Abstract
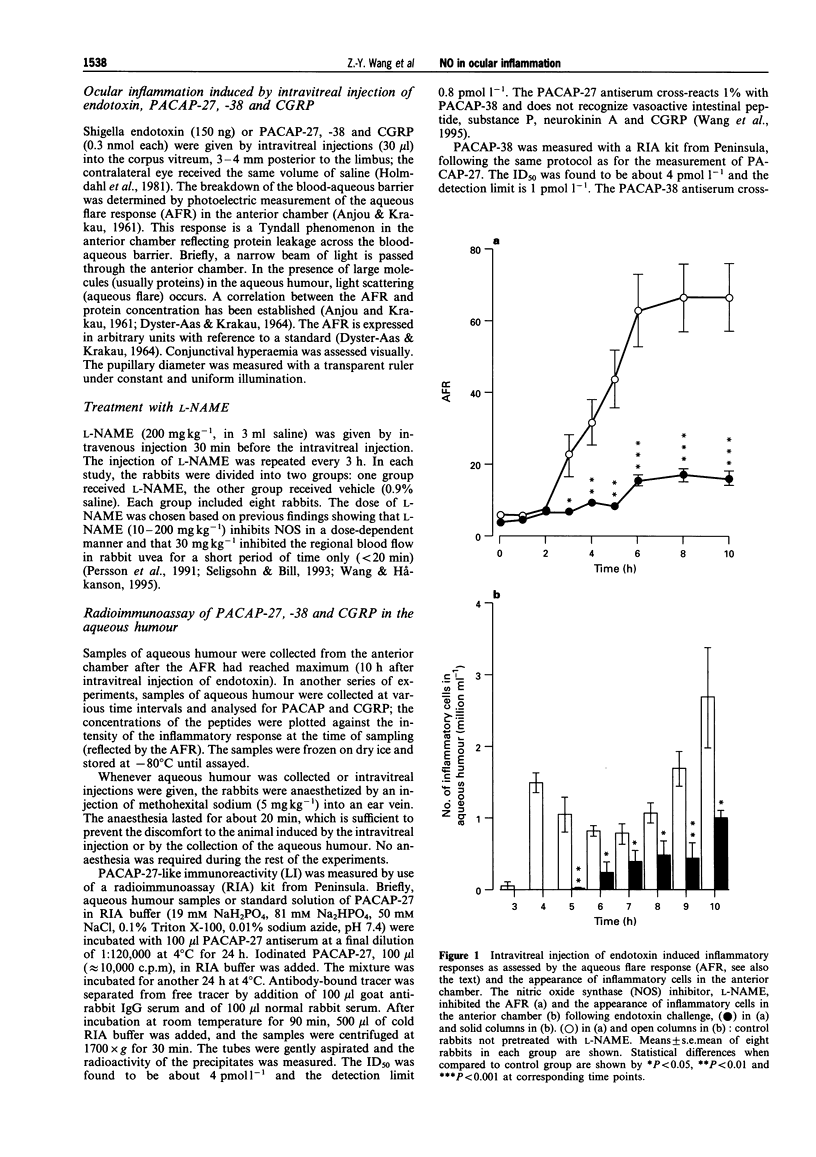
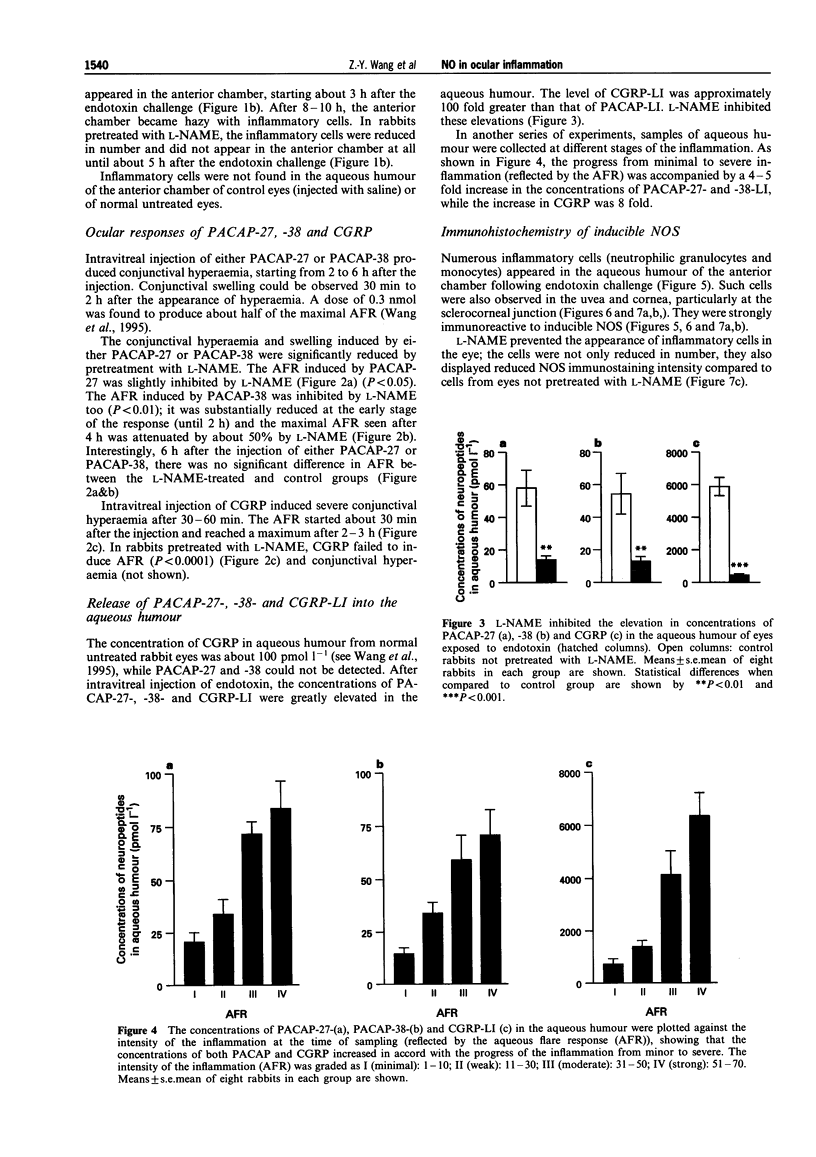
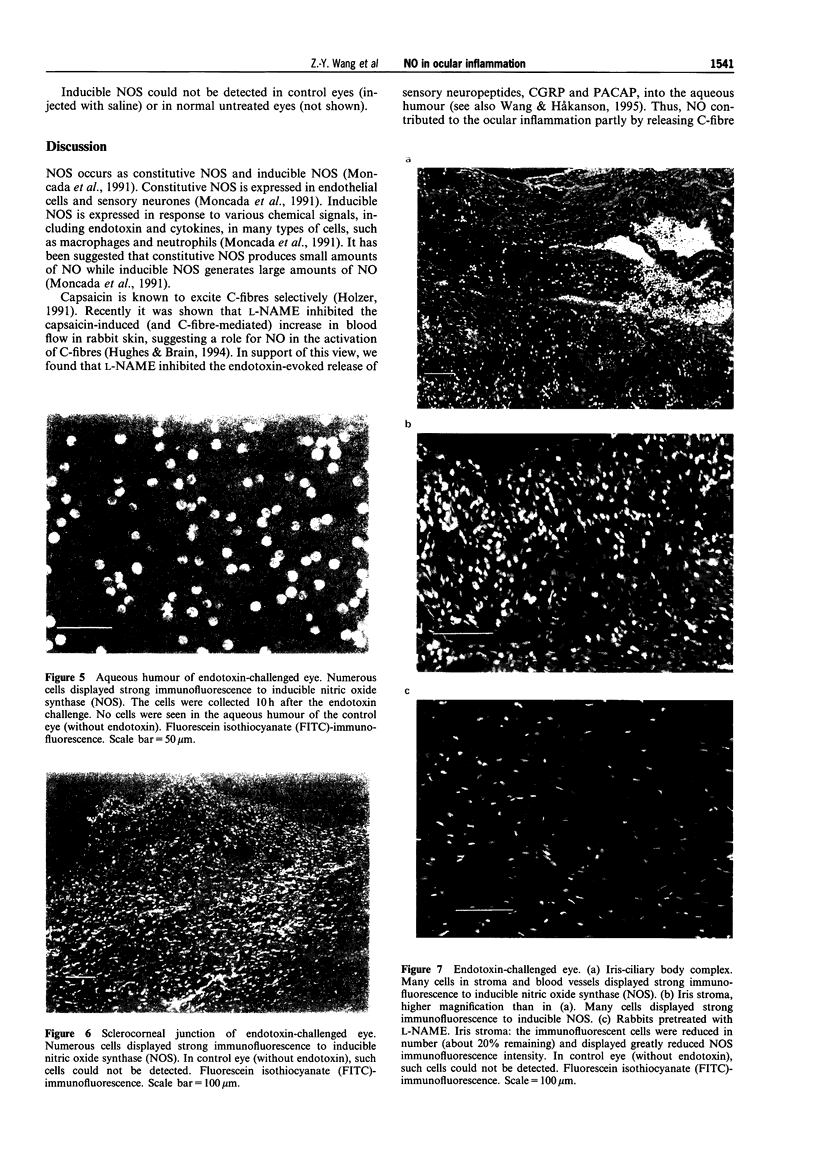
1. The actions of nitric oxide (NO) have been investigated in an endotoxin-evoked ocular inflammatory model in the rabbit, with particular emphasis on the relationship between NO, sensory nerves (C-fibres) and the C-fibre neuropeptides, calcitonin gene-related peptide (CGRP) and pituitary adenylate cyclase activating peptide (PACAP). 2. Endotoxin, injected intravitreally, evoked inflammatory responses, i.e. conjunctival hyperaemia, miosis and protein extravasation, reflected by the aqueous flare response (AFR). In control rabbits, the maximum AFR was 66.5 +/- 9.5 (arbitrary units). Pretreatment with the NO synthase (NOS) inhibitor, NG-nitro-L-arginine (L-NAME, 200 mg kg-1) given by intravenous injection, inhibited the endotoxin-evoked responses; the AFR was 16.5 +/- 1.9 (n = 8, P < 0.001) and the conjunctival hyperaemia was abolished. 3. Endotoxin-evoked ocular inflammation is associated with the release of CGRP and PACAP from C-fibres. In the eyes challenged with endotoxin, the concentrations of PACAP-27, -38 and CGRP in the aqueous humour were 58.2 +/- 10.9, 54.4 +/- 12.4 and 5526 +/- 519 (pmoll'), respectively. L-NAME inhibited the release of PACAP-27, -38 and CGRP; the concentrations were 14.3 +/- 2.5, 13.5 +/- 2.5 and 510 +/- 67 (pmoll-1), respectively (n = 8, P < 0.01 or 0.001). 4. Intravitreal injection of 0.3 nmol CGRP induced conjunctival hyperaemia and AFR; the maximum AFR was 140.2 +/- 11.4. L-NAME suppressed the response induced by CGRP; the AFR was 23.4 +/- 5.5 (n = 8, P < 0.001). L-NAME abolished the conjunctival hyperaemia induced by PACAP-27 and -38 (0.3 nmol) and reduced the AFR. 5. The inflammatory cells that infiltrated the uvea, cornea and aqueous humour in large numbers in response to intravitreal injection of endotoxin were found to express inducible NOS. L-NAME prevented the appearance of such cells. 6. Our findings suggest that NO plays an important role in the endotoxin-evoked ocular inflammation in the rabbit: NO activates C-fibres causing release of C-fibre neuropeptides into the aqueous humour. In addition, NO mediates scme of the ocular effects of CGRP and PACAP, since L-NAME suppressed the AFR induced by these peptides.
Full text
PDF
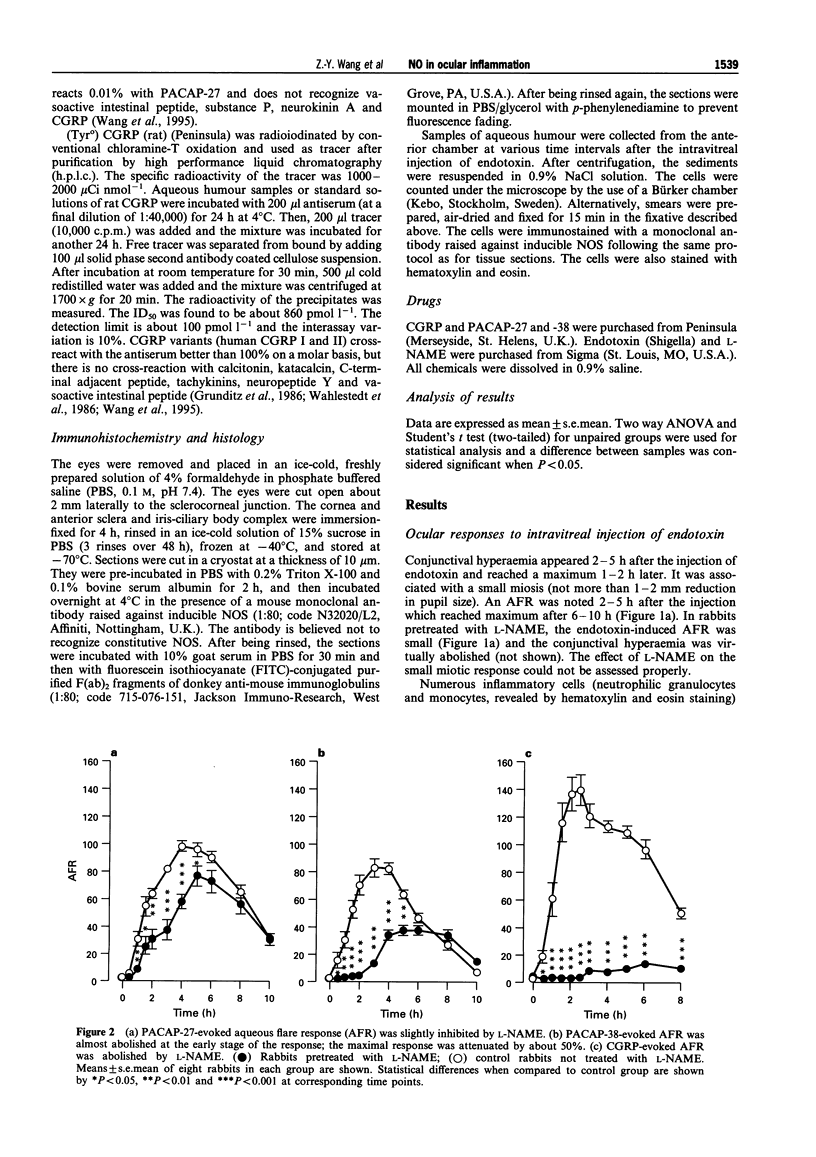


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- ANJOU C. I., KRAKAU C. E. Aqueous flare and protein content in the anterior chamber of normal rabbits' eyes. Acta Ophthalmol (Copenh) 1961;39:95–101. doi: 10.1111/j.1755-3768.1961.tb00244.x. [DOI] [PubMed] [Google Scholar]
- Andersson S. E. Glibenclamide and L-NG-nitro-arginine methyl ester modulate the ocular and hypotensive effects of calcitonin gene-related peptide. Eur J Pharmacol. 1992 Nov 24;224(1):89–91. doi: 10.1016/0014-2999(92)94823-e. [DOI] [PubMed] [Google Scholar]
- Bito L. Z. Inflammatory effects of endotoxin-like contaminants in commonly used protein preparations. Science. 1977 Apr 1;196(4285):83–85. doi: 10.1126/science.557238. [DOI] [PubMed] [Google Scholar]
- DYSTER-AAS K., KRAKAU C. E. AQUEOUS FLOW DETERMINATION IN THE RABBIT BY MEANS OF A MINIMAL EYE TRAUMA. Invest Ophthalmol. 1964 Apr;3:127–134. [PubMed] [Google Scholar]
- Grunditz T., Ekman R., Håkanson R., Rerup C., Sundler F., Uddman R. Calcitonin gene-related peptide in thyroid nerve fibers and C cells: effects on thyroid hormone secretion and response to hypercalcemia. Endocrinology. 1986 Nov;119(5):2313–2324. doi: 10.1210/endo-119-5-2313. [DOI] [PubMed] [Google Scholar]
- Holmdahl G., Håkanson R., Leander S., Rosell S., Folkers K., Sundler F. A substance P antagonist, [D-Pro2, D-Trp7,9]SP, inhibits inflammatory responses in the rabbit eye. Science. 1981 Nov 27;214(4524):1029–1031. doi: 10.1126/science.6171036. [DOI] [PubMed] [Google Scholar]
- Holzer P. Capsaicin: cellular targets, mechanisms of action, and selectivity for thin sensory neurons. Pharmacol Rev. 1991 Jun;43(2):143–201. [PubMed] [Google Scholar]
- Howes E. L., Jr, Cole P. W., Adair T. M., Cruse V. K., Pollycove M. Cellular and vascular responses in acute experimental ocular inflammation. Invest Ophthalmol Vis Sci. 1994 Nov;35(12):4031–4038. [PubMed] [Google Scholar]
- Hughes S. R., Brain S. D. Nitric oxide-dependent release of vasodilator quantities of calcitonin gene-related peptide from capsaicin-sensitive nerves in rabbit skin. Br J Pharmacol. 1994 Feb;111(2):425–430. doi: 10.1111/j.1476-5381.1994.tb14752.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moncada S., Palmer R. M., Higgs E. A. Nitric oxide: physiology, pathophysiology, and pharmacology. Pharmacol Rev. 1991 Jun;43(2):109–142. [PubMed] [Google Scholar]
- Osborne N. N., Barnett N. L., Herrera A. J. NADPH diaphorase localization and nitric oxide synthetase activity in the retina and anterior uvea of the rabbit eye. Brain Res. 1993 May 7;610(2):194–198. doi: 10.1016/0006-8993(93)91400-m. [DOI] [PubMed] [Google Scholar]
- Persson M. G., Wiklund N. P., Gustafsson L. E. Nitric oxide requirement for vasomotor nerve-induced vasodilatation and modulation of resting blood flow in muscle microcirculation. Acta Physiol Scand. 1991 Jan;141(1):49–56. doi: 10.1111/j.1748-1716.1991.tb09043.x. [DOI] [PubMed] [Google Scholar]
- Rosenbaum J. T., Raymond W. Monocyte chemotactic activity induced by intravitreal endotoxin. Invest Ophthalmol Vis Sci. 1985 Sep;26(9):1267–1273. [PubMed] [Google Scholar]
- Seligsohn E. E., Bill A. Effects of NG-nitro-L-arginine methyl ester on the cardiovascular system of the anaesthetized rabbit and on the cardiovascular response to thyrotropin-releasing hormone. Br J Pharmacol. 1993 Aug;109(4):1219–1225. doi: 10.1111/j.1476-5381.1993.tb13752.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stone R. A., Kuwayama Y., Laties A. M. Regulatory peptides in the eye. Experientia. 1987 Jul 15;43(7):791–800. doi: 10.1007/BF01945357. [DOI] [PubMed] [Google Scholar]
- Unger W. G. Review: mediation of the ocular response to injury. J Ocul Pharmacol. 1990 Winter;6(4):337–353. doi: 10.1089/jop.1990.6.337. [DOI] [PubMed] [Google Scholar]
- Wahlestedt C., Beding B., Ekman R., Oksala O., Stjernschantz J., Håkanson R. Calcitonin gene-related peptide in the eye: release by sensory nerve stimulation and effects associated with neurogenic inflammation. Regul Pept. 1986 Dec 22;16(2):107–115. doi: 10.1016/0167-0115(86)90054-6. [DOI] [PubMed] [Google Scholar]
- Wang Z. Y., Alm P., Håkanson R. Distribution and effects of pituitary adenylate cyclase-activating peptide in the rabbit eye. Neuroscience. 1995 Nov;69(1):297–308. doi: 10.1016/0306-4522(95)00258-k. [DOI] [PubMed] [Google Scholar]
- Wang Z. Y., Håkanson R. Role of nitric oxide (NO) in ocular inflammation. Br J Pharmacol. 1995 Nov;116(5):2447–2450. doi: 10.1111/j.1476-5381.1995.tb15094.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamamoto R., Bredt D. S., Snyder S. H., Stone R. A. The localization of nitric oxide synthase in the rat eye and related cranial ganglia. Neuroscience. 1993 May;54(1):189–200. doi: 10.1016/0306-4522(93)90393-t. [DOI] [PubMed] [Google Scholar]