Abstract
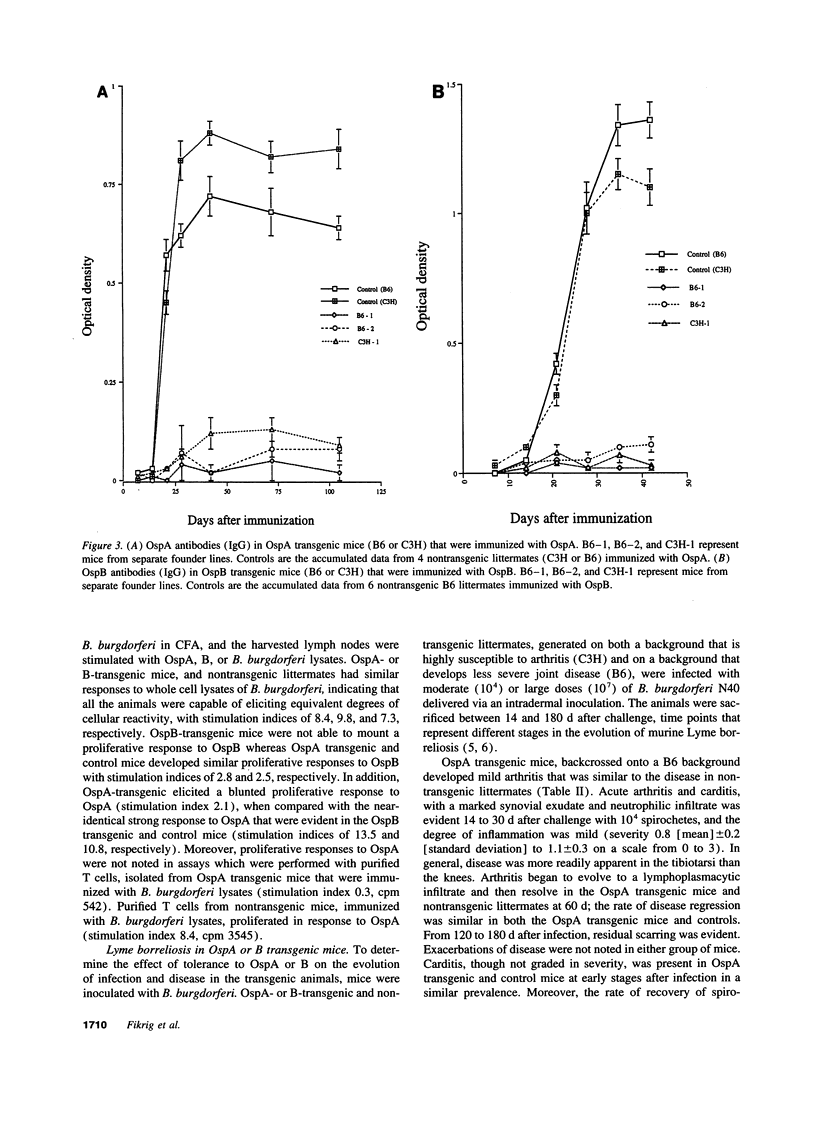
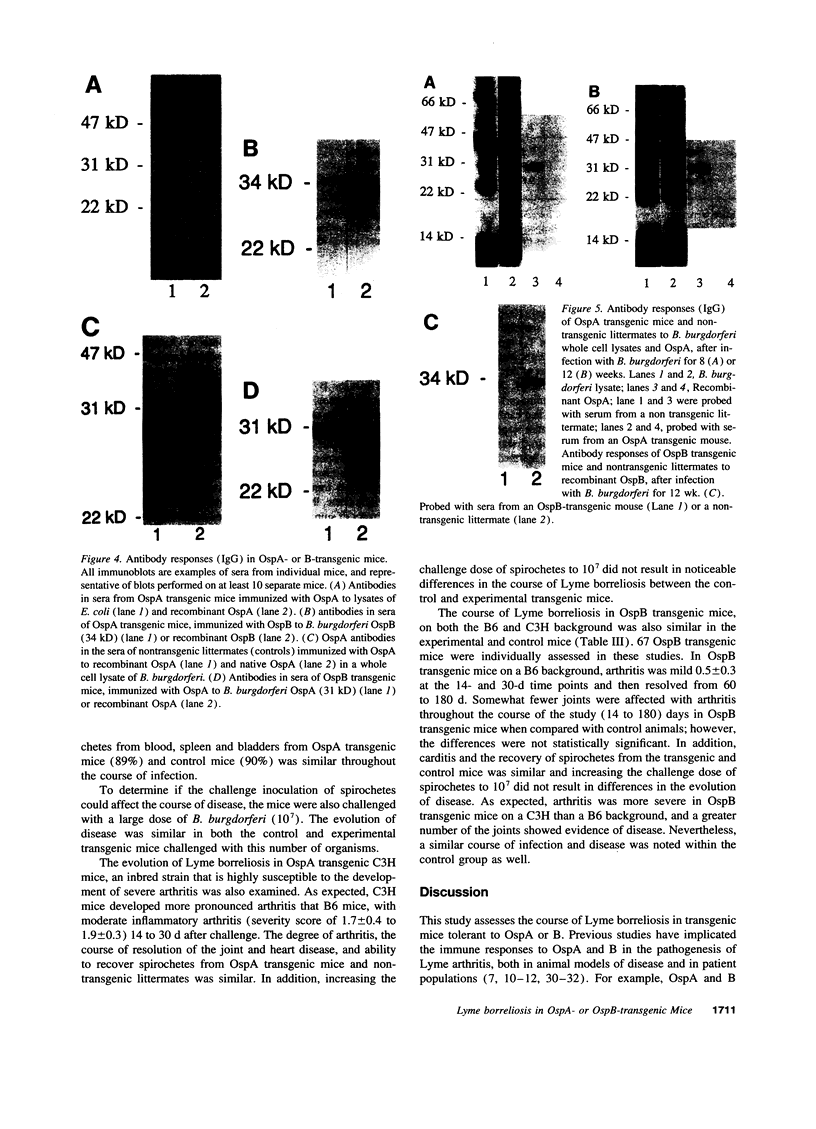
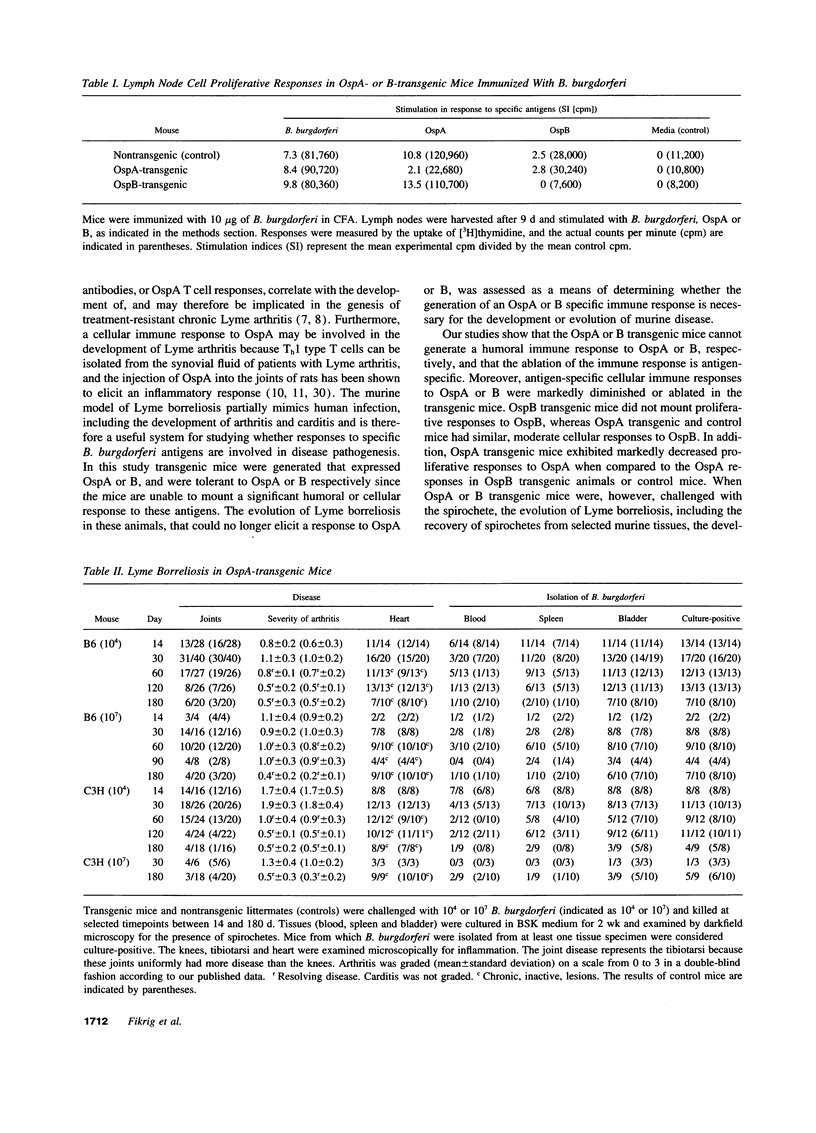
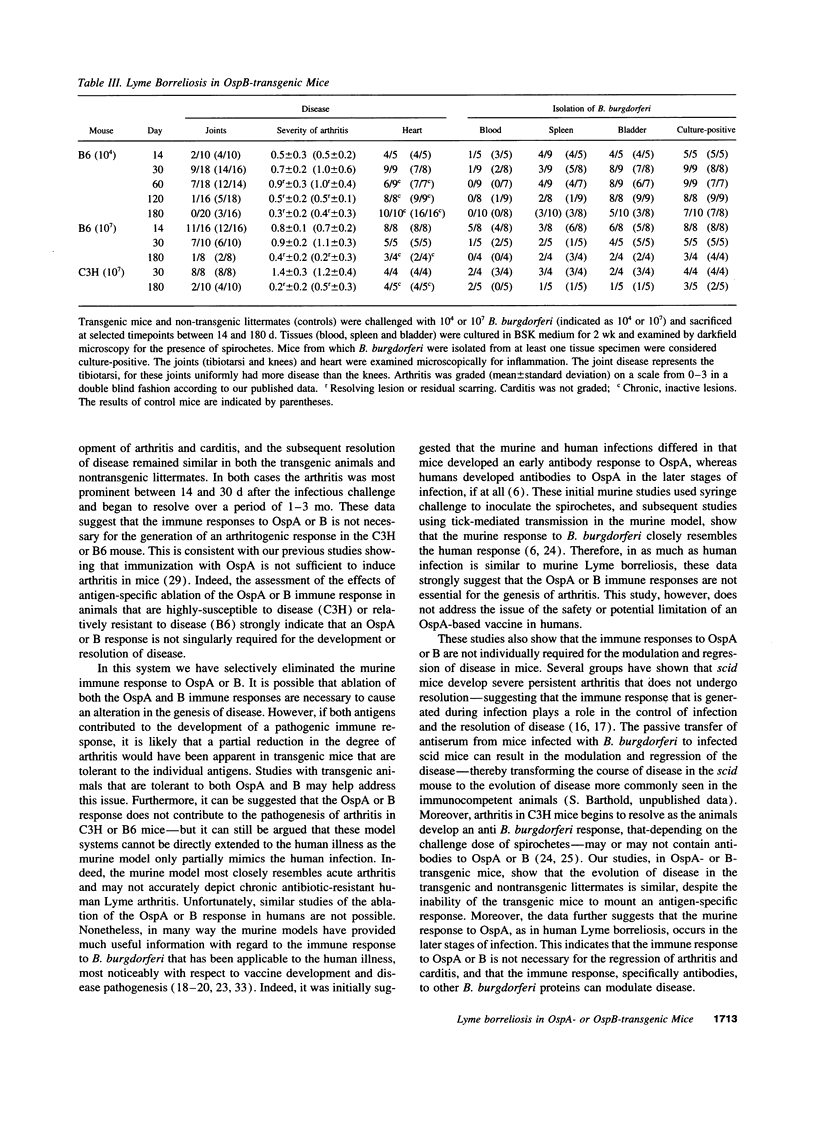
The evolution of Lyme borreliosis in transgenic mice tolerant to Borrelia burgdorferi outer surface proteins (Osps) A or B was assessed to investigate the role of immunity to OspA or B in infection and pathogenesis of Lyme disease. Antibodies to OspA or B protect immunocompetent C3H/HeJ or C.B.17 severe combined immunodeficient (scid) mice from challenge with B. burgdorferi. Moreover, arthritis in infected C3H mice resolves with the rise of high titers of B. burgdorferi specific antibodies, including OspA and B, whereas disease persists in scid mice--suggesting that the regression of arthritis may be due to the development of borreliacidal OspA or B antibodies. To evaluate the course of Lyme borreliosis in OspA or B tolerant mice we developed transgenic mice that expressed OspA or B under control of the major histocompatibility complex (MHC) class I promoter. Mice carrying OspA or B transgenes on a C3H/HeJ (C3H, disease-susceptible) or C57BL/6 (B6, disease-resistant) background, immunized with OspA or B, did not mount a humoral or cellular immune response to OspA or B, respectively, but responded normally to other B. burgdorferi antigens. The evolution of Lyme borreliosis, including infection and the development of arthritis and carditis, was similar in transgenic and nontransgenic littermates suggesting that an OspA or B immune response is not singularly involved in either the genesis or regression of Lyme disease in C3H or B6 mice.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bachmann M. F., Rohrer U. H., Kündig T. M., Bürki K., Hengartner H., Zinkernagel R. M. The influence of antigen organization on B cell responsiveness. Science. 1993 Nov 26;262(5138):1448–1451. doi: 10.1126/science.8248784. [DOI] [PubMed] [Google Scholar]
- Barthold S. W., Beck D. S., Hansen G. M., Terwilliger G. A., Moody K. D. Lyme borreliosis in selected strains and ages of laboratory mice. J Infect Dis. 1990 Jul;162(1):133–138. doi: 10.1093/infdis/162.1.133. [DOI] [PubMed] [Google Scholar]
- Barthold S. W., Persing D. H., Armstrong A. L., Peeples R. A. Kinetics of Borrelia burgdorferi dissemination and evolution of disease after intradermal inoculation of mice. Am J Pathol. 1991 Aug;139(2):263–273. [PMC free article] [PubMed] [Google Scholar]
- Barthold S. W., Sidman C. L., Smith A. L. Lyme borreliosis in genetically resistant and susceptible mice with severe combined immunodeficiency. Am J Trop Med Hyg. 1992 Nov;47(5):605–613. doi: 10.4269/ajtmh.1992.47.605. [DOI] [PubMed] [Google Scholar]
- Barthold S. W., de Souza M. S., Janotka J. L., Smith A. L., Persing D. H. Chronic Lyme borreliosis in the laboratory mouse. Am J Pathol. 1993 Sep;143(3):959–971. [PMC free article] [PubMed] [Google Scholar]
- Dunn J. J., Lade B. N., Barbour A. G. Outer surface protein A (OspA) from the Lyme disease spirochete, Borrelia burgdorferi: high level expression and purification of a soluble recombinant form of OspA. Protein Expr Purif. 1990 Nov;1(2):159–168. doi: 10.1016/1046-5928(90)90011-m. [DOI] [PubMed] [Google Scholar]
- Erdile L. F., Brandt M. A., Warakomski D. J., Westrack G. J., Sadziene A., Barbour A. G., Mays J. P. Role of attached lipid in immunogenicity of Borrelia burgdorferi OspA. Infect Immun. 1993 Jan;61(1):81–90. doi: 10.1128/iai.61.1.81-90.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fikrig E., Barthold S. W., Flavell R. A. OspA vaccination of mice with established Borrelia burgdorferi infection alters disease but not infection. Infect Immun. 1993 Jun;61(6):2553–2557. doi: 10.1128/iai.61.6.2553-2557.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fikrig E., Barthold S. W., Kantor F. S., Flavell R. A. Protection of mice against the Lyme disease agent by immunizing with recombinant OspA. Science. 1990 Oct 26;250(4980):553–556. doi: 10.1126/science.2237407. [DOI] [PubMed] [Google Scholar]
- Fikrig E., Berland R., Chen M., Williams S., Sigal L. H., Flavell R. A. Serologic response to the Borrelia burgdorferi flagellin demonstrates an epitope common to a neuroblastoma cell line. Proc Natl Acad Sci U S A. 1993 Jan 1;90(1):183–187. doi: 10.1073/pnas.90.1.183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fikrig E., Bockenstedt L. K., Barthold S. W., Chen M., Tao H., Ali-Salaam P., Telford S. R., Flavell R. A. Sera from patients with chronic Lyme disease protect mice from Lyme borreliosis. J Infect Dis. 1994 Mar;169(3):568–574. doi: 10.1093/infdis/169.3.568. [DOI] [PubMed] [Google Scholar]
- Fikrig E., Tao H., Kantor F. S., Barthold S. W., Flavell R. A. Evasion of protective immunity by Borrelia burgdorferi by truncation of outer surface protein B. Proc Natl Acad Sci U S A. 1993 May 1;90(9):4092–4096. doi: 10.1073/pnas.90.9.4092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gondolf K. B., Mihatsch M., Curschellas E., Dunn J. J., Batsford S. R. Induction of experimental allergic arthritis with outer surface proteins of Borrelia burgdorferi. Arthritis Rheum. 1994 Jul;37(7):1070–1077. doi: 10.1002/art.1780370713. [DOI] [PubMed] [Google Scholar]
- Kalish R. A., Leong J. M., Steere A. C. Association of treatment-resistant chronic Lyme arthritis with HLA-DR4 and antibody reactivity to OspA and OspB of Borrelia burgdorferi. Infect Immun. 1993 Jul;61(7):2774–2779. doi: 10.1128/iai.61.7.2774-2779.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lahesmaa R., Shanafelt M. C., Allsup A., Soderberg C., Anzola J., Freitas V., Turck C., Steinman L., Peltz G. Preferential usage of T cell antigen receptor V region gene segment V beta 5.1 by Borrelia burgdorferi antigen-reactive T cell clones isolated from a patient with Lyme disease. J Immunol. 1993 May 1;150(9):4125–4135. [PubMed] [Google Scholar]
- Lengl-Janssen B., Strauss A. F., Steere A. C., Kamradt T. The T helper cell response in Lyme arthritis: differential recognition of Borrelia burgdorferi outer surface protein A in patients with treatment-resistant or treatment-responsive Lyme arthritis. J Exp Med. 1994 Dec 1;180(6):2069–2078. doi: 10.1084/jem.180.6.2069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Probert W. S., LeFebvre R. B. Protection of C3H/HeN mice from challenge with Borrelia burgdorferi through active immunization with OspA, OspB, or OspC, but not with OspD or the 83-kilodalton antigen. Infect Immun. 1994 May;62(5):1920–1926. doi: 10.1128/iai.62.5.1920-1926.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schaible U. E., Kramer M. D., Eichmann K., Modolell M., Museteanu C., Simon M. M. Monoclonal antibodies specific for the outer surface protein A (OspA) of Borrelia burgdorferi prevent Lyme borreliosis in severe combined immunodeficiency (scid) mice. Proc Natl Acad Sci U S A. 1990 May;87(10):3768–3772. doi: 10.1073/pnas.87.10.3768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schaible U. E., Kramer M. D., Wallich R., Tran T., Simon M. M. Experimental Borrelia burgdorferi infection in inbred mouse strains: antibody response and association of H-2 genes with resistance and susceptibility to development of arthritis. Eur J Immunol. 1991 Oct;21(10):2397–2405. doi: 10.1002/eji.1830211016. [DOI] [PubMed] [Google Scholar]
- Sears J. E., Fikrig E., Nakagawa T. Y., Deponte K., Marcantonio N., Kantor F. S., Flavell R. A. Molecular mapping of Osp-A mediated immunity against Borrelia burgdorferi, the agent of Lyme disease. J Immunol. 1991 Sep 15;147(6):1995–2000. [PubMed] [Google Scholar]
- Shanafelt M. C., Anzola J., Soderberg C., Yssel H., Turck C. W., Peltz G. Epitopes on the outer surface protein A of Borrelia burgdorferi recognized by antibodies and T cells of patients with Lyme disease. J Immunol. 1992 Jan 1;148(1):218–224. [PubMed] [Google Scholar]
- Shanafelt M. C., Hindersson P., Soderberg C., Mensi N., Turck C. W., Webb D., Yssel H., Peltz G. T cell and antibody reactivity with the Borrelia burgdorferi 60-kDa heat shock protein in Lyme arthritis. J Immunol. 1991 Jun 1;146(11):3985–3992. [PubMed] [Google Scholar]
- Steere A. C., Dwyer E., Winchester R. Association of chronic Lyme arthritis with HLA-DR4 and HLA-DR2 alleles. N Engl J Med. 1990 Jul 26;323(4):219–223. doi: 10.1056/NEJM199007263230402. [DOI] [PubMed] [Google Scholar]
- Steere A. C., Grodzicki R. L., Kornblatt A. N., Craft J. E., Barbour A. G., Burgdorfer W., Schmid G. P., Johnson E., Malawista S. E. The spirochetal etiology of Lyme disease. N Engl J Med. 1983 Mar 31;308(13):733–740. doi: 10.1056/NEJM198303313081301. [DOI] [PubMed] [Google Scholar]
- Steere A. C. Lyme disease. N Engl J Med. 1989 Aug 31;321(9):586–596. doi: 10.1056/NEJM198908313210906. [DOI] [PubMed] [Google Scholar]
- Stover C. K., Bansal G. P., Hanson M. S., Burlein J. E., Palaszynski S. R., Young J. F., Koenig S., Young D. B., Sadziene A., Barbour A. G. Protective immunity elicited by recombinant bacille Calmette-Guerin (BCG) expressing outer surface protein A (OspA) lipoprotein: a candidate Lyme disease vaccine. J Exp Med. 1993 Jul 1;178(1):197–209. doi: 10.1084/jem.178.1.197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang L., Ma Y., Schoenfeld R., Griffiths M., Eichwald E., Araneo B., Weis J. J. Evidence for B-lymphocyte mitogen activity in Borrelia burgdorferi-infected mice. Infect Immun. 1992 Aug;60(8):3033–3041. doi: 10.1128/iai.60.8.3033-3041.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang L., Weis J. H., Eichwald E., Kolbert C. P., Persing D. H., Weis J. J. Heritable susceptibility to severe Borrelia burgdorferi-induced arthritis is dominant and is associated with persistence of large numbers of spirochetes in tissues. Infect Immun. 1994 Feb;62(2):492–500. doi: 10.1128/iai.62.2.492-500.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yssel H., Nakamoto T., Schneider P., Freitas V., Collins C., Webb D., Mensi N., Soderberg C., Peltz G. Analysis of T lymphocytes cloned from the synovial fluid and blood of a patient with Lyme arthritis. Int Immunol. 1990;2(11):1081–1089. doi: 10.1093/intimm/2.11.1081. [DOI] [PubMed] [Google Scholar]
- Yssel H., Shanafelt M. C., Soderberg C., Schneider P. V., Anzola J., Peltz G. Borrelia burgdorferi activates a T helper type 1-like T cell subset in Lyme arthritis. J Exp Med. 1991 Sep 1;174(3):593–601. doi: 10.1084/jem.174.3.593. [DOI] [PMC free article] [PubMed] [Google Scholar]