Abstract
We compared ciprofloxacin, rifampin, and gentamicin treatments, alone and in combination, for 5 days in the therapy of experimental aortic valve endocarditis in rats caused by a clinical isolate of vancomycin-resistant Enterococcus faecium. The MICs and MBCs of vancomycin, ciprofloxacin, rifampin, and gentamicin were 250 and > 1,000, 3.1 and 6.3, 0.098 and 1.6, and 12.5 and > 50 micrograms/ml, respectively. Infected rats were sacrificed after completing 5 days of therapy. Additional rats within each treatment group were followed for 5 days beyond the last dose of antibiotic therapy. Although survivals in the different groups were not significantly different after 5 days of therapy, survival was significantly better 5 days beyond the last dose of antibiotic therapy in rats treated with rifampin-containing regimens. The combination of ciprofloxacin and gentamicin was bactericidal in vitro and in vegetations from rats with enterococcal endocarditis. Rifampin alone was similarly bactericidal in vivo, but it was not significantly better than rifampin in combination with other antibiotics. Subpopulations resistant to rifampin, but not ciprofloxacin, were detected in the inoculum and in most vegetations during therapy. However, the combination of ciprofloxacin plus both gentamicin and rifampin reduced both the rifampin-susceptible and -resistant population in vegetations of 9 of 10 animals below the level of detection after 5 days of therapy. Nevertheless, a residual enterococcal population apparently remained in numbers of < 2 log10 CFU/g after 5 days of therapy, which resulted in relapse. Perhaps a longer course of therapy would have eliminated this residual population and improved efficacy.
Full text
PDF
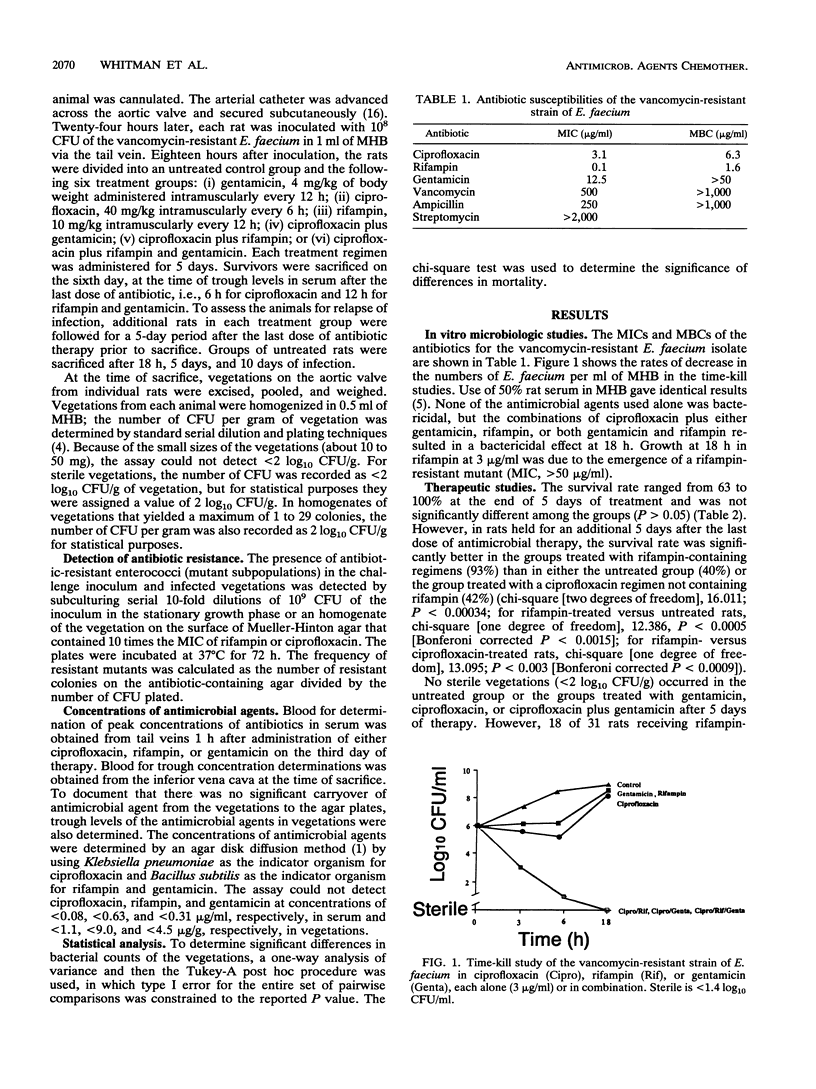
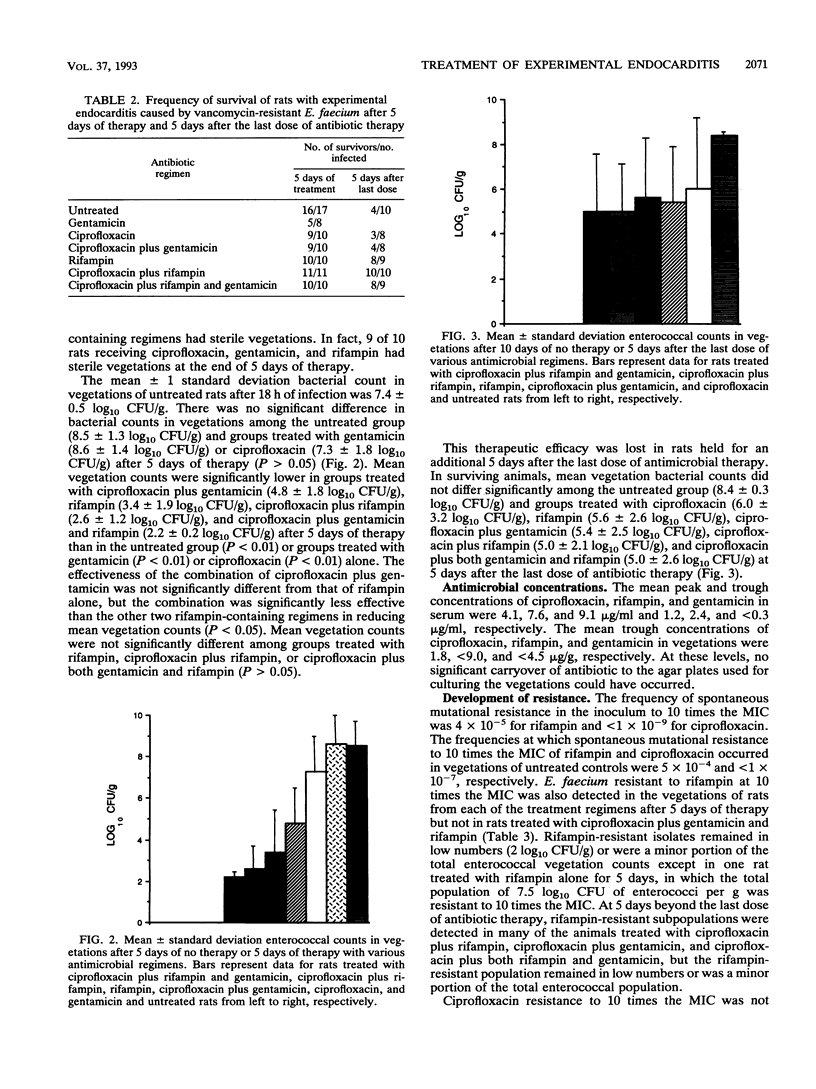


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bisno A. L., Dismukes W. E., Durack D. T., Kaplan E. L., Karchmer A. W., Kaye D., Rahimtoola S. H., Sande M. A., Sanford J. P., Watanakunakorn C. Antimicrobial treatment of infective endocarditis due to viridans streptococci, enterococci, and staphylococci. JAMA. 1989 Mar 10;261(10):1471–1477. [PubMed] [Google Scholar]
- Bush L. M., Calmon J., Cherney C. L., Wendeler M., Pitsakis P., Poupard J., Levison M. E., Johnson C. C. High-level penicillin resistance among isolates of enterococci. Implications for treatment of enterococcal infections. Ann Intern Med. 1989 Apr 1;110(7):515–520. doi: 10.7326/0003-4819-110-7-515. [DOI] [PubMed] [Google Scholar]
- Carrizosa J., Kaye D. Antibiotic synergism in enterococcal endocarditis. J Lab Clin Med. 1976 Jul;88(1):132–141. [PubMed] [Google Scholar]
- Gold M. J., Calmon J., Wendeler M., Levison M. E., Johnson C. C. Synergistic bactericidal activity of rat serum with vancomycin against enterococci. J Infect Dis. 1991 Jun;163(6):1358–1361. doi: 10.1093/infdis/163.6.1358. [DOI] [PubMed] [Google Scholar]
- Ingerman M., Pitsakis P. G., Rosenberg A., Hessen M. T., Abrutyn E., Murray B. E., Levison M. E. beta-Lactamase production in experimental endocarditis due to aminoglycoside-resistant Streptococcus faecalis. J Infect Dis. 1987 Jun;155(6):1226–1232. doi: 10.1093/infdis/155.6.1226. [DOI] [PubMed] [Google Scholar]
- Kaatz G. W., Seo S. M., Barriere S. L., Albrecht L. M., Rybak M. J. Ciprofloxacin and rifampin, alone and in combination, for therapy of experimental Staphylococcus aureus endocarditis. Antimicrob Agents Chemother. 1989 Aug;33(8):1184–1187. doi: 10.1128/aac.33.8.1184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaplan A. H., Gilligan P. H., Facklam R. R. Recovery of resistant enterococci during vancomycin prophylaxis. J Clin Microbiol. 1988 Jun;26(6):1216–1218. doi: 10.1128/jcm.26.6.1216-1218.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim M. J., Weiser M., Gottschall S., Randall E. L. Identification of Streptococcus faecalis and Streptococcus faecium and susceptibility studies with newly developed antimicrobial agents. J Clin Microbiol. 1987 May;25(5):787–790. doi: 10.1128/jcm.25.5.787-790.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leclercq R., Derlot E., Duval J., Courvalin P. Plasmid-mediated resistance to vancomycin and teicoplanin in Enterococcus faecium. N Engl J Med. 1988 Jul 21;319(3):157–161. doi: 10.1056/NEJM198807213190307. [DOI] [PubMed] [Google Scholar]
- Livornese L. L., Jr, Dias S., Samel C., Romanowski B., Taylor S., May P., Pitsakis P., Woods G., Kaye D., Levison M. E. Hospital-acquired infection with vancomycin-resistant Enterococcus faecium transmitted by electronic thermometers. Ann Intern Med. 1992 Jul 15;117(2):112–116. doi: 10.7326/0003-4819-117-2-112. [DOI] [PubMed] [Google Scholar]
- Moellering R. C., Jr, Wennersten C. Therapeutic potential of rifampin in enterococcal infections. Rev Infect Dis. 1983 Jul-Aug;5 (Suppl 3):S528–S532. doi: 10.1093/clinids/5.supplement_3.s528. [DOI] [PubMed] [Google Scholar]
- Murray B. E., Mederski-Samaroj B. Transferable beta-lactamase. A new mechanism for in vitro penicillin resistance in Streptococcus faecalis. J Clin Invest. 1983 Sep;72(3):1168–1171. doi: 10.1172/JCI111042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sahm D. F., Koburov G. T. In vitro activities of quinolones against enterococci resistant to penicillin-aminoglycoside synergy. Antimicrob Agents Chemother. 1989 Jan;33(1):71–77. doi: 10.1128/aac.33.1.71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santoro J., Levison M. E. Rat model of experimental endocarditis. Infect Immun. 1978 Mar;19(3):915–918. doi: 10.1128/iai.19.3.915-918.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shlaes D. M., Bouvet A., Devine C., Shlaes J. H., al-Obeid S., Williamson R. Inducible, transferable resistance to vancomycin in Enterococcus faecalis A256. Antimicrob Agents Chemother. 1989 Feb;33(2):198–203. doi: 10.1128/aac.33.2.198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uttley A. H., Collins C. H., Naidoo J., George R. C. Vancomycin-resistant enterococci. Lancet. 1988 Jan 2;1(8575-6):57–58. doi: 10.1016/s0140-6736(88)91037-9. [DOI] [PubMed] [Google Scholar]
- Williamson R., le Bouguénec C., Gutmann L., Horaud T. One or two low affinity penicillin-binding proteins may be responsible for the range of susceptibility of Enterococcus faecium to benzylpenicillin. J Gen Microbiol. 1985 Aug;131(8):1933–1940. doi: 10.1099/00221287-131-8-1933. [DOI] [PubMed] [Google Scholar]
- Wolfson J. S., Hooper D. C. Bacterial resistance to quinolones: mechanisms and clinical importance. Rev Infect Dis. 1989 Jul-Aug;11 (Suppl 5):S960–S968. doi: 10.1093/clinids/11.supplement_5.s960. [DOI] [PubMed] [Google Scholar]