Abstract
Thrombopoietin (TPO) acts as a lineage-specific late-acting factor to stimulate megakaryocyte and platelet formation. However, analysis of mice lacking either the cytokine or its receptor, c-Mpl, also revealed deficiencies in progenitor cells of multiple hematopoietic lineages, suggesting that TPO signaling may play an important role in the regulation of the hematopoietic stem cell compartment. To investigate this hypothesis, we determined preprogenitor and colony forming unit-spleen (CFU-S) numbers and analyzed the long-term hematopoietic repopulating capacity of bone marrow cells from mpl−/− mice. mpl−/− mice had 4- to 12-fold fewer preprogenitor cells than wild-type mice. In irradiated normal recipients, mpl−/− bone marrow generated 8- to 10-fold fewer spleen colonies than wild-type marrow at both 8 and 12 days after transplantation. This defect was intrinsic to the transplanted hematopoietic cells, as the microenvironment of mpl−/− recipients supported similar CFU-S growth to that observed in wild-type recipients. In definitive assays of stem cell function, bone marrow cells from mpl−/− mice failed to compete effectively with normal cells for long-term reconstitution of the hematopoietic organs of irradiated recipients, even when transplanted in 10-fold excess. Serial transplantation studies further suggested that stem cell self-renewal also may be compromised in mpl−/− mice. These data imply that TPO, signaling through c-Mpl, plays a vital physiological role in the regulation of hematopoietic stem cell production and function.
Thrombopoietin (TPO) is a key regulator of megakaryocyte and platelet formation (1). In semisolid cultures of bone marrow, spleen, or cord blood cells, TPO specifically stimulates the formation of small colonies of mature megakaryocytes (2–5). This lineage-restricted action also is observed in vivo, where administration of TPO elevates the number of megakaryocytes and circulating platelets without affecting the levels of erythroid or white cells (5–8). The magnitude of the thrombocytosis induced by TPO is the largest yet observed for an administered cytokine, and the indispensible physiological role of TPO and its receptor in thrombopoiesis has been confirmed by analysis of mice that lack the genes for TPO or its receptor, c-mpl. The bone marrow and spleen of TPO−/− or mpl−/− mice are largely devoid of megakaryocytes, and the mice are severely thrombocytopenic (9–11). Thus, consistent with the effects on administration of the purified cytokine, under physiological conditions within the mature hematopoietic compartment, intact TPO signaling is required specifically for megakaryocytopoiesis.
Despite the selectivity of TPO action on mature hematopoietic cells, the defects in c-mpl and TPO-deficient mice are not restricted to the megakaryocytic lineage. In vitro clonogenic assays revealed that the number of hematopoietic progenitor cells committed to multiple lineages in the bone marrow of mpl−/− mice was less than half the normal value (10). The spleens of mpl−/− mice were similarly deficient and the reduction in hematopoietic progenitor cells was also evident in TPO−/− mice (10, 12). Together with evidence suggesting that c-Mpl is expressed on a subset of immature hematopoietic cells (13, 14), and that v-mpl, a constitutively active receptor form, induces a myeloproliferative disease characterized by proliferation of factor-independent progenitor cells of multiple lineages (15), the deficiency in progenitor cells evident in mpl- and TPO-deficient mice may reflect an important role for TPO signaling in multipotential hematopoietic cells. Recent in vitro studies are consistent with such a role, having demonstrated stimulatory effects of TPO, in concert with other early-acting cytokines, on the growth, survival, and multilineage differentiation of hematopoietic populations enriched for primitive cells (16–22).
To investigate directly the role of TPO signaling within the primitive hematopoietic compartment in vivo, the numbers of preprogenitor cells and colony forming unit-spleen (CFU-S) in the bone marrow of mpl−/− mice were compared with those of wild-type animals, and the effect of c-mpl deficiency on hematopoietic stem cells was examined in competitive long-term repopulating assays. The number of cells in each of these immature hematopoietic populations was dramatically reduced in mpl−/− mice, indicating an essential role for TPO signaling in the regulation of hematopoietic stem cell production and function.
MATERIALS AND METHODS
Mice.
The generation of mpl−/− mice has been described previously (10). Wild-type and mpl−/− mice on a purely 129/Sv genetic background were raised in a pathogen-free animal facility and used in all transplantation experiments at 2–4 months of age.
In Vitro Cultures.
Semisolid 1-ml agar cultures containing 5 × 104 bone marrow cells were prepared by using DMEM with a final concentration of 20% newborn calf serum and 0.3% agar. Blast colony formation by preprogenitor cells (23) was stimulated by using each of the following purified recombinant murine stimuli: 100 ng of stem cell factor (SCF), 500 ng of Flk-2-ligand (FL) plus 10 ng of leukemia inhibitory factor (LIF), or 10 ng of interleukin (IL) 3. Cultures were incubated for 7 days at 37°C in a fully humidified atmosphere of 10% CO2 in air, fixed with 1 ml of 2.5% glutaraldehyde, floated onto glass slides, and stained in sequence for acetylcholinesterase then Luxol fast blue and hematoxylin. All colonies in each culture were typed at ×100 or ×400 magnifications.
CFU-S Studies.
Bone marrow cells from 129/Sv mpl−/− mice or their wild-type littermates were washed in serum-free DMEM and injected intravenously via the retro-orbital sinus into either wild-type 129/Sv or 129/Sv mpl−/− recipients. Several hours before transplantation, the recipient mice were irradiated with 11 Gy of γ-irradiation in two equal doses given 3 hr apart from a 137Cs source (Atomic Energy, Ottawa) at a dose rate of 31 cGy/min. Transplanted mice were maintained on oral antibiotic (1.1 g/liter of neomycin sulfate; Sigma). Spleens were removed after 8, 9, or 12 days and fixed in Carnoy’s solution (60% ethanol, 30% chloroform, 10% acetic acid), and the numbers of macroscopic colonies were counted. Histological sections from a representative sample of recipient spleens were prepared, stained with hematoxylin and eosin, and examined microscopically.
Competitive Long-Term Repopulating Stem Cells Assays.
To examine the capacity of long-term repopulating stem cells in mpl−/− bone marrow, 2 × 105 bone marrow cells from wild-type 129/Sv female mice (competitor cells) were coinjected with increasing numbers (5 × 104, 2 × 105, or 2 × 106) of male mpl−/− or wild-type bone marrow cells (test cells) into each of five female wild-type 129/Sv recipients. To control for the contribution of residual host stem cells in the recipient mice, 2 × 105 bone marrow cells from wild-type female 129/Sv mice were transplanted into 129/Sv wild-type male recipients. Before transplantation, the recipient mice were irradiated as described above. Two experiments, each involving transplantation of bone marrow cells from independent wild-type and mpl−/− mice into five recipients, were performed, with analysis at 3 months or 5 months (two mice from each group) and at 9 months or 10 months (three mice) after primary transplantation, respectively.
For serial bone marrow transplantation, secondary transplants were performed at the first time of analysis of primary recipients (see above) with tertiary transplantations an additional 3 months later. Bone marrow from two individuals from each group were pooled, and the equivalent of one-half of one femur was injected into each of three new irradiated recipients.
At analysis, genomic DNA was extracted from the bone marrow, thymus, and spleen of each mouse. Five micrograms of DNA was digested with 10 units of NcoI, separated in 0.8% agarose gels, transferred to nylon membrane (GeneScreen Plus, Life Science, Boston, MA) and hybridized with a male-specific DNA probe (720-bp insert from the Y chromosome-specific plasmid pY2) (24). The intensities of the male-specific signals were quantitated by PhosPhorimage analysis (ImageQuant, Molecular Dynamics) and the contribution to each organ was determined by comparison with a standard curve constructed from defined mixtures of male and female DNA. To correct the data for differences in the amounts of DNA loaded, male-specific DNA probes were stripped, and the membranes were hybridized with a single copy DNA probe (1.4-kb XhoI fragment from the murine c-myc cDNA) (25).
RESULTS
Preprogenitor Cells.
The two major classes of clonogenic preprogenitor cells able to form blast colonies containing progenitor cells can be enumerated in cultures stimulated either by SCF (26) or by FL plus LIF (27). Levels of the more mature lineage-committed progenitor cells can be established from parallel cultures stimulated by IL-3 (28).
As shown in Table 1, the frequency of lineage-committed progenitor cells in mpl−/− marrow populations was 50% of that in wild-type marrow populations, in agreement with previous observations (10). The frequency of preprogenitor cells in mpl−/− marrow populations was more severely reduced to 11% of controls for SCF-responsive cells, 26% of controls for FL plus LIF-responsive cells, and 9% of controls for IL-3-responsive cells.
Table 1.
Frequency of preprogenitor and lineage-committed progenitor cells in mpl+/+ and mpl−/− mice
| Stimulus | Colony type | mpl+/+ | mpl−/− |
|---|---|---|---|
| SCF | Blast | 36 ± 8 | 4 ± 3 |
| Granulocytic | 89 ± 23 | 34 ± 10 | |
| FL + LIF | Blast | 19 ± 2 | 5 ± 5 |
| IL-3 | Blast | 35 ± 13 | 3 ± 3 |
| Granulocytic | 77 ± 14 | 33 ± 3 | |
| GM | 57 ± 14 | 33 ± 13 | |
| Macrophage | 56 ± 14 | 33 ± 3 | |
| Eosinophil | 15 ± 7 | 7 ± 3 | |
| Megakaryocytic | 37 ± 17 | 17 ± 12 |
Frequencies of clonogenic cells are expressed per 105 bone marrow cells. Mean data ± SD from replicate cultures of three 8-week-old mice of each genotype. GM, granulocyte-macrophage.
CFU-S Studies.
Cells in the primitive hematopoietic compartment of mpl−/− mice were examined by comparing the number of day-12 CFU-S in the bone marrow of mpl-deficient mice with those of wild-type controls. Initial experiments injecting irradiated wild-type mice with increasing numbers of wild-type or mpl−/− bone marrow cells revealed a linear relationship between the number of injected cells and resultant CFU-S in the range of 104 to 105 donor cells, but dramatically fewer colonies in spleens of recipients of mpl−/− marrow (Fig. 1A). To extend this observation, bone marrow cells from each of six mpl−/− and five wild-type mice were injected into five wild-type recipients. Whereas wild-type bone marrow cells yielded on average 17.8 ± 2.4 CFU-Sd12 per 1.5 × 105 bone marrow cells, only 1.8 ± 0.7 spleen colonies developed after 12 days in mice that received an equivalent number of mpl−/− cells (Fig. 1B). Because the cellularity of the bone marrow is similar in normal and mpl−/− mice (10), these data reveal an overall 10-fold reduction in CFU-Sd12 in mpl-deficient marrow. A range of both large and small spleen colonies were generated by cells from both marrow populations, and microscopic examination of histological sections revealed no differences in gross colony composition between the two donor types. To investigate whether the CFU-S deficit in mpl−/− marrow reflected reduced cell numbers or altered rates of maturation of cells in this compartment, CFU-Sd8 were examined. A similar 8- to 10-fold deficit in CFU-Sd8 was observed within the bone marrow of mpl−/− mice, suggesting an overall reduction in the number of CFU-S within the mutant marrow.
Figure 1.
Reduced numbers of CFU-S in mpl−/− mice. (A) Appearance of spleens from irradiated wild-type recipient mice 12 days after intravenous injection of 7.5 × 104 wild-type (mpl+/+) or 1.5 × 105 mpl−/− bone marrow cells. (B) Spleen colony numbers per 1.5 × 105 wild-type or mpl−/− bone marrow cells transplanted into normal irradiated recipient mice. Data are the means ± SDs from five or six (CFU-Sd12) or two (CFU-Sd8) individual donor mice of each genotype, each transplanted into five recipients. (C) Spleen colony numbers after transplantation of 7.5 × 104 wild-type bone marrow cells into normal or mpl-deficient recipients. The means ± SDs of data from five recipients receiving marrow from each of four mpl−/− recipients or three wild-type recipients donor mice.
To investigate whether the CFU-S deficiency in mpl−/− mice was a direct effect of the absence of TPO signaling in multipotential hematopoietic cells or was contributed to by a defective mpl−/− hematopoietic microenvironment, the development of CFU-S from normal bone marrow cells was compared in irradiated wild-type and mpl−/− recipient mice. Probably reflecting their severe deficiency in platelets, several mpl−/− mice became moribund 8–9 days after irradiation, and spleen colonies therefore were analyzed at this time. Consistent with a direct action of TPO on multipotential cells in vivo, no difference in the number of CFU-S developing from identical transplants of normal marrow were observed between normal and mpl−/− recipients (Fig. 1C).
Competitive Long-Term Reconstitution Assays.
The capacity for long-term reconstitution of the hematopoietic system of a transplanted host in which endogenous hematopoiesis has been ablated is the most stringent functional assay for stem cells. To examine repopulating cells in mpl−/− mice, the capacity of the bone marrow from these animals to compete with normal marrow for hematopoietic reconstitution of irradiated mice was determined. A competitive repopulation design was chosen to allow direct comparison of normal and mpl-deficient stem cell potential (29, 30). Increasing doses of bone marrow cells from individual male mpl−/− or wild-type control mice were mixed with 2 × 105 wild-type female bone marrow cells and transplanted into lethally irradiated normal female recipients. At various times after transplantation, the spleen, thymus, and bone marrow were removed from two to three mice from each group, and the proportion of male DNA present in these tissues was determined in Southern blot analyses as a measure of the capacity of the stem cells in the test graft to repopulate the hematopoietic system. An example of this analysis, for the bone marrow of a group of primary recipient mice 3 months after transplantation, is shown in Fig. 2. Standard curves were constructed by using mixtures of male and female DNA and variations in DNA loading, and transfer was controlled by reanalyzing the Southern blot filters with a single copy c-myc probe after the male-specific pY2 DNA probe. An additional control group, in which normal male recipients were transplanted with 2 × 105 wild-type female cells also was analyzed. On average over two experiments, the proportion of male DNA in the bone marrow, spleen, and thymus of these recipients was 5%, 5%, and 13%, respectively, at 3–5 months after transplantation and 9% and 8% in the spleen and thymus after 9–10 months. Thus, the irradiation regimen used was sufficient to effectively prevent significant recovery of endogenous hematopoiesis in the recipient mice.
Figure 2.
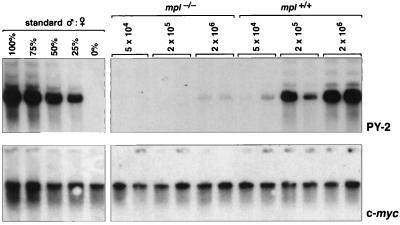
Analysis of inferior competitive long-term repopulating capacity of transplanted mpl−/− bone marrow cells. Southern blots of genomic DNA extracted from bone marrow cells from pairs of primary recipient mice that received 2 × 105 wild-type female competitor bone marrow cells mixed with 5 × 104, 2 × 105, or 2 × 106 test cells from male mpl−/− or wild-type (mpl+/+) mice (experiment 1). The filters were hybridized with the Y-chromosome specific pY2 probe, and the contribution of test cells to the bone marrow was determined in each case by comparison with a standard curve constructed from defined mixtures of male and female DNA. To correct the data for differences in the amounts of DNA loaded, male-specific DNA probes were stripped, and the membranes hybridized with a single copy c-myc DNA probe.
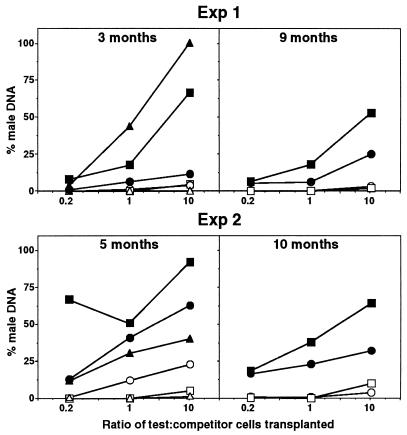
The reconstitution of hematopoietic tissues of primary recipients in two experiments with independent wild-type and mpl−/− donor mice is shown in Fig. 3. As expected, normal male bone marrow cells competed effectively with their female counterparts for hematopoietic reconstitution, with an increasing proportion of male DNA detected in the bone marrow, spleen, and thymus of recipient mice as the ratio of test-to-competitor cell numbers increased. In sharp contrast, the hematopoietic repopulating activity of mpl−/− bone marrow was dramatically impaired. In the first experiment, when mixed in equal proportion with normal competitor bone marrow, 3 months after transplantation the contribution of wild-type test cells to the bone marrow, spleen, and thymus was 44%, 6%, and 18%, respectively. An equivalent number of mpl−/− bone marrow cells reconstituted 0%, 1.4%, and 0% of the same organs (Fig. 3). Even when cotransplanted in 10-fold excess over normal marrow, mpl−/− cells contributed to less than 5% of these hematopoietic organs, whereas the bone marrow, spleen, and thymus of recipients receiving a similar excess of wild-type test cells contained 100%, 12%, and 66% male DNA, respectively. Similar results also were observed in the second experiment, where 5 months after transplantation the contribution of mpl−/− bone marrow to hematopoiesis in primary recipients was again significantly lower than that of wild-type marrow for each organ and at all ratios of test to competitor cells analyzed (Fig. 3).
Figure 3.
Inferior competitive long-term repopulating capacity of mpl−/− stem cells in primary recipients. Contribution of transplanted bone marrow cells to the bone marrow (▵, ▴), spleen (○, •), and thymus (□, ▪) of primary recipients that received 2 × 105 wild-type female competitor bone marrow cells mixed with 5 × 104, 2 × 105, or 2 × 106 test cells (test/competitor ratios 0.2, 1, and 10) from male mpl−/− (open symbols) or wild-type (filled symbols) mice. Each point represents the mean percentage of male DNA in the organs of 2–3 recipient mice determined by Southern blot analysis. Experiments 1 and 2 involved transplantation of bone marrow cells from independent wild-type and mpl−/− donor mice, the recipients of which were analyzed at 3 and 9 months and 5 and 10 months, respectively.
The deficiency in repopulating stem cells of mpl−/− mice was also evident when reconstitution of recipient animals was measured at 9–10 months after transplantation. Although occlusion of the marrow space, probably a long-term consequence of irradiation, prevented collection of sufficient cells from this organ, analysis of the spleen and thymus in both experiments revealed no contribution from mpl−/− cells in recipients transplanted at test/competitor cell ratios of 0.2:1 or 1:1 (Fig. 3). Only at the ratio of 10:1 was a mpl−/− contribution evident, but at very low levels (experiment 1: spleen 3%, thymus 2%; experiment 2: spleen 4%, thymus 10%). In contrast, stem cells from wild-type marrow contributed to hematopoietic repopulation at all test/competitor cell ratios analyzed and when in 10-fold excess over competitor cells resulted in reconstitution of 25% and 53% (experiment 1) and 32% and 64% (experiment 2) of the spleen and thymus, respectively (Fig. 3). Indeed, the repopulating capacity of wild-type stem cells was greater at 0.2:1 test/competitor cell ratio than that of mpl−/− stem cells transplanted at 10-fold excess over normal competitor cells.
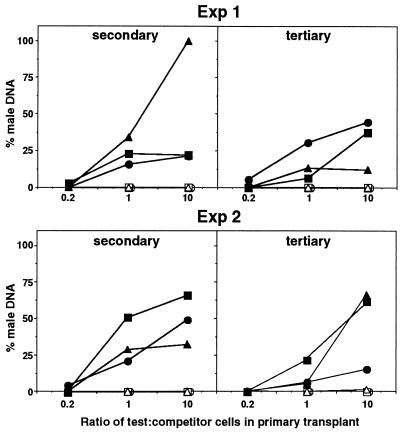
Bone marrow cells from the primary recipient mice were transplanted into secondary recipients. After 3 months, the contribution of test cells to hematopoietic organs of these secondary hosts were analyzed, and an additional serial marrow transplant was conducted into tertiary recipients, which were themselves analyzed 2–3 months posttransplantation. As expected, at original test/competitor cell ratios of 1:1 and 10:1, stem cells from wild-type bone marrow exhibited sufficient self-renewal capacity to contribute significantly to hematopoiesis in secondary and tertiary recipients (Fig. 4). In contrast, there was no detectable contribution of mpl−/− stem cells to the repopulation of the bone marrow, spleen, or thymus in secondary or tertiary recipients at any primary test/competitor ratio, even when mpl−/− marrow cells were present at 10-fold excess over normal competitor cells in the primary transplant and contributing to hematopoiesis in these mice, albeit at low levels (Fig. 4).
Figure 4.
Inferior competitive long-term repopulating capacity of mpl−/− cells in secondary and tertiary recipients. Contribution to the bone marrow (▵, ▴), spleen (○, •), and thymus (□, ▪) of secondary and tertiary recipients of transplanted bone marrow cells from primary recipient mice that received 2 × 105 wild-type female competitor bone marrow cells mixed with 5 × 104, 2 × 105, or 2 × 106 test cells (test/competitor ratios 0.2, 1, and 10) from male mpl−/− (open symbols) or wild-type (filled symbols) mice. Each point represents the mean percentage of male DNA in the organs of three recipient mice determined by Southern blot analysis. Experiments 1 and 2 involved transplantation of bone marrow cells from independent wild-type and mpl−/− donor mice to the primary recipients. Secondary and tertiary transplant recipients were analyzed 2–3 months after each preceding transplant.
DISCUSSION
We document here that in mice lacking c-mpl, the receptor for thrombopoietin, multilineage deficiencies are present in the bone marrow that increase in severity in progressively less mature subpopulations. Apart from a deficit in megakaryocytes, total marrow cellularity and mature cell complement in mpl−/− mice are normal (10). However, there is a 50% deficiency in lineage-committed progenitor cells (10), and the present data indicate a deficiency of up to 90% in preprogenitor cells and spleen colony-forming cells as well as a deficiency in repopulating cells of even greater magnitude.
The ability of irradiated mpl−/− mice to support normal hematopoietic colony formation by wild-type CFU-S suggests that the hematopoietic defects in mpl−/− mice are based on an intrinsic defect in the behavior of hematopoietic cells in the absence of TPO signaling. However, as c-mpl is expressed in only a minority of immature hematopoietic cells, apparently excluding most committed nonmegakaryocytic progenitors (13, 14), a direct in vivo requirement for TPO by all committed progenitor cells is unlikely. Rather, our results suggest that TPO acts on cells within the stem cell compartment, and that loss of this action prevents the production of normal numbers of more committed progeny in mpl−/− mice. The most severe deficiencies are evident within megakaryocytopoiesis because this lineage remains TPO-dependent at all maturation stages. A stimulatory action of TPO on hematopoietic stem cells also could account for the accelerated recovery of white blood cells and erythrocytes, in addition to platelets, noted after TPO administration following myelosuppression (31, 32).
The deficiency in stem cells revealed by the incapacity of mpl−/− bone marrow to effectively compete with normal marrow for the long-term repopulation of the hematopoietic organs of primary recipient mice, implies that c-mpl is indispensible for the in vivo regulation of stem cell production, survival, appropriate commitment and differentiation, or a combination of these processes. Although we cannot entirely distinguish between these possibilities at the stem cell level, our analyses of CFU-S indicated that although the numbers of both CFU-Sd8 and CFU-Sd12 were deficient in mpl−/− mice, no dramatic differences in size or composition were evident between spleen colonies that were formed by normal or mpl-deficient cells. This finding suggests that c-mpl is necessary for the generation of normal numbers of CFU-S but not for their subsequent differentiation or maturation. However, hematopoietic stem cells lacking c-mpl also may have defective self-renewal properties because the meager capacity of mpl−/− bone marrow cells to contribute to hematopoiesis in primary recipient mice was completely extinguished on serial transfer into secondary and tertiary hosts (33).
Despite the marked stem cell deficiency evident from our analysis of mpl−/− mice, sufficient hematopoietic function does exist in these animals for the ultimate development of normal numbers of mature hematopoietic cells in those lineages where later maturation stages are TPO-independent (9, 10). Although compensatory mechanisms may exist to boost the efficiency of mature cell production by restricted numbers of stem cells, a low, but sustained, level of stem cell formation does occur in mpl−/− mice. The regulation of hematopoietic stem cells is a complex process in which a number of growth factors and cytokines have been implicated. In vitro, the survival and proliferation of hematopoietic populations enriched for stem cells are responsive to the actions of many cytokines, some of which also have been shown to amplify this compartment in vivo (34). The physiological roles of several growth factors in hematopoietic stem cell regulation have been addressed by analysis of mutant mice deficient in these regulators or their receptors. LIF, IL-6, FL, and SCF all are required for the maintenance of normal CFU-S and/or repopulating stem cell number or function (35–38). The stem cell deficiency observed in mpl−/− mice is substantially greater than that observed in mice lacking LIF, IL-6, or FL (35–37). Nevertheless, because stem cells appear to require multiple stimuli for efficient function, the residual stem cell activity in mpl-deficient mice may reflect a reduced net signal to all stem cells or alternatively may be because of a pool of TPO-independent cells regulated exclusively by alternative cytokines. Analysis of compound mouse mutants lacking multiple regulators of stem cell function will help define the nature of the cytokine network controlling hematopoietic stem cell function.
In summary, our analyses of the in vivo deficiencies of hematopoietic stem cells lacking c-Mpl add to recent in vitro studies showing direct actions of TPO on the proliferation, survival, and multilineage differentiation of hematopoietic populations enriched for primitive cells (16–22). They establish that in addition to the well-defined role of TPO in the production of megakaryocytes and platelets, in which the cytokine acts at all differentiation stages of the lineage, TPO signaling is also a prominent and essential physiological component of normal hematopoietic stem cell regulation. As such, TPO may prove to be a potent agent in clinical strategies requiring the stimulation or ex vivo expansion of hematopoietic stem cells.
Acknowledgments
We thank Sandra Mifsud, Ladina Di Rago, Angela D’Amico, and Angela Milligan for excellent technical assistance and Jodie deWinter, Louise Gilmore, and Anne Chow for animal husbandry. This work was supported by the National Health and Medical Research Council, Canberra, the Anti-Cancer Council of Victoria, the National Institutes of Health Grant No. CA22556, the Australian Cooperative Research Centres Program, and by support to S. K. by the Naito Foundation.
ABBREVIATIONS
- TPO
thrombopoietin
- SCF
stem cell factor
- FL
Flk-2-ligand
- LIF
leukemia inhibitory factor
- IL
interleukin
- CFU-S
colony forming unit-spleen
References
- 1.Alexander, W. S. & Begley, C. G. (1998) Cytokines Cell. Mol. Ther., in press. [PubMed]
- 2.Broudy V C, Lin N L, Kaushansky K. Blood. 1995;85:1719–1726. [PubMed] [Google Scholar]
- 3.Hunt P, Li Y-S, Nichol J L, Hokom M M, Bogenberger J M, et al. Blood. 1995;86:540–547. [PubMed] [Google Scholar]
- 4.Banu N, Wang J F, Deng B, Groopman J E, Avraham H. Blood. 1995;86:1331–1338. [PubMed] [Google Scholar]
- 5.Kaushansky K, Lok S, Holly R D, Broudy V C, Lin N, Bailey M C, Forstrom J W, Buddle M M, Oort P J, Hagen F S, Roth G J, Papayannopoulou T, Foster D C. Nature (London) 1994;369:568–571. doi: 10.1038/369568a0. [DOI] [PubMed] [Google Scholar]
- 6.Lok S, Kaushansky K, Holly R D, Kuijper J L, Lofton-Day C E, et al. Nature (London) 1994;369:565–568. doi: 10.1038/369565a0. [DOI] [PubMed] [Google Scholar]
- 7.Farese A M, Hunt P, Boone T, MacVittie T J. Blood. 1995;86:54–59. [PubMed] [Google Scholar]
- 8.Basser R L, Rasko J E J, Clarke K, Cebon J, Green M D, Hussein S, Alt C, Menchaca D, Tomita D, Marty J, Fox R M, Begley C G. Lancet. 1996;348:1279–1281. doi: 10.1016/S0140-6736(96)04471-6. [DOI] [PubMed] [Google Scholar]
- 9.Gurney A L, Carver-Moore K, de Sauvage F J, Moore M W. Science. 1994;265:1445–1447. doi: 10.1126/science.8073287. [DOI] [PubMed] [Google Scholar]
- 10.Alexander W S, Roberts A R, Nicola N A, Li R, Metcalf D. Blood. 1996;87:2162–2170. [PubMed] [Google Scholar]
- 11.de Sauvage F J, Carver-Moore K, Luoh S-M, Ryan A, Dowd M, Eaton D L, Moore M W. J Exp Med. 1996;183:651–656. doi: 10.1084/jem.183.2.651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Carver-Moore K, Broxmeyer H E, Luoh S-M, Cooper S, Peng J, Burstein S A, Moore M W, de Sauvage F J. Blood. 1996;88:803–808. [PubMed] [Google Scholar]
- 13.Debili N, Wendling F, Cosman D, Titeux M, Florindo C, Dusanter-Fourt I, Schooley K, Methia N, Charon M, Nador R, Bettaieb A, Vainchenker W. Blood. 1995;85:391–401. [PubMed] [Google Scholar]
- 14.Beradi A C, Wang A, Levine J D, Lopez P, Scadden D T. Science. 1995;267:104–108. doi: 10.1126/science.7528940. [DOI] [PubMed] [Google Scholar]
- 15.Wendling F, Varlet P, Charon M, Tambourin P. Virology. 1986;149:242–246. doi: 10.1016/0042-6822(86)90125-x. [DOI] [PubMed] [Google Scholar]
- 16.Ku H, Yonemura Y, Kaushansky K, Ogawa M. Blood. 1996;87:4544–4551. [PubMed] [Google Scholar]
- 17.Sitnicka E, Lin N, Priestly G V, Fox N, Broudy V C, Wolf N S, Kaushansky K. Blood. 1996;87:4998–5005. [PubMed] [Google Scholar]
- 18.Kobayashi M, Laver J H, Kato T, Miyazaki H, Ogawa M. Blood. 1996;88:429–436. [PubMed] [Google Scholar]
- 19.Ramsfjell V, Borge O J, Veiby O P, Cardier J, Murphy M J, Jr, Lyman S D, Lok S, Jacobsen S E W. Blood. 1996;88:4481–4492. [PubMed] [Google Scholar]
- 20.Young J C, Bruno E, Luens K M, Wu S, Backer M, Murray L J. Blood. 1996;88:1619–1631. [PubMed] [Google Scholar]
- 21.Borge O J, Ramsfjell V, Cui L, Jacobsen S E W. Blood. 1997;90:2282–2292. [PubMed] [Google Scholar]
- 22.Tanimukai S, Kimura T, Sakabe H, Ohmizono Y, Kato T, Miyazaki H, Yamagishi H, Sonoda Y. Exp Hematol. 1997;25:1025–1033. [PubMed] [Google Scholar]
- 23.Metcalf, D. (1998) Leukemia, in press.
- 24.Lamar E E, Palmer E. Cell. 1984;37:171–177. doi: 10.1016/0092-8674(84)90312-x. [DOI] [PubMed] [Google Scholar]
- 25.Bernard O, Cory S, Gerondakis S, Webb E, Adams J M. EMBO J. 1983;2:2375–2383. doi: 10.1002/j.1460-2075.1983.tb01749.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Metcalf D. Proc Natl Acad Sci USA. 1991;88:11310–11314. doi: 10.1073/pnas.88.24.11310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Metcalf D. Proc Natl Acad Sci USA. 1997;94:11552–11556. doi: 10.1073/pnas.94.21.11552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Metcalf D, Begley C G, Nicola N A, Johnson G R. Exp Hematol. 1987;15:288–295. [PubMed] [Google Scholar]
- 29.Harrison D E. Blood. 1980;55:77–81. [PubMed] [Google Scholar]
- 30.Szilvassy S J, Humphries R K, Lansdorp P M, Eaves A C, Eaves C J. Proc Natl Acad Sci USA. 1990;87:8736–8740. doi: 10.1073/pnas.87.22.8736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Grossmann A, Lenox J, Ren H P, Humes J M, Forstrom J W, Kaushansky K, Sprugel K H. Exp Hematol. 1996;24:1238–1246. [PubMed] [Google Scholar]
- 32.Harker L A, Marzec U M, Kelly A B, Cheung E, Tomer A, Nichol J L, Hanson S R, Stead R B. Blood. 1997;89:155–165. [PubMed] [Google Scholar]
- 33.Harrison D E, Astle C M. J Exp Med. 1982;156:1767–1779. doi: 10.1084/jem.156.6.1767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ogawa M. Blood. 1993;81:2844–2853. [PubMed] [Google Scholar]
- 35.Escary J-L, Perreau J, Dumenil D, Ezine S, Brulet P. Nature (London) 1993;363:361–364. doi: 10.1038/363361a0. [DOI] [PubMed] [Google Scholar]
- 36.Bernad A, Kopf M, Kulbacki R, Weich N, Koehler G, Gutierrez-Ramos J C. Immunity. 1994;1:725–731. doi: 10.1016/s1074-7613(94)80014-6. [DOI] [PubMed] [Google Scholar]
- 37.Mackarehtschian K, Hardin J D, Moore K A, Boast S, Goff S P, Lemischka I R. Immunity. 1995;3:147–161. doi: 10.1016/1074-7613(95)90167-1. [DOI] [PubMed] [Google Scholar]
- 38.Russell E S. Adv Genet. 1979;20:357–459. [PubMed] [Google Scholar]