Abstract
Background
Cancers of the pancreas originate from both the endocrine and exocrine elements of the organ, and represent a major cause of cancer-related death. This study provides a comprehensive assessment of gene expression for pancreatic tumors, the normal pancreas, and nonneoplastic pancreatic disease.
Methods/Results
DNA microarrays were used to assess the gene expression for surgically derived pancreatic adenocarcinomas, islet cell tumors, and mesenchymal tumors. The addition of normal pancreata, isolated islets, isolated pancreatic ducts, and pancreatic adenocarcinoma cell lines enhanced subsequent analysis by increasing the diversity in gene expression profiles obtained. Exocrine, endocrine, and mesenchymal tumors displayed unique gene expression profiles. Similarities in gene expression support the pancreatic duct as the origin of adenocarcinomas. In addition, genes highly expressed in other cancers and associated with specific signal transduction pathways were also found in pancreatic tumors.
Conclusion
The scope of the present work was enhanced by the inclusion of publicly available datasets that encompass a wide spectrum of human tissues and enabled the identification of candidate genes that may serve diagnostic and therapeutic goals.
Introduction
The aim of this study is to provide a global assessment of gene expression patterns for the major neoplastic diseases of the pancreas. We demonstrate that the inclusion of a diverse source of diseased and normal pancreatic tissues for analysis using DNA microarrays enhances the identification of gene expression patterns that can be attributed to specific cell types and pancreatic diseases.
Pancreatic adenocarcinoma represents a significant health burden in industrialized countries and represents one of the most lethal human cancers. It is the fourth-leading cause of cancer-related death in the United States with a five-year survival rate of 4% [1]. Oligonucleotide microarrays, cDNA microarrays, and serial analysis of gene expression technologies have been used to characterize the gene expression profile of pancreatic adenocarcinomas [2]–[5]. These studies have identified candidate genes that may play a role in disease pathogenesis or may serve as clinical markers of disease. The analysis of gene expression data for pancreatic cancer, however, is problematic because of the pronounced desmoplasia associated with the disease. The actual neoplastic cells often comprise a minority of the total cell population within pancreatic tumors, with the majority belonging to stromal elements. When pancreatic adenocarcinomas have been compared to normal pancreatic tissues, the robust desmoplastic response produced by stromal cells contributes to a gene expression profile that may dominate over that contributed by the neoplastic cells themselves. Because other pancreatic diseases are also known to result in significant desmoplasia, further confounding the interpretation of the patterns seen in cancers, this study approached the problem by expanding the tissues examined to include a wide variety of diseased and normal pancreatic tissues. The present study used samples derived from pancreatic tumors of exocrine, endocrine, and mesenchymal origin. In addition, isolated pancreatic islets and ducts from normal tissues as well as pancreatic cancer cell lines were used to enhance analysis of the gene expression profile. Each pancreatic tissue and cell line contributes to an interpretative framework that facilitates identification of genes whose expression is most characteristic of specific cell types and diseases. For example, genes that participate in the desmoplastic response can be distinguished because their expression is a shared feature in a variety of different pancreatic diseases in which this response is seen.
Islet cell tumors represent a second major pancreatic tumor whose pathogenesis is poorly understood and for which effective therapies are lacking. In contrast to previously published studies that compared gene expression between islet tumors and normal islets, the current study also examines gene expression in islet tumors in the context of the whole pancreas and other pancreatic diseases [6]–[8].
The expanding collection of publicly accessible gene expression data also provides an opportunity to compare and enhance the dataset provided by this study. For example, the gene expression profiles of a wide spectrum of normal human tissues [9] provide an opportunity to compare gene expression profiles of pancreatic cancer to a detailed overview of the cells and tissues of the human body, and thereby focus on those features most likely to provide diagnostic markers for detection of disease.
Materials and Methods
Samples and RNA isolation
All participating patients provided consent prior to surgery. The resected tissue was immediately frozen in liquid nitrogen, and stored at −80°C. A portion of the sample was processed for histopathology. Normal pancreatic islets and pancreatic ducts were obtained from organ donors. Samples were obtained with approved human subjects protocols from Stanford University, Samsung Medical Center (Seoul, South Korea), University of Washington, and the Cooperative Human Tissue Network. Data derived from 11 adenocarcinoma samples and 5 non-tumor pancreatic samples included in this report were previously published in collaboration with Johns Hopkins University [5]. The present study represents the addition of 91 microarray studies that includes additional adenocarcinomas, islet cell tumors, other non-adenocarcinomas, benign pancreatic disease, normal isolated pancreatic islets and ducts, and pancreatic cancer cell lines.
Pancreatic cancer cell lines were obtained from the American Type Culture Collection (Manassas, VA) and included AsPC-1 (#CRL-1682), BxPC-3 (#CRL-1687), Capan-1 (#HTB-79), Capan-2 (#HTB-80), CFPAC-1 (#CRL-1918), Hs 766T (#HTB-134), MIA PaCa-2 (#CRL-1420), PANC-1 (#CRL-1469), SU.86.86 (#CRL-1837). The cells were grown to 80–90% confluence in media specified for each cell type. Islets were isolated from pancreata as previously described [10]. Cells derived from human pancreatic ducts were initially expanded as a primary culture and only early passage cells that retained responsiveness to secretin stimulation were used [11].
Total RNA for all samples was extracted using Trizol (Invitrogen, Carlsbad, CA) followed by mRNA isolation using FastTrack® mRNA (Invitrogen, Carlsbad, CA). RNA amplification was performed for the isolated pancreatic duct cells and islets, 3 adenocarcinomas, and the reference mRNA standards using the procedure described by Wang et al. [12]. Two rounds of amplifications were used.
Microarray Procedure
DNA microarrays containing 41,125 clones, representing approximately 24,473 unique genes were printed on microscope slides and processed for hybridization as previously described (http://cmgm.stanford.edu/pbrown/protocols/index.html) [13], [14]. The sample mRNA was labeled with Cy5-UDP and a common reference RNA derived from a panel of cell lines was labeled with Cy3-UDP (Amersham Biosciences, Piscataway, NJ) [14]. The arrays were scanned using a GenePix 4000A scanner (Axon Instruments, Molecular Devices Corp., Palo Alto, CA).
Features containing artifacts or blemishes were flagged for exclusion from further analysis using GenePix Pro 5.0 (Axon Instruments). The raw data was deposited in the Stanford Microarray Database (http://genome-www5.stanford.edu) [15]. A total of 9,259 unique genes (represented by 16,959 spots) satisfied subsequent filtering by exhibiting a mean signal intensity at least 1.5 fold greater than background for either the Cy5 or Cy3 channels. Only genes for which acceptable data were obtained in at least 70% of the samples and that showed at least a 1.5-fold difference in expression from the mean for at least 1 sample were retained. The expression data were visualized using Treeview [16]. Analysis included a hierarchical clustering algorithm that was applied to both the genes and arrays, and a supervised analysis by multi-class comparison using Significance Analysis of Microarrays (SAM) software [16], [17]. Identification of genes capable of discriminating between classes using shrunken centroids employed Prediction Analysis of Microarrays (PAM) software [18]. Gene annotations were obtained using SOURCE (http://source.stanford.edu) [19]. Genes predicted to encode secretory and membrane proteins were identified using the SignalIP software program that analyzes amino acid sequences for signal peptides (HMM/S mean score method)[20] or the TMHMM program that identifies transmembrane domains[21]. Additional putative secretory and membrane proteins were identified by interrogating an empirically-derived dataset[22]. Differentially expressed genes in pancreatic adenocarcinoma that are associated with areas of chromosome gains or loss were identified using comparative genomic hybridization data of the 6 cell lines used in the present study[23]. A high resolution map for regions of DNA copy number gains and losses was obtained using the CLuster Along Chromosome (CLAC) method[24].
The entire primary dataset is available at the Stanford Microarray Database (http://genome-www5.stanford.edu/) or the Lowe laboratory website (http://www.stanford.edu/group/lowelab/)
Immunohistochemistry and In-situ hybridization
Immunohistochemistry was performed using Dako Envision Plus (Glostrup, Denmark) following the manufacturer's instruction. Rabbit anti-axin2 serum was from Zymed (Invitrogen, Carlsbad, CA). Tissue microarrays constructed to evaluate axin2 expression contained 13 normal pancreata and 26 pancreatic adenocarcinomas. Rabbit antibodies against LGALS4 were produced against peptide sequence NH2-PPYPGPGHCHQQLNS, and for MUC13 peptide sequence NH2-DPEEKHSMAYQDLHSEC (AnaSpec, Inc., San Jose, CA).
In situ hybridization was performed as previously described using paraffin-embedded sections [25].
RESULTS
Gene expression profiles were obtained using cDNA microarrays for a total of 80 surgically removed pancreatic tissue samples (Table 1). The majority of the tumors removed were determined by pathology to be adenocarcinomas. Other pancreatic tissues collected included islet cell tumors, rare pancreatic tumors, chronic pancreatitis, and cystic disease. When available, non-tumor tissue was also collected. Normal pancreatic tissue was obtained from 9 organ donors.
Table 1. Samples used in the study.
| Adenocarcinoma * | 43 | |
| Islet cell tumors (4 patients) | 6 | |
| Other tumors | 4 | |
| osteoclast giant cell tumor (1 patient) | 2 | |
| liposarcoma | 1 | |
| lymphoma | 1 | |
| Benign disease | 5 | |
| chronic pancreatitis | 3 | |
| pseudocyst | 1 | |
| mucinous cyst | 1 | |
| Non-tumor and normals | 23 | |
| Pancreatic cancer cell lines | 9 | |
| Primary pancreatic ducts | 5 | |
| Primary pancreatic islets | 9 |
3 adenocarciinomas were used solely as amplified samples.
Additional samples included primary cultures of human pancreatic duct cells, purified human islets from normal donors, and nine commercially available pancreatic cancer cell lines. The human pancreatic duct cells and human islets were harvested from 5 and 9 different donors, respectively, and were processed separately and not pooled. RNA derived from the pancreatic duct cells and purified islets required antisense RNA amplification for the microarray analysis because of lower amounts of starting material.
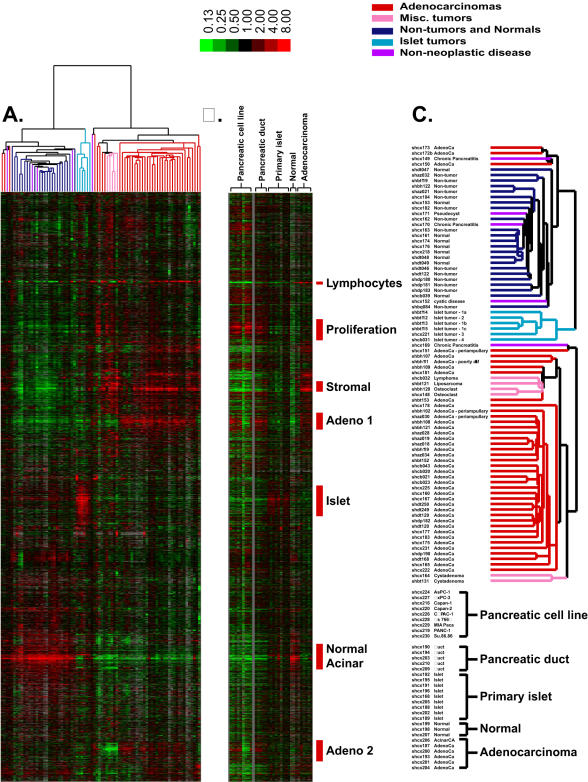
We first used an unsupervised hierarchical clustering approach to organize and explore the data and highlight groups of genes and groups of samples with similar gene expression patterns[16]. Based on similarities in their overall gene expression profiles, most samples were clustered in a manner consistent with their original pathologic classification, yielding distinct clusters of adenocarcinomas, islet cell tumors, and normal pancreatic tissues (Figure 1A). Thirty-seven of 40 adenocarcinoma samples were grouped in one of the two major branches of the clustering dendrogram, of which 31 shared very similar expression profiles and were clustered within the same node. Six adenocarcinomas clustered in an adjoining node with lymphoma and other mesenchymal tumors, including 3 adenocarcinomas (shbh111, shbh109, shbt153, see Data S1 for clinical details) with atypical pathologic features. These adenocarcinomas were distinguished by higher expression of genes associated with proliferation and lower expression of the genes in the “Adeno” clusters 1 and 2. Three adenocarcinomas that clustered away from the majority of adenocarcinomas (shcx173, shcx172b, and shcx150) were distinguished by significant expression of genes encoding digestive enzymes, such as pancreatic amylase (AMY2A) and elastase (ELA3A), suggesting the presence of normal acinar cells in addition to the neoplastic component. The gene expression profiles for most of the adenocarcinomas were distinct from non-neoplastic tissues as well as other pancreatic tumors, including lymphomas, osteoclast tumors, liposarcomas, and islet-cell tumors. Among the adenocarcinomas, no significant segregation was observed on this analysis that would suggest subclassification into distinct molecular subtypes that might be associated with differences in tumor or cellular staging criteria. Clinical outcomes following surgery were known for 16/39 adenocarcinomas in this dataset (Data S1). The disease-free interval for almost all patients was less than 2 years. Among these samples, there was no indication that hierarchical clustering resulted in any classification by survival length.
Figure 1.
Unsupervised hierarchical clustering of the patterns of variation in expression for 9,259 genes (represented by 16,959 cDNA) in 80 pancreatic tissue specimens (A). The image uses a color code to represent relative expression levels. Red represents expression levels greater than the mean for a given gene across all samples. Green represents expression levels less than the mean across samples. A color bar (top) relates color code to the magnitude of the differences in gene expression relative to the all-sample means for each gene. Grey indicates missing or excluded data. (B) Image representing gene expression in pancreatic cell lines, primary pancreatic duct and islet cells, normal pancreas, and adenocarcinomas. The cell lines and amplified samples were each mean-centered separately. The genes presented are organized and displayed in an identical manner to the genes shown in panel (A). (C) Enlarged view of the array dendrogram shown in panel A along with sample identification.
Islet cell tumors were derived from four patients. Three samples from one patient representing separate metastatic lesions are included. Although immunohistochemistry revealed different secretory products for 3 patients, the tumors shared sufficient similarity in gene expression profiles to be clustered together.
Other pancreatic tumors that did not cluster with the adenocarcinomas or islet cell tumors included a pancreatic sarcoma, lymphoma, and osteoclast-like giant cell tumor. Although only single cases were represented for these rare tumors, they clustered together with the major common feature consisting of genes characteristic of the desmoplastic response, and relatively high expression of genes associated with proliferation.
Non-tumor and normal whole pancreatic tissues consistently clustered separately from the diseased tissues. The analysis did not distinguish between uninvolved non-tumor tissue from patients with cancer and pancreatic tissue from normal donors without any history of pancreatic disease.
Unsupervised hierarchical clustering also highlighted clusters of genes specifically associated with adenocarcinomas, islet cell tumors, and normal pancreata. The diversity of tumors and cell types in this sample set gave rise to a diversity of gene expression patterns, many of which appear to represent specific cell types. Defining the source of the distinctive gene expression patterns was facilitated by projecting the expression data for clonal pancreatic cancer cell lines, primary pancreatic duct cultures, and isolated pancreatic islets alongside the data derived from the previous clustering analysis (Figure 1B).
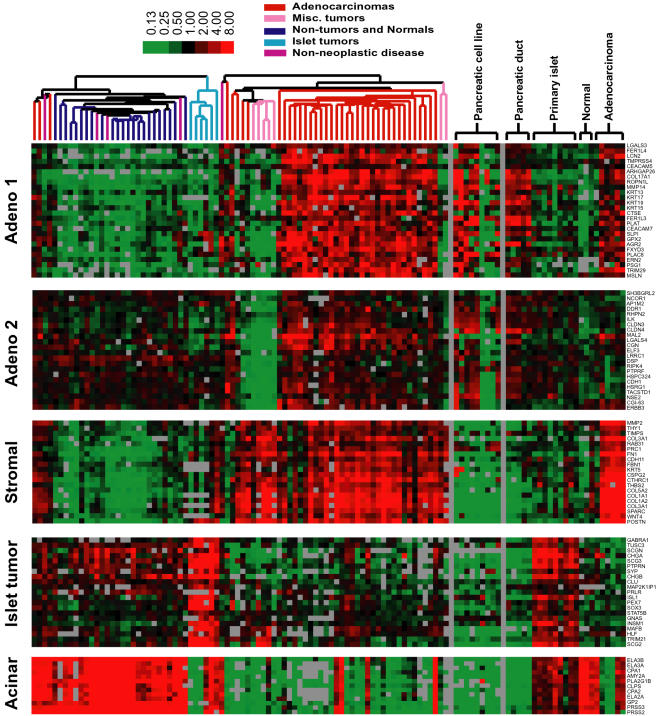
Of particular interest in this study was the segregation of genes associated with adenocarcinomas cells themselves from those involved in the desmoplastic response. Two gene clusters, labeled “Adeno 1” (297 genes) and “Adeno 2” (88 genes) (Figure 2), were characterized by higher expression in adenocarcinomas compared to other tumors or normal pancreatic tissues. The separation into 2 gene clusters is due to the higher relative expression of genes in adenocarcinomas compared to normal tissues in the “Adeno 1” cluster, as compared to the “Adeno 2” gene cluster. The specificity of genes within the adenocarcinoma clusters is supported by their lower expression in lymphoma, sarcoma, osteoclast-like cells, and islet cell tumors. Contained within each cluster are genes whose expression was previously associated with pancreatic adenocarcinoma, including keratin 19 (KRT19), anterior gradient 2 homolog (AGR2), v-erb-b2 avian erythroblastic leukemia viral oncogene homolog 3 (ERBB3), mesothelin (MSLN), and prostate stem cell antigen (PSCA) [5], [26]–[28]. Evidence that hierarchical clustering successfully distinguished between genes expressed predominantly in neoplastic epithelial cells and in stromal cells, respectively, is illustrated by two previously characterized matrix metalloproteinases, MMP14 and MMP2[25]. Expression of MMP14 by adenocarcinoma cells has been previously established by in situ hybridization, which is consistent with its clustering within the “Adeno 1” cluster. MMP2 is likewise associated with a separate cluster consistent with its expression by stromal cells (Figure 2). In situ hybridization or immunocytochemistry was performed for selected genes from each “Adeno” cluster and confirmed that the enhanced gene expression was indeed derived from the neoplastic cells (Figure 3).
Figure 2.
Expanded view of gene clusters shown in Figure 1A .Color code and experimental conditions are as described for Figure 1.
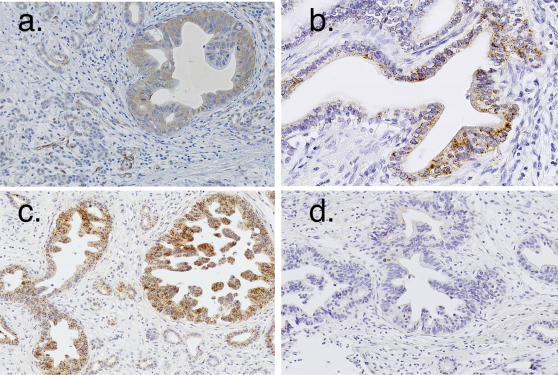
Figure 3.
Immunohistochemistry of paraffin embedded pancreatic adenocarcinomas. Anti-galectin 4 (LGALS4) antibody (panel a). In situ hybridization for collagen 17A1 (COL17A1) (panel b) and mucin 13 (MUC13) (panel c). In situ hybridization sense control for MUC13 (panel d).
We used significance analysis of microarrays (SAM), a supervised approach of analysis that incorporates an assessment of statistical significance, to identify genes that are differentially expressed in adenocarcinomas compared to the normal pancreas and other tumors in the dataset [17]. A total of 293 genes with an average expression at least 3-fold or higher in adenocarcinomas compared to non-adenocarcinoma samples were identified, with a false discovery rate of<.00001%. These genes were ranked based on the statistical significance of their differential expression in pancreatic adenocarcinoma versus other samples (Data S2). Among the 293 ranked genes, 176 are associated with the previously identified “Adeno 1” and “Adeno 2” clusters, and 87 genes are associated with the Stromal cluster. These genes include many not previously associated with pancreatic adenocarcinoma, such as galectin-4 (LGALS4), mucin 13 (MUC13), secretory leukocyte protease inhibitor (SLPI), collagen 17A1 (COL17A1), and ropporin 1-like (ROPN1L). Available annotations with respect to function and subcellular location revealed genes that function in signal transduction, the cytoskeleton, the extracellular matrix, and cellular metabolism (Data S2). Recent studies using comparative genomic hybridization of genomic DNA have produced a high resolution map for regions of DNA gains and losses for pancreatic cancer cell lines, including the 6 used in the present study. When used as a guide, 31 highly expressed genes and 26 genes expressed at lower levels in adenocarcinomas were also associated with corresponding areas of frequent DNA copy gains and losses (Data S3). Supporting the ductal origin of pancreatic adenocarcinomas, the majority of genes clustered in “Adeno 1” and “Adeno 2” were similarly highly expressed in isolated pancreatic duct cells. The same genes also showed higher expression in clonal pancreatic cancer cell lines. Among the commonly available adenocarcinoma cell lines, only a subset consisting of AsPC1, BXPC3, Capan-1, Capan-2, and CFPAC-1, consistently exhibited higher expression of genes that were similarly elevated in pancreatic adenocarcinomas (Figure 2). In contrast, genes in the stromal cluster exhibited much lower expression in the pancreatic adenocarcinoma cell lines and primary pancreatic duct cells. Significance analysis was also used to identify genes differentially expressed between primary duct and acinar cells. Because acinar cells comprise ∼90% of the pancreas, differences in gene expression between pancreatic duct and acinar cells were determined by comparing expression patterns of normal isolated pancreatic ducts with that of the normal whole pancreas derived from organ donors. A total of 1,476 genes with at least a 3-fold difference in expression between the two classes were identified with a false discovery rate of 0.05%. Again, genes with enhanced expression in the pancreatic ducts were also similarly expressed in adenocarcinomas (Data S4).
Islet tumors were identified by a cluster of 311 genes with elevated expression (correlation ≥0.75). Consistent with their endocrine origin, these tumors were characterized by elevated levels of transcripts encoding chromogranins A and B (CHGA, CHGB), secretogranin II (SCG2), synaptophysin (SYP), peptidylglycine α-amidating monooxygenase (PAM), dopa decarboxylase (DDC), and somatostatin receptor 1 (SSTR1) (Data S5). Genes in the islet tumor cluster were generally expressed at high levels in the isolated islet cells but not in isolated pancreatic duct cells (Figure 2). Significance analysis identified genes differentially expressed in islet tumors compared to all other tissue samples in the dataset (Data S5). Two hundred genes were identified with a false discovery rate of 3.5%, of which 106 genes showed relatively high expression in the islet tumors. Consistent with previously published findings, genes with significantly elevated expression in islet cell tumors included insulinoma-associated 1 (INSM1), glutaminyl-peptide cyclotransferase (QPCT), fibrinogen B beta polypeptide (FGB), peroxisomal biogenesis factor 7 (PEX7). All of these genes identified by the SAM analysis were within the islet tumor cluster. Genes not previously described in islet cell tumors, but exhibiting elevated specific expression, include the AXL receptor tyrosine kinase (AXL) and the transcription factor V-maf musculoaponeurotic fibrosarcoma oncogene homolog B (MAFB). AXL expression is also elevated in leukemia, melanoma, prostate, colon, endometrial, and thyroid cancers[29]–[32]. Increased MAFB gene expression has been observed in myeloma [33]. INSM1, AXL, and MAFB are all located on chromosome 20 where comparative genomic hybridization studies have detected high-level amplification[34], [35].
A group of genes was expressed at higher levels in non-tumor and normal pancreatic tissues than in either islet cell tumors or adenocarcinomas. Acinar cells represent 90% of the cellular composition of the pancreas and are expected to dominate the expression profile of the normal pancreas. Genes encoding digestive enzymes produced by acinar cells such as amylase (AMY2A), elastase (ELA3A, ELA3B), carboxypeptidase A (CPA2), pancreatic lipase (PNLIP), and trypsin (PRSS2) were enriched in this cluster (Figures 1, “Acinar” cluster).
Genes known to be associated with the desmoplastic response in pancreatic cancer were clustered separately from the adenocarcinoma clusters. These genes included osteonectin (SPARC), fibronectin (FN1), versican (CSPG2), and collagen type 1A2 (COL1A2), tissue inhibitor of metalloproteinase 1 (TIMP1), biglycan (BGN), matrix metalloproteinase 2 (MMP2), and stromelysin-1 (MMP3) [5], [36]–[38]. The desmoplasia associated gene expression pattern was not seen in islet cell tumors, normal pancreatic tissues, pancreatic adenocarcinoma cell lines, or isolated pancreatic ducts, but was seen in other tumors such as lymphoma, sarcoma, osteoclast-like giant cell tumors, and adenocarcinomas. The expression of the stromal genes in tumors other than adenocarcinomas likely contributed to their segregation in a gene cluster distinct from the “Adeno” clusters, where expression was specifically associated with adenocarcinomas.
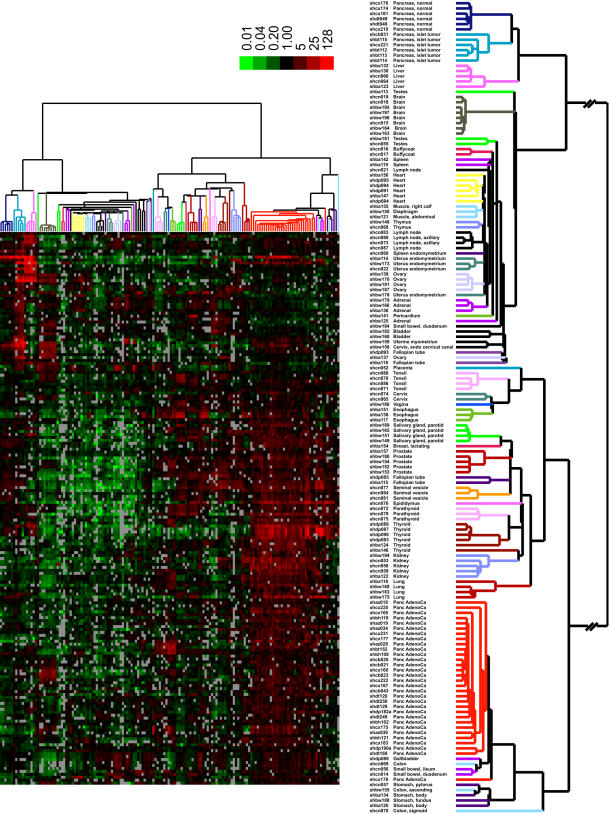
Additional analysis was performed to identify genes potentially useful for diagnostic and therapeutic purposes. As secretory or membrane proteins may be especially useful as diagnostic markers or therapeutic targets, each gene enriched in pancreatic cancer or islet cell tumors was further analyzed using algorithms to identify signal peptide or transmembrane domains. For pancreatic cancer, only genes within the “Adeno” clusters were analyzed because they are enriched in adenocarcinoma cells and do not include the stromal genes common to other pancreatic diseases. Of the 259 genes in the “Adeno” clusters, 126 were classified as secretory or membrane proteins by amino acid sequence analysis. An additional 71 genes, for a total of 197, were identified using a recently defined dataset by Diehn et al. in which a genome-wide screen for secretory proteins was empirically determined using similar DNA microarrays [22]. For the 107 genes identified by significance analysis of microarrays with high expression in islet cell tumors, 32 were classified using sequence analysis as encoding secretory or membrane proteins, and an additional 20 genes were identified using the empirically derived dataset. These putative membrane/secreted protein encoding genes identified for pancreatic cancer and islet cell tumors were then examined using unsupervised hierarchical clustering in the context of a diverse collection of normal human tissues. The approach utilized the recently published gene expression profiles obtained from a diverse collection of normal human tissues, obtained using technologies and methods identical to those used in the present study [9]. The resultant set of putative secretory and membrane protein genes were able to distinctly cluster the pancreatic adenocarcinomas, islet cell tumors, and each of the normal tissues (Figure 4). None of the genes examined were uniquely expressed in pancreatic adenocarcinomas or islet cell tumors as each was detectably expressed in one or more normal tissues. Prediction analysis of microarrays (PAM), an algorithm that employs shrunken centroids [18] to identify genes capable of discriminating between different predetermined classes, identified a set of 16 genes whose expression pattern was capable of distinguishing 31 of 31 adenocarcinomas and a set of 12 genes whose expression pattern was capable of distinguishing 6 of 6 islet cell tumor samples from all normal tissues (Table 2 and Data S6). Four out of 121 non-adenocarcinomas were misclassified with the adenocarcinomas. None of the 150 non-islet cell tumor tissues were misclassified as tumors.
Figure 4.
Cluster analysis of secretory and membrane proteins. Unsupervised hierarchical cluster analysis of expression of 187 genes encoding putative membrane or secreted proteins in normal pancreas, pancreatic adenocarcinoma, islet cell tumor tissues and a panel of normal tissues [9]. Only genes for which 80% of spots met quality criteria were used. The figure shows 187 genes representing secretory or membrane-bound proteins that show enhanced expression in pancreatic adenocarcinomas or islet cell tumors. An enlarged view of the array dendrogram along with sample identification is shown on the right. A color bar (top) relates color code to the magnitude of the differences in gene expression relative to the all-sample means for each gene. Grey indicates missing or excluded data.
Table 2. Genes that discriminate adenocarcinoma and islet cell tumors from normal human tissues.
| Gene Symbol | Name | Clone ID |
| Pancreatic Adenocarcinoma | ||
| LAMC2 | Transcribed locus, homologous to laminin, gamma 2 | IMAGE:460403 |
| CTSE | Cathepsin E | IMAGE:243202 |
| GPX2 | Glutathione peroxidase 2 (gastrointestinal) | IMAGE:587847 |
| LGALS4 | Lectin, galactoside-binding, soluble, 4 (galectin 4) | IMAGE:511068 |
| GPRC5A | G protein-coupled receptor, family C, group 5, member A | IMAGE:595037 |
| MMP14 | Matrix metallopeptidase 14 (membrane-inserted) | IMAGE:270505 |
| ITGA2 | Integrin, alpha 2 (CD49B, alpha 2 subunit of VLA-2 receptor | IMAGE:525246 |
| AGR2 | Anterior gradient 2 homolog (Xenopus laevis) | IMAGE:2321113 |
| COL17A1 | Collagen, type XVII, alpha 1 | IMAGE:252259 |
| TSPAN8 | Tetraspanin 8 | IMAGE:509731 |
| GPRC5A | G protein-coupled receptor, family C, group 5, member A | IMAGE:1457276 |
| CEACAM7 | Carcinoembryonic antigen-related cell adhesion molecule 7 | IMAGE:509688 |
| Islet Cell Tumors | ||
| SCG2 | Secretogranin II (chromogranin C) | IMAGE:174627 |
| RASSF7 | Ras association (RalGDS/AF-6) domain family 7 | IMAGE:1573778 |
| TTR | Transthyretin (prealbumin, amyloidosis type I) | IMAGE:1868551 |
| SGNE1 | Neuroendocrine protein 1 (putative) | IMAGE:878836 |
| INSM1 | Insulinoma-associated 1 | IMAGE:22895 |
| PCSK2 | Proprotein convertase subtilisin/kexin type 2 | IMAGE:24254 |
| QPCT | Glutaminyl-peptide cyclotransferase | IMAGE:711918 |
| FGB | **Fibrinogen beta chain | IMAGE:84713 |
| PEX7 | Peroxisomal biogenesis factor 7 | IMAGE:2018758 |
| PTPRN2 | Protein tyrosine phosphatase, receptor type | IMAGE: 812968 |
| SERPINA1 | Serpin peptidase inhibitor | IMAGE:294578 |
| ARF3 | ADP-ribosylation factor 3 | IMAGE:291097 |
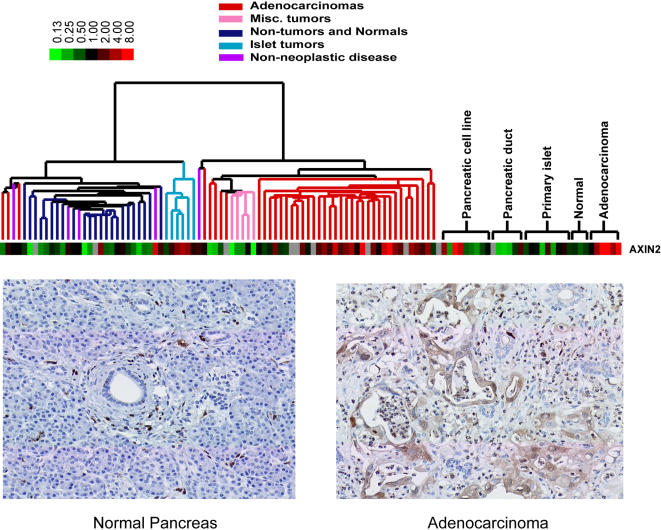
Mutational activation of the Wnt pathway is one of the most common genetic steps in the pathogenesis of gastrointestinal cancers. Expression of AXIN2, a marker for Wnt pathway activation [39]–[41], was generally higher in pancreatic adenocarcinomas compared to primary pancreatic duct cells (Figure 5). AXIN2 expression was also elevated in islet cell tumors, which likely contributed to its clustering away from the previously identified adenocarcinoma gene clusters. Immunocytochemistry using tissue microarrays of pancreatic tissues revealed positive axin2 staining in 15 of 26 adenocarcinomas. No staining was seen in 13 normal pancreata.
Figure 5.
Axin 2 expression in islet cell tumors and adenocarcinomas. Pseudocolored image of axin 2 expression (top). Image represents mean centered values of sample/reference ratios. Representative immunohistochemistry with rabbit anti-axin2 antibodies in normal pancreas (bottom left) and pancreatic adenocarcinoma (bottom right).
Discussion
DNA microarray analysis of global gene expression patterns in a diverse collection of normal and diseased pancreatic tissues allowed identification of specific gene expression profiles for pancreatic adenocarcinomas, islet cell tumors, and the normal pancreas. Interpretation of the gene expression profiles was facilitated by the parallel analysis of gene expression in primary pancreatic ducts and islets, and pancreatic cancer cell lines. The approach identified genes that are specifically expressed by neoplastic or stromal cells. Supporting the approach is the identification of many genes previously associated with pancreatic adenocarciomas or islet cell tumors. Many other genes not previously associated with these cancers were also newly identified. The availability of publicly available gene expression data provided an opportunity to compare the gene expression patterns of pancreatic adenocarcinomas and islet cell tumors to those of many other normal tissues. Other available datasets enabled the association of differentially expressed genes with areas of genomic DNA gains and losses. Similar gene expression datasets for other adenocarcinomas including hepatic, gastric, and breast tumors provide an opportunity for additional comparisons[9], [14], [42], [43]. In addition, the identification of secretory and membrane proteins using computational algorithms or annotations derived from genome-wide empirical assessments of mRNA localization further enhance of our ability to identify likely candidates for diagnostic and therapeutic purposes.
The present study also provides a characterization of the gene expression profile for islet cell tumors in the context of the whole pancreas. The gene expression profile for neuroendocrine tumors in patients with the MEN1 syndrome was recently reported [44]. The syndrome is characterized by neuroendocrine tumors of the parathyroids, pituitary, and pancreatic islets. Of the 6 patients with islet cell tumors studied, 2 exhibited insulinomas, which were compared to that of purified human islets from normal subjects. The differentially expressed genes in the MEN1 syndrome were not similar to those identified in this study, and suggest a different pathogenesis for spontaneous islet cell tumors. Our analysis identified additional genes, including AXL, MFAB, and AXIN2, whose elevated expression in islet cell tumors suggests signal transduction pathways important in their development.
Among pancreatic tumors, the adenocarcinomas are the most common and the most deadly. Distinct and characteristic differences in gene expression patterns have revealed subclasses of common tumors, such as breast cancer, that also differ with respect to their natural history or response to therapy[45]. In contrast, patients afflicted with pancreatic adenocarcinoma generally follow a similar course. Consistent with the uniformity of the clinical course, the gene expression profile for the majority of adenocarcinomas in the present study was quite uniform, suggesting that the disease is relatively homogenous. Although the present study is biased toward patients with surgically resectable tumors, differences in gene expression profiles would have been expected if significant biological differences among pancreatic adenocarcinomas were common.
Our results provide evidence that the Wnt pathway may often be activated in pancreatic adenocarcinoma and islet cell tumors. Evidence for Wnt pathway activation in pancreatic cancer has been inconclusive because nuclear staining for β-catenin, a mediator of Wnt signaling, is observed in less than 5% of cases [46]. Wnt pathway activation has been previously inferred from the altered cellular distribution of β-catenin in pancreatic adenocarcinomas, with decreased plasma membrane and increased cytoplasmic staining compared to normal pancreatic ducts and acinar cells by immunohistochemistry [46]. Recent studies have established that Axin2 expression is induced by Wnt pathway activation and serves as a useful marker. Higher expression of Axin2 in pancreatic adenocarcinomas and islet tumors compared to their non-neoplastic counterparts supports the potential importance of Wnt pathway activation in these tumors. A systematic search for mutations in genes involved in the Wnt pathway may yield new insights into the pathogenesis of these cancers.
The present study provides a comprehensive analysis of gene expression in pancreatic tumors. The analysis of a broad spectrum of pancreatic tissues in conjunction with publicly available datasets and software tools enhanced the identification of genes that may participate in disease pathogenesis, or may serve as preferred targets for diagnostic or therapeutic strategies.
Supporting Information
List of genes within the Adeno 1 and 2 clusters. Annotated gene list identified by significance analysis of microarrays versus all other non-adenocarcinoma pancreatic tissues. Only the 31 adenocarcinomas grouped together in Figure 1 were used for the analysis. Worksheets also contain analysis for the identification of putative secretory and membrane proteins and genes that are associated with DNA copy gains and losses as determined by CGH. A table of contents is contained in the first Excel worksheet.
(1.91 MB XLS)
Table of differentially expressed genes identified by SAM in adenocarcinomas whose genomic location showed high DNA copy gains or losses.
(0.03 MB XLS)
List of genes differentially expressed between primary pancreatic duct cells and whole normal pancreatic tissue obtained using SAM as described in the Methods.
(2.12 MB XLS)
List of genes within the islet cell tumor cluster. Annotated list of genes identified by SAM that are differentially expressed in islet tumors compared to all other pancreatic tissues in the dataset.
(0.43 MB XLS)
Identification of candidate genes capable of discriminating pancreatic adenocarcinomas or islet cell tumors from a diverse collection of normal human tissues. Putative secretory or membrane proteins enriched in pancreatic adenocarcinomas or islet cell tumors in the current study were examined in the context of a wide variety of normal tissues reported in reference [9].
(0.05 MB XLS)
Acknowledgments
The authors wish to acknowledge the contributions of Ward Trueblood, Augusto Bastidas, Mark Vierra, Sherry Wren, Nancy Kaduk, George Yang, Janet Mitchell, and Kelli Montgomery from the Departments of Surgery and Pathology at Stanford University, and Suet Y. Leung (University of Hong Kong, Hong Kong, China) for assistance in tissue acquisition; and to Christopher Savard for assistance in isolating the pancreatic ducts and islets.
Footnotes
Competing Interests: The authors have declared that no competing interests exist.
Funding: The Susan E. Riley Family Foundation (AWL and POB), The Lustgarten Foundation (AWL), NCI grant CA77097 and HHMI (POB), General Medical Research Service of the Department of Veterans Affairs (SPL). POB is an investigator of the Howard Hughes Medical Institute. The study sponsors had no role in the study design, data collection, analysis, interpretation of the data, writing of the paper, or the decision to submit for publication.
References
- 1.Kern S, Hruban R, Hollingsworth MA, Brand R, Adrian TE, et al. A white paper: the product of a pancreas cancer think tank. Cancer Res. 2001;61:4923–4932. [PubMed] [Google Scholar]
- 2.Ryu B, Jones J, Blades NJ, Parmigiani G, Hollingsworth MA, et al. Relationships and differentially expressed genes among pancreatic cancers examined by large-scale serial analysis of gene expression. Cancer Res. 2002;62:819–826. [PubMed] [Google Scholar]
- 3.Terris B, Blaveri E, Crnogorac-Jurcevic T, Jones M, Missiaglia E, et al. Characterization of gene expression profiles in intraductal papillary-mucinous tumors of the pancreas. Am J Pathol. 2002;160:1745–1754. doi: 10.1016/S0002-9440(10)61121-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Logsdon CD, Simeone DM, Binkley C, Arumugam T, Greenson JK, et al. Molecular profiling of pancreatic adenocarcinoma and chronic pancreatitis identifies multiple genes differentially regulated in pancreatic cancer. Cancer Res. 2003;63:2649–2657. [PubMed] [Google Scholar]
- 5.Iacobuzio-Donahue CA, Maitra A, Olsen M, Lowe AW, Van Heek NT, et al. Exploration of global gene expression patterns in pancreatic adenocarcinoma using cDNA microarrays. Am J Pathol. 2003;162:1151–1162. doi: 10.1016/S0002-9440(10)63911-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wang XC, Xu SY, Wu XY, Song HD, Mao YF, et al. Gene expression profiling in human insulinoma tissue: genes involved in the insulin secretion pathway and cloning of novel full-length cDNAs. Endocr Relat Cancer. 2004;11:295–303. doi: 10.1677/erc.0.0110295. [DOI] [PubMed] [Google Scholar]
- 7.Maitra A, Hansel DE, Argani P, Ashfaq R, Rahman A, et al. Global expression analysis of well-differentiated pancreatic endocrine neoplasms using oligonucleotide microarrays. Clin Cancer Res. 2003;9:5988–5995. [PubMed] [Google Scholar]
- 8.Hansel DE, Rahman A, House M, Ashfaq R, Berg K, et al. Met proto-oncogene and insulin-like growth factor binding protein 3 overexpression correlates with metastatic ability in well-differentiated pancreatic endocrine neoplasms. Clin Cancer Res. 2004;10:6152–6158. doi: 10.1158/1078-0432.CCR-04-0285. [DOI] [PubMed] [Google Scholar]
- 9.Shyamsundar R, Kim YH, Higgins JP, Montgomery K, Jorden M, et al. A DNA microarray survey of gene expression in normal human tissues. Genome Biol. 2005;6:R22. doi: 10.1186/gb-2005-6-3-r22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sweet IR, Cook DL, Wiseman RW, Greenbaum CJ, Lernmark A, et al. Dynamic perifusion to maintain and assess isolated pancreatic islets. Diabetes Technol Ther. 2002;4:67–76. doi: 10.1089/15209150252924111. [DOI] [PubMed] [Google Scholar]
- 11.Oda D, Savard CE, Nguyen TD, Swenson ER, Lee SP. Culture of human main pancreatic duct epithelial cells. In Vitro CellDevBiolAnim. 1998;34:211–216. doi: 10.1007/s11626-998-0126-6. [DOI] [PubMed] [Google Scholar]
- 12.Wang E, Miller LD, Ohnmacht GA, Liu ET, Marincola FM. High-fidelity mRNA amplification for gene profiling. NatBiotechnol. 2000;18:457–459. doi: 10.1038/74546. [DOI] [PubMed] [Google Scholar]
- 13.Alizadeh AA, Eisen MB, Davis RE, Lossos IS, Rosenwald A, et al. Distinct types of diffuse large B-cell lymphoma identified by gene expression profiling. Nature. 2000;403:503–511. doi: 10.1038/35000501. [DOI] [PubMed] [Google Scholar]
- 14.Perou CM, Sorlie T, Eisen MB, van de Rijn M, Jeffrey SS, et al. Molecular portraits of human breast tumours. Nature. 2000;406:747–752. doi: 10.1038/35021093. [DOI] [PubMed] [Google Scholar]
- 15.Sherlock G, Hernandez-Boussard T, Kasarskis A, Binkley G, Matese JC, et al. The Stanford Microarray Database. Nucleic Acids Res. 2001;29:152–155. doi: 10.1093/nar/29.1.152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Eisen MB, Spellman PT, Brown PO, Botstein D. Cluster analysis and display of genome-wide expression patterns. ProcNatlAcadSciUSA. 1998;95:14863–14868. doi: 10.1073/pnas.95.25.14863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tusher VG, Tibshirani R, Chu G. Significance analysis of microarrays applied to the ionizing radiation response. Proc Natl Acad Sci U S A. 2001;98:5116–5121. doi: 10.1073/pnas.091062498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tibshirani R, Hastie T, Narasimhan B, Chu G. Diagnosis of multiple cancer types by shrunken centroids of gene expression. Proc Natl Acad Sci U S A. 2002;99:6567–6572. doi: 10.1073/pnas.082099299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Diehn M, Sherlock G, Binkley G, Jin H, Matese JC, et al. SOURCE: a unified genomic resource of functional annotations, ontologies, and gene expression data. Nucleic Acids Res. 2003;31:219–223. doi: 10.1093/nar/gkg014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bendtsen JD, Nielsen H, von Heijne G, Brunak S. Improved prediction of signal peptides: SignalP 3.0. J Mol Biol. 2004;340:783–795. doi: 10.1016/j.jmb.2004.05.028. [DOI] [PubMed] [Google Scholar]
- 21.Krogh A, Larsson B, von Heijne G, Sonnhammer EL. Predicting transmembrane protein topology with a hidden Markov model: application to complete genomes. J Mol Biol. 2001;305:567–580. doi: 10.1006/jmbi.2000.4315. [DOI] [PubMed] [Google Scholar]
- 22.Diehn M, Bhattacharya R, Botstein D, Brown PO. Genome-Scale Identification of Membrane-Associated Human mRNAs. PLoS Genet. 2006;2:e11. doi: 10.1371/journal.pgen.0020011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bashyam MD, Bair R, Kim YH, Wang P, Hernandez-Boussard T, et al. Array-based comparative genomic hybridization identifies localized DNA amplifications and homozygous deletions in pancreatic cancer. Neoplasia. 2005;7:556–562. doi: 10.1593/neo.04586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wang P, Kim Y, Pollack J, Narasimhan B, Tibshirani R. A method for calling gains and losses in array CGH data. Biostatistics. 2005;6:45–58. doi: 10.1093/biostatistics/kxh017. [DOI] [PubMed] [Google Scholar]
- 25.Iacobuzio-Donahue CA, Ryu B, Hruban RH, Kern SE. Exploring the host desmoplastic response to pancreatic carcinoma: gene expression of stromal and neoplastic cells at the site of primary invasion. Am J Pathol. 2002;160:91–99. doi: 10.1016/S0002-9440(10)64353-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Friess H, Wang L, Zhu Z, Gerber R, Schroder M, et al. Growth factor receptors are differentially expressed in cancers of the papilla of vater and pancreas. Ann Surg. 1999;230:767–774. doi: 10.1097/00000658-199912000-00005. discussion 774–765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Argani P, Rosty C, Reiter RE, Wilentz RE, Murugesan SR, et al. Discovery of new markers of cancer through serial analysis of gene expression: prostate stem cell antigen is overexpressed in pancreatic adenocarcinoma. Cancer Res. 2001;61:4320–4324. [PubMed] [Google Scholar]
- 28.Argani P, Iacobuzio-Donahue C, Ryu B, Rosty C, Goggins M, et al. Mesothelin is overexpressed in the vast majority of ductal adenocarcinomas of the pancreas: identification of a new pancreatic cancer marker by serial analysis of gene expression (SAGE). Clin Cancer Res. 2001;7:3862–3868. [PubMed] [Google Scholar]
- 29.Sainaghi PP, Castello L, Bergamasco L, Galletti M, Bellosta P, et al. Gas6 induces proliferation in prostate carcinoma cell lines expressing the Axl receptor. J Cell Physiol. 2005;204:36–44. doi: 10.1002/jcp.20265. [DOI] [PubMed] [Google Scholar]
- 30.Ito T, Ito M, Naito S, Ohtsuru A, Nagayama Y, et al. Expression of the Axl receptor tyrosine kinase in human thyroid carcinoma. Thyroid. 1999;9:563–567. doi: 10.1089/thy.1999.9.563. [DOI] [PubMed] [Google Scholar]
- 31.Sun WS, Fujimoto J, Tamaya T. Coexpression of growth arrest-specific gene 6 and receptor tyrosine kinases Axl and Sky in human uterine endometrial cancers. Ann Oncol. 2003;14:898–906. doi: 10.1093/annonc/mdg257. [DOI] [PubMed] [Google Scholar]
- 32.O'Bryan JP, Frye RA, Cogswell PC, Neubauer A, Kitch B, et al. axl, a transforming gene isolated from primary human myeloid leukemia cells, encodes a novel receptor tyrosine kinase. Mol Cell Biol. 1991;11:5016–5031. doi: 10.1128/mcb.11.10.5016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wang PW, Eisenbart JD, Cordes SP, Barsh GS, Stoffel M, et al. Human KRML (MAFB): cDNA cloning, genomic structure, and evaluation as a candidate tumor suppressor gene in myeloid leukemias. Genomics. 1999;59:275–281. doi: 10.1006/geno.1999.5884. [DOI] [PubMed] [Google Scholar]
- 34.Stumpf E, Aalto Y, Hoog A, Kjellman M, Otonkoski T, et al. Chromosomal alterations in human pancreatic endocrine tumors. Genes Chromosomes Cancer. 2000;29:83–87. doi: 10.1002/1098-2264(2000)9999:9999<::aid-gcc1011>3.0.co;2-z. [DOI] [PubMed] [Google Scholar]
- 35.Terris B, Meddeb M, Marchio A, Danglot G, Flejou JF, et al. Comparative genomic hybridization analysis of sporadic neuroendocrine tumors of the digestive system. Genes Chromosomes Cancer. 1998;22:50–56. doi: 10.1002/(sici)1098-2264(199805)22:1<50::aid-gcc7>3.0.co;2-6. [DOI] [PubMed] [Google Scholar]
- 36.Weber CK, Sommer G, Michl P, Fensterer H, Weimer M, et al. Biglycan is overexpressed in pancreatic cancer and induces G1-arrest in pancreatic cancer cell lines. Gastroenterology. 2001;121:657–667. doi: 10.1053/gast.2001.27222. [DOI] [PubMed] [Google Scholar]
- 37.Bramhall SR, Stamp GW, Dunn J, Lemoine NR, Neoptolemos JP. Expression of collagenase (MMP2), stromelysin (MMP3) and tissue inhibitor of the metalloproteinases (TIMP1) in pancreatic and ampullary disease. Br J Cancer. 1996;73:972–978. doi: 10.1038/bjc.1996.190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.West RB, Nuyten DS, Subramanian S, Nielsen TO, Corless CL, et al. Determination of Stromal Signatures in Breast Carcinoma. PLoS Biol. 2005;3:e187. doi: 10.1371/journal.pbio.0030187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Leung JY, Kolligs FT, Wu R, Zhai Y, Kuick R, et al. Activation of AXIN2 expression by beta-catenin-T cell factor. A feedback repressor pathway regulating Wnt signaling. J Biol Chem. 2002;277:21657–21665. doi: 10.1074/jbc.M200139200. [DOI] [PubMed] [Google Scholar]
- 40.Lustig B, Jerchow B, Sachs M, Weiler S, Pietsch T, et al. Negative feedback loop of Wnt signaling through upregulation of conductin/axin2 in colorectal and liver tumors. Mol Cell Biol. 2002;22:1184–1193. doi: 10.1128/MCB.22.4.1184-1193.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Jho EH, Zhang T, Domon C, Joo CK, Freund JN, et al. Wnt/beta-catenin/Tcf signaling induces the transcription of Axin2, a negative regulator of the signaling pathway. Mol Cell Biol. 2002;22:1172–1183. doi: 10.1128/MCB.22.4.1172-1183.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Chen X, Cheung ST, So S, Fan ST, Barry C, et al. Gene expression patterns in human liver cancers. Mol Biol Cell. 2002;13:1929–1939. doi: 10.1091/mbc.02-02-0023.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Leung SY, Chen X, Chu KM, Yuen ST, Mathy J, et al. Phospholipase A2 group IIA expression in gastric adenocarcinoma is associated with prolonged survival and less frequent metastasis. Proc Natl Acad Sci U S A. 2002;99:16203–16208. doi: 10.1073/pnas.212646299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Dilley WG, Kalyanaraman S, Verma S, Cobb JP, Laramie JM, et al. Global gene expression in neuroendocrine tumors from patients with the MEN1 syndrome. Mol Cancer. 2005;4:9. doi: 10.1186/1476-4598-4-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Sorlie T, Perou CM, Tibshirani R, Aas T, Geisler S, et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci U S A. 2001;98:10869–10874. doi: 10.1073/pnas.191367098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Lowy AM, Fenoglio-Preiser C, Kim OJ, Kordich J, Gomez A, et al. Dysregulation of beta-catenin expression correlates with tumor differentiation in pancreatic duct adenocarcinoma. Ann Surg Oncol. 2003;10:284–290. doi: 10.1245/aso.2003.05.003. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
List of genes within the Adeno 1 and 2 clusters. Annotated gene list identified by significance analysis of microarrays versus all other non-adenocarcinoma pancreatic tissues. Only the 31 adenocarcinomas grouped together in Figure 1 were used for the analysis. Worksheets also contain analysis for the identification of putative secretory and membrane proteins and genes that are associated with DNA copy gains and losses as determined by CGH. A table of contents is contained in the first Excel worksheet.
(1.91 MB XLS)
Table of differentially expressed genes identified by SAM in adenocarcinomas whose genomic location showed high DNA copy gains or losses.
(0.03 MB XLS)
List of genes differentially expressed between primary pancreatic duct cells and whole normal pancreatic tissue obtained using SAM as described in the Methods.
(2.12 MB XLS)
List of genes within the islet cell tumor cluster. Annotated list of genes identified by SAM that are differentially expressed in islet tumors compared to all other pancreatic tissues in the dataset.
(0.43 MB XLS)
Identification of candidate genes capable of discriminating pancreatic adenocarcinomas or islet cell tumors from a diverse collection of normal human tissues. Putative secretory or membrane proteins enriched in pancreatic adenocarcinomas or islet cell tumors in the current study were examined in the context of a wide variety of normal tissues reported in reference [9].
(0.05 MB XLS)