Abstract
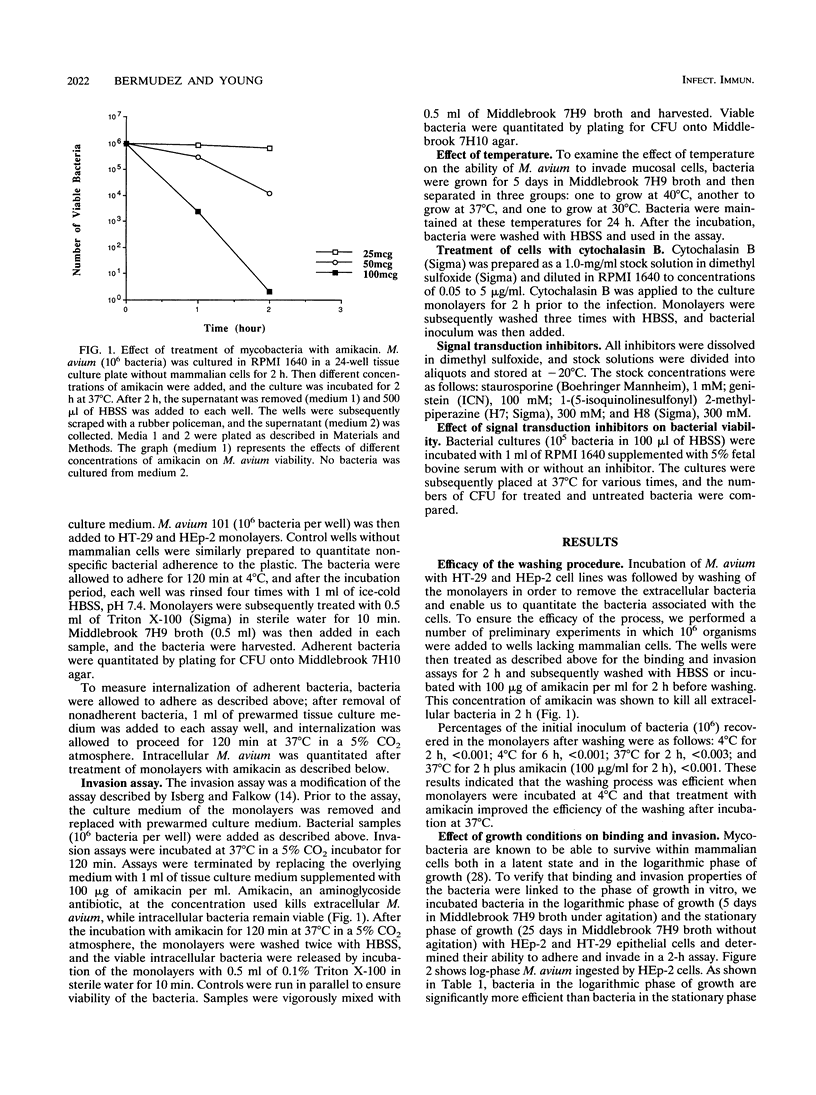
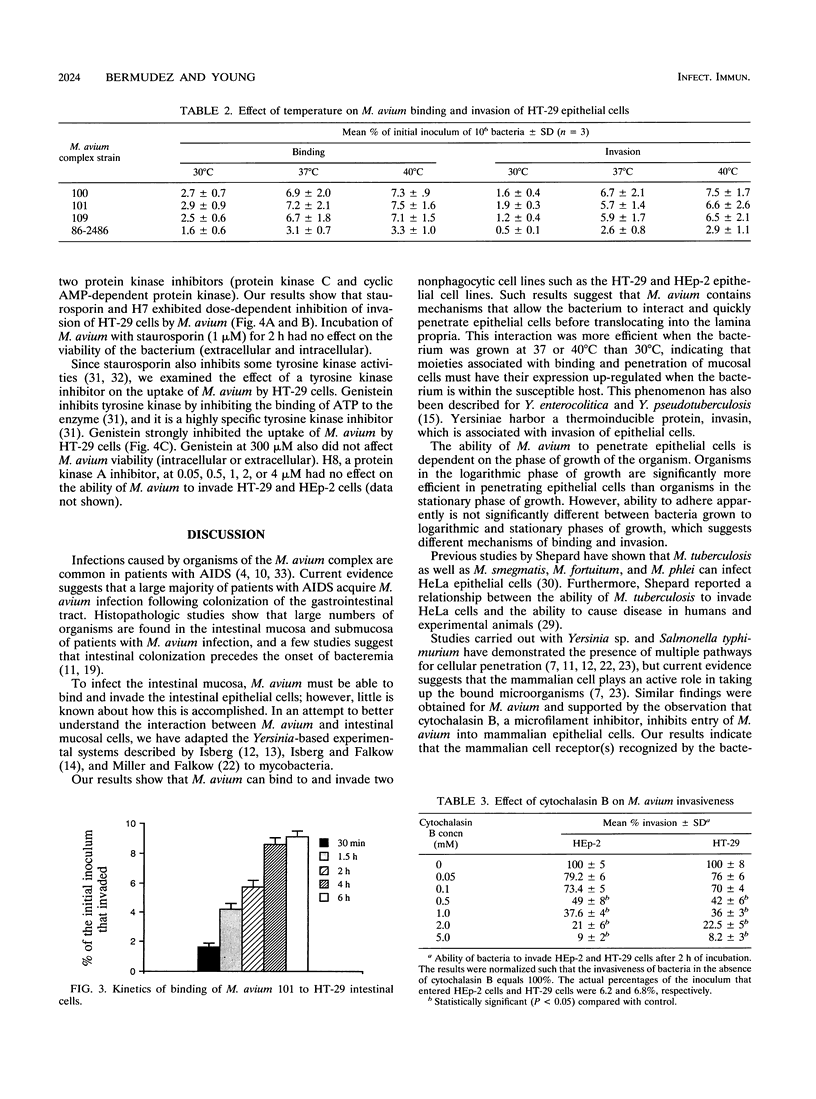
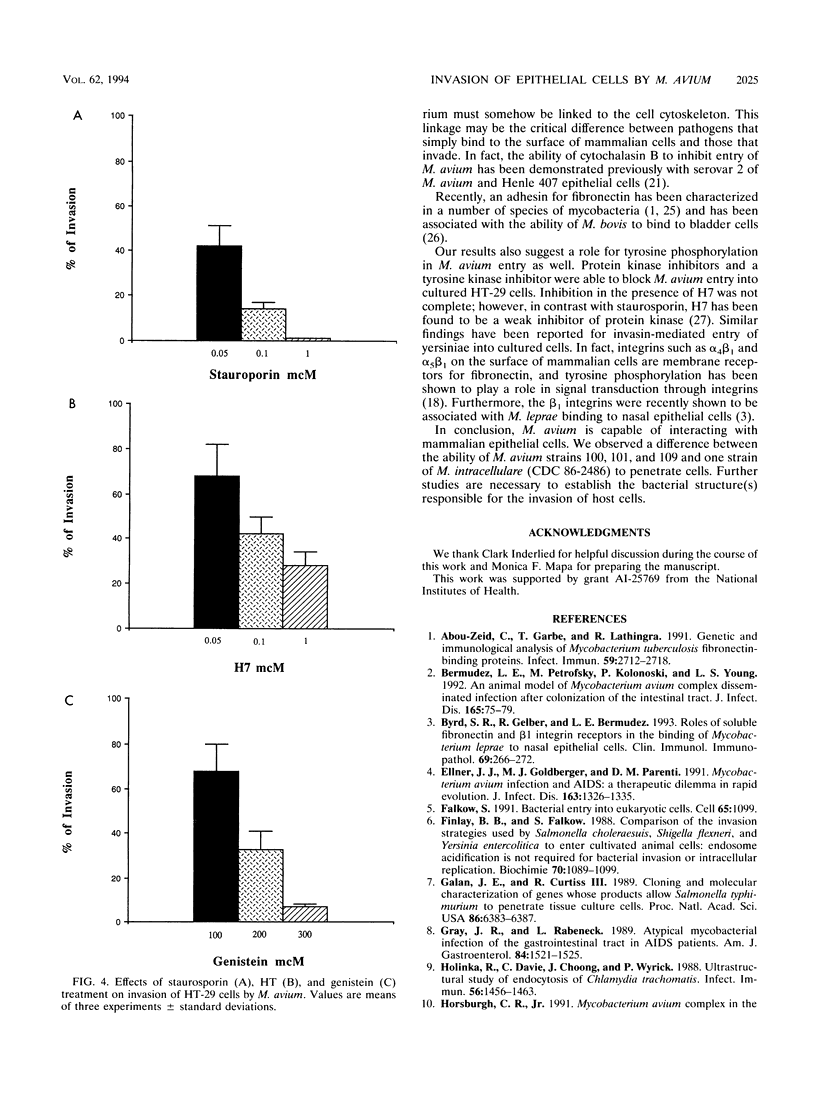
Organisms of the Mycobacterium avium complex cause disseminated blood-borne infection in patients with AIDS, who acquire the infection mainly through the gastrointestinal tract. Prior to causing infection, M. avium must colonize and invade the intestinal mucosa. This study examined the ability of several serovars of the M. avium complex to bind to and invade the HT-29 intestinal mucosal cell line and the HEp-2 laryngeal cell line. Logarithmic-phase M. avium was more efficient in binding and invasion than organisms in the stationary phase of growth. Bacteria incubated at 37 and 40 degrees C adhered to and invaded HT-29 cells more efficiently than bacteria cultured at 30 degrees C. The ability of M. avium to invade HT-29 and HEp-2 cells was inhibited when the cells were incubated with cytochalasin B prior to exposure to the bacterium, suggesting active participation of the mammalian cell in the process of internalization. Two protein kinase inhibitors, staurosporine and H7, blocked invasion of M. avium, and a specific tyrosine protein kinase inhibitor, genistein, also blocked the internalization but not the binding of bacteria. The findings suggest that M. avium binds to a specific receptor(s) on the epithelial cells and uses the cytoskeleton of the mammalian cell to become internalized.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abou-Zeid C., Garbe T., Lathigra R., Wiker H. G., Harboe M., Rook G. A., Young D. B. Genetic and immunological analysis of Mycobacterium tuberculosis fibronectin-binding proteins. Infect Immun. 1991 Aug;59(8):2712–2718. doi: 10.1128/iai.59.8.2712-2718.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bermudez L. E., Petrofsky M., Kolonoski P., Young L. S. An animal model of Mycobacterium avium complex disseminated infection after colonization of the intestinal tract. J Infect Dis. 1992 Jan;165(1):75–79. doi: 10.1093/infdis/165.1.75. [DOI] [PubMed] [Google Scholar]
- Byrd S. R., Gelber R., Bermudez L. E. Roles of soluble fibronectin and beta 1 integrin receptors in the binding of Mycobacterium leprae to nasal epithelial cells. Clin Immunol Immunopathol. 1993 Dec;69(3):266–271. doi: 10.1006/clin.1993.1179. [DOI] [PubMed] [Google Scholar]
- Ellner J. J., Goldberger M. J., Parenti D. M. Mycobacterium avium infection and AIDS: a therapeutic dilemma in rapid evolution. J Infect Dis. 1991 Jun;163(6):1326–1335. doi: 10.1093/infdis/163.6.1326. [DOI] [PubMed] [Google Scholar]
- Falkow S. Bacterial entry into eukaryotic cells. Cell. 1991 Jun 28;65(7):1099–1102. doi: 10.1016/0092-8674(91)90003-h. [DOI] [PubMed] [Google Scholar]
- Finlay B. B., Falkow S. Comparison of the invasion strategies used by Salmonella cholerae-suis, Shigella flexneri and Yersinia enterocolitica to enter cultured animal cells: endosome acidification is not required for bacterial invasion or intracellular replication. Biochimie. 1988 Aug;70(8):1089–1099. doi: 10.1016/0300-9084(88)90271-4. [DOI] [PubMed] [Google Scholar]
- Galán J. E., Curtiss R., 3rd Cloning and molecular characterization of genes whose products allow Salmonella typhimurium to penetrate tissue culture cells. Proc Natl Acad Sci U S A. 1989 Aug;86(16):6383–6387. doi: 10.1073/pnas.86.16.6383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gray J. R., Rabeneck L. Atypical mycobacterial infection of the gastrointestinal tract in AIDS patients. Am J Gastroenterol. 1989 Dec;84(12):1521–1524. [PubMed] [Google Scholar]
- Hodinka R. L., Davis C. H., Choong J., Wyrick P. B. Ultrastructural study of endocytosis of Chlamydia trachomatis by McCoy cells. Infect Immun. 1988 Jun;56(6):1456–1463. doi: 10.1128/iai.56.6.1456-1463.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Isberg R. R. Discrimination between intracellular uptake and surface adhesion of bacterial pathogens. Science. 1991 May 17;252(5008):934–938. doi: 10.1126/science.1674624. [DOI] [PubMed] [Google Scholar]
- Isberg R. R., Falkow S. A single genetic locus encoded by Yersinia pseudotuberculosis permits invasion of cultured animal cells by Escherichia coli K-12. Nature. 1985 Sep 19;317(6034):262–264. doi: 10.1038/317262a0. [DOI] [PubMed] [Google Scholar]
- Isberg R. R. Pathways for the penetration of enteroinvasive Yersinia into mammalian cells. Mol Biol Med. 1990 Feb;7(1):73–82. [PubMed] [Google Scholar]
- Isberg R. R., Swain A., Falkow S. Analysis of expression and thermoregulation of the Yersinia pseudotuberculosis inv gene with hybrid proteins. Infect Immun. 1988 Aug;56(8):2133–2138. doi: 10.1128/iai.56.8.2133-2138.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Isberg R. R., Voorhis D. L., Falkow S. Identification of invasin: a protein that allows enteric bacteria to penetrate cultured mammalian cells. Cell. 1987 Aug 28;50(5):769–778. doi: 10.1016/0092-8674(87)90335-7. [DOI] [PubMed] [Google Scholar]
- Jacobson M. A., Hopewell P. C., Yajko D. M., Hadley W. K., Lazarus E., Mohanty P. K., Modin G. W., Feigal D. W., Cusick P. S., Sande M. A. Natural history of disseminated Mycobacterium avium complex infection in AIDS. J Infect Dis. 1991 Nov;164(5):994–998. doi: 10.1093/infdis/164.5.994. [DOI] [PubMed] [Google Scholar]
- Juliano R. L., Haskill S. Signal transduction from the extracellular matrix. J Cell Biol. 1993 Feb;120(3):577–585. doi: 10.1083/jcb.120.3.577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klatt E. C., Jensen D. F., Meyer P. R. Pathology of Mycobacterium avium-intracellulare infection in acquired immunodeficiency syndrome. Hum Pathol. 1987 Jul;18(7):709–714. doi: 10.1016/s0046-8177(87)80242-3. [DOI] [PubMed] [Google Scholar]
- Lee C. A., Falkow S. The ability of Salmonella to enter mammalian cells is affected by bacterial growth state. Proc Natl Acad Sci U S A. 1990 Jun;87(11):4304–4308. doi: 10.1073/pnas.87.11.4304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mapother M. E., Songer J. G. In vitro interaction of Mycobacterium avium with intestinal epithelial cells. Infect Immun. 1984 Jul;45(1):67–73. doi: 10.1128/iai.45.1.67-73.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller V. L., Falkow S. Evidence for two genetic loci in Yersinia enterocolitica that can promote invasion of epithelial cells. Infect Immun. 1988 May;56(5):1242–1248. doi: 10.1128/iai.56.5.1242-1248.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller V. L., Finlay B. B., Falkow S. Factors essential for the penetration of mammalian cells by Yersinia. Curr Top Microbiol Immunol. 1988;138:15–39. [PubMed] [Google Scholar]
- Orme I. M., Furney S. K., Roberts A. D. Dissemination of enteric Mycobacterium avium infections in mice rendered immunodeficient by thymectomy and CD4 depletion or by prior infection with murine AIDS retroviruses. Infect Immun. 1992 Nov;60(11):4747–4753. doi: 10.1128/iai.60.11.4747-4753.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ratliff T. L., McCarthy R., Telle W. B., Brown E. J. Purification of a mycobacterial adhesin for fibronectin. Infect Immun. 1993 May;61(5):1889–1894. doi: 10.1128/iai.61.5.1889-1894.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ratliff T. L., Palmer J. O., McGarr J. A., Brown E. J. Intravesical Bacillus Calmette-Guérin therapy for murine bladder tumors: initiation of the response by fibronectin-mediated attachment of Bacillus Calmette-Guérin. Cancer Res. 1987 Apr 1;47(7):1762–1766. [PubMed] [Google Scholar]
- Rosenshine I., Duronio V., Finlay B. B. Tyrosine protein kinase inhibitors block invasin-promoted bacterial uptake by epithelial cells. Infect Immun. 1992 Jun;60(6):2211–2217. doi: 10.1128/iai.60.6.2211-2217.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SHEPARD C. C. Growth characteristics in HeLa cells of the rapidly growing acid fast bacteria, Mycobacterium fortuitum, Mycobacterium phlei, and Mycobacterium smegmatis. J Bacteriol. 1957 Jun;73(6):722–726. doi: 10.1128/jb.73.6.722-726.1957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SHEPARD C. C. Growth characteristics of tubercle bacilli and certain other mycobacteria in HeLa cells. J Exp Med. 1957 Jan 1;105(1):39–48. doi: 10.1084/jem.105.1.39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tamaoki T., Nakano H. Potent and specific inhibitors of protein kinase C of microbial origin. Biotechnology (N Y) 1990 Aug;8(8):732–735. doi: 10.1038/nbt0890-732. [DOI] [PubMed] [Google Scholar]
- Watson S. P., McNally J., Shipman L. J., Godfrey P. P. The action of the protein kinase C inhibitor, staurosporine, on human platelets. Evidence against a regulatory role for protein kinase C in the formation of inositol trisphosphate by thrombin. Biochem J. 1988 Jan 15;249(2):345–350. doi: 10.1042/bj2490345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young L. S., Inderlied C. B., Berlin O. G., Gottlieb M. S. Mycobacterial infections in AIDS patients, with an emphasis on the Mycobacterium avium complex. Rev Infect Dis. 1986 Nov-Dec;8(6):1024–1033. doi: 10.1093/clinids/8.6.1024. [DOI] [PubMed] [Google Scholar]



