Abstract
The anticancer agent 5-fluorouracil (FU) frequently induces cutaneous lupus erythematosus (LE) lesions on sun exposed sites. Based on this observation, we have tried to establish a cutaneous LE model of C57BL/6 J (B6) mice, B6 T cell receptor (TCR)-α–/– mice and B6 TCR-δ–/– mice treated with FU and/or ultraviolet B light (UVBL) in order to clarify the role of T cells and the cytokine profile of cutaneous lupus lesions. Cutaneous LE-like skin lesions could be induced in TCR-α–/– mice with low FU (0·2 mg) plus UVBL, and in B6 mice treated with a high dose of FU (2·0 mg) plus UVBL. In contrast, low FU plus UVBL induced such skin lesions in TCR-δ–/– mice at a very low incidence. Specifically, the skin lesions of TCR-α–/– mice with low FU plus UVBL appeared more rapidly and were more severe than lesions in B6 mice. The former had the common characteristic features of human chronic cutaneous LE such as typical histology, positive IgG at the dermoepidermal junction, low antinuclear antibody and low mortality. Furthermore, a Th1 response was induced in the development of drug-induced cutaneous LE. FU and UVBL-induced cutaneous LE-like eruption is an excellent model for better understanding the pathomechanisms of skin lesion development in LE.
Keywords: drug-induced cutaneous LE, fluorouracil, TCR-α KO mouse, ultraviolet light
INTRODUCTION
Skin lesions are one of the common manifestations in lupus erythematosus disorders such as systemic lupus erythematosus (SLE) and discoid LE (DLE) [1]. The aetiology of cutaneous lupus erythematous (cutaneous LE) is not fully understood, although immune complexes, antibody-dependent cellular cytotoxicity and immunocompetent cells have all been proposed to play a role in the erythematous lesions of SLE, subacute cutaneous LE (SCLE) and neonatal LE (NLE) [2]. The distribution of clinical skin eruptions and histopathological findings suggest that environmental factors including ultraviolet light (UVL) and biological stressors may also be involved [2,3]. In addition cytokines’ effects might be important in the development of cutaneous lupus lesions [4].
It is well known that SLE can be induced by many kinds of drugs such as anti-arrhythmic, antimicrobial, anticonvulsant and antihypertensitive agents [5–8]. In contrast, there is little information regarding drug-induced cutaneous lupus lesions. Previously, we reported that 5-fluorouracil (FU), an anticancer agent for treatment of gastrointestinal adenocarcinoma, frequently induces DLE-like eruptions on sun exposed sites [9]. Because the trigger is clear in drug-induced LE, the model of drug-induced cutaneous LE, if present, has advantages for investigating the disease.
Skin lesions of cutaneous LE have a number of infiltrating T cells, and these T cells and related cytokines are involved in the development of the lesions. Recently, the role of αβ and γδT cells have been investigated with regard to systemic autoimmune diseases [10–13]. γδT cells show a specific tissue distribution in contrast to αβT cells. γδT cells accumulate at epithelial surfaces in the gut, epidermis and reproductive organs, but are present at only a low density in classical lymphoid tissues in humans and mice [14]. Thus γδT cells have been studied and verified to be involved in host defence against infections. However, it remains unclear whether γδT cells are involved in autoimmune skin lesions.
Based on these observations, we designed immunopathological studies to establish a mouse model for drug-induced cutaneous LE using FU and ultraviolet B light (UVBL), and proposed to investigate the mechanism of development of cutaneous lupus.
MATERIALS AND METHODS
Mice
Mice were purchased from the Jackson Laboratory (Bar Harbor, ME, USA). C57BL/6 J (B6) mice, TCR-α–/– mice of the B6 background and TCR-δ–/– mice of the B6 background were used in our experiments [15,16]. These mice were used as the parental strain in order to study the role of αβ or γδT cell in lupus-prone mice [12,13]. All mice were virgin females and 8 weeks old at the start of each experiment.
5-Fluorouracil injection
FU was purchased from Waco Pure Chemical Industries Ltd (Osaka, Japan). FU was dissolved with phosphate-buffered saline (PBS) at two different concentrations, 1 mg/ml (low dose) and 10 mg/ml (high dose). Intraperitoneal injections of 0·2 ml of FU solution were administered to each mouse at the age of 8 weeks, every 2 days for 12 weeks. The dose of FU was calculated on the basis of results in our previous report of human DLE induced by FU [9]. In cases administered the high FU dose, the total FU dose for 12 weeks was almost equal to that of human patients. The total low FU dose was too small to induce DLE-like lesions in human cases.
UV irradiation
The UVBL source was a Torex FL-20S.E UV lamp (Toshiba Medical Supply, Tokyo, Japan). The energy emitted from these lamps is within the UVBL range (260–400 nm), and the peak emission is at 315 nm. The average irradiance of the source was approximately 10 W/m2 at 21-cm distance, as measured by a UVR-305/365, D (II) UV radiometer (Tokyo Optical Co., Japan).
We divided each strain into six groups: PBS injection group, FU (low or high dose) injection groups, UVBL irradiation group and the groups of FU (low or high dose) injection plus UVBL irradiation (Table 1). Among the six groups, as shown in Table 1, mice in groups 4, 5 and 6 were irradiated on dorsal skin (200 mJ/cm2 UVBL irradiation five times a week). The dorsal skin of mice was shaved twice a week. When mice in groups 5 and 6 had received a 12-week FU injection, UVBL irradiation was then initiated along with further FU injections for an additional 12 weeks. Groups 2 and 3 received a 24-week UVBL irradiation in total. Mice were sacrificed at the end of the FU and/or UVBL irradiation period.
Table 1.
Experimental groups
| Group | PBS | Low FU (1 mg/ml) | High FU (10 mg/ml) | UVBL (200mJ/cm2) |
|---|---|---|---|---|
| 1 | + | non | non | non |
| 2 | non | + | non | non |
| 3 | non | non | + | non |
| 4 | non | non | non | + |
| 5 | non | + | non | + |
| 6 | non | non | + | + |
+Treatment done. UVBL irradiation started 12 weeks after the beginning of experiments in each group.
We examined several autoimmune traits including skin eruptions, haematoxylin–eosin (HE) staining of skin and kidney and skin immunoglobulin deposits by direct immunofluorescence (IF) testing. Proteinuria, spleen weight, body weight, antinuclear antibody, anti-DNA antibodies and mRNA expression of several cytokines in the skin were also evaluated.
UVA, as well as UVBL treatment, was performed. The UVA source was a FL 20 SBL UV lamp (Sankyo Electric, Tokyo, Japan). The energy emitted from this lamps was within the UVA range (315–400 nm), and the peak emission was at 360 nm. The UVA irradiation group was irradiated 10 J/cm2 UVA five times a week for 1 month, according to previous methods [17].
Light microscopic observation
Skin specimens were taken from the upper back region because skin lesions appeared on this site on B6 mice and TCR-α–/– mice. The specimens were fixed in 10% formalin and stained with haematoxylin–eosin (HE). The kidney was also examined in the same way.
IF study
Specimens of both skin and kidney were prepared for direct IF examination. All specimens were frozen immediately in OCT embedding medium (Sakura Finetechnical Co. Ltd, Tokyo, Japan) and stored at −80°C until use. Cryostat sections (6 µm thick) were prepared and stained with FITC-conjugated antimouse IgG, IgM and C3 (Jackson Immuno Research, Pennsylvania, USA).
Serum levels of antinuclear antibody (ANA) were determined by the fluoro HEPANA test (MBL, Nagoya, Japan) in which Hep-2 cells were used as substrates. The serum of each mouse was diluted 20-, 40-, 80- and 160-fold. The specimens were evaluated as negative (–: fluorescence was not seen), intermediate (±: fluorescence was seen a little, but the staining pattern was not clear) and positive (+: fluorescence and clear staining pattern). Dilutions exceeding 40-fold were regarded as positive based on results with control mouse sera.
Indirect complement immunofluorescence tests were carried out according to the method of Beutner et al. [18].
The specimens were investigated by a fluorescence microscope (Nikon, Tokyo, Japan) in a ‘blind fashion’ by three researchers (TY, AH and FF) without prior knowledge of specimen identification.
Immunohistochemical studies
For immunohistochemical analyses, frozen skin sections were fixed in acetone for 10 min at 4°C and air-dried. At all incubation steps, sections were washed with Tris-buffered saline (TBS, 0·05 m, pH 7·6). Non-specific binding was blocked by an avidin–biotin blocking kit solution (Vector Laboratories, Burlingame, CA, USA), 2% normal bovine serum (NBS) and 2% normal goat serum (NGS) in TBS. Subsequently, the specimens were incubated with the primary antibodies which were diluted at 1 : 500 in TBS containing 2% NBS at room temperature (RT) for 1 h. After washing, sections were incubated with the secondary antibody, diluted 1 : 150 in TBS containing 4% mouse normal serum and 2% NGS for 30 min at RT. After washing, a routine staining method for avidin–biotin complex labelled with alkaline phosphatase was used (Vector Laboratories) and counterstained with Mayer's haematoxylin. Negative controls were performed by replacement of primary antibody with normal rat IgG (Dianova, Hamburg, Germany). The following antibodies were used as primary antibodies: anti-CD3, anti-CD4 (Pharmingen, San Diego, CA, USA), anti-CD8 (Southern Biotechnology, Birmingham, AL, USA), H57-597 or GL3 (anti-TCR-Cβ and TCR-γδ, Pharmingen). A biotinylated goat antirat IgG (Dianova) was used as the secondary antibody. The number of infiltrating cells was assessed semiquantitatively according to the following scoring system: no positive cells/no aberrant expression (–); staining of isolated cells/involvement of individual epidermis and/or dermis (+); staining of a mild cell infiltrate/involvement of multiple epidermis and/or dermis (+ +); staining of an intense cell infiltrate/involvement of most epidermis and/or dermis (+ + +) [19].
Measurements of anti-DNA antibodies in sera
Enzyme-linked immunosorbent assays (ELISA) for antibodies to double-stranded (ds) DNA and single-stranded (ss) DNA were carried out according to our previous reports [20,21]. An ANA method is described above.
Cytokine analysis
Cytokine analysis was performed according to methods described in our previous report [22]. Each primer was purchased from Kurabo Medical Ltd (Okayama, Japan). Levels of mRNA for tumour necrosis factor (TNF)-α, interferon (IFN)-γ, interleukin (IL)-2, IL-4, IL-10 and IL-12 were determined by reverse transcription–polymerase chain reaction (RT-PCR) in each group of TCR-α–/– mice. After mice were sacrificed, the dorsal skin was removed and frozen immediately in liquid nitrogen. Total RNA was prepared by RNeasy Mini Kit (Qiagen, Tokyo, Japan). Single-stranded cDNA was reverse-transcribed using each RNA sample and amplified by PCR (Takara PCR Thermal Cycler, Takara, Otsu, Japan). PCR was performed by incubation for 5 min at 94°C, followed by 40 cycles each consisting of 1 min at 94°C, 1 min at 60°C and 1·5 min at 72°C. PCR products were electrophoresed on a 2% agarose gel and visualized by ethidium bromide staining. All assays were performed in triplicate according to the manufacturer's instructions.
Proteinuria
Proteinuria was tested to monitor the onset of renal disease by using Eimus urine test paper (Viel-Sankyo, Tokyo, Japan) in examined mice, and was evaluated as negative (0–30 mg/dl), 1 + (30–100 mg/dl), 2 + (100–300 mg/dl) and 3 + (300–1000 mg/dl) [22].
Cell transfer
Spleen cells were obtained when TCR-α–/– mice were sacrificed. T cell-enriched fractions of splenocytes of each experimental group (Table 1) were prepared according to an established method [23,24]. Spleen cells were haemolysed with 0·17 m ammonium chloride at 37°C for 5 min and incubated on a 5-cm plastic dish 37°C for 90 min to remove dish-adherent cells. The non-adherent cells were collected by gentle shaking and applied to nylon wool column to enrich the T cells. The T cell-enriched fraction was used for flow cytometry and transfer experiments. These cells were stained with FITC- and/or PE-conjugated monoclonal antibodies for 30 min at 4°C. After washing three times, the cells were analysed with a flow cytometer (FACScan, Becton Dickinson, Oxnard, CA, USA).
To assess disease induction, non-treated young (2 months old) B6 mice were injected i.v. with 2 × 106γδ T cells (more than 96% purity) from TCR-α–/– mice with UV and/or FU treatment as shown in Table 1. One group of recipient mice was irradiated with UVBL (200 mJ/cm2, five times a week) for 8 weeks, and the other group received no irradiation. Clinical skin lesions were monitored every day.
Statistical analysis
Significance was calculated using the two-tailed Student's t-test; P < 0·05 was estimated to be a significant difference.
RESULTS
Macroscopic findings
The incidence of skin lesion appearance at the end of the experiments is shown in Table 2. Alopecia and erythema skin lesions with slight scaling appeared in TCR-α–/– mice treated with low dose FU plus UVBL (60%), B6 mice treated with high dose FU plus UVBL (80%) and TCR-δ–/– mice treated with low dose FU plus UVBL (15%). In other groups such as B6 mice treated with either high FU (17%) or UVBL (9%) and TCR-δ–/– mice treated with UVBL (25%) a slight scaling was found, but alopecia and erythema were not demonstrated. Knock-out mice treated with high dose FU were excluded from this experiment because many mice died due to the side effects of the agent. Autopsy findings of these mice revealed severe damage of the liver and kidney.
Table 2.
Incidence of skin lesion of examined micea
| C57BL/6 J | TCR-α–/– | TCR-δ–/– | ||||
|---|---|---|---|---|---|---|
| Group | No. examined | Skin lesion | No. examined | Skin lesion | No. examined | Skin lesion |
| 1 PBS | 6 | 0/6 | 6 | 0/6 | 6 | 0/6 |
| 2 Low FU (1 mg/ml) | 11 (1) | 0/10 | 11 (3) | 0/8 | 8 (2) | 0/6 |
| 3 High FU (10 mg/ml) | 25 (13) | 2/12 | n.d. | n.d. | n.d. | n.d. |
| 4 UVBL (200 mJ/cm2) | 11 | 1/11 | 9 (1) | 2/8 | 12 | 3/12 |
| 5 Low FU plus UVBL | 6 | 0/6 | 18 (3) | 9/15 | 13 | 2/13 |
| 6 High FU plus UVBL | 6 (1) | 4/5 | n.d. | n.d. | n.d. | n.d. |
The incidence of skin lesion was estimated when mice were sacrificed. (): number of dead mice during experiments. n.d.: Data were excluded because of high mortality.
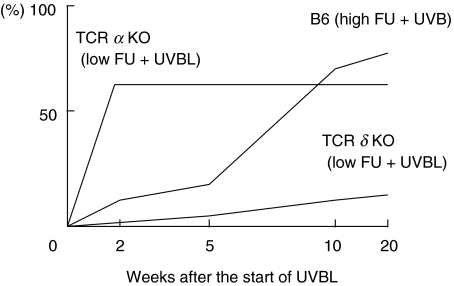
The time-dependent incidence of alopecia and erythema is shown in Fig. 1. TCR-α–/– mice treated with low dose FU plus UVBL developed skin lesions on their backs more rapidly and the lesions were more severe than in the other mouse groups (Fig. 2a). Among B6 mice groups, skin eruptions were found most frequently in the high dose FU plus UVBL group. These skin lesions were similar to those of TCR-α–/– mice treated with low dose FU plus UVBL, but the degree of skin changes was slightly decreased (Fig. 2c). TCR-δ–/– mice had a low incidence of skin lesions (Table 2), and the skin lesions were only slightly erythematous. Mouse groups treated with UVAL alone did not show any skin or systemic manifestations (data not shown).
Fig. 1.
Time-dependent incidence of alopecia and erythema.
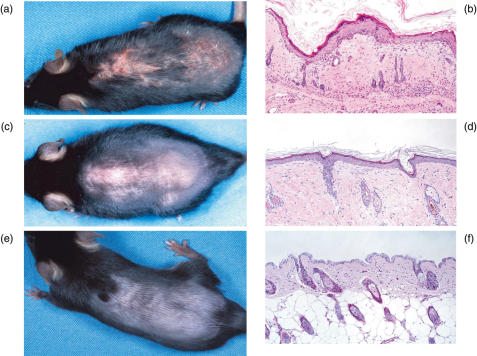
Fig. 2.
Macroscopic and microscopic changes in examined mice. (a) TCR-α–/– mice treated with low dose FU plus UVBL. Erythema and alopecia with scaling were observed. (b) Histological findings in TCR-α–/– mice treated with low dose FU plus UVBL. Hyperkeratosis, acanthosis, plugging, liquefaction-like degeneration and perivascular mononuclear cell infiltration were observed. (c) B6 mouse treated with high FU plus UVBL. Alopecia and slight erythema were observed. The degree of skin changes was slightly decreased compared to that of TCR-α–/– mice in Fig. 2a. (d) Histological findings in a B6 mouse treated with high FU plus UVBL. Hyperkeratosis, acanthosis, plugging and mononuclear cell infiltration were observed, but these changes were much milder than those of TCR-α–/– mice in Fig. 2a,b. (e) Control B6 mouse treated with PBS. (f) Histological findings in a control B6 mouse treated with PBS. Marked changes were not observed.
Histopathological changes
Five characteristic changes, which are common in human DLE, including hyperkeratosis, plugging, acanthosis, liquefaction degeneration and mononuclear cell infiltration were investigated (Table 3). In TCR-α–/– mice treated with low FU plus UVBL, hyperkeratosis, plugging and mononuclear cell infiltration were found in all mice, and acanthosis and liquefaction were observed in 78% and 44%, respectively. The histological findings in B6 mice treated with high FU plus UVBL were similar to those of TCR-α–/– mice treated with low FU plus UVBL. However, as shown in Fig. 2b, d the pathological changes in TCR-α–/– mice treated with low FU plus UVBL were more severe than those in B6 mice treated with high FU plus UVBL. The incidence of these changes in TCR-δ–/– mice treated with low FU plus UVBL were somewhat lower than in B6 and TCR-α–/– mice showing alopecia and erythema.
Table 3.
Histological findings of skin lesion in examined mice
| Mice | Treatment | Hyperkeratosis | Plugging | Acanthosis | Liquefaction degeneration | Mononuclear cell infiltration |
|---|---|---|---|---|---|---|
| C57BL/6 J | 1 PBS | 0 | 0 | 0 | 0 | 0 |
| 2 Low FU (1 mg/ml) | 10 | 10 | 20 | 0 | 10 | |
| 3 High FU (10 mg/ml) | 17 | 25 | 8 | 8 | 67 | |
| 4 UVBL (200 mJ/cm2) | 82 | 45 | 82 | 18 | 73 | |
| 5 Low FU plus UVBL | 100 | 67 | 33 | 33 | 100 | |
| 6 High FU plus UVBL | 80 | 100 | 80 | 60 | 80 | |
| TCR-α–/–mice | 1 PBS | 0 | 0 | 0 | 0 | 0 |
| 2 Low FU (1 mg/ml) | 67 | 50 | 0 | 0 | 83 | |
| 3 High FU (10 mg/ml) | n.d. | n.d. | n.d. | n.d. | n.d. | |
| 4 UVBL (200 mJ/cm2) | 100 | 67 | 50 | 17 | 67 | |
| 5 Low FU plus UVBL | 100 | 100 | 78 | 44 | 100 | |
| 6 High FU plus UVBL | n.d. | n.d. | n.d. | n.d. | n.d. | |
| TCR-δ–/– mice | 1 PBS | 17 | 0 | 17 | 0 | 50 |
| 2 Low FU (1 mg/ml) | 0 | 67 | 67 | 17 | 83 | |
| 3 High FU (10 mg/ml) | n.d. | n.d. | n.d | n.d. | n.d. | |
| 4 UVBL (200 mJ/cm2) | 42 | 42 | 58 | 25 | 83 | |
| 5 Low FU plus UVBL | 67 | 75 | 100 | 58 | 92 | |
| 6 High FU plus UVBL | n.d. | n.d. | n.d. | n.d. | n.d. |
Specimens were obtained from the back of killed and/or autopsied mice and stained with haematoxylin eosin solution. Each figure represents the percentage (%). n.d.: Data were excluded because of high mortality. Five to 14 mice were examined in each treatment.
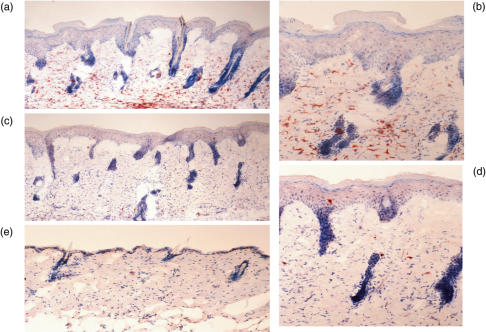
In TCR-α–/– mice treated with low FU plus UVBL, a significant number of CD4+ T cells appeared in the dermis (Fig. 3a, b). The infiltration score ranged from mild (+ +) to intense (+ + +). There was no infiltration of CD4+ T cells in the epidermis. A small amount of CD8+ T cell infiltration was also seen in the dermis (score +) (Fig. 3c, d). No positive cells were found using the anti-TCR-Cβ antibody. Most cells infiltrating cells showed a negative reaction against the anti-TCR-γδ antibody, although a small number had weak positivity from (+/–) to (+). Control TCR-α–/– mice showed no marked infiltration of CD4+ (Fig. 3e) and CD8+ T cells in the skin. Infiltration of CD3+ cells was similar to that of CD4+ cells, and the infiltration score was highest in TCR-α–/– mice treated with low FU plus UVBL.
Fig. 3.
Photographs of immunohistochemical staining. (a, b) CD4+ T cells in TCR-α–/– mice treated with low dose FU plus UVBL (a; × 100, b; × 200). (c, d) CD8+T cells in TCR-α–/–mice treated with low dose FU plus UVBL (c; × 100, d; × 200). (e) CD4+T cells in control TCR-α–/– mice ( × 100).
Subepidermal immunoglobulin deposits
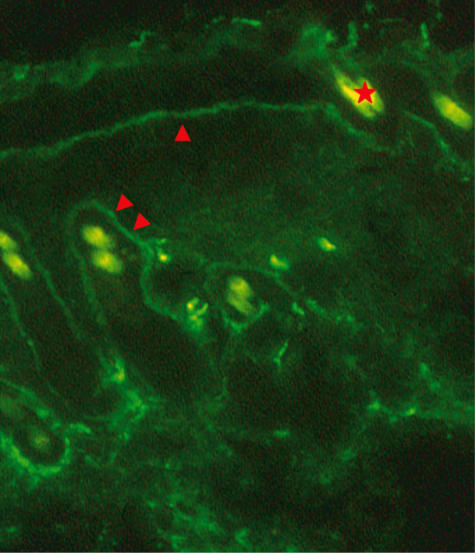
As immunoglobulin deposits at the dermo–epidermal junction (DEJ) is a common and specific finding in human DLE, a direct IF study was performed. As shown in Table 4, TCR-α–/– mice treated with low FU plus UVBL showed a high incidence of IgG deposition at the DEJ. In some specimens IgG was observed at the basement membrane zone (BMZ) of the hair follicle as well as at the DEJ (Fig. 4). Nuclear staining as found in aged MRL/n mice [21] was not found in any of the mice. A similarly high incidence of IgG deposition was also found in B6 mice treated with high FU plus UVBL. IgG deposition was always associated with C3. IgM deposition was observed frequently in mice, irrespective of the absence or presence of skin eruptions. Indirect complement immunofluorescence tests were negative.
Table 4.
Positive rate of subepidermal immunoglobulin deposition in examined mice
| C57BL/6 J | TCR-α–/– | TCR-δ–/– | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Group | No. examined | IgG (%) | IgM (%) | No. examined | IgG (%) | IgM (%) | No. examined | IgG (%) | IgM (%) |
| 1 PBS | (6) | 0 | 0 | (6) | 0 | 0 | (6) | 0 | 0 |
| 2 Low FU (1 mg/ml) | (7) | 0 | 0 | (6) | 66 | 83 | (6) | 33 | 66 |
| 3 High FU (10 mg/ml) | (7) | 14 | 29 | (0) | n.d. | n.d. | (0) | n.d. | n.d. |
| 4 UVBL (200 mJ/cm2) | (6) | 17 | 17 | (6) | 0 | 100 | (6) | 33 | 100 |
| 5 Low FU plus UVBL | (6) | 66 | 100 | (8) | 100 | 100 | (7) | 14 | 100 |
| 6 High FU plus UVBL | (5) | 80 | 80 | (0) | n.d. | n.d. | (0) | n.d. | n.d. |
n.d.: Data were excluded because of high mortality.
Fig. 4.
Fluorescence photograph of IgG deposition. IgG deposition is observed at the dermo–epidermal junction (arrowhead) and at the basement membrane zone of the hair follicle (double arrowheads). Autofluorescence of hair is seen, one of which is indicated by an asterisk (original magnification × 100).
Antinuclear antibody (ANA) and anti-DNA antibodies in sera
As shown in Table 5, among the TCR-α–/– mice treated with low FU plus UVBL 38% were positive for IgG ANA and 13% for IgM ANA. In contrast, B6 mice treated with high FU plus UVBL, which showed lupus-like skin lesions, had no ANA in their sera. ANA was not associated with the absence or presence of skin eruptions. Table 6 shows the levels of anti-DNA antibodies (IgG). Anti-dsDNA antibody was not observed in any groups. Anti-ssDNA antibody levels were slightly elevated in mice treated with UVBL or UBL plus low FU. However, these titres were extremely low when compared with those of autoimmune-prone MRL/lpr mice.
Table 5.
Positive rate of ANA in serum of examined mice
| C57BL/6 J | TCR-δ–/– | TCR-α–/– | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Group | No. examined | IgG (%) | IgM (%) | No. examined | IgG (%) | IgM (%) | No. examined | IgG (%) | IgM (%) |
| 1 PBS | (6) | 0 | 0 | (6) | 33 | 0 | (6) | 0 | 0 |
| 2 Low FU (1 mg/ml) | (7) | 0 | 14 | (8) | 25 | 0 | (6) | 17 | 0 |
| 3 High FU (10 mg/ml) | (10) | 10 | 10 | (0) | n.d. | n.d. | (0) | n.d. | n.d. |
| 4 UVBL (200 mJ/cm2) | (10) | 20 | 0 | (6) | 33 | 17 | (8) | 13 | 0 |
| 5 Low FU plus UVBL | (6) | 50 | 17 | (8) | 38 | 13 | (11) | 45 | 9 |
| 6 High FU plus UVBL | (5) | 0 | 0 | (0) | n.d. | n.d. | (0) | n.d. | n.d. |
n.d.: Data were excluded because of high mortality.
Table 6.
Anti-DNA antibodies (Ig G) in sera of examined mice
| C57BL/6 J | TCR-α–/– | TCR-δ–/– | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Group | No. examined | dsDNA | ssDNA | No. examined | dsDNA | ssDNA | No. examined | dsDNA | ssDNA |
| 1 PBS | (6) | <0·05 | <0·05 | (6) | <0·05 | <0·05 | (6) | <0·05 | <0·05 |
| 2 Low FU (1 mg/ml) | (7) | <0·05 | <0·05 | (8) | <0·05 | <0·05 | (6) | <0·05 | <0·05 |
| 3 High FU (10 mg/ml) | (10) | <0·05 | <0·05 | (0) | n.d. | n.d. | (0) | n.d. | n.d. |
| 4 UVBL (200 mJ/cm2) | (10) | <0·05 | 0·11 (0·07) | (6) | <0·05 | 0·21 (0·11) | (8) | <0·05 | 0·17 (0·09) |
| 5 Low FU plus UVBL | (6) | <0·05 | 0·07 (0·14) | (8) | <0·05 | 0·18 (0·09) | (11) | <0·05 | 0·16 (0·15) |
| 6 High FU plus UVBL | (5) | <0·05 | <0·05 | (0) | n.d. | n.d. | (0) | n.d. | n.d. |
n.d.: Data were excluded because of high mortality. The levels of anti-DNA antibodies are expressed as the mean values of the optical density (standard deviation). Positive controls (pooled sera form aged MRL/lpr mice) showed 0·24 (0·09) for antidsDNA antibody and 1·29 (0·43) for antissDNA antibody.
Analysis of cytokine mRNA expression in the skin by the RT-PCR method
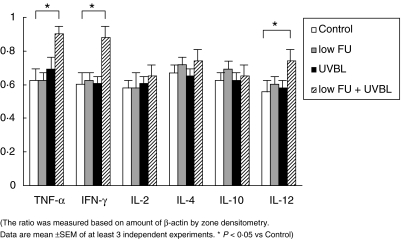
In each TCR-α–/– group, significant differences in mRNA expression were seen (Fig. 5). Low FU plus UVBL treated mice showed predominantly Th1 type cytokine production. Specifically, the transcriptional levels of TNF-α, IFN-γ and IL-12 were remarkably increased in this group. TGF-β, IL-10 and IL-18 were induced in each group but there were no differences between the examined mice (data not shown).
Fig. 5.
Expression of cytokine mRNA in the skin of TCR-α–/– mice by the RT-PCR method. The ratio was meausred based on amount of β-actin by zone densitometry. Data are mean ±s.e.m. of at least three independent experiments. *P < 0⋅05 versus control.
Proteinuria
There was no evidence of proteinuria in the examined groups (data not shown).
Body weight
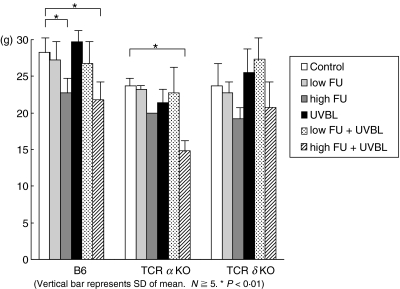
As shown in Fig. 6, high FU induced a significant reduction in body weight among the examined mice. TCR-α–/– mice that received low FU plus UVBL showed similar body weights to control TCR-α–/– mice.
Fig. 6.
Body weight changes of examined mice. Vertical bar represents s.d. of mean. N = ≥5; P < 0·01.
Cell transfer
Skin lesions were not induced in our experimental systems, irrespective of UVBL irradiation, in which non-treated young (2 months old) B6 mice were injected i.v. with 2 × 106γδT cells (more than 96% purity) from TCR-α–/– mice with UV and/or FU treatment, as shown in Table 1.
DISCUSSION
The aetiology and pathogenesis of autoimmune diseases cannot be analysed readily without appropriate animal models, although no single animal model mimics a human disease perfectly. Animal models are used commonly to study the genetic, environmental and pathogenic aspects of autoimmune diseases. There are several experimental groups, including the inbred mice, which develop spontaneously a disease similar to human SLE, the chronic graft versus host diseases induced in F1 hybrid mice injected with lymphoid parental cells, UVL-irradiated mice immunized with some components of DNA, immunodeficient mice inoculated or engrafted with immunocompetent cells or tissues and gene-manipulated mice such as transgenic or knock out mice [25,26]. In fact, experimental models and inbred lupus-prone mice are tools that enable a better understanding of the photosensitivity or photocytotoxicity phenomena in relation to autoimmunity models [26,27]. Genetic studies of MRL/lpr mice revealed that the appearance of macroscopic LE-like skin lesions needs the lpr mutation plus an additional factor in an autosomal-dominant fashion [18]. The candidate for the additional factor is UVBL light, susceptibility to which is regulated by the genetic background [17, 21, 28].
Skin lesions of cutaneous LE have a number of infiltrating T cells, and it is these T cells and the related cytokines that are involved in the development of the lesions. Recently, the role of αβ and γδT cells has been investigated with regard to systemic autoimmune diseases [10–13]. Therefore, we considered that the TCR knock-out mouse might be a good model to investigate the role of T cells and cytokines in the pathogenesis of cutaneous LE as the trigger is clear in drug-induced LE, unlike the situation in spontaneously autoimmune mice.
In our present studies, cutaneous LE-like skin lesions could be induced in TCR-α–/– mice with low FU plus UVBL, and in B6 mice treated with a high dose of FU plus UVBL. In contrast, low FU plus UVBL induced such skin lesions in TCR-δ–/– mice at only a very low incidence. Specifically, the skin lesions of TCR-α–/– mice treated with low FU plus UVBL appeared more rapidly and were more severe than the lesions in B6 mice. Furthermore, the former had the common characteristic features of chronic cutaneous LE, such as typical histology, positive IgG at the DEJ, low ANA and low mortality [1]. Therefore, we considered that TCR-α–/– mice may be a better drug-induced cutaneous lupus model than B6 mice in our present system.
FU is an antimetabolite agent, which is converted to FdUMP and FUTP in vivo[29]. FU inhibits the synthesis of DNA and induces dysfunctional RNA. It is likely that FU affects the basal layer of the epidermis, which is the primary location of mitotically active cells in the epidermis [30]. The first step in the development of the present drug-induced cutaneous LE is injury to the basal cells induced by FU. However, FU alone induced no marked changes either clinically or histologically.
The involvement of UVBL in this mouse model is very similar to the idiopathic and/or drug-induced human DLE lesions observed previously because the skin lesions in these patients appear on sun exposed sites [1,9]. UVBL has several adverse effects, such as photocarcinogenesis, local and systemic immune suppression as well as immunomodulatory effects [31,32]. The close association of UVBL and cutaneous LE has been reported in humans as well as mouse models [27]. UVBL is considered to be one of the major triggers of pathogenesis through the translocation of SS-A/Ro antigens to the surface of keratinocytes [33]. In our study, mice treated with UVBL alone showed no remarkable clinical changes, although some pathological changes were seen in these mice. For the induction of cutaneous LE-like lesions, both FU and UVBL were required continuously in B6 and TCR-α–/– mice, which was also confirmed by the results of cell transfer experiments. Therefore, it is likely that the effects of UVBL on keratinocytes damaged by FU are of primary importance in the development of drug-induced cutaneous LE.
In this study, the dramatic formation of cutaneous LE-like lesions was observed in low FU- and UVBL-treated TCR-α–/– mice, suggesting that αβT cells are required to protect against the development of drug-induced cutaneous LE. This assumption is supported by the observation that only weak formation of cutaneous LE-like skin lesions was seen in low FU- and UVBL-treated TCR-δ–/– mice. Regulatory T (Tr) cells, which have a CD4+ CD25+ TCR-αβ+ phenotype, are the candidate suppressor cells in the process of cutaneous LE formation. It has been reported that Tr cells act as inhibitors in the development of autoreactive Th1 cells in experimental tissue-specific autoimmune diseases [34] and in the induction of tumoricidal immunity mediated by Th1 cells and CTLs [24]. In addition, in vitro induction of Tr cells using UVBL-treated dendritic cells (DCs) shows clearly that UVBL and Tr cell induction are closely related [35]. Further examinations using CD4KO mice are necessary to clarify the role this down-regulatory mechanism has on the generation of drug-induced cutaneous LE.
γδT cells play an important role in many immunological phenomena such as host defence against infections [36] and the down-regulation of epithelial malignancy [37]. Concerning autoimmune diseases, a lupus-like phenomenon in Fas-defect MRL mice has been attributed strongly to αβT cell proliferation and autoantibody production [38–40]. However, recent studies have demonstrated significant roles for non-αβT cells in systemic autoimmune disease [10, 11, 41]. In our studies, the TCR-α–/– mouse was more likely to develop drug-induced cutaneous LE than the wild-type B6 mice. This suggests that γδT cells were accelerators or αβT cells were decelerators in the development of cutaneous LE. UVBL may potentially enhance the functions of γδT cells. Similarly, an impaired balance of γδT cells and αβT cells might be associated with the development of cutaneous LE. γδT cells have also been reported to participate in both the regulation and propagation of murine lupus [12]. In addition to the regulatory role in autoimmunity, γδT cells classified into the Th1-, Th2- or Tr-type are generally known to participate in immunoregulation of oral tolerance, contact hypersensitivity, transplantation, bacterial infection and tumour regression [42]. Thus γδT cells perform complex modulatory functions in the pathogenesis of systemic autoimmunity, which involves the up- and down-regulation of both pathogenic αβT cells and pathogenic autoantibody-producing B cells. In the development of drug-induced cutaneous LE, TCR-α–/– mice might develop autoimmune skin lesions predominantly through the function of γδT cells. In fact certain T-cell subsets, including Thy-1+ dendritic epidermal cells, have been reported to be involved in the spontaneous development of LE-like skin lesions in autoimmune-prone MRL/lpr mice [43].
Immune staining of drug-induced cutaneous LE in TCR-α–/– mice revealed mild to intense CD4+ T cell infiltration in the dermis and revealed the presence of CD8+ T cells in the basal cell layer, which accompany liquefaction degeneration. In human skin lesions of LE, the proportion of CD8+ T cells was two- to threefold higher than that of CD4+ T cells in the lesions of SLE. In contrast, a predominance of CD4+ T cells was observed in DLE/chronic cutaneous LE [44,45]. Although it is possible that CD8+ cytotoxic T cells were activated by antigens derived from damaged keratinocytes, our study suggests that both CD4+ CD8-γδT cells and CD4- CD8+ γδT cells would be induced in the pathomechanisms of drug-induced cutaneous LE.
γδT cells have been shown to accumulate in the skin lesions of chronic cutaneous LE [46] and also in clinically normal skin of SLE patients [47]. Furthermore, the percentage of γδT cells correlated with the activity of the SLE [47]. As γδT cells recognize antigens expressed by injured keratinocytes [48] and dendritic epidermal T cells recognize self-antigens in immune surveillance [49], it is likely that the γδT cells in the present study directly recognize the antigens of keratinocytes damaged by FU and UVBL, and thereby participate in the development of cutaneous LE-like lesion.
Most studies dealing with immunomodulation by UVBL have indicated that Th1 mediated immune responses are particularly sensitive to UVBL exposure [50,51]. The effects of UVBL exposure on Th-2-mediated immune responses have received limited consideration and the few results available are somewhat contradictory. UVBL exposure has been found to suppress both Th1- and Th2-mediated immune responses [52]. However, in our experiments, a Th1 response was induced in the development of drug-induced cutaneous LE. DLE/chronic cutaneous LE in humans is associated with Th1 cytokines characterized by the expression of IL-2 and IFN-γ[53]. Our experimental observations regarding cytokines are in accordance with the results reported in humans. The TNF-α in drug-induced cutaneous LE may be produced by UVBL-irradiated keratinocytes; however, IFN-γ is not considered to be produced by keratinocytes. It is possible that the production of IL-12 produced by keratinocytes or γδT cells may promote Th1-type mediated immune responses. As a result, such eruptions would be more likely to develop in TCR-α–/– mice than B6 mice. In addition to Th1-type cytokines, Th2 and other cytokines including TNF-α, IL-1 and TGF-β may be induced in murine lupus. The differences in the cytokine types could be accounted for on the basis of variations in study design, including methods of administration, dose and timing of treatment initiation [54].
We believe that the FU and UVBL-induced cutaneous LE-like eruptions are an excellent model for understanding better the pathomechanisms of development of discoid lesions in LE, and that further study may provide new insights into autoimmune interface dermatitis.
Acknowledgments
This work was supported in part by the Japanese Ministry of Welfare, Health and Labor and Cosmetology Foundation.
REFERENCES
- 1.Elder D, Elenitsas R, Jaworsky C, et al. Connective tissue diseases. In: Elder D, Elenitsas R, Jaworsky C, Johnson B Jr, editors. Lever's histopathology of the skin. 8. Philadelphia: Lippincott-Raven Publishers; 1997. pp. 253–85. [Google Scholar]
- 2.Norris DA. Pathomechanisms of photosensitive lupus erythematosus. J Invest Dermatol. 1993;100:58s–68s. doi: 10.1111/1523-1747.ep12355599. [DOI] [PubMed] [Google Scholar]
- 3.Hasen T, Nyberg F, Stephansson E, et al. Photosensitivity in lupus erythematosus, UV photoprovocation results compared with history of photosensitivity and clinical findings. Br J Dermatol. 1997;136:699–705. [PubMed] [Google Scholar]
- 4.Stein LF, Saed GM, Fivenson DP. T-cell cytokine network in cutaneous lupus erythematosus. J Am Acad Dermatol. 1997;36:191–6. doi: 10.1016/s0190-9622(97)70279-2. [DOI] [PubMed] [Google Scholar]
- 5.Hoffman BJ. Sensitivity to sulfadazine resembling acute disseminated lupus erythematosus. Arch Dermatol Syph. 1945;51:190–2. [Google Scholar]
- 6.Gold S. Role of sulfonamides and penicillin in the pathogenesis of systemic lupus erythematosus. Lancet. 1951;i:268–72. doi: 10.1016/s0140-6736(51)93319-3. [DOI] [PubMed] [Google Scholar]
- 7.Pavlidakey GP, Hashimoto K, Heller GL, et al. Chlorpromazine-induced lupus-like disease: case report and review of the literature. J Am Acad Dermatol. 1985;13:109–15. doi: 10.1016/s0190-9622(85)70152-1. [DOI] [PubMed] [Google Scholar]
- 8.Monestier M, Kotzin BL. Antibodies to histones in systemic lupus erythematosus and drug-induced lupus syndromes. Rheum Dis Clin North Am. 1992;18:415–36. [PubMed] [Google Scholar]
- 9.Yoshimasu T, Hiroi A, Uede K, et al. Discoid lupus erythematosus (DLE)-like lesion induced by uracil-tegafur (UFT) Eur J Dermatol. 2001;11:54–7. [PubMed] [Google Scholar]
- 10.Peng SL, Madaio MP, Hughes DPM, et al. Murine lupus in the absence of αβ T cells. J Immunol. 1996;156:4041–9. [PubMed] [Google Scholar]
- 11.Peng SL, Cappadona J, Mcniff JM, et al. Pathogenesis of autoimmunity in αβ T cell-deficient lupus-prone mice. Clin Exp Immunol. 1998;111:107–16. doi: 10.1046/j.1365-2249.1998.00424.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Peng SL, Madaio MP, Hayday AC, et al. Propagation and regulation of systemic autoimmunity by γ/δT-cells. J Immunol. 1996;157:5689–98. [PubMed] [Google Scholar]
- 13.Fujii T, Okada M, Craft J. Regulation of T cell-dependent autoantibody production by a γ/δ T cell line derived from lupus-prone mice. Cell Immunol. 2002;217:23–35. doi: 10.1016/s0008-8749(02)00509-9. [DOI] [PubMed] [Google Scholar]
- 14.Ziegler HK, Skeen MJ, Pearce KM. Role of α/βΤ and γ/δΤ cells in innate and acquired immunity. Ann NY Acad Sci. 1994;730:53–70. doi: 10.1111/j.1749-6632.1994.tb44239.x. [DOI] [PubMed] [Google Scholar]
- 15.Mombaerts P, Clarke AR, Rudnicki MA, et al. Mutations in T-cell antigen receptor genes alpha and beta block thymocyte development at different stages. Nature. 1992;360:225–31. doi: 10.1038/360225a0. [DOI] [PubMed] [Google Scholar]
- 16.Itohara S, Mombaerts P, Lafaille J, et al. T cell receptor δ gene mutant mice: independent generation of αβ T cells and programmed rearrangements of γδ TCR genes. Cell. 1993;72:337–48. doi: 10.1016/0092-8674(93)90112-4. [DOI] [PubMed] [Google Scholar]
- 17.Horiguchi Y, Furukawa F, Ohshio G, et al. Effects of ultraviolet light irradiation on the skin of the MRL/1 mice. Arch Dermatol Res. 1987;279:478–83. doi: 10.1007/BF00412595. [DOI] [PubMed] [Google Scholar]
- 18.Beutner EH, Kumar V, Krasny SA, Chorzelski TP. Defined immunofluorescence in immunodermatology. In: Beutner EH, Kumar V, Chorzelski TP, editors. Immunopathology of the skin. 3. New York: John Wiley & Sons; 1987. pp. 3–40. [Google Scholar]
- 19.Wakita H, Takigawa M. E-selectin and vascular cell adhesion molecule-1 are critical for initial trafficking of helper-inducer/memory T cells in psoriatic plaques. Arch Dermatol. 1994;130:457–63. [PubMed] [Google Scholar]
- 20.Furukawa F, Ohshio G, Imamura S. Pathogenesis of lupus dermatoses in autoimmune mice. XIX. Attempts to induce subepidermal immunoglobulin deposition in MRL/Mp-+/+ mice. Arch Dermatol Res. 1993;285:20–6. doi: 10.1007/BF00370818. [DOI] [PubMed] [Google Scholar]
- 21.Furukawa F, Kanauchi H, Wakita H, et al. Spontaneous autoimmune skin lesions of MRL/n mice: autoimmune disease-prone genetic background in relation to Fas-defect MRL/lpr mice. J Invest Dermatol. 1996;107:95–100. doi: 10.1111/1523-1747.ep12298305. [DOI] [PubMed] [Google Scholar]
- 22.Ito T, Seo N, Yagi H, et al. Unique therapeutic effects of the Japanese-Chinese herbal medicine, Sairei-to, on Th1/Th2 balance of the autoimmunity of MRL/lpr mice. J Dermatol Sci. 2002;28:198–210. doi: 10.1016/s0923-1811(01)00161-x. [DOI] [PubMed] [Google Scholar]
- 23.Seo N, Tokura Y, Furukawa F, Takigawa M. Down-regulation of tumoricidal NK and NK T cell activities by MHC Kb molecules expressed on Th2-type γδ and αβ T cells coinfiltrating in early B16 melanoma lesions. J Immunol. 1998;161:4138–45. [PubMed] [Google Scholar]
- 24.Seo N, Hayakawa S, Takigawa M, et al. Interleukin-10 expressed at early tumour sites induces subsequent generation of CD4; T-regulatory cells and systemic collapse of antitumour immunity. Immunology. 2001;103:449–57. doi: 10.1046/j.1365-2567.2001.01279.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Izui S, Merino R, Iwamoto M, et al. Mechanisms of genetic control of murine systemic lupus erythematosus. Springer Semin Immunopathol. 1994;16:133–52. doi: 10.1007/BF00197514. [DOI] [PubMed] [Google Scholar]
- 26.Furukawa F. Photosensitivity in cutaneous lupus erythematosus: lessons from mice and men. J Dermatol Sci. 2003;33:81–9. doi: 10.1016/j.jdermsci.2003.08.005. [DOI] [PubMed] [Google Scholar]
- 27.Furukawa F. Animal models of cutaneous lupus erythematosus and lupus erythematosus photosensitivity. Lupus. 1997;6:193–202. doi: 10.1177/096120339700600215. [DOI] [PubMed] [Google Scholar]
- 28.Furukawa F, Lyons MB, Norris DA. Susceptible cytotoxicity to ultraviolet B light in fibroblasts and keratinocytes cultured from autoimmune-prone MRL/Mp-lpr/lpr mice. Clin Immunol Immunopathol. 1989;52:460–72. doi: 10.1016/0090-1229(89)90160-8. [DOI] [PubMed] [Google Scholar]
- 29.Takiuchi H, Ajani JA. Uracil-Tegafur in gastric carcinoma: a comprehensive review. J Clin Oncol. 1998;16:2877–85. doi: 10.1200/JCO.1998.16.8.2877. [DOI] [PubMed] [Google Scholar]
- 30.Bickenbach JR. Identification and behavior of label-retaining cells in oral mucosa and skin. J Dent Res. 1981;60:1611–20. doi: 10.1177/002203458106000311011. [DOI] [PubMed] [Google Scholar]
- 31.McMichael AJ. Does immunosuppressive ultraviolet radiation explain the latitude gradient for multiple sclerosis? Epidemiology. 1997;8:642–5. doi: 10.1097/00001648-199710000-00005. [DOI] [PubMed] [Google Scholar]
- 32.Kripke ML, Cox PA, Alas LG, et al. Pyrimidine dimers in DNA initiate systemic immunosuppression in UV-irradiated mice. Proc Natl Acad Sci USA. 1992;89:7516–20. doi: 10.1073/pnas.89.16.7516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Furukawa F, Itoh T, Wakita H, et al. Keratinocytes from patients with lupus erythematosus show enhanced cytotoxicity to ultraviolet radiation and to antibody-mediated cytotoxicity. Clin Exp Immunol. 1999;118:164–70. doi: 10.1046/j.1365-2249.1999.01026.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Groux H, O'Garra A, Bigler M, et al. A CD4+ T-cell subset inhibits antigen-specific T-cell responses and prevents colitis. Nature. 1997;389:737–42. doi: 10.1038/39614. [DOI] [PubMed] [Google Scholar]
- 35.Simon JC, Hara H, Denfeld RW, et al. UVBL-irradiated dendritic cells induce nonproliferating, regulatory type T cells. Skin Pharmacol Appl Skin Physiol. 2002;15:330–4. doi: 10.1159/000064537. [DOI] [PubMed] [Google Scholar]
- 36.Rosat JP, MacDonald HR, Louis JA. A role for γδ T-cells during experimental infection of mice with Leishmania major. J Immunol. 1993;150:550–5. [PubMed] [Google Scholar]
- 37.Girardi M, Oppenheim DE, Steele CR, et al. Regulation of cutaneous malignancy by γδ T cells. Sciencexpress. 2001;20:1–5. doi: 10.1126/science.1063916. [DOI] [PubMed] [Google Scholar]
- 38.Santoro TJ, Portanova JP, Kotzin BL. The contribution of L3T4+ T cells to lymphoproliferation and autoantibody production in MRL-lpr/lpr mice. J Exp Med. 1988;167:1713–8. doi: 10.1084/jem.167.5.1713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Jevnika AM, Grusby MJ, Glimcher LH. Prevention of nephritis in major histocompatibility complex class II-deficient MRL-/lpr mice. J Exp Med. 1994;179:1137–43. doi: 10.1084/jem.179.4.1137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Koh DR, Ho A, Rahemutulla A, et al. Murine lupus in MRL/lpr mice lacking CD4 or CD8 T cells. Eur J Immunol. 1995;25:2558–62. doi: 10.1002/eji.1830250923. [DOI] [PubMed] [Google Scholar]
- 41.Rajagopalan S, Zordan T, Tsokos GC, et al. Pathogenic anti-DNA autoantibody-inducing T helper cell lines from patients with active lupus nephritis: isolation of CD4-8- T helper lines from patients with active antigen receptor. Proc Natl Acad Sci USA. 1990;87:7020–4. doi: 10.1073/pnas.87.18.7020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Seo N. Suppressor lymphocytes participating in collapse of antitumor immunity. Rec Res Dev Immunol. 2001;3:61–74. [Google Scholar]
- 43.Kanauchi H, Furukawa F, Imamura S. Characterization of cutaneous infiltrates in MRL/lpr mice monitored from onset to the full development of lupus erythematosus-like skin lesions. J Invest Dermatol. 1991;96:478–83. doi: 10.1111/1523-1747.ep12470176. [DOI] [PubMed] [Google Scholar]
- 44.Kohchiyama A, Oka D, Ueki H. T-cell subsets in lesions of systemic and discoid lupus erythematosus. J Cutan Pathol. 1985;12:493–9. doi: 10.1111/j.1600-0560.1985.tb00448.x. [DOI] [PubMed] [Google Scholar]
- 45.Viljaranta S, Ranki A, Kariniemi AL, et al. Distribution of natural killer cells and lympohocyte subclasses in Jessner's lymphocytic infiltration of the skin and in cutaneous lesions of discoid and systemic lupus erythematosus. Br J Dermatol. 1987;116:831–8. doi: 10.1111/j.1365-2133.1987.tb04902.x. [DOI] [PubMed] [Google Scholar]
- 46.Volc-Platzer B, Anegg B, Milota S, et al. Accumulation of γδ T cells in chronic cutaneous lupus erythematosus. J Invest Dermatol. 1993;100:84s–91s. doi: 10.1111/1523-1747.ep12356084. [DOI] [PubMed] [Google Scholar]
- 47.Robak E, Niewiadomska H, Robak T, et al. Lymphocytes Tγδ in clinically normal skin and peripheral blood of patients with systemic lupus erythematosus and their correlation with disease activity. Med Inflamm. 2001;10:179–89. doi: 10.1080/09629350124724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Jameson J, Ugarte K, Chen N, et al. A role for skin γδ T cells in wound repair. Science. 2002;296:747–9. doi: 10.1126/science.1069639. [DOI] [PubMed] [Google Scholar]
- 49.Havran WL, Chien YH, Allison JP. Recognition of self antigens by skin-derived T cells with invariant γδ antigen receptors. Science. 1991;252:1430–2. doi: 10.1126/science.1828619. [DOI] [PubMed] [Google Scholar]
- 50.Araneo BA, Dowel T, Moon HB, et al. Regulation of murine lymphokine production in vivo. Ultraviolet radiation exposure depresses IL-2 and enhances IL-4 production by T cells through an IL-1 dependent mechanism. J Immunol. 1989;143:1737–44. [PubMed] [Google Scholar]
- 51.Simon JC, Tigelaar RE, Bergstresser PR, et al. Ultraviolet B radiation converts Langerhans cells from immunogenic to tolerogenic antigen-presenting cells. Induction of specific clonal anergy in CD4+ T helper 1 cells. J Immunol. 1991;146:485–91. [PubMed] [Google Scholar]
- 52.Garssen J, Vandebriel RJ, De Gruijl FR, et al. UVBL exposure-induced systemic modulation of Th1- and Th2- mediated immune responses. Immunology. 1999;97:506–14. doi: 10.1046/j.1365-2567.1999.00801.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Toro JR, Finlay D, Dou X, et al. Detection of type 1 cytokines in discoid lupus erythematosus. Arch Dermatol. 2000;136:1497–501. doi: 10.1001/archderm.136.12.1497. [DOI] [PubMed] [Google Scholar]
- 54.Theofilopoulos AN, Lawson BR. Tumor necrosis factor and other cytokines in murine lupus. Ann Rheum Dis. 1999;58:149–55. doi: 10.1136/ard.58.2008.i49. [DOI] [PMC free article] [PubMed] [Google Scholar]