Abstract
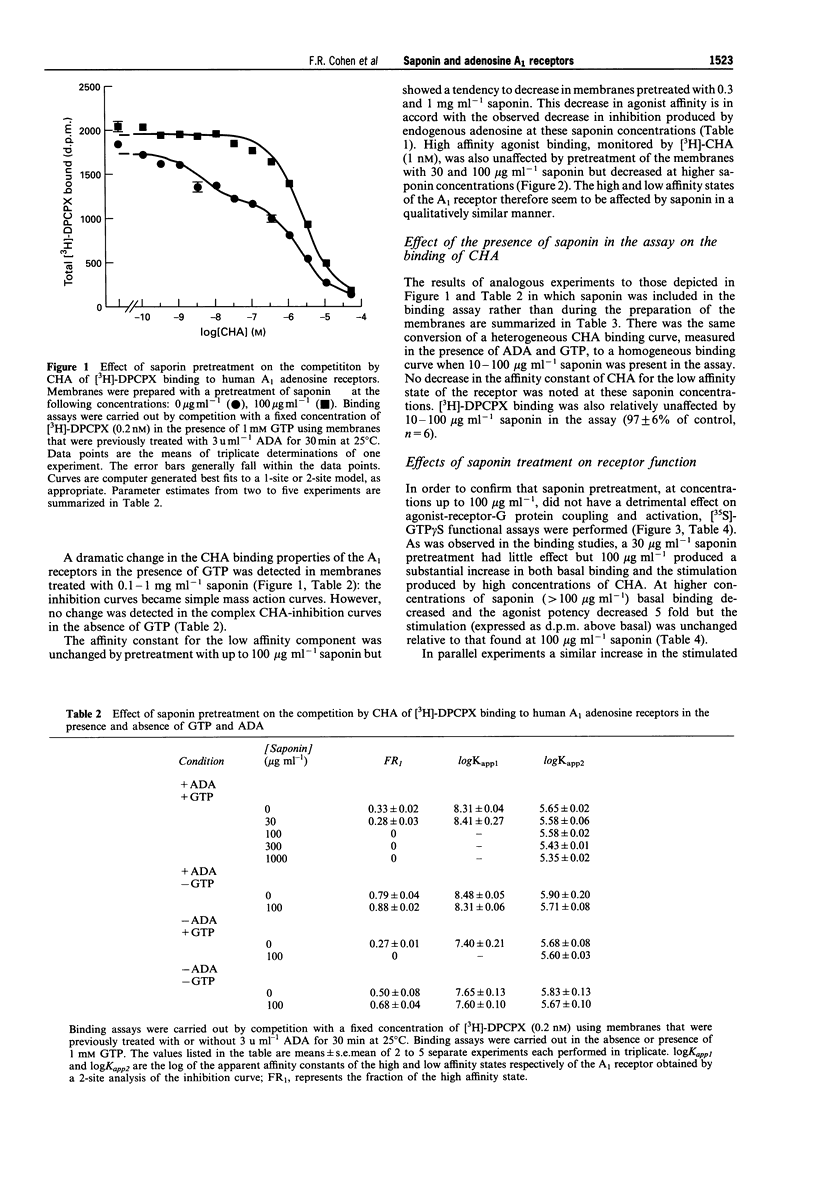
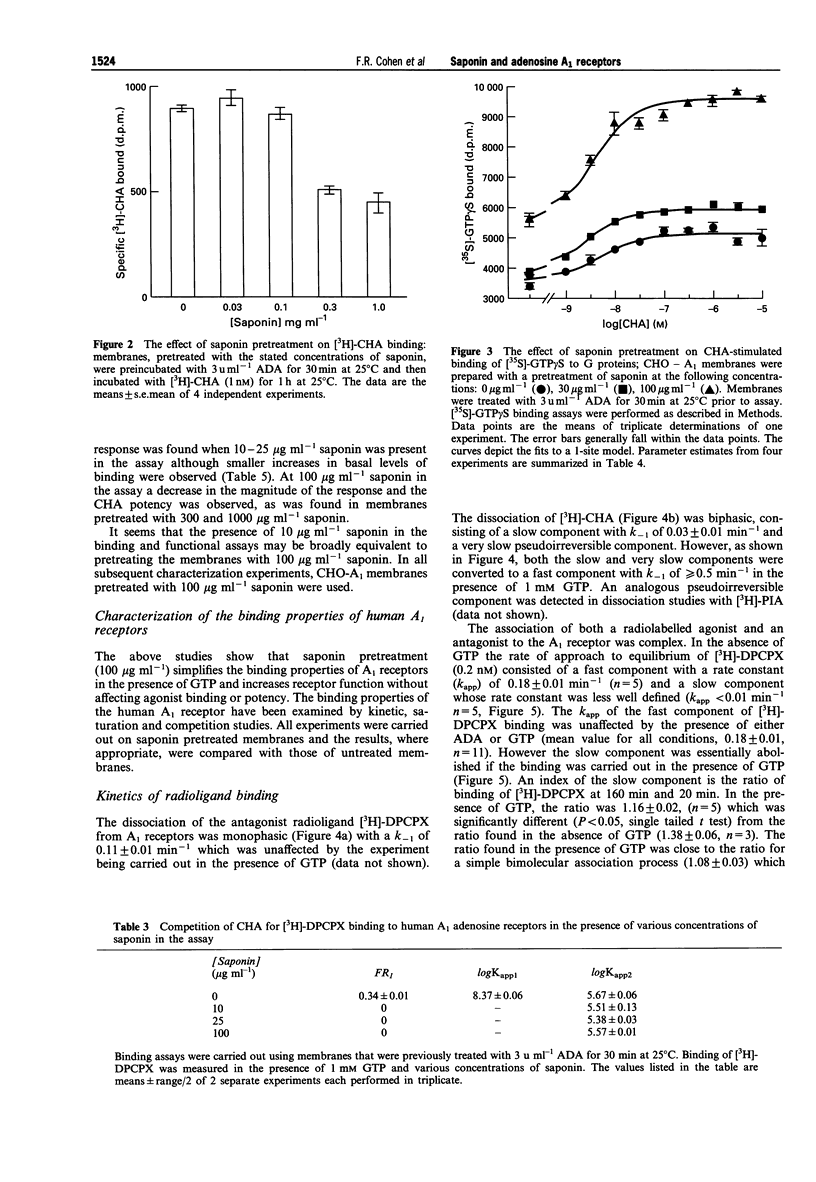
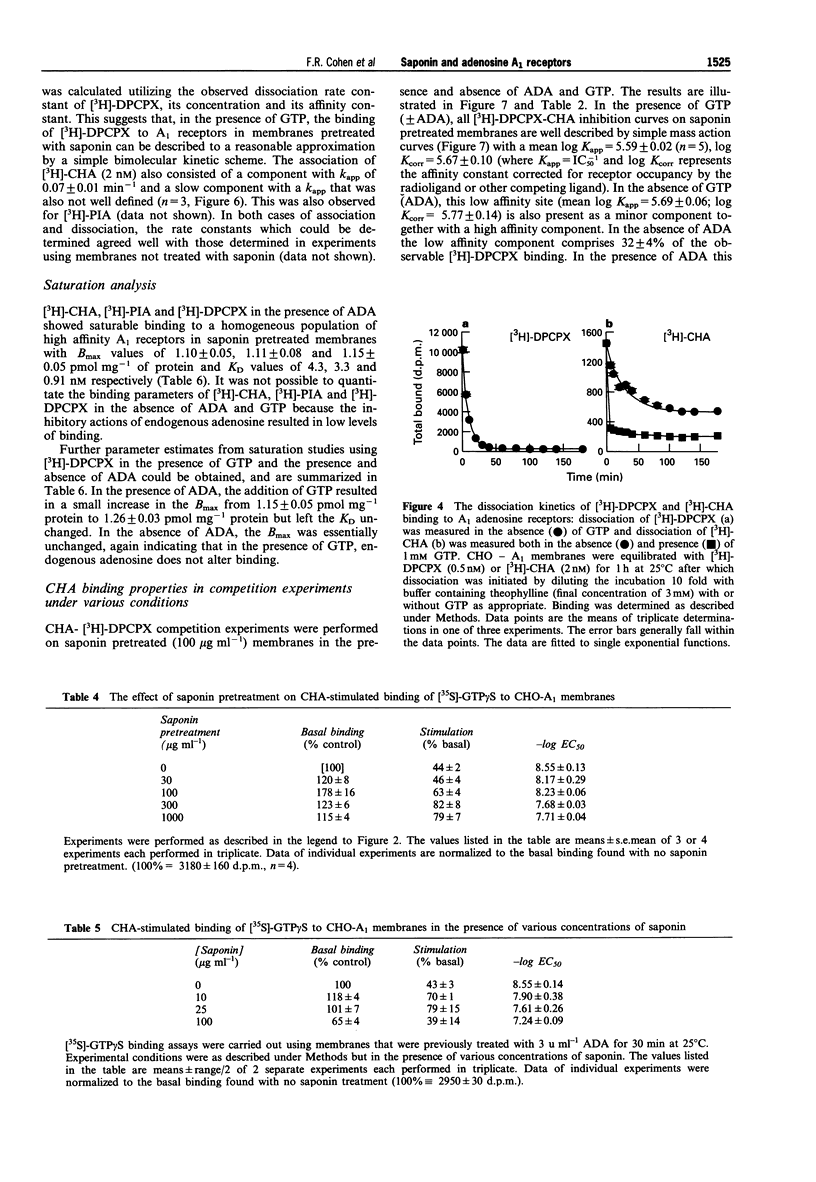
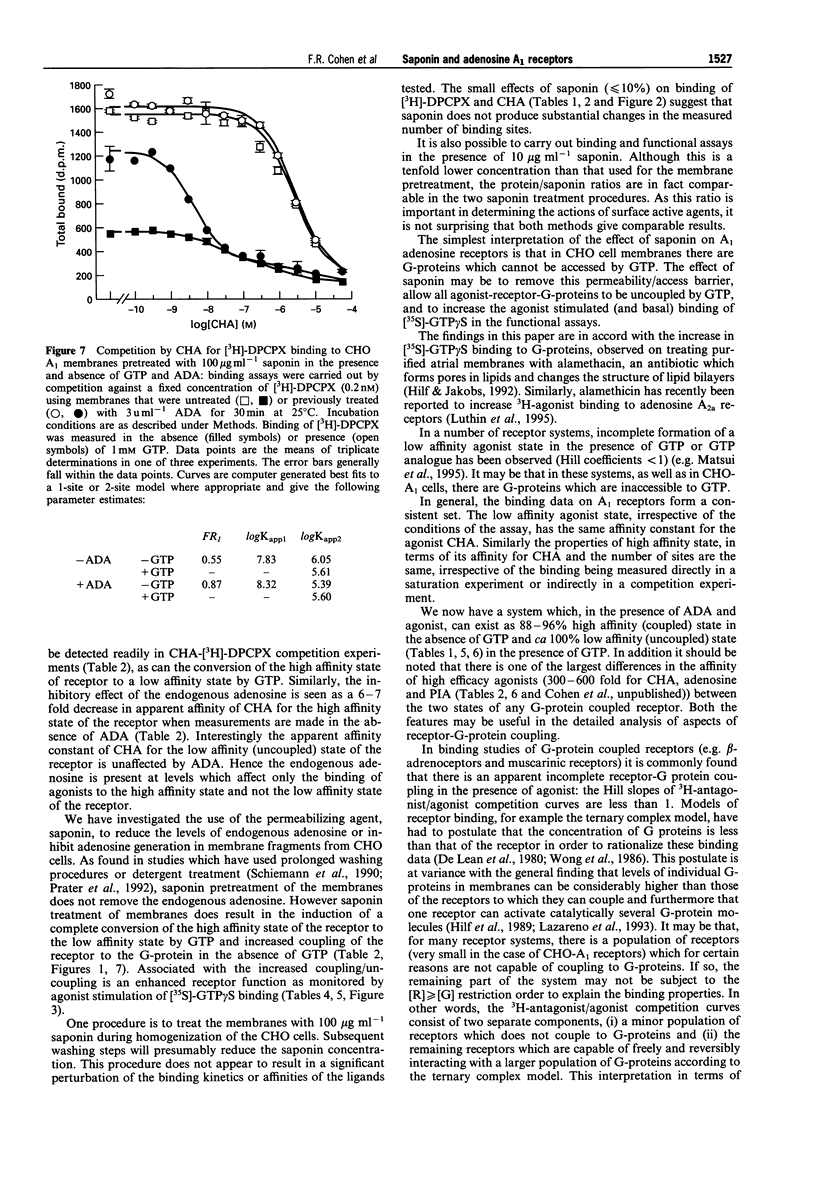
1. Experiments with adenosine deaminase suggest that adenosine is present in membrane preparations from CHO cells bearing adenosine A1 receptors. 2. Pretreatment of the membranes (ca 0.6 mg protein ml-1) with the permeabilizing agent saponin (100 micrograms ml-1) or addition of saponin (10 micrograms ml-1) to the membranes (0.02-0.08 mg protein ml-1) in the assay, generates homogeneous low affinity agonist binding curves in the presence of GTP and an increased function, assessed by agonist stimulation of [35S]-GTP gamma S binding. The affinity constants for the binding of an agonist and an antagonist are not affected by this saponin treatment. Saponin facilitates the interaction of guanine nucleotides with receptor G-protein complexes, possibly by removing a permeability barrier to access of G-proteins by GTP. However, adenosine is still present in the binding assays after saponin treatment. 3. The agonist binding properties of the human A1 receptor have been characterized. In saponin pretreated membranes, 80-90% of the A1 receptors are capable of forming agonist-receptor-G protein complexes in the absence of GTP. These complexes have a 300-600 fold higher affinity than uncoupled receptors for N6-cyclohexyladenosine. 4. A very slow component is observed in the association and dissociation kinetics of the agonist [3H]-N6-cyclohexyladenosine ([3H]-CHA) and in the association but not dissociation kinetics of the antagonist [3H]-8-cyclopentyl-1,3-dipropylxanthine ([3H]-DPCPX). The slow association component of [3H]-DPCPX is essentially absent when incubations are carried out in the presence of GTP. The slow dissociation component of [3H]-CHA binding is rapidly disrupted by GTP. 5. It is hypothesized that long-lasting adenosine-receptor-G protein complexes are present in the CHO membrane preparations. The existence of these complexes, resistant to the action of adenosine deaminase but sensitive to GTP, may rationalize the observed kinetics and the increase in 3H-antagonist binding produced by GTP which has been observed in essentially all studies of A1 receptors and has been ascribed previously to precoupling of A1 receptors to G-proteins in the absence of agonists.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Burgess G. M., McKinney J. S., Fabiato A., Leslie B. A., Putney J. W., Jr Calcium pools in saponin-permeabilized guinea pig hepatocytes. J Biol Chem. 1983 Dec 25;258(24):15336–15345. [PubMed] [Google Scholar]
- De Lean A., Stadel J. M., Lefkowitz R. J. A ternary complex model explains the agonist-specific binding properties of the adenylate cyclase-coupled beta-adrenergic receptor. J Biol Chem. 1980 Aug 10;255(15):7108–7117. [PubMed] [Google Scholar]
- Hilf G., Gierschik P., Jakobs K. H. Muscarinic acetylcholine receptor-stimulated binding of guanosine 5'-O-(3-thiotriphosphate) to guanine-nucleotide-binding proteins in cardiac membranes. Eur J Biochem. 1989 Dec 22;186(3):725–731. doi: 10.1111/j.1432-1033.1989.tb15266.x. [DOI] [PubMed] [Google Scholar]
- Hilf G., Jakobs K. H. Agonist-independent inhibition of G protein activation by muscarinic acetylcholine receptor antagonists in cardiac membranes. Eur J Pharmacol. 1992 Mar 12;225(3):245–252. doi: 10.1016/0922-4106(92)90026-r. [DOI] [PubMed] [Google Scholar]
- Jones P. G., Curtis C. A., Hulme E. C. The function of a highly-conserved arginine residue in activation of the muscarinic M1 receptor. Eur J Pharmacol. 1995 Feb 15;288(3):251–257. doi: 10.1016/0922-4106(95)90036-5. [DOI] [PubMed] [Google Scholar]
- Klotz K. N., Keil R., Zimmer F. J., Schwabe U. Guanine nucleotide effects on 8-cyclopentyl-1,3-[3H]dipropylxanthine binding to membrane-bound and solubilized A1 adenosine receptors of rat brain. J Neurochem. 1990 Jun;54(6):1988–1994. doi: 10.1111/j.1471-4159.1990.tb04902.x. [DOI] [PubMed] [Google Scholar]
- Lazareno S., Farries T., Birdsall N. J. Pharmacological characterization of guanine nucleotide exchange reactions in membranes from CHO cells stably transfected with human muscarinic receptors m1-m4. Life Sci. 1993;52(5-6):449–456. doi: 10.1016/0024-3205(93)90301-i. [DOI] [PubMed] [Google Scholar]
- Leung E., Jacobson K. A., Green R. D. Analysis of agonist-antagonist interactions at A1 adenosine receptors. Mol Pharmacol. 1990 Jul;38(1):72–83. [PMC free article] [PubMed] [Google Scholar]
- Linden J. Adenosine deaminase for removing adenosine: how much is enough? Trends Pharmacol Sci. 1989 Jul;10(7):260–262. doi: 10.1016/0165-6147(89)90021-7. [DOI] [PubMed] [Google Scholar]
- Luthin D. R., Olsson R. A., Thompson R. D., Sawmiller D. R., Linden J. Characterization of two affinity states of adenosine A2a receptors with a new radioligand, 2-[2-(4-amino-3-[125I]iodophenyl)ethylamino]adenosine. Mol Pharmacol. 1995 Feb;47(2):307–313. [PubMed] [Google Scholar]
- Matsui H., Lazareno S., Birdsall N. J. Probing of the location of the allosteric site on m1 muscarinic receptors by site-directed mutagenesis. Mol Pharmacol. 1995 Jan;47(1):88–98. [PubMed] [Google Scholar]
- Parkinson F. E., Fredholm B. B. Magnesium-dependent enhancement of endogenous agonist binding to A1 adenosine receptors: a complicating factor in quantitative autoradiography. J Neurochem. 1992 Mar;58(3):941–950. doi: 10.1111/j.1471-4159.1992.tb09347.x. [DOI] [PubMed] [Google Scholar]
- Prater M. R., Taylor H., Munshi R., Linden J. Indirect effect of guanine nucleotides on antagonist binding to A1 adenosine receptors: occupation of cryptic binding sites by endogenous vesicular adenosine. Mol Pharmacol. 1992 Nov;42(5):765–772. [PubMed] [Google Scholar]
- Schiemann W. P., Walther J. M., Buxton I. L. On the ability of endogenous adenosine to regulate purine nucleoside receptor binding of antagonists in smooth muscle membranes. J Pharmacol Exp Ther. 1990 Nov;255(2):886–892. [PubMed] [Google Scholar]
- Severne Y., Ijzerman A., Nerme V., Timmerman H., Vauquelin G. Shallow agonist competition binding curves for beta-adrenergic receptors: the role of tight agonist binding. Mol Pharmacol. 1987 Jan;31(1):69–73. [PubMed] [Google Scholar]
- Stiles G. L. A1 adenosine receptor-G protein coupling in bovine brain membranes: effects of guanine nucleotides, salt, and solubilization. J Neurochem. 1988 Nov;51(5):1592–1598. doi: 10.1111/j.1471-4159.1988.tb01129.x. [DOI] [PubMed] [Google Scholar]
- Wassler M., Jonasson I., Persson R., Fries E. Differential permeabilization of membranes by saponin treatment of isolated rat hepatocytes. Release of secretory proteins. Biochem J. 1987 Oct 15;247(2):407–415. doi: 10.1042/bj2470407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Willingham M. C., Yamada S. S., Pastan I. Ultrastructural antibody localization of alpha2-macroglobulin in membrane-limited vesicles in cultured cells. Proc Natl Acad Sci U S A. 1978 Sep;75(9):4359–4363. doi: 10.1073/pnas.75.9.4359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong H. M., Sole M. J., Wells J. W. Assessment of mechanistic proposals for the binding of agonists to cardiac muscarinic receptors. Biochemistry. 1986 Nov 4;25(22):6995–7008. doi: 10.1021/bi00370a037. [DOI] [PubMed] [Google Scholar]


