied by E. C. Beyer (University of Chicago, Chicago). Previous studies have demonstrated that 32P-labeled probes synthesized from this rat Cx43 cDNA template hybridize specifically with their respective human mRNA (21).
SDS/PAGE and Immunoblotting.
Astrocytes grown to confluence in 100-mm tissue culture dishes (Falcon) and treated as described in Fig. 4A were lysed in 200 μl of 10 mM Tris⋅HCl hypotonic lysis buffer (4°C, pH 7.5), sonicated, and centrifuged (10,000 × g, 10 min), and the pellets were resuspended in 2× SDS/PAGE loading buffer and separated by using 12% acrylamide gels, followed by Western blotting onto poly(vinylidene difluoride) membrane (Bio-Rad). Blots were probed with monoclonal mouse anti-human Cx43 antibody (Chemicon) (1:1000) followed by horseradish peroxidase-conjugated goat anti-mouse IgG (Bio-Rad) (1:1000) and developed by using the SuperSignal detection system (Pierce).
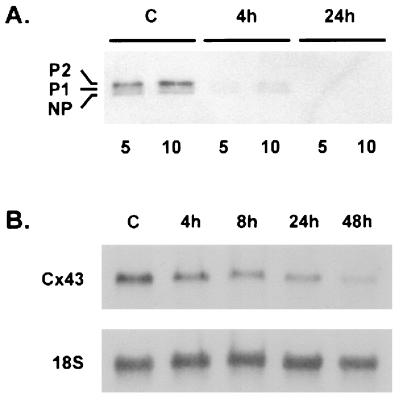
Figure 4.
Effect of IL-1β on Cx43 expression. (A) Immunoblot analysis of Cx43 protein expression. Membrane extracts were prepared from control cultures (C) and cultures that had been treated with IL-1β for 4 or 24 hr. Five-and ten-microgram samples were loaded and subjected to SDS/PAGE and immunoblot analysis performed for Cx43. Note that of the three forms (NP, P1, and P2) detected, P2 predominated, and that treatment with IL-1β resulted in loss of all three forms from the membrane. Data are representative of four independent experiments. (B) Northern blot analysis of Cx43 mRNA expression in astrocytes. Total RNA was extracted from untreated cultures (C) and cultures treated with IL-1β for the times indicated, separated by electrophoresis, and hybridized with a 32P-labeled probe specific for Cx43. The blots were then stripped and reprobed for 18S rRNA. Note that treatment with IL-1β led to progressive down-regulation of the Cx43 signal. Data are representative of four independent experiments.
Electrophysiology.
Recordings of junctional conductance (gj) were obtained from freshly dissociated astrocyte pairs from control or IL-1β-treated cultures. Experiments were performed at room temperature in bathing solution containing 140 mM NaCl, 2 mM CsCl, 2 mM CaCl2, 1 mM MgCl2, 5 mM Hepes, 4 mM KCl, 5 mM dextrose, 2 mM pyruvate, and 1 mM BaCl2, pH 7.2. Each cell of the pair was voltage clamped with heat-polished patch pipettes (3–7 MΩ) filled with internal solution (130 mM CsCl, 0.5 mM CaCl2, 2 mM Na2ATP, 3 mM MgATP, 10 mM Hepes, 10 mM EGTA, pH 7.2); gj was determined by dividing the junctional current by the voltage step applied to the other cell, and is presented as the mean ± SEM (22).
Lucifer Yellow Microinjection.
Astrocytes were plated on uncoated glass microwells (Mat-Tek) at a density of 2.5 × 105 cells/ml. At 7–30 days after plating, confluent control cultures or cultures treated with 10 ng/ml IL-1β for 24 hr were rinsed three times with PBS, and individual cells were iontophoretically injected with a 5% (wt/vol in 150 mM LiCl) solution of Lucifer Yellow dilithium salt (Sigma) through drawn glass capillaries. Cells were photographed 2 min after injection by using FITC epi-illumination and excitation filters.
Semiquantitative Reverse Transcription–PCR.
At indicated times, total RNA was isolated from confluent control or IL-1β-treated cultures by using Trizol. RNA (10 μg per sample) was treated with DNAse-1 (Promega), and first strand cDNA was prepared by using oligo(dT) and the SuperScript II preamplification system (Life Technologies), and amplified directly by PCR using Pfu (Stratagene). As a control, RNA samples were also subjected to the first strand cDNA preparation protocol without reverse transcriptase. Primers were based on the reported corrected sequence of the human P2Y2 receptor (23), and have been published previously (24): forward, 5′-CTC TAC TTT GTC ACC ACC AGC GCG-3′; reverse, 5′-TTC TGC TCC TAC AGC CGA ATG TCC-3′. Primers used to amplify beta-actin were obtained commercially (CLONTECH): forward, 5′-ATC TGG CAC CAC ACC TTC TAC AAT GAG CTG CG-3′; reverse, 5′-CGT CAT ACT CCT GCT TGC TGA TCC ACA TCT GC-3′. The number of cycles at which a linear relationship was maintained between the quantity of input RNA and the final PCR product was determined for P2Y2 and beta-actin. Conditions applied for PCR whereas follows: 95°C for 5 min, followed by 28 cycles of 1 min at 95°C, 1 min at 70°C, and 1 min at 72°C, followed by 72°C for 7 min. Samples were separated by electrophoresis in ethidium bromide-impregnated 1.6% agarose gels.
Cloning of P2Y2 Receptor Gene.
Reverse transcription–PCR of human astrocyte total RNA yielded a single 632-bp fragment, which was excised from a 1.6% agarose gel and purified by using the Geneclean II kit (Bio 101). An adenine nucleotide was added to each end by using Taq (Life Technologies) at 72°C for 10 min, and the fragment was ligated into pCRII-TOPO (InVitrogen) and transformed into ONE-Shot ultracompetent Escherichia coli. Both strands from five independent clones were sequenced.
Statistics.
Statistical analyses were performed using unpaired t tests, and P values < 0.05 were considered significant.
RESULTS
Calcium Waves Are Transmitted Between Human Fetal Astrocytes via Both the Extracellular, P2 Receptor-Mediated Pathway, and the Intercellular, Gap Junction-Mediated Pathway.
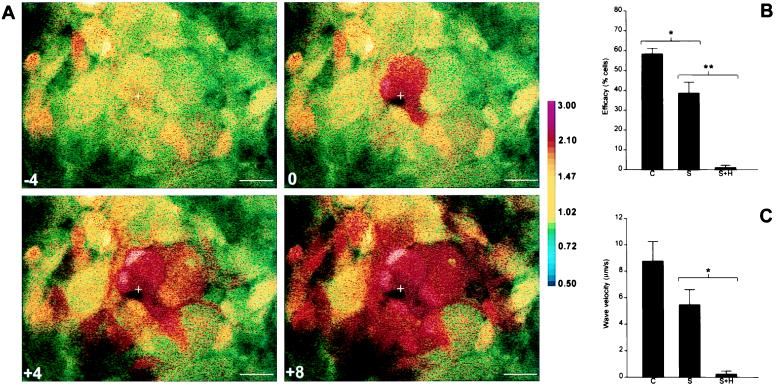
Gentle mechanical stimulation caused a transient increase in intracellular calcium concentration in the stimulated cell, followed by an intercellular calcium wave (Fig. 1A). To determine whether a P2 receptor-mediated pathway contributed to the transmission of calcium waves between cells, the P2 receptor blocker suramin (100 μM) was added to the cultures. The efficacy (Fig. 1B) and velocity (Fig. 1C) of mechanically evoked calcium waves were reduced after addition of suramin. A reduction in calcium wave efficacy and velocity was also observed in cultures treated either with the more selective P2X/P2Y antagonist pyridoxalphosphate-6-azophenyl-2′,4′-disulfonic acid, tetrasodium (50 μM) or with the ATP-degrading enzyme apyrase (5 units/ml at 37°C) (data not shown). To determine whether a gap junction-mediated pathway was responsible for the remaining transmission of calcium waves in suramin-treated cultures, the gap junction blocking agent heptanol (3 mM) was added. Heptanol effectively blocked intercellular calcium wave transmission in suramin-treated cultures (Fig. 1).
Figure 1.
Intercellular calcium wave transmission in cultured human fetal astrocytes. Confluent cultures were loaded with Indo-1AM (A), and a single cell (marked by crosshairs) was mechanically stimulated during ratiometric confocal imaging. The pseudocolor map indicates Indo-1 fluorescence ratio, representative of [Ca2+]i. Time (sec) before and after stimulation is indicated on each panel. (Scale bar = 25 μm.) Data shown are representative of five independent experiments. Efficacy (B) and velocity (C) of wave transmission are shown for representative control cultures, suramin-treated cultures (S), and cultures treated with suramin and heptanol (S+H). Data shown are derived from three separate confluent fields (approximately 15 cells per field) for each condition. ∗, P < 0.05; ∗∗, P < 0.01.
IL-1β Differentially Regulates Calcium Wave Transmission via the P2 Receptor-Mediated Pathway Versus the Gap Junction-Mediated Pathway.
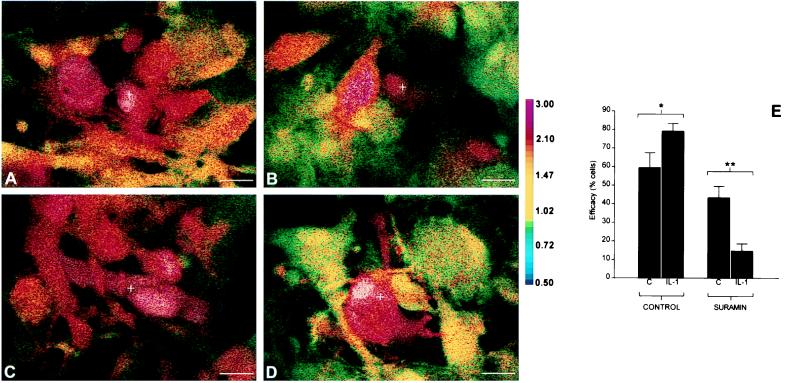
Treatment of cultures with 10 ng/ml IL-1β for 24 hr produced striking differential effects on the efficacy of wave transmission in the absence or presence of suramin. Calcium wave transmission in the absence of suramin was potentiated; the efficacy of wave transmission between cells was increased in IL-1β-treated cultures compared with controls in all experiments (Fig. 2 C versus A, and E). However, addition of suramin to IL-1β-treated cultures immediately produced a marked reduction in efficacy; most strikingly, in all experiments the efficacy of calcium wave transmission after addition of suramin to IL-1β-treated cultures was reduced to a level substantially below that observed after addition of the same concentration of suramin to control cultures (Fig. 2 D versus B, and E). Wave velocity was not altered in IL-1β-treated cultures compared with controls (data not shown). These data indicate that in human fetal astrocytes, treatment with IL-1β caused a potentiation in efficacy of intercellular calcium wave transmission via the suramin-sensitive P2 receptor-mediated pathway, and a reduction in the contribution of the suramin-resistant gap junction-mediated pathway.
Figure 2.
Effect of IL-1β treatment on calcium wave transmission in cultured human fetal astrocytes. Confluent cultures were loaded with Indo-1AM (A–D) and subjected to mechanical stimulation as described for Fig. 1. The spread of calcium wave in control cultures in the absence (A) or presence (B) of suramin is shown. The spread in cultures treated with 10 ng/ml IL-1β for 24 hr in the absence (C) or presence (D) of suramin is also shown. Before stimulation, the Indo-1 fluorescence ratio was between 0.8 and 1.0 for all cells in all conditions, and images shown were captured 10 sec after mechanical stimulation, corresponding to maximal extent of wave spread for all conditions. Treatment with IL-1β had striking differential effects on the efficacy of wave transmission in the absence or presence of suramin (E). Note the potentiation of efficacy of wave transmission by IL-1β (∗, P < 0.05), and the striking reduction in wave transmission via the suramin-insensitive pathway (∗∗, P < 0.01). Data shown are derived from six separate confluent fields (approximately 15 cells per field) for each condition and are representative of three independent experiments.
Down-Regulation of Calcium Wave Transmission via the Gap Junction-Mediated Pathway Is Associated with Down-Regulation of Cx43 mRNA, and Loss of Cx43 Protein and Functional Coupling.
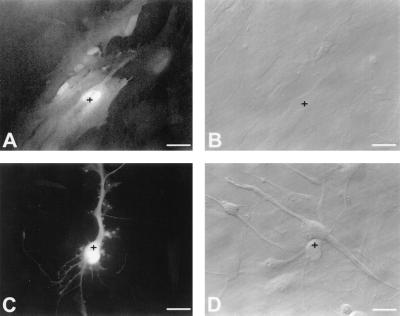
Measurement of junctional conductance using the double whole-cell recording technique demonstrated that the unitary conductance of single gap junction channels in human fetal astrocytes was approximately 60–90 pS, similar to that found in cell types expressing Cx43 (data not shown). Moreover, junctional conductance observed for gap junctions in human fetal astrocytes was relatively voltage insensitive, which is also characteristic of Cx43 (25). In control cultures, mean junctional conductance was 1.69 ± 0.98 nS (n = 12 cell pairs). However, at 24 hr after treatment of cultures with IL-1β, junctional conductance was reduced to 0.061 ± 0.02 nS (n = 14 cell pairs). Microinjection of the gap junction-permeable fluorescent dye Lucifer Yellow was also used to compare gap junction-mediated coupling in control and IL-1β-treated cultures; while in control cultures transfer to 3–5 additional cells was typically observed within 2 min of Lucifer Yellow injection (Fig. 3 A and B), in cultures treated with 10 ng/ml IL-1β for 24 hr, no spread of dye was observed from any injected cell (Fig. 3 C and D). These data indicate that gap junctions composed of Cx43 subunits are the major functional form of gap junction in human fetal astrocytes, and that gap junction-mediated intercellular coupling is markedly reduced after treatment of cultures with IL-1β.
Figure 3.
Effect of IL-1β on dye coupling. Individual cells (marked by crosshairs) in confluent control or IL-1β-treated cultures were iontophoretically injected with Lucifer Yellow and photographed 2 min after injection. A and B show matched fluorescence (A) and phase contrast (B) images of injected control cultures, and C and D show matched images from IL-1β-treated cultures. Note the lack of dye transfer from the injected cell in IL-1β-treated cultures. Note also the change in cell morphology induced by IL-1β. Data shown are representative of three independent experiments, at least four cells injected per condition in each experiment. (Scale bar = 25 μm.)
Immunoblotting experiments using a commercially available anti-Cx43 monoclonal antibody demonstrated the expression of three distinct bands at 42, 44, and 46 kDa on immunoblots of astrocyte homogenates corresponding to the three isoforms of Cx43 (NP, P1, and P2) previously described (26). On immunoblots of preparations of membrane from human fetal astrocytes, levels of the 46-kDa band were substantially higher than levels of the other two bands (Fig. 4A), indicating that the functional form of Cx43 predominates on the plasmalemma of human fetal astrocytes (27). Down-regulation of all three Cx43 bands was observed within 4 hr after treatment with IL-1β, and none of the three bands could be detected by immunoblotting 24 hr after IL-1β treatment. Northern blot analysis of human fetal astrocyte RNA demonstrated a single Cx43 mRNA band identical in size to that previously observed in RNA samples from human cells (21). Progressive down-regulation of Cx43 transcript was observed from 4 hr and continuing beyond 24 hr after IL-1β treatment (Fig. 4B).
Up-Regulation of Calcium Wave Transmission via the P2 Receptor-Mediated Pathway Is Associated with Potentiation of the Efficacy of Response to UTP but Not to 2-MeSATP, and Follows Up-Regulation of P2Y2 mRNA Expression.
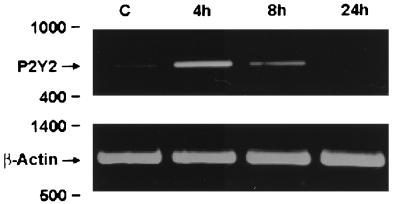
Because our results (Fig. 2) demonstrated that IL-1β treatment for 24 hr led to a potentiation of calcium wave transmission in human fetal astrocytes that could be blocked by P2 receptor antagonists, we compared in confluent control and treated cultures the effect of selective P2 receptor agonists (selective antagonists for the various P2 receptors are not yet available). A marked increase in the number of cells responding to UTP, a selective agonist at P2Y2 and P2Y4 receptors (28, 29), was noted in cultures treated with 10 ng/ml IL-1β for 24 hr compared with controls in all experiments (n = 3 independent experiments, three separate confluent fields analyzed for each condition per experiment). In control cultures, 35 ± 10% of cells responded to 100 μM UTP, compared with 64 ± 11% of cells in IL-1β-treated cultures. In contrast, although 59 ± 6% of cells in control cultures responded to the selective P2Y1 agonist 2-MeSATP [10 μM (30)], no increase in the number of cells responding to 2-MeSATP was noted in IL-1β-treated cultures (55 ± 5% of cells responded). No response to the selective P2X agonist α,β-methyleneadenosine 5′-triphosphate [10 and 50 μM (31)] was observed in control or IL-1β-treated cultures. These data suggest that treatment with IL-1β selectively up-regulates a UTP-responsive pyrimidinergic pathway. To determine whether this reflected the acquisition or up-regulation of a UTP-dependent pyrimidinergic receptor in astrocytes, we examined expression of the P2Y2 receptor in control and IL-1β-treated cells by using a PCR-based method designed to quantitate relative differences in P2Y2 gene expression. In control untreated human fetal astrocytes, a faint band at 632 bp was detected for the P2Y2 receptor, whereas in the IL-1β-treated cultures, a more robust expression was detected 4 hr after stimulation that had decreased by 8 hr and returned to baseline values by 24 hr (Fig. 5). No band was detected in samples subjected to the first strand cDNA preparation protocol in the absence of reverse transcriptase and then used for PCR. To confirm the identity of the PCR product, the band was excised from the gel, cloned, and sequenced. Data obtained from three separate clones gave sequences that were >97% identical to the published corrected sequence of the P2Y2 receptor cloned from human airway and colonic epithelia (23).
Figure 5.
PCR analysis of P2Y2 mRNA expression. Total RNA was extracted from untreated cultures (C) and cultures treated with IL-1β for the times indicated, and subjected to reverse transcription–PCR for P2Y2 and beta-actin as described in the text. Note that treatment with IL-1β led to a transient up-regulation of the signal for the P2Y2 receptor, which was verified by cloning and sequencing. Data are representative of three independent experiments.
DISCUSSION
In this study, we have examined the effect of the proinflammatory cytokine IL-1β on the cell–cell communication pathways utilized by human fetal cortical astrocytes in culture. In rodent systems, it has been shown that astrocytes communicate with each other (and potentially other cell types) via two distinct pathways that mobilize intracellular calcium: a gap junction-mediated pathway (11) and an extracellular pathway mediated by nucleotides such as ATP that activate P2 receptors (12, 13). Our data for human astrocytes are substantially in agreement with these findings and further show that treatment with IL-1β profoundly affects astrocyte-to-astrocyte communication pathways by significantly down-regulating gap junction connectivity and potentiating the P2 receptor-dependent pathway.
In human fetal cortical astrocytes, we found that functional gap junction channels detected in whole cell recording experiments possessed electrophysiological properties characteristic of gap junctions composed of Cx43 subunits. This is consistent with what has been reported for rodent astrocytes, where Cx43 is by far the most abundant connexin (22). In cultures treated with IL-1β, there was a fundamental loss of intercellular gap junction-mediated coupling, as assessed both by double whole-cell recording and dye-coupling studies, loss of Cx43 protein (as assessed by immunoblotting) and down-regulation of Cx43 mRNA (Northern blotting). Kinetic analyses of these events strongly suggested that loss of protein preceded loss of mRNA. Gap junctions allow intercellular exchange of ions and small molecules (32), and given the central role for intercellular small molecule and electrical signaling in CNS function, inactivation of glial gap junction channels may be expected to significantly affect cell–cell communication in the CNS (25). Our observation that intercellular calcium wave transmission occurs in human fetal cortical astrocytes when gap junction pathways have been eliminated is in agreement with studies in Cx43−/− mice (13) and in rat glial cells treated with the sleep-inducing lipid oleamide, a selective inhibitor of Cx43 gap junction-mediated coupling in astrocytes (26). Our results indicate that, while in control cultures gap junctions play a major role in transmission of intercellular calcium waves, in IL-1β-treated cultures, calcium wave transmission relies on the extracellular, P2 receptor-mediated pathway. In this context, we have noted a recent report indicating a role for gap junctions in the transmission of apoptotic signals after an ischemic insult to cells in culture (33). Because IL-1β has been shown to be expressed in ischemic lesions, we suggest that the strongly down-regulatory effects of IL-1β on connexin expression and function of gap junctions may serve to limit the extent of bystander cell death in such lesions.
Analysis of astrocyte responses to P2 agonists in control and IL-1β-treated cultures indicated that treatment with IL-1β selectively potentiated the response to UTP, while having no effect on the response to 2-MeSATP. Because it is not yet possible for most P2 receptors to use traditional methods, such as ligand binding or antibody binding, to study receptor number at the cell surface, we determined whether IL-1β modulated expression of the UTP-sensitive P2Y2 receptor at the mRNA level. The results showed a striking transient up-regulation of mRNA for P2Y2 in IL-1β-treated cells, suggesting that the promoter for this receptor is regulated by cytokine signaling. A similar conclusion has been reached recently in mouse astrocytes (34) and the THP-1 monocyte cell line (35). Specific antagonists for the various P2 receptors are not yet available, and so we were not able to determine whether the up-regulation of P2Y2 mRNA was solely responsible for the potentiation of P2 receptor-mediated intercellular calcium wave transmission we observed in our experiments. However, our results demonstrate that the potentiation of calcium wave transmission induced by IL-1β is accompanied by an increase in mRNA expression of at least one P2Y receptor. In this context, we have noted with interest the recent observation that in the mouse macrophage cell line J774, binding of UTP to pyrimidinoceptors potentiates the activation of the inducible form of nitric oxide synthase (36). We suggest that the selective potentiation of pathways activated by specific nucleotides may alter the repertoire of responses that the cell can make to changes in the extracellular environment. Because a number of studies have also demonstrated interaction of glia and neuronal circuits, modulation of the ability of astrocytes to respond to changes in the extracellular environment may in turn alter the data transmitted from such glial networks to neuronal circuits in the CNS.
In the normal CNS, ATP is a neurotransmitter and as such its release and breakdown in the extracellular space is tightly regulated. Similarly, IL-1 is expressed at very low levels in the normal CNS but can be readily detected in a number of different inflammatory and degenerative conditions. For example, in tissues from patients with multiple sclerosis, a strong signal for IL-1 is detected in both acute and chronic-active lesions, with particularly prominent expression at the lesion edge (37). In Alzheimer’s disease, IL-1β is expressed by activated microglial cells associated with senile plaques (38). In HIV, associated encephalitis immunoreactivity for IL-1β colocalizes with HIV to microglial nodules (39), and in stroke, IL-1β is expressed in the ischemic tissue (17). These studies indicate that IL-1β is expressed both by resident glia and blood-derived macrophages early in the course of a number of pathological conditions, and animal models have also strongly implicated a role for IL-1 in disease pathogenesis in many of these disorders.
In conclusion, we have demonstrated that a suramin-sensitive, P2 receptor-mediated pathway and a suramin-resistant, heptanol-sensitive gap junction-mediated pathway both contribute to calcium wave transmission in primary human fetal astrocyte cultures. Although there has been debate surrounding the relative contribution of the two pathways to calcium wave transmission between mammalian cells, our data indicate that in primary human astrocytes, a single proinflammatory stimulus differentially regulates wave transmission via the P2 receptor-mediated pathway versus the gap junction-mediated pathway. Because in inflammatory conditions of the human CNS IL-1β is produced both by resident glia and by invading cells of the immune system, our data would suggest that inflammatory events may have significant effects on the coordination of astrocytic function and on information processing in the human CNS.
Acknowledgments
We thank Dr. Karen Weidenheim, Director of the Human Fetal Tissue Repository, for tissue collection; Dr. Meng-Liang Zhao and Wa Shen for culture preparations; and Liwei Hua and Marcia Urban for assistance with Northern blotting. This work was supported by U.S. Public Health Service Grants MH55477 (S.C.L.), NS11920 (C.F.B.), T32GM07288 (J.S.H.L.), and NS34931 and NS07512 (D.C.S.); American Paralysis Association Grant 9802-2 (E.S.); National Multiple Sclerosis Society Grant RG2771 (S.C.L.); Fundação de Amparo à Pesquisa do Estado de Sao Paulo Grant 97/2379-2 (S.O.S.); and an Albert Einstein College of Medicine Pathology Department Pilot Project Grant (G.R.J.).
ABBREVIATIONS
- CNS
central nervous system
- Cx43
connexin43
- 2-MeSATP
2-methythioadenosine 5′-triphosphate
References
- 1.Sanderson M J, Charles A C, Boitano S, Dirksen E R. Mol Cell Endocrinol. 1994;98:173–187. doi: 10.1016/0303-7207(94)90136-8. [DOI] [PubMed] [Google Scholar]
- 2.Saez J C, Connor J A, Spray D C, Bennett M V. Proc Natl Acad Sci USA. 1989;86:2708–2712. doi: 10.1073/pnas.86.8.2708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Boitano S, Dirksen E R, Sanderson M J. Science. 1992;258:292–295. doi: 10.1126/science.1411526. [DOI] [PubMed] [Google Scholar]
- 4.Osipchuk Y, Cahalan M. Nature (London) 1992;359:241–244. doi: 10.1038/359241a0. [DOI] [PubMed] [Google Scholar]
- 5.Yuste R, Nelson D, Rubin W, Katz L C. Neuron. 1995;14:7–17. doi: 10.1016/0896-6273(95)90236-8. [DOI] [PubMed] [Google Scholar]
- 6.Cornell-Bell A H, Finkbeiner S M, Cooper M S, Smith S J. Science. 1990;247:470–473. doi: 10.1126/science.1967852. [DOI] [PubMed] [Google Scholar]
- 7.Charles A C, Merrill J E, Dirksen E R, Sanderson M J. Neuron. 1991;6:983–992. doi: 10.1016/0896-6273(91)90238-u. [DOI] [PubMed] [Google Scholar]
- 8.Takeda M, Nelson D J, Soliven B. Glia. 1995;14:225–236. doi: 10.1002/glia.440140308. [DOI] [PubMed] [Google Scholar]
- 9.Dani J W, Chernjavsky A, Smith S J. Neuron. 1992;8:429–440. doi: 10.1016/0896-6273(92)90271-e. [DOI] [PubMed] [Google Scholar]
- 10.Newman E A, Zahs K R. Science. 1997;275:844–847. doi: 10.1126/science.275.5301.844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Finkbeiner S. Neuron. 1992;8:1101–1108. doi: 10.1016/0896-6273(92)90131-v. [DOI] [PubMed] [Google Scholar]
- 12.Hassinger T D, Guthrie P B, Atkinson P B, Bennett M V L, Kater S B. Proc Natl Acad Sci USA. 1996;93:13268–13273. doi: 10.1073/pnas.93.23.13268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Scemes E, Dermietzel R, Spray D C. Glia. 1998;24:65–73. doi: 10.1002/(sici)1098-1136(199809)24:1<65::aid-glia7>3.0.co;2-#. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Parpura V, Basarsky T, Liu F, Jeftinija K, Jeftinija S, Haydon P. Nature (London) 1994;369:744–747. doi: 10.1038/369744a0. [DOI] [PubMed] [Google Scholar]
- 15.Liu J S, Zhao M-L, Brosnan C F, Lee S C. J Immunol. 1996;157:3569–3576. [PubMed] [Google Scholar]
- 16.Breder C D, Dinarello C A, Saper C B. Science. 1988;240:321–324. doi: 10.1126/science.3258444. [DOI] [PubMed] [Google Scholar]
- 17.Hopkins S J, Rothwell N J. Trends Neurosci. 1995;18:83–88. [PubMed] [Google Scholar]
- 18.Lee S C, Liu W, Brosnan C F, Dickson D W. Lab Invest. 1992;67:465–476. [PubMed] [Google Scholar]
- 19.Delhase M, Hayakawa M, Chen Y, Karin M. Science. 1999;284:309–313. doi: 10.1126/science.284.5412.309. [DOI] [PubMed] [Google Scholar]
- 20.Ninomiya-Tsuji J, Kishimoto K, Hiyama A, Inoue J, Cao Z, Matsumoto K. Nature (London) 1999;398:252–256. doi: 10.1038/18465. [DOI] [PubMed] [Google Scholar]
- 21.Kanter H L, Saffitz J E, Beyer E C. J Mol Cell Cardiol. 1994;26:861–868. doi: 10.1006/jmcc.1994.1103. [DOI] [PubMed] [Google Scholar]
- 22.Dermetziel R, Hertzberg E L, Kessler J A, Spray D C. J Neurosci. 1991;11:1421–1432. doi: 10.1523/JNEUROSCI.11-05-01421.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Parr C E, Sullivan D M, Paradiso A M, Lazarowski E R, Burch L H, Olsen J C, Erb L, Weisman G A, Boucher R C, Turner J T. Proc Natl Acad Sci USA. 1994;91:3275–3279. doi: 10.1073/pnas.91.8.3275. , and erratum (1994) 91, 13067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gorodeski G I, Burfeind P, Gan S U, Pal D, Abdul-Karim F W. Am J Physiol. 1998;275:C758–C765. doi: 10.1152/ajpcell.1998.275.3.C758. [DOI] [PubMed] [Google Scholar]
- 25.Spray D C. In: Gap Junctions in the Nervous System. Spray D C, Dermetziel R, editors. New York: R. G. Landes Company and Chapman & Hall; 1996. pp. 39–60. [Google Scholar]
- 26.Guan X, Cravatt B F, Ehring G R, Hall J E, Boger D L, Lerner R A, Gilula N B. J Cell Biol. 1997;139:1785–1792. doi: 10.1083/jcb.139.7.1785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Musil L S, Cunningham B A, Edelman G M, Goodenough D A. J Cell Biol. 1990;111:2077–2088. doi: 10.1083/jcb.111.5.2077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lustig K D, Shiau A K, Brake A J, Julius D. Proc Natl Acad Sci USA. 1993;90:5113–5117. doi: 10.1073/pnas.90.11.5113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Communi D, Pirotton S, Parmentier M, Boeynaems J. J Biol Chem. 1995;270:30849–30852. doi: 10.1074/jbc.270.52.30849. [DOI] [PubMed] [Google Scholar]
- 30.Webb T E, Simon J, Krishek B J, Bateson A N, Smart T G, King B F, Burnstock G, Barnard E A. FEBS Lett. 1993;324:219–225. doi: 10.1016/0014-5793(93)81397-i. [DOI] [PubMed] [Google Scholar]
- 31.King B F, Neary J T, Zhu Q, Wang S, Norenberg M D, Burnstock G. Neuroscience. 1996;74:1187–1196. doi: 10.1016/0306-4522(96)00209-6. [DOI] [PubMed] [Google Scholar]
- 32.Rose C R, Ransom B R. Glia. 1997;20:299–307. doi: 10.1002/(sici)1098-1136(199708)20:4<299::aid-glia3>3.0.co;2-1. [DOI] [PubMed] [Google Scholar]
- 33.Lin J H, Weigel H, Cotrina M L, Liu S, Bueno E, Hansen A J, Hansen T W, Goldman S, Nedergaard M. Nat Neurosci. 1998;1:494–500. doi: 10.1038/2210. [DOI] [PubMed] [Google Scholar]
- 34.Stella N, Estellés A, Siciliano J, Tencé M, Desagher S, Piomelli D, Glowinski J, Prémont J. J Neurosci. 1997;17:2939–2946. doi: 10.1523/JNEUROSCI.17-09-02939.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Martin K A, Kertesy S B, Dubyak G R. Mol Pharmacol. 1997;51:97–108. doi: 10.1124/mol.51.1.97. [DOI] [PubMed] [Google Scholar]
- 36.Chen B-C, Chou C-F, Lin W-W. J Biol Chem. 1998;273:29754–29763. doi: 10.1074/jbc.273.45.29754. [DOI] [PubMed] [Google Scholar]
- 37.Cannella B, Raine C S. Ann Neurol. 1995;37:424–435. doi: 10.1002/ana.410370404. [DOI] [PubMed] [Google Scholar]
- 38.Griffin W S, Stanley L C, Ling C, White L, MacLeod V, Perrot L J, White C L, 3rd, Araoz C. Proc Natl Acad Sci USA. 1989;86:7611–7615. doi: 10.1073/pnas.86.19.7611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Tyor W R, Glass J D, Griffin J W, Becker P S, McArther J C, Bezman L, Griffin D E. Ann Neurol. 1992;31:349–360. doi: 10.1002/ana.410310402. [DOI] [PubMed] [Google Scholar]