Abstract
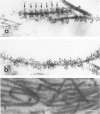
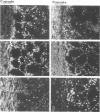
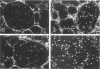
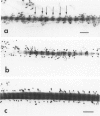
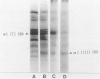
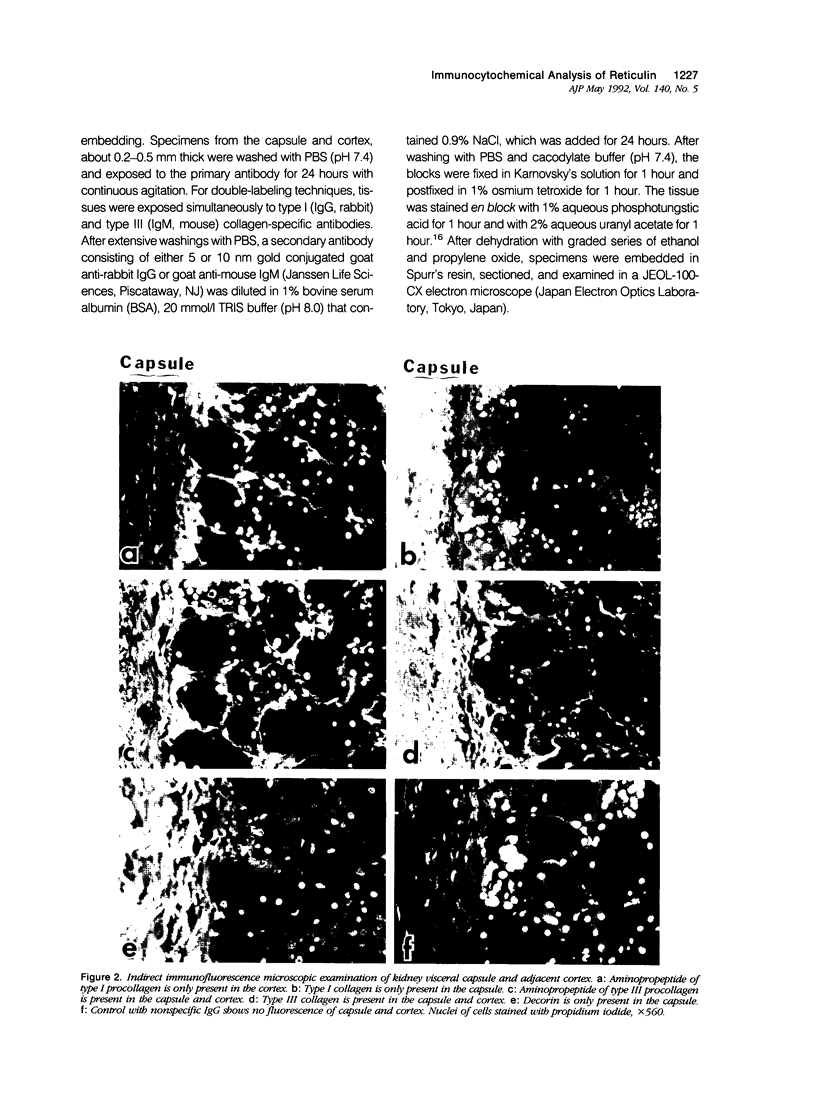
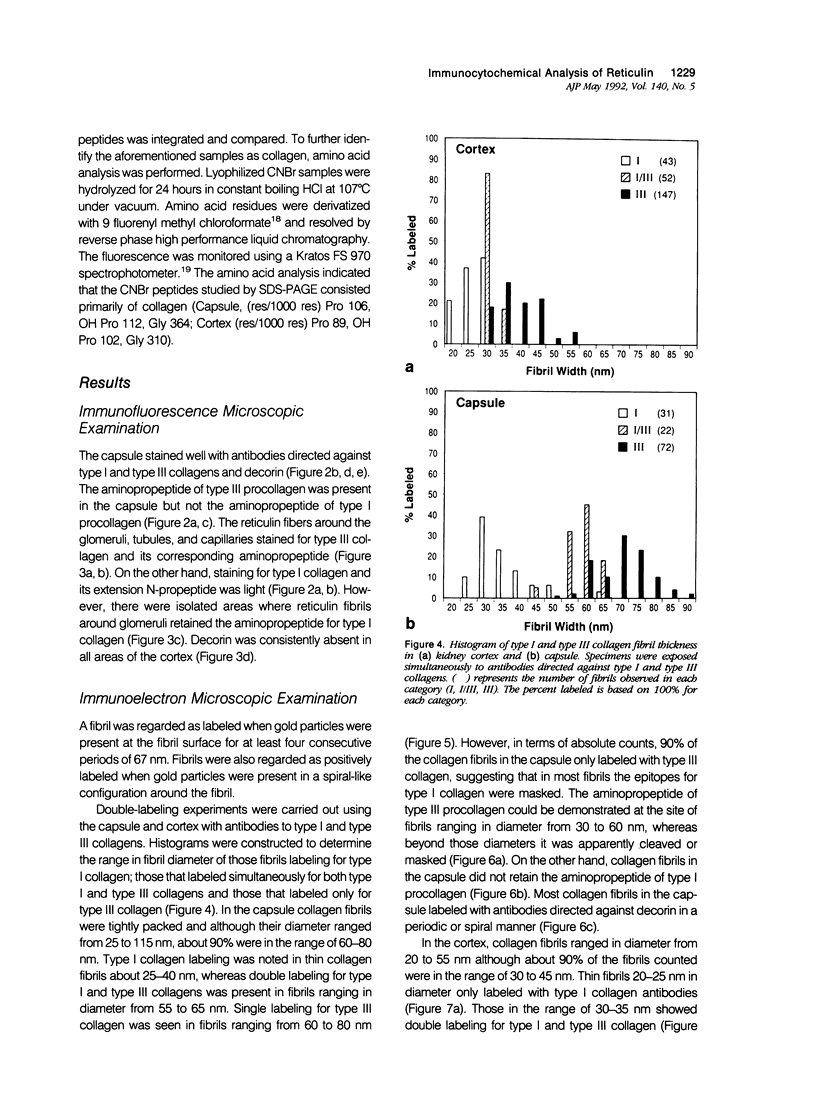
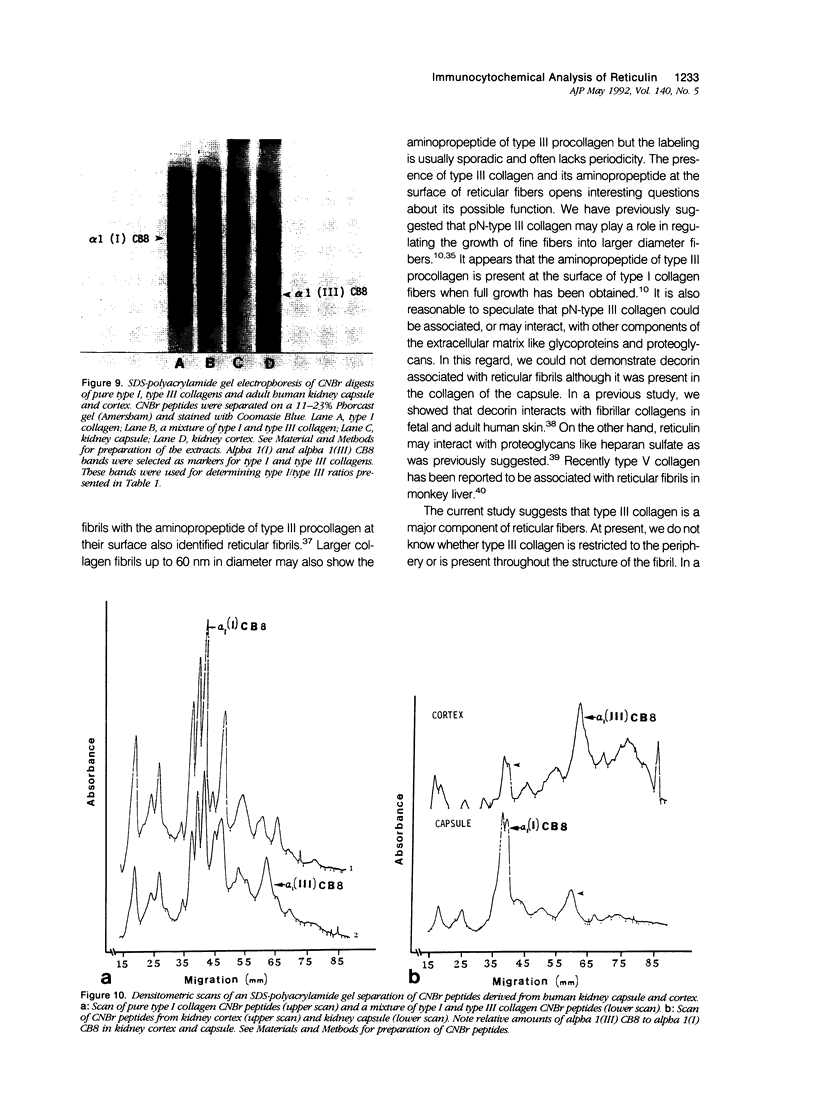
This study characterized the nature of reticulin fibrils from human kidney cortex by immunochemical analysis. Controls consisted of type I collagen fibrils derived from the kidney parietal capsule. Most of the fibrils in the capsule ranged in diameter from 60 to 80 nm whereas reticulin fibrils from the cortex ranged from 30-45 nm. Immunochemistry by light and electron microscopic examinations was carried out with antibodies directed against type I and type III collagens, their corresponding aminopropeptides, and decorin (PG-II). The ratio of type I to type III collagen was determined by cyanogen bromide peptide digests. This study showed that reticulin fibrils are hybrids of type I and type III collagens. Double immunoelectron microscopic examination showed that fibrils 20-25 nm consisted mainly of type I collagen some of which retained their aminopropeptide. Larger fibrils 30-35 nm labeled simultaneously for type I and type III collagens. However, most fibrils with diameters between 40-55 nm labeled for type III collagen and its corresponding aminopropeptide. No decorin was detected at the surface of reticulin fibrils. Purified reticulin consisted of 82% type III and 18% type I collagen whereas collagen derived from the capsule revealed 76% type I and 24% type III. The presence of the aminopropeptide of type III procollagen in reticulin fibrils is a striking feature and may play a role in regulating their diameter.
Full text
PDF







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adachi E., Hayashi T., Hashimoto P. H. A comparison of the immunofluorescent localization of collagen types I, III, and V with the distribution of reticular fibers on the same liver sections of the snow monkey (Macaca fuscata). Cell Tissue Res. 1991 Apr;264(1):1–8. doi: 10.1007/BF00305716. [DOI] [PubMed] [Google Scholar]
- Apaja-Sarkkinen M., Alavaikko M., Karttunen T., Autio-Harmainen H. Basement membrane proteins in the spleen: immunohistochemical demonstration and relation to reticulin. Histopathology. 1986 Mar;10(3):295–302. doi: 10.1111/j.1365-2559.1986.tb02483.x. [DOI] [PubMed] [Google Scholar]
- Becker U., Nowack H., Gay S., Timpl R. Production and specificity of antibodies against the aminoterminal region in type III collagen. Immunology. 1976 Jul;31(1):57–65. [PMC free article] [PubMed] [Google Scholar]
- Bernard M. P., Chu M. L., Myers J. C., Ramirez F., Eikenberry E. F., Prockop D. J. Nucleotide sequences of complementary deoxyribonucleic acids for the pro alpha 1 chain of human type I procollagen. Statistical evaluation of structures that are conserved during evolution. Biochemistry. 1983 Oct 25;22(22):5213–5223. doi: 10.1021/bi00291a023. [DOI] [PubMed] [Google Scholar]
- Fisher L. W., Hawkins G. R., Tuross N., Termine J. D. Purification and partial characterization of small proteoglycans I and II, bone sialoproteins I and II, and osteonectin from the mineral compartment of developing human bone. J Biol Chem. 1987 Jul 15;262(20):9702–9708. [PubMed] [Google Scholar]
- Fleischmajer R., Fisher L. W., MacDonald E. D., Jacobs L., Jr, Perlish J. S., Termine J. D. Decorin interacts with fibrillar collagen of embryonic and adult human skin. J Struct Biol. 1991 Feb;106(1):82–90. doi: 10.1016/1047-8477(91)90065-5. [DOI] [PubMed] [Google Scholar]
- Fleischmajer R., Gay S., Meigel W. N., Perlish J. S. Collagen in the cellular and fibrotic stages of scleroderma. Arthritis Rheum. 1978 May;21(4):418–428. doi: 10.1002/art.1780210404. [DOI] [PubMed] [Google Scholar]
- Fleischmajer R., MacDonald E. D., Perlish J. S., Burgeson R. E., Fisher L. W. Dermal collagen fibrils are hybrids of type I and type III collagen molecules. J Struct Biol. 1990 Oct-Dec;105(1-3):162–169. doi: 10.1016/1047-8477(90)90110-x. [DOI] [PubMed] [Google Scholar]
- Fleischmajer R., Olsen B. R., Timpl R., Perlish J. S., Lovelace O. Collagen fibril formation during embryogenesis. Proc Natl Acad Sci U S A. 1983 Jun;80(11):3354–3358. doi: 10.1073/pnas.80.11.3354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fleischmajer R., Perlish J. S., Burgeson R. E., Shaikh-Bahai F., Timpl R. Type I and type III collagen interactions during fibrillogenesis. Ann N Y Acad Sci. 1990;580:161–175. doi: 10.1111/j.1749-6632.1990.tb17927.x. [DOI] [PubMed] [Google Scholar]
- Fleischmajer R., Perlish J. S., Olsen B. R. Amino and carboxyl propeptides in bone collagen fibrils during embryogenesis. Cell Tissue Res. 1987 Jan;247(1):105–109. doi: 10.1007/BF00216552. [DOI] [PubMed] [Google Scholar]
- Fleischmajer R., Perlish J. S., Timpl R. Collagen fibrillogenesis in human skin. Ann N Y Acad Sci. 1985;460:246–257. doi: 10.1111/j.1749-6632.1985.tb51172.x. [DOI] [PubMed] [Google Scholar]
- Fleischmajer R., Timpl R., Tuderman L., Raisher L., Wiestner M., Perlish J. S., Graves P. N. Ultrastructural identification of extension aminopropeptides of type I and III collagens in human skin. Proc Natl Acad Sci U S A. 1981 Dec;78(12):7360–7364. doi: 10.1073/pnas.78.12.7360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gay S., Fietzek P. P., Remberger K., Eder M., Kühn K. Liver cirrhosis: immunofluorescence and biochemical studies demonstrate two types of collagen. Klin Wochenschr. 1975 Mar 1;53(5):205–208. doi: 10.1007/BF01468808. [DOI] [PubMed] [Google Scholar]
- Huang W. Chemical and histochemical studies of human alveolar collagen fibers. Am J Pathol. 1977 Jan;86(1):81–98. [PMC free article] [PubMed] [Google Scholar]
- Hulmes D. J., Jesior J. C., Miller A., Berthet-Colominas C., Wolff C. Electron microscopy shows periodic structure in collagen fibril cross sections. Proc Natl Acad Sci U S A. 1981 Jun;78(6):3567–3571. doi: 10.1073/pnas.78.6.3567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- KORTING G. W., HOLZMANN H., KUHN K. [Electron microscopic studies on the behavior of periodate-sensitive substances of collagen fibrils in progressive scleroderma]. Arch Klin Exp Dermatol. 1959;209:66–75. [PubMed] [Google Scholar]
- Karttunen T., Alavaikko M., Apaja-Sarkkinen M., Autio-Harmainen H. An immunohistochemical study of laminin, type-IV collagen and type-III pN-collagen with relation to reticular fibres in Hodgkin's disease. Int J Cancer. 1988 Jan 15;41(1):52–58. doi: 10.1002/ijc.2910410111. [DOI] [PubMed] [Google Scholar]
- Karttunen T., Apaja-Sarkkinen M., Alavaikko M., Blanco G., Aine R., Järvinen M., Autio-Harmainen H. Characterization of extracellular matrix in non-Hodgkin's lymphomas. Pathol Res Pract. 1988 Nov;183(6):735–746. doi: 10.1016/S0344-0338(88)80059-1. [DOI] [PubMed] [Google Scholar]
- Karttunen T., Sormunen R., Risteli L., Risteli J., Autio-Harmainen H. Immunoelectron microscopic localization of laminin, type IV collagen, and type III pN-collagen in reticular fibers of human lymph nodes. J Histochem Cytochem. 1989 Mar;37(3):279–286. doi: 10.1177/37.3.2918219. [DOI] [PubMed] [Google Scholar]
- Keene D. R., Sakai L. Y., Burgeson R. E., Bächinger H. P. Direct visualization of IgM antibodies bound to tissue antigens using a monoclonal anti-type III collagen IgM as a model system. J Histochem Cytochem. 1987 Mar;35(3):311–318. doi: 10.1177/35.3.3546481. [DOI] [PubMed] [Google Scholar]
- Keene D. R., Sakai L. Y., Bächinger H. P., Burgeson R. E. Type III collagen can be present on banded collagen fibrils regardless of fibril diameter. J Cell Biol. 1987 Nov;105(5):2393–2402. doi: 10.1083/jcb.105.5.2393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kramer R. H., Rosen S. D., McDonald K. A. Basement-membrane components associated with the extracellular matrix of the lymph node. Cell Tissue Res. 1988 May;252(2):367–375. doi: 10.1007/BF00214379. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- McCurley T. L., Gay R. E., Gay S., Glick A. D., Haralson M. A., Collins R. D. The extracellular matrix in "sclerosing" follicular center cell lymphomas: an immunohistochemical and ultrastructural study. Hum Pathol. 1986 Sep;17(9):930–938. doi: 10.1016/s0046-8177(86)80644-x. [DOI] [PubMed] [Google Scholar]
- Montes G. S., Krisztán R. M., Shigihara K. M., Tokoro R., Mourão P. A., Junqueira L. C. Histochemical and morphological characterization of reticular fibers. Histochemistry. 1980 Feb;65(2):131–141. doi: 10.1007/BF00493161. [DOI] [PubMed] [Google Scholar]
- Nowack H., Gay S., Wick G., Becker U., Timpl R. Preparation and use in immunohistology of antibodies specific for type I and type III collagen and procollagen. J Immunol Methods. 1976;12(1-2):117–124. doi: 10.1016/0022-1759(76)90101-0. [DOI] [PubMed] [Google Scholar]
- Pras M., Glynn L. E. Isolation of a non-collagenous reticulin component and its primary characterization. Br J Exp Pathol. 1973 Aug;54(4):449–456. [PMC free article] [PubMed] [Google Scholar]
- Pras M., Johnson G. D., Holborow E. J., Glynn L. E. Antigenic properties of a non-collagenous reticulin component of normal connective tissue. Immunology. 1974 Sep;27(3):469–478. [PMC free article] [PubMed] [Google Scholar]
- Puchtler H., Waldrop F. W. Silver impregnation methods for reticulum fibers and reticulin: a re-investigation of their origins and specificity. Histochemistry. 1978 Sep 15;57(3):177–187. doi: 10.1007/BF00492078. [DOI] [PubMed] [Google Scholar]
- Schwartz E. Connective tissue alterations in the skin of ultraviolet irradiated hairless mice. J Invest Dermatol. 1988 Aug;91(2):158–161. doi: 10.1111/1523-1747.ep12464405. [DOI] [PubMed] [Google Scholar]
- Stenman S., Vaheri A. Distribution of a major connective tissue protein, fibronectin, in normal human tissues. J Exp Med. 1978 Apr 1;147(4):1054–1064. doi: 10.1084/jem.147.4.1054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Unsworth D. J., Scott D. L., Almond T. J., Beard H. K., Holborow E. J., Walton K. W. Studies on reticulin. I: Serological and immunohistological investigations of the occurrence of collagen type III, fibronectin and the non-collagenous glycoprotein of Pras and Glynn in reticulin. Br J Exp Pathol. 1982 Apr;63(2):154–166. [PMC free article] [PubMed] [Google Scholar]
- WINDRUM G. M., KENT P. W., EASTOE J. E. The constitution of human renal reticulin. Br J Exp Pathol. 1955 Feb;36(1):49–59. [PMC free article] [PubMed] [Google Scholar]