Abstract
Zinc is involved in virtually all aspects of cellular and molecular biology as a catalytic, structural, and regulatory cofactor in over 1000 proteins. Zinc binding to proteins requires an adequate supply of zinc and intact molecular mechanisms for redistributing zinc ions to make them available at the right time and location. Several dozen gene products participate in this process, in which interactions between zinc and sulfur donors determine the mobility of zinc and establish coupling between cellular redox state and zinc availability. Specifically, the redox properties of metallothionein and its apoprotein thionein are critical for buffering zinc ions and for controlling fluctuations in the range of picomolar concentrations of “free” zinc ions in cellular signaling. Metallothionein and other proteins with sulfur coordination environments are sensitive to redox perturbations and can render cells susceptible to injury when oxidative stress compromises the cellular redox and zinc buffering capacity in chronic diseases. The implications of these fundamental principles for zinc metabolism in type 2 diabetes are briefly discussed.
By and large, the significance of the essential nutrient zinc in medicine has been limited to relatively rare cases of severe deficiency, which results in growth retardation, hypogonadism, dermatitis, and immune dysfunction (1). In contrast, mild zinc deficiency is believed to be widespread. Although difficult to diagnose owing to a lack of suitable clinical tests, mild zinc deficiency is thought to be associated with clinically important functional deficits. Much more than anecdotal evidence links zinc deficiency to poor neuropsychological performance, abnormalities of fetal development, and the risk of developing cancer of the upper aerodigestive tract (2–4). Moreover, zinc has been implicated in the etiology of specific diseases, such as Acrodermatitis enteropathica, a rare genetic disorder that leads to severe zinc deficiency due to mutations in the human Zip4 (SLC39A4) importer (5,6) and transient neonatal zinc deficiency, in which zinc in the mother’s milk is low due to a mutation in the human ZnT-2 (SLC30A2) exporter (7). Other gene defects and genetic variability in zinc homeostatic proteins likely exist because at least two dozen gene products regulate cellular zinc. A case in point is a polymorphism in the human MT-1a gene, which affects zinc ion release from the protein and is associated with longevity in an Italian population (8). Another area where zinc has been implicated in pathogenesis is based on the hypothesis that increases of intracellular “free” zinc ions above a certain threshold are neurotoxic (9). In this case, it is not a deficiency of zinc but rather too much available zinc that causes cellular injury. To evaluate the consequences of such cellular imbalances of zinc, one of the key challenges is to understand the largely unknown molecular processes that control availability and redistribution of zinc. Like most transition metals, zinc is not readily available in the cell as its aquocomplex. Families of membrane zinc transporters, zinc sensors, such as metal response element (MRE)-binding transcription factor-1 (MTF-1), and metallothioneins (MT) are part of a cellular zinc homeostatic system with specific biological mechanisms in supplying thousands of proteins with zinc. Thus, a significant number of proteins in the human genome is dedicated exclusively to the task of regulating cellular zinc. We focus in this article on quantitative information regarding the redox control of the availability of zinc ions at the cellular and molecular level and a role of MT in zinc buffering.
Metallothionein
The Zn3S9 and Zn4S11 zinc/thiolate clusters in MT bind zinc rather tightly and yet can make it available. Experiments in vitro demonstrated that a fraction of zinc in MT can be transferred to the apoforms of enzymes (10), and that the apoprotein (reduced thionein, TR), which is present under normal physiological conditions in tissues and in neoplastic cell lines (11–13), removes zinc from zinc-inhibited enzymes (14).
Functions of zinc-MT are based on reversible dissociation of its zinc ions and oxidoreduction of the cysteine sulfur donors in the zinc/thiolate clusters (15). This property is unusual for zinc proteins as it challenges the conventional description of zinc coordination environments in proteins being redox-inert. It provides a mechanism whereby zinc availability and redox metabolism are linked (16). When oxidants react with MT, zinc ions are released and the oxidized protein is formed. Assays were developed to determine the amount of oxidized protein (thionin, T0) in tissues (13,17). T0 is indeed present in tissues and cell lines under normal physiological conditions and increases under conditions of oxidative stress (13,18). Thus, MT is highly dynamic with regard to its zinc load and redox state. The protein with 20 reduced cysteinyl residues and seven bound divalent metal ions, which is the only form for which a high resolution structure has been determined, is not the only physiologically important state of the protein. The new assays allow determination of the metal load with the holoprotein/apoprotein (MT/TR) ratio and the oxidation state with the reduced/oxidized protein (TR/T0) ratio. Fluorogenic zinc chelating agents make it possible to determine simultaneously the amount of “free” zinc in cells.
Cellular Zinc Buffering Capacity and “Free” Zinc
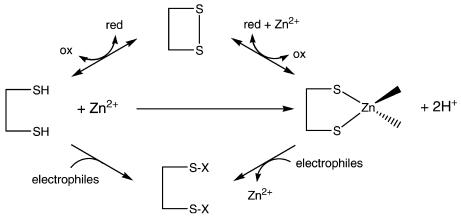
Total zinc concentrations of eukaryotic cells are a few hundred micromolar and thus relatively high. How much of it is in the “free” form depends on the zinc buffering capacity, which has never been determined. Investigations with human colon carcinoma (HT-29) cells demonstrated that about 28 μM of cellular ligands do not have a bound zinc ion but bind added zinc ions tightly with an average dissociation constant of 83 pM (19). Given a total concentration of 264 μM in these cells, 28 μM corresponds to an 11% surplus of cellular ligands. The discovery of these sites that can bind zinc ions with relatively high affinity but are not occupied with zinc demonstrates tight zinc buffering. About 30% of this zinc buffering capacity is due to sulfur donors (thiols), and thus, serves at the same time as redox (oxidant) buffering capacity (20). A condition such as oxidative stress or the presence of reactive compounds, such as electrophiles, can have the dual effects of oxidizing or modifying cysteine sulfur ligands that are essential for zinc buffering and releasing zinc ions from proteins (Figure 1). In both instances, “free” zinc concentrations increase. The molecular identity of the zinc buffering sites is unknown, but they comprise TR. When cellular “free” zinc increases, zinc modulates, or may regulate, the biological functions of proteins with such sites. Candidates for zinc regulation are inhibitory zinc-binding sites on enzymes with nanomolar or lower affinity for zinc, such as protein tyrosine phosphatases and caspase-3 (14), or interfacial zinc sites, where zinc binding determines the quaternary or quinary structure of proteins (21). The results suggest regulatory roles of zinc ions at picomolar concentrations, a range that hitherto was inaccessible to biochemical exploration.
Figure 1.
Competition among oxidation or covalent modification of thiols, zinc ion binding, and oxidation or covalent modification of zinc/thiolates determines the availability of “free” zinc ions.
In HT-29 cells, the concentration of “free” zinc ions determined with the fluorogenic chelating agent FluoZin-3 (“dye”) is 784 pM. This concentration is many orders of magnitude lower than that of total zinc. The value was established with a new procedure, in which data are collected at different dye concentrations and then extrapolated to a zero dye concentration to correct for the interference of the dye with cellular zinc buffering (19). Significantly, the dye response and hence the calculated amount of “free” zinc ions depend on the zinc buffering capacity of the cell. The amount of “free” zinc is 614 pM in resting, 1.25 nM in differentiated, and > 2.5 nM in apoptotic cells. These cellular states have different redox potentials for the glutathione/glutathione disulfide pair (22). More oxidized conditions correlate with increased amounts of T0 and “free” zinc (20). Thus, in the same cell line, physiological changes of the glutathione redox potential affect the redox state of MT and the amount of “free” zinc, clearly linking the availability of zinc, MT, and redox metabolism.
Many factors induce MT expression and thereby alter the zinc and redox buffering capacity; the protein varies at least 400-fold in different cell lines (23). The protein that is induced, however, is thionein, not metallothionein. Induced thionein is a strong chelating and reducing agent and will either lower zinc availability, if the protein is not immediately oxidized under conditions of high oxidative stress, or increase the cellular reducing capacity, if the binding sites of the protein are not immediately saturated by zinc ions under conditions of high zinc availability.
In summary, the redox dynamics of MT establish multiple ways of zinc-dependent cellular regulation. MT and T function as signal transducers for both zinc and redox signals at a central node in the cellular signaling network.
Perturbations of Zinc and Redox Buffering
Chemical reactions with zinc buffering ligands enable fluctuations of available “free” zinc ions with important consequences for the physiology of a cell because zinc ions are very potent effectors of the functions of some proteins at picomolar and low nanomolar concentrations. Buffering can be perturbed by redox agents that modify the surplus high-affinity zinc-binding sites, xenobiotics that bind zinc or cysteinyl sulfur, or thiophilic metal ions, such as cadmium, mercury, or others that either bind to the surplus ligands or displace zinc from its binding sites. Thus, one toxicologic mechanism during heavy metal poisoning may actually be the release of cytotoxic amounts of zinc ions (24). Changes of zinc buffering would make either more or less zinc available. As a result, “free” zinc ions will fluctuate with higher amplitudes and affect biological systems that normally would not respond. Exceeding physiological zinc buffering will elicit zinc signals with much larger amplitudes, and presumably, pleiotropic effects.
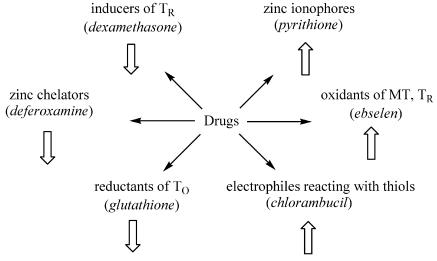
The relationships among zinc buffering, available zinc, and zinc-dependent functions are significant for the mechanisms of action of drugs (Figure 2). A drug with zinc-chelating properties will interfere with cellular zinc buffering and lower the availability of zinc, while a drug with redox properties can affect the thiol/disulfide redox state and change the availability of zinc.
Figure 2.
Drugs change the availability of “free” zinc by several mechanisms. Zinc chelation, induction of TR or generation of TR by reduction of T0 decrease zinc (left). Oxidation or other covalent modification of thiols and compounds that serve as carriers of zinc into the cell increase zinc (right).
Implications for Diseases with Particular Emphasis on Diabetes
Induced release of zinc ions from proteins with sulfur coordination environments is a general pathophysiological pathway with implications for many diseases. In this discussion, we focus on type 2 diabetes, where oxidative stress perturbs both zinc metabolism and MT levels and where proper control of zinc availability is essential for normal function, because zinc is involved in many aspects of insulin metabolism and signaling (25). Type 2 diabetes risk is associated with a polymorphism of the pancreatic β-cell-specific zinc transporter ZnT-8 (SLC30A8), which provides zinc for insulin maturation and/or storage in these cells (26,27). Antidiabetogenic and insulinomimetic properties of zinc are supported by the results from zinc supplementation in animals and humans (28,29) and are now explored therapeutically by synthesizing and testing zinc-containing drugs (30). Because cardiovascular disease is the leading cause of mortality in type 2 diabetes, there is a major research focus on the diabetic heart. “Free” zinc increases from 520 pM in normal to 870 pM in diabetic rat cardiomyocytes concomitant with increased reactive species, whereas levels of metallothionein and reduced glutathione and activities of enzymes of the antioxidant defense decrease (31). Treatment with selenium compounds that catalytically couple MT/T and glutathione/glutathione disulfide redox cycles prevent these effects (31,32). Also, zinc supplementation prevents the development of cardiomyopathy in mice through induction of MT, which has an antioxidant function in the heart (33,34). It had been noticed for some time that cardiovascular disease is associated with hyperhomocysteinemia. Homocysteinylation of MT releases zinc ions that activate transcription factor EGR-1 (early growth response-1) (35). Because the modified MT can no longer scavenge superoxide ions, concentrations of reactive species increase even in the presence of reduced glutathione or cysteine. The resulting impairment of redox and zinc homeostasis is thought to lead to endothelial dysfunction and atherothrombotic disease. The significance of metabolic functions of MT for diabetes is reinforced by the observations that MT levels are reduced in the plasma and muscle of humans with type 2 diabetes, and do not increase in skeletal muscle with exercise training (36), and that MT-1/-2 knock-out mice become mildly obese and develop hyperinsulinemia (37).
An additional pathway for both zinc ion release and interference with the functions of MT involves aldehydes (38). In diabetes, decomposition of AGEs (advanced glycation end-products) and lipid peroxides formed under oxidative stress increases the concentrations of endogenous aldehydes, such as acrolein, 4-hydroxynonenal, and malondialdehyde. These aldehydes react with zinc proteins and increase cellular “free” zinc ions, which in turn inhibit protein tyrosine phosphatases, in particular PTP 1B, the major phosphatase controlling the phosphorylation state of the insulin receptor (38–40). Zinc inhibition of PTP 1B enhances phosphorylation signaling of insulin and is believed to account for the known insulinomimetic effects of zinc (41). Moreover, it was suggested that development of local (tissue-specific) zinc deficiencies abolishes tonic zinc inhibition and modulation of protein tyrosine phosphatases and leads to insulin resistance (41).
In conclusion, cysteinyl sulfur is the ligand of zinc in a large number of proteins. Oxidation or other covalent modification of the sulfur donor affects the availability of zinc ions. Depending on the amplitudes of fluctuations of “free” zinc ions, the outcome can be either cytoprotection, for example, in lung injury (42), or cytotoxicity, for example, in neuronal injury (43). What role zinc supplementation, either alone or in combination with antioxidants, has in prevention and therapy requires thorough scrutiny (44). Zinc supplementation protects against the hepatotoxic effects of alcohol (ethanol) (45). It appears that rather small differences of picomolar “free” zinc – changes that could be readily achieved by nutrients, drugs, or toxins – determine the transition from physiology to pathology. Clearly, further exploration of the role of zinc buffering in oxidative and chemical cell injury in various organs and in chronic diseases is warranted.
ACKNOWLEDGMENT
This work was supported in part by a grant from the National Institutes of Health (GM 065388 to WM).
Footnotes
Online address: http://www.molmed.org
REFERENCES
- 1.Hambidge M. Human zinc deficiency. J Nutr. 2000;130:1344S–9S. doi: 10.1093/jn/130.5.1344S. [DOI] [PubMed] [Google Scholar]
- 2.Sandstead HH, Frederickson CJ, Penland JG. History of zinc related to brain function. J Nutr. 2000;130:496S–502S. doi: 10.1093/jn/130.2.496S. [DOI] [PubMed] [Google Scholar]
- 3.Frederickson CJ, Koh J-Y, Bush AI. The neurobiology of zinc in health and disease. Nat Rev Neurosci. 2005;6:449–62. doi: 10.1038/nrn1671. [DOI] [PubMed] [Google Scholar]
- 4.Fong LYY, Jiang YB, Farber JL. Zinc deficiency potentiates induction and progression of lingual and esophageal tumors in p53-deficient mice. Carcinogenesis. 2006;27:1489–96. doi: 10.1093/carcin/bgl012. [DOI] [PubMed] [Google Scholar]
- 5.Wang K, et al. A novel member of a zinc transporter family is defective in acrodermatitis enteropathica. Am J Hum Genet. 2002;71:66–73. doi: 10.1086/341125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Küry S, et al. Identification of SLC39A4, a gene involved in acrodermatitis enteropathica. Nat Genet. 2002;31:239–40. doi: 10.1038/ng913. [DOI] [PubMed] [Google Scholar]
- 7.Chowanadisai W, Lönnerdal B, Kelleher SL. Identification of a mutation in SLC30A2 (ZnT-2) in women with low milk zinc concentration that results in transient neonatal zinc deficiency. J Biol Chem. 2006;281:39699–707. doi: 10.1074/jbc.M605821200. [DOI] [PubMed] [Google Scholar]
- 8.Cipriano C, et al. Polymorphisms in MT1a gene coding region are associated with longevity in Italian Central female population. Biogerontology. 2006;7:357–65. doi: 10.1007/s10522-006-9050-x. [DOI] [PubMed] [Google Scholar]
- 9.Sensi SL, Jeng JM. Rethinking the excito-toxic ionic milieu: the emerging role of Zn2+ in ischemic neuronal injury. Curr Mol Med. 2004;4:87–111. doi: 10.2174/1566524043479211. [DOI] [PubMed] [Google Scholar]
- 10.Jacob C, Maret W, Vallee BL. Control of zinc transfer between thionein, metallothionein and zinc proteins. Proc Natl Acad Sci USA. 1998;95:3489–94. doi: 10.1073/pnas.95.7.3489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Pattanaik A, et al. Basal metallothionein in tumors: Widespread presence of apoprotein. J Inorg Biochem. 1994;54:91–105. doi: 10.1016/0162-0134(94)80023-5. [DOI] [PubMed] [Google Scholar]
- 12.Yang Y, Maret W, Vallee BL. Differential fluorescence labeling of cysteinyl clusters uncovers high tissue levels of thionein. Proc Natl Acad Sci USA. 2001;98:5556–9. doi: 10.1073/pnas.101123298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Krê¿el A, Maret W. Different redox states of metallothionein/thionein in biological tissue. Biochem J. 2007;402:551–8. doi: 10.1042/BJ20061044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Maret W, et al. Inhibitory sites in enzymes: Zinc removal and reactivation by thionein. Proc Natl Acad Sci USA. 1999;96:1936–40. doi: 10.1073/pnas.96.5.1936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Maret W, Vallee BL. Thiolate ligands in metallothionein confer redox activity on zinc clusters. Proc Natl Acad Sci USA. 1998;95:3478–82. doi: 10.1073/pnas.95.7.3478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Maret W. Zinc coordination environments in proteins as redox sensors and signal transducers. Antioxid Redox Signal. 2006;8:1419–41. doi: 10.1089/ars.2006.8.1419. [DOI] [PubMed] [Google Scholar]
- 17.Haase H, Maret W. A differential assay for the reduced and oxidized states of metallothionein and thionein. Anal Biochem. 2004;333:19–26. doi: 10.1016/j.ab.2004.04.039. [DOI] [PubMed] [Google Scholar]
- 18.Feng W, et al. Metallothionein disulfides are present in metallothionein-overexpressing transgenic mouse heart and increase under conditions of oxidative stress. J Biol Chem. 2006;281:681–7. doi: 10.1074/jbc.M506956200. [DOI] [PubMed] [Google Scholar]
- 19.Krê¿el A, Maret W. Zinc buffering capacity of a eukaryotic cell at physiological pZn. J Biol Inorg Chem. 2006;11:1049–62. doi: 10.1007/s00775-006-0150-5. [DOI] [PubMed] [Google Scholar]
- 20.Krężel A, Hao Q, Maret W. The zinc/thiolate redox biochemistry of metallothionein and the control of zinc ion fluctuations in cell signaling. Arch Biochem Biophys. 2007 doi: 10.1016/j.abb.2007.02.017. [DOI] [PubMed] [Google Scholar]
- 21.Maret W. Protein interface zinc sites: A role of zinc in the supramolecular assembly of proteins and in transient protein-protein interactions. In: Messerschmidt A, Bode W, Cygler M, editors. Handbook of Metalloproteins. Vol. 3. John Wiley; Chichester: 2004. pp. 432–41. [Google Scholar]
- 22.Kirlin WG, et al. Glutathione redox potential in response to differentiation and enzyme inducers. Free Radic Biol Med. 1999;27:1208–18. doi: 10.1016/s0891-5849(99)00145-8. [DOI] [PubMed] [Google Scholar]
- 23.Woo ES, Monks A, Watkins SC, et al. Diversity of metallothionein content and sub-cellular localization in the National Cancer Institute tumor panel. Cancer Chemother Pharmacol. 1997;4:61–8. doi: 10.1007/s002800050708. [DOI] [PubMed] [Google Scholar]
- 24.Haase H, et al. Flow cytometric measurements of labile zinc in peripheral blood mononuclear cells. Anal Biochem. 2006;352:222–30. doi: 10.1016/j.ab.2006.02.009. [DOI] [PubMed] [Google Scholar]
- 25.Maret W. Zinc and diabetes. BioMetals. 2005;18:293–4. doi: 10.1007/s10534-005-3707-9. [DOI] [PubMed] [Google Scholar]
- 26.Sladek R, et al. A genome-wide association study identifies novel risk loci for type 2 diabetes. Nature. 2007;445:881–5. doi: 10.1038/nature05616. [DOI] [PubMed] [Google Scholar]
- 27.Chimienti F, Favier A, Seve M. ZnT-8, a pancreatic beta-cell-specific transporter. BioMetals. 2005;18:313–7. doi: 10.1007/s10534-005-3687-9. [DOI] [PubMed] [Google Scholar]
- 28.Roussel A-M, et al. Antioxidant effects of zinc supplementation in Tunisians with type 2 diabetes mellitus. J Am Coll Nutr. 2003;22:316–1. doi: 10.1080/07315724.2003.10719310. [DOI] [PubMed] [Google Scholar]
- 29.Schott-Ohly P, et al. Prevention of spontaneous and experimentally induced diabetes in mice with zinc sulfate-enriched drinking water is associated with activation and reduction of NF-κB and AP-1 in islets, respectively. Exp Biol Med. 2004;229:1177–85. doi: 10.1177/153537020422901113. [DOI] [PubMed] [Google Scholar]
- 30.Adachi Y, et al. Oral administration of a zinc complex improves type 2 diabetes and metabolic syndromes. Biochem Biophys Res Commun. 2006;351:165–70. doi: 10.1016/j.bbrc.2006.10.014. [DOI] [PubMed] [Google Scholar]
- 31.Ayaz M, Turan B. Selenium prevents diabetes-induced alterations in [Zn2+]i and metallothionein level of rat heart via restoration of cell redox cycle. Am J Physiol Heart Circ Physiol. 2006;290:1071–80. doi: 10.1152/ajpheart.00754.2005. [DOI] [PubMed] [Google Scholar]
- 32.Chen Y, Maret W. Catalytic selenols couple the redox cycles of metallothionein and glutathione. Eur J Biochem. 2001;268:3346–53. doi: 10.1046/j.1432-1327.2001.02250.x. [DOI] [PubMed] [Google Scholar]
- 33.Wang J, et al. Cardiac metallothionein induction plays the major role in the prevention of diabetic cardiomyopathy by zinc supplementation. Circulation. 2006;113:544–54. doi: 10.1161/CIRCULATIONAHA.105.537894. [DOI] [PubMed] [Google Scholar]
- 34.Kang YJ. The antioxidant function of metallothionein in the heart. Proc Soc Exp Biol Med. 1999;222:263–73. doi: 10.1046/j.1525-1373.1999.d01-143.x. [DOI] [PubMed] [Google Scholar]
- 35.Barbato JC, et al. Targeting of metallothionein by L-homocysteine. A novel mechanism for disruption of zinc and redox homeostasis. Arterioscler Thromb Vasc Biol. 2007;27:49–54. doi: 10.1161/01.ATV.0000251536.49581.8a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Scheede-Bergdahl C, et al. Metallothionein-mediated antioxidant defense system and its response to exercise training are impaired in human type 2 diabetes. Diabetes. 2005;54:3089–94. doi: 10.2337/diabetes.54.11.3089. [DOI] [PubMed] [Google Scholar]
- 37.Beattie, et al. Obesity and hyperleptinemia in metallothionein (-I and –II) null mice. Proc Natl Acad Sci USA. 1998;95:358–63. doi: 10.1073/pnas.95.1.358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hao Q, Maret W. Aldehydes release zinc from proteins. A pathway from oxidative stress/lipid peroxidation to cellular functions of zinc. FEBS J. 2006;273:4300–10. doi: 10.1111/j.1742-4658.2006.05428.x. [DOI] [PubMed] [Google Scholar]
- 39.Haase H, Maret W. Intracellular zinc fluctuations modulate protein tyrosine phosphatase activity in insulin/insulin-like growth factor-1 signaling. Exp Cell Res. 2003;291:289–98. doi: 10.1016/s0014-4827(03)00406-3. [DOI] [PubMed] [Google Scholar]
- 40.Haase H, Maret W. Fluctuations of cellular, available zinc modulate phosphorylation signaling. J Trace Elem Med Biol. 2005;19:37–42. doi: 10.1016/j.jtemb.2005.02.004. [DOI] [PubMed] [Google Scholar]
- 41.Haase H, Maret W. Protein tyrosine phosphatases as targets of the combined insulinomimetic effects of zinc and oxidants. BioMetals. 2005;18:333–8. doi: 10.1007/s10534-005-3707-9. [DOI] [PubMed] [Google Scholar]
- 42.St. Croix CM, et al. Nitric oxide and zinc homeostasis in acute lung injury. Proc Am Thor Soc. 2005;2:236–42. doi: 10.1513/pats.200501-007AC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Frederickson CJ, Maret W, Cuajungco MP. Zinc and excitotoxic brain injury: A new model. Neuroscientist. 2004;10:18–25. doi: 10.1177/1073858403255840. [DOI] [PubMed] [Google Scholar]
- 44.Maret W, Sandstead HH. Zinc requirements and the risks and benefits of zinc supplementation. J Trace Elem Med Biol. 2006;20:3–18. doi: 10.1016/j.jtemb.2006.01.006. [DOI] [PubMed] [Google Scholar]
- 45.Kang YJ, Zhou Z. Zinc prevention and treatment of alcoholic liver disease. Mol Asp Med. 2005;26:391–404. doi: 10.1016/j.mam.2005.07.002. [DOI] [PubMed] [Google Scholar]