Abstract
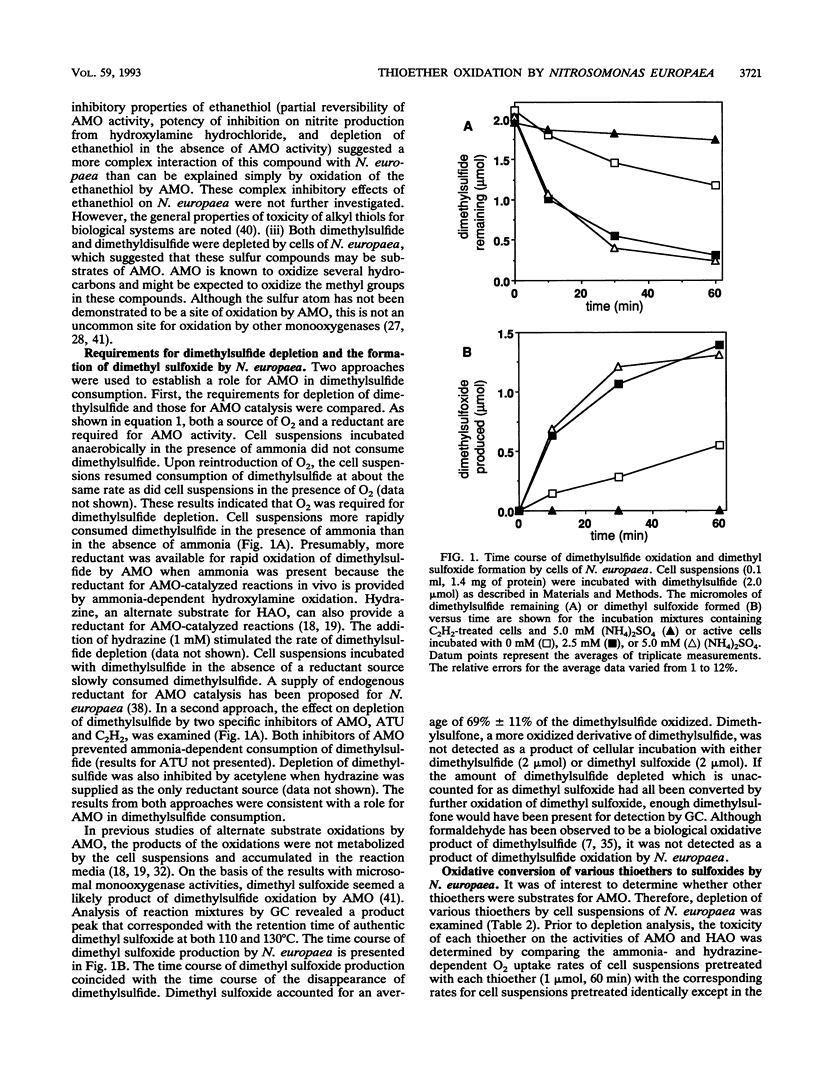
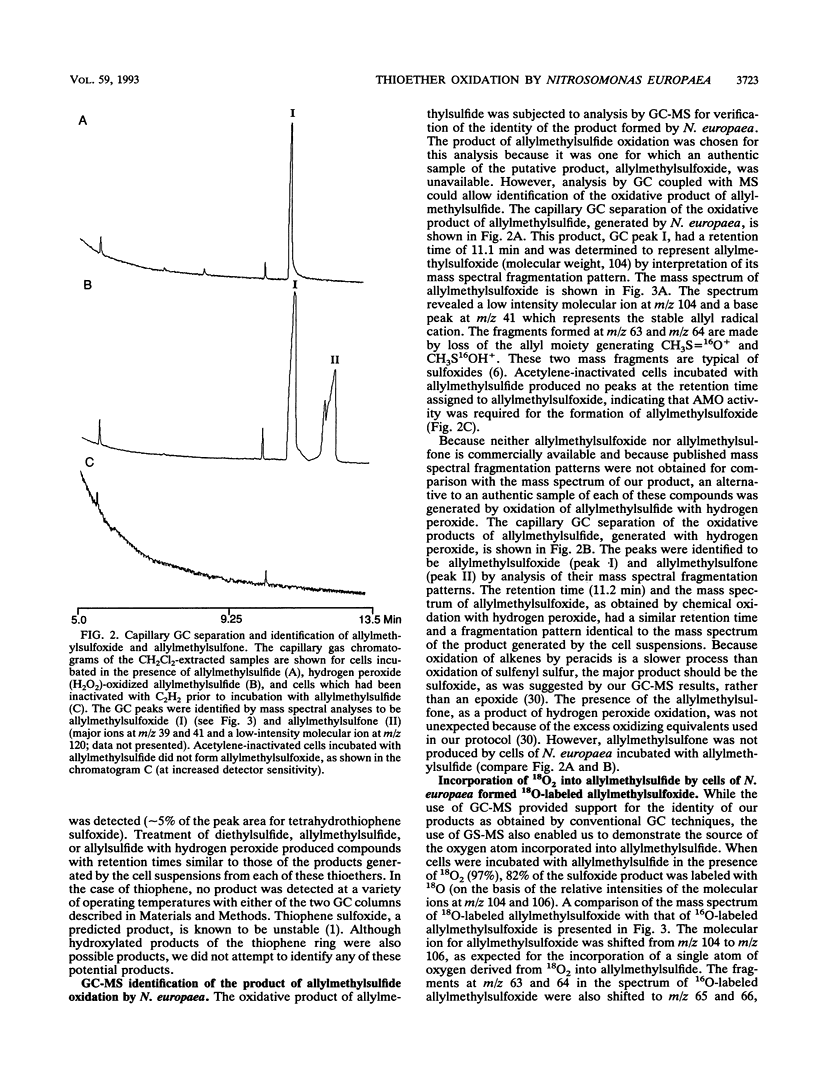
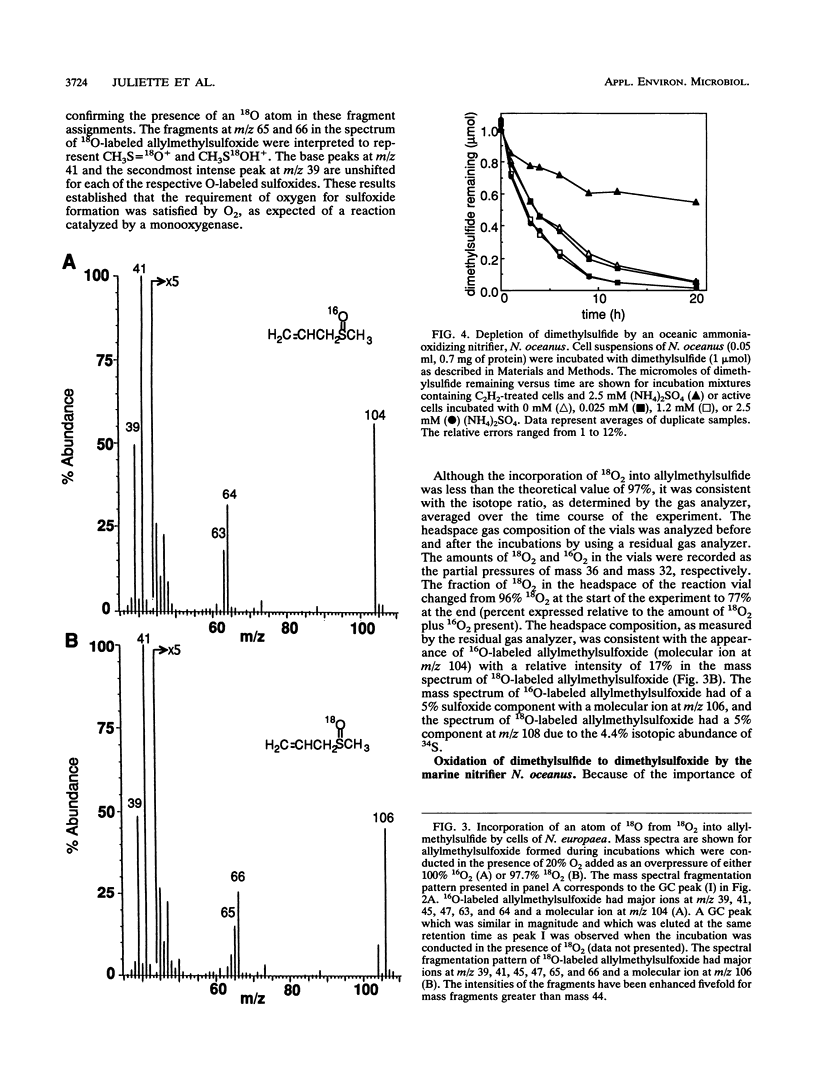
Organic sulfur compounds are well-known nitrification inhibitors. The inhibitory effects of dimethylsulfide, dimethyldisulfide, and ethanethiol on ammonia oxidation by Nitrosomonas europaea were examined. Both dimethylsulfide and dimethyldisulfide were weak inhibitors of ammonia oxidation and exhibited inhibitory characteristics typical of substrates for ammonia monooxygenase (AMO). Depletion of dimethylsulfide required O2 and was prevented with either acetylene or allylthiourea, two inhibitors of AMO. The inhibition of ammonia oxidation by dimethylsulfide was examined in detail. Cell suspensions incubated in the presence of ammonia oxidized dimethylsulfide to dimethyl sulfoxide. Depletion of six other thioethers was also prevented by treating cell suspensions with either allylthiourea or acetylene. The oxidative products of three thioethers were identified as the corresponding sulfoxides. The amount of sulfoxide formed accounted for a majority of the amount of sulfide depleted. By using gas chromatography coupled with mass spectrometry, allylmethylsulfide was shown to be oxidized to allylmethylsulfoxide by N. europaea with the incorporation of a single atom of 18O derived from 18O2 into the sulfide. This result supported our conclusion that a monooxygenase was involved in the oxidation of allylmethylsulfide. The thioethers are concluded to be a new class of substrates for AMO. This is the first report of the oxidation of the sulfur atom by AMO in whole cells of N. europaea. The ability of N. europaea to oxidize dimethylsulfide is not unique among the ammonia-oxidizing bacteria. Nitrosococcus oceanus, a marine nitrifier, was also demonstrated to oxidize dimethylsulfide to dimethyl sulfoxide.
Full text
PDF






Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BROWN W. T., QUASTEL J. H., SCHOLEFIELD P. G. Effects of mercapto compounds on soil nitrification. Appl Microbiol. 1954 Jul;2(4):235–239. doi: 10.1128/am.2.4.235-239.1954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bédard C., Knowles R. Physiology, biochemistry, and specific inhibitors of CH4, NH4+, and CO oxidation by methanotrophs and nitrifiers. Microbiol Rev. 1989 Mar;53(1):68–84. doi: 10.1128/mr.53.1.68-84.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HOFMAN T., LEES H. The biochemistry of the nitrifying organisms. IV. The respiration and intermediary metabolism of Nitrosomonas. Biochem J. 1953 Jul;54(4):579–583. doi: 10.1042/bj0540579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hyman M. R., Arp D. J. 14C2H2- and 14CO2-labeling studies of the de novo synthesis of polypeptides by Nitrosomonas europaea during recovery from acetylene and light inactivation of ammonia monooxygenase. J Biol Chem. 1992 Jan 25;267(3):1534–1545. [PubMed] [Google Scholar]
- Hyman M. R., Arp D. J. Quantification and removal of some contaminating gases from acetylene used to study gas-utilizing enzymes and microorganisms. Appl Environ Microbiol. 1987 Feb;53(2):298–303. doi: 10.1128/aem.53.2.298-303.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hyman M. R., Kim C. Y., Arp D. J. Inhibition of ammonia monooxygenase in Nitrosomonas europaea by carbon disulfide. J Bacteriol. 1990 Sep;172(9):4775–4782. doi: 10.1128/jb.172.9.4775-4782.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hyman M. R., Murton I. B., Arp D. J. Interaction of Ammonia Monooxygenase from Nitrosomonas europaea with Alkanes, Alkenes, and Alkynes. Appl Environ Microbiol. 1988 Dec;54(12):3187–3190. doi: 10.1128/aem.54.12.3187-3190.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hyman M. R., Wood P. M. Methane oxidation by Nitrosomonas europaea. Biochem J. 1983 Apr 15;212(1):31–37. doi: 10.1042/bj2120031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hyman M. R., Wood P. M. Suicidal inactivation and labelling of ammonia mono-oxygenase by acetylene. Biochem J. 1985 May 1;227(3):719–725. doi: 10.1042/bj2270719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kiene R. P. Dimethyl sulfide production from dimethylsulfoniopropionate in coastal seawater samples and bacterial cultures. Appl Environ Microbiol. 1990 Nov;56(11):3292–3297. doi: 10.1128/aem.56.11.3292-3297.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LEES H. The biochemistry of the nitrifying organisms. I. The ammonia oxidizing systems of Nitrosomonas. Biochem J. 1952 Sep;52(1):134–139. doi: 10.1042/bj0520134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- May S. W., Phillips R. S., Mueller P. W., Herman H. H. Dopamine beta-hydroxylase. Comparative specificities and mechanisms of the oxygenation reactions. J Biol Chem. 1981 Aug 25;256(16):8470–8475. [PubMed] [Google Scholar]
- Rasche M. E., Hicks R. E., Hyman M. R., Arp D. J. Oxidation of monohalogenated ethanes and n-chlorinated alkanes by whole cells of Nitrosomonas europaea. J Bacteriol. 1990 Sep;172(9):5368–5373. doi: 10.1128/jb.172.9.5368-5373.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rasche M. E., Hyman M. R., Arp D. J. Factors Limiting Aliphatic Chlorocarbon Degradation by Nitrosomonas europaea: Cometabolic Inactivation of Ammonia Monooxygenase and Substrate Specificity. Appl Environ Microbiol. 1991 Oct;57(10):2986–2994. doi: 10.1128/aem.57.10.2986-2994.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shears J. H., Wood P. M. Spectroscopic evidence for a photosensitive oxygenated state of ammonia mono-oxygenase. Biochem J. 1985 Mar 1;226(2):499–507. doi: 10.1042/bj2260499. [DOI] [PMC free article] [PubMed] [Google Scholar]


