Abstract
Pax5-deficient progenitor B (pro-B) cells are thought to be severely defective for recombination of all immunoglobulin heavy chain (IgH) V gene segments, but the mechanism by which Pax5 regulates this process has not been defined. To address this issue, we have examined the assembly of the IgH locus in Pax5-deficient pro-B cells and find, unexpectedly, that 3′ IgH V gene segments, which lie closest to the D-J-Cμ region, recombine efficiently, but progressively more distal V gene segments recombine progressively less efficiently. Histone acetylation and germ-line transcription correlate strongly with an open or an accessible chromatin structure thought to be permissive for V(D)J recombination, and defects in recombination are typically accompanied by deficits in these processes. We were therefore surprised to observe that distal VH gene segments in Pax5−/− pro-B cells exhibit no defect in these measures of accessibility. The finding of transcribed, histone acetylated gene segments that fail to recombine suggests that a Pax5-dependent regulatory mechanism is required in addition to standard constraints of accessibility to control VH gene recombination.
Keywords: V(D)J recombination, Pax5, accessibility, lymphocyte development, locus control
During lymphocyte development, V(D)J recombination assembles the variable region of antigen receptor loci from individual V, D, and J gene segments. This reaction is initiated by binding of the RAG1 and RAG2 proteins to conserved recombination signal sequences (RSSs) that flank each of these gene segments. After binding to two RSS elements, the RAG1/RAG2 complex introduces DNA double-strand breaks between the RSSs and their flanking gene segments. Importantly, the two RSSs must first be brought into close proximity and assembled into a synaptic complex before cleavage can occur (Fugmann et al. 2000; Gellert 2002), ensuring that the recombining partners are juxtaposed before potentially dangerous DNA breaks are created. After DNA cleavage, the RAG proteins and DNA repair factors process and join the ends to complete the recombination reaction.
Temporal and developmental specificity of V(D)J recombination are achieved in part by regulating the accessibility of RSSs to the recombination machinery (Yancopoulos and Alt 1985; Hesslein and Schatz 2001). Although the underlying structural basis of accessibility has not been determined, V(D)J recombination of the antigen receptor loci is tightly correlated with locus changes such as nuclease sensitivity, germ-line (sterile) transcription, DNA demethylation, and histone acetylation (Krangel 2001). Cis-acting transcriptional elements found in the antigen receptor loci have been implicated in the regulation of gene rearrangement, but the mechanisms by which these elements control V(D)J recombination are unknown.
Bone marrow B-cell development has been divided into distinct stages based on cell size and surface markers by Hardy (Hardy et al. 1991) and Rolink and Melchers (Rolink et al. 1994). The earliest B lineage cells (Hardy fractions A and B or Rolink and Melcher pro/pre-B-I cells) express c-kit and B220 and actively undergo DH-to-JH recombination (in fraction B), the first step in assembly of a functional immunoglobulin heavy chain (IgH) gene. In addition, up to one-quarter of IgH alleles in these cells undergo V-to-DJ recombination (Ehlich et al. 1994; ten Boekel et al. 1995). In the next fraction (fraction C), c-kit is down-regulated, V-to-DJ recombination is completed, and cells with productive IgH rearrangements undergo extensive pre-B-cell receptor-directed proliferation and further maturation. Although IgH D-to-J recombination also occurs in thymocytes, VH-to-DJH recombination is B-cell-specific and is considered a hallmark of B-cell lineage commitment. Little is known concerning the cis-acting elements that regulate IgH V-to-DJ recombination except that deletion of the IgH intronic enhancer reduces its levels (Serwe and Sablitzky 1993; Sakai et al. 1999). Studies of other antigen receptor gene loci have implicated cis-acting elements within the V gene cluster in regulating V gene recombination (Hesslein and Schatz 2001), but their mechanism of action is not known.
Gene-targeting studies have identified numerous transcription factors required for B-cell development. Among these, PU.1, E2A-encoded proteins, and EBF have been shown to bind to cis-acting elements of the IgH locus and/or activate recombination of the DH-JH cluster when expressed in cell culture systems (Goebel et al. 2001; Hesslein and Schatz 2001), but as yet, none of these factors has been implicated in regulating VH-to-DJ recombination. In contrast, both the transcription factor BSAP (encoded by the Pax5 gene) and signaling through the interleukin 7 receptor (IL-7R) have been linked to IgH V-to-DJ recombination. In Pax5-deficient mice, bone marrow B-cell development is blocked in Hardy fraction B, and although the progenitor B (pro-B) cells perform D-to-JH rearrangement normally, they were reported to contain almost undetectable levels of V-to-DJH rearrangements for several different VH families (7183, Q52, and J558; Urbanek et al. 1994; Nutt et al. 1997).
A deficiency in IL-7R signaling results in normal D-to-JH recombination and an intriguing gradient effect on V-to-DJH recombination, in which progressively more 5′ VH gene families exhibit a progressively more severe recombination defect (Peschon et al. 1994; Corcoran et al. 1998). Defective 5′ VH recombination was mirrored by a defect in germ-line transcription of these VH gene segments (Corcoran et al. 1998). More recently, IL-7R signaling has been shown to enhance histone acetylation in the 5′ portion of the VH cluster (Chowdhury and Sen 2001), supporting the model that IL-7 plays a key role in controlling the accessibility of this portion of the locus (Corcoran et al. 1998).
We have analyzed IgH locus recombination in Pax5-deficient pro-B cells, and find that whereas D-to-J recombination is normal, VH gene usage exhibits a progressive 3′-to-5′ defect very similar to that reported for IL-7Rα-deficient mice. Recombination of proximal VH gene families (7183, Q52) occurs at levels approaching that seen in wild-type, but recombination of the more distal families (3609, J558) is strongly reduced. In striking contrast to previous findings in IL-7Rα-deficient mice, however, accessibility, as measured by sterile transcription and histone acetylation, of both 3′ and 5′ VH genes was equivalent in wild-type and Pax5−/− pro-B cells. Hence, 5′ VH genes are apparently accessible but recombine very inefficiently in Pax5−/− pro-B cells. Pax5 appears to play a role in regulating V(D)J recombination of the IgH locus at a level distinct from histone acetylation and germ-line transcription.
Results and Discussion
Normal recombination and sterile transcription of IgH D and J gene segments in Pax5−/− Pro-B cells
We isolated progenitor B cells from the bone marrow of 14- to 16-day-old Pax5-deficient mice by flow cytometry. Non-B-lineage cells were eliminated by negative sorting for the markers Mac-1, Gr-1, Thy-1.2, and Ter-119; and B220+ c-kit+ (fraction A/B) pro-B cells were positively sorted and isolated (see Materials and Methods). The same procedure was applied to bone marrow from age-matched C57BL/6 wild-type littermates to ensure that cell populations from equivalent developmental stages were compared (data not shown). After removal of non-B-lineage cells by negative sorting (as described above), the only B220+ cells in the bone marrow of Pax5-deficient mice were also c-kit+ (data not shown), consistent with a development block in Hardy fraction B as described previously (Nutt et al. 1997).
One of the earliest events in IgH locus activation is the appearance of the Iμ and μ0 germ-line transcripts in the D-J-Cμ portion of the gene (Fig. 1A). Levels of these transcripts were measured with a reverse transcriptase–polymerase chain reaction (RT–PCR) assay using RNA isolated from sorted wild-type and Pax5-deficient pro-B cells. The levels of these transcripts were not affected by the absence of Pax5 [Fig. 1B; detection of hypoxanthine phosphoribosyl transferase (HPRT) transcripts controls for the quality and amount of input cDNA in each sample]. Using a standard PCR assay, we also assessed D-to-JH recombination in DNA from the sorted pro-B cells. No differences in recombination levels were found in wild-type and Pax5−/− cells (Fig. 1C), consistent with previous findings (Nutt et al. 1997). Thus, loss of Pax5 does not appear to affect activation or recombination of the D-J-Cμ portion of the IgH locus.
Figure 1.
Transcription and recombination of the D-JH locus. (A) Schematic of the IgH D-J locus with gene segments represented by rectangles and the intronic enhancer represented by a black circle. There are 13 D and 4 J gene segments. The start sites and direction of the μ0 and Iμ sterile transcripts are represented as lines with arrows. Not to scale. (B) RT–PCR detection of μ0 (31 cycles), Iμ (28 cycles), and HPRT (30 cycles) transcripts. RNA from kidney (lanes 2,3), RAG1−/− B220+ cells (lanes 12,13), and total wild-type (WT) bone marrow (lanes 14,15) served as controls. The experimental RT reactions from Pax5−/− and WT bone marrow c-kit+ B220+ cells were serially diluted fivefold (lanes 5–7,9–11). (C) PCR detection of D-to-J recombination (36 cycles). DNA from HEK 293T cells was used as a negative control. Approximately 3000 cell equivalents of DNA from B220+ c-kit+ cells were used in lanes 6 and 10, respectively. Lanes 3–6 and 7–10 represent threefold serial dilutions.
Recombination of IgH V gene segments in Pax5−/− Pro-B cells
The murine IgH locus occupies ∼3 Mb (Chevillard et al. 2002), with the V gene segments organized into families (defined by levels of sequence similarity) at the 5′ end of the locus (Fig. 2A). We used standard PCR assays to measure VH-to-DJ recombination of different V gene families in DNA from sorted pro-B cells from wild-type and Pax5-deficient mice. Recombination of the J558 V gene family was severely reduced in Pax5-deficient cells but was easily detected in wild-type cells (Fig. 2B), consistent with the previous estimate of a >50-fold deficit of J558 VH gene rearrangements in Pax5−/− mice (Nutt et al. 1997). Surprisingly, we found that rearrangements of the V7183 gene family are abundant and readily detectable in Pax5-deficient pro-B cells (Fig. 2B). We then investigated recombination of three other VH gene families (Fig. 2A), and observed that as the distance of a VH gene family increases from the D-J gene cluster, recombination of that VH gene family progressively decreases. The results of four experiments (each from an independent preparation of sorted pro-B cells) are summarized in Figure 2C, with levels of recombination in Pax5-deficient cells displayed as a percentage of that observed in wild-type cells assayed in parallel. The failure of 5′ VH gene segments to rearrange is unlikely to be caused by defects in recombinase activity because D-to-J and V7183-to-DJ recombination occur at wild-type, or nearly wild-type, levels.
Figure 2.
Pax5-dependent VH-to-DJ recombination. (A) Schematic of the murine V gene cluster with the length of each V gene family approximately equal to the size of each gene family. Only the VH gene families assayed are depicted. Not to scale. (B) PCR detection of VH-to-DJ recombination (36–38 cycles). See legend for Figure 1C for additional details. (C) Quantitation of VH-to-DJ recombination in Pax5−/− pro-B cells for five different VH gene families from four independent preparations of sorted cells. Results are expressed as a percent of the recombination detected in wild-type pro-B cells.
The defect in VH gene recombination we observe for Pax5−/− mice is strikingly similar to that described for IL-7Rα-deficient mice (Corcoran et al. 1998). We therefore isolated DNA from B220+ c-kit+ bone marrow cells from IL-7Rα−/− mice and confirmed that such cells have a similar pattern of VH gene recombination to Pax5−/− pro-B cells (data not shown).
IgH gene expression in Pax5−/− pro-B cells
We determined whether rearranged IgH alleles are transcribed normally in Pax5−/− pro-B cells using RT–PCR (Fig. 3A). For both D-to-J and V7183-to-DJ rearranged alleles, transcript levels were quite similar in Pax5-deficient and wild-type pro-B cells, whereas VJ558-to-DJ transcripts were strongly reduced in the absence of Pax5 (Fig. 3B). Hence, transcript levels mirror rearrangement levels, indicating that the absence of Pax5 does not affect transcription of partially or fully recombined IgH alleles. The block in bone marrow B-cell development in Pax5−/− mice is therefore not caused by a defect in IgH recombination or expression, consistent with previous findings that an IgH transgene does not support developmental progression of Pax5−/− pro-B cells (Thevenin et al. 1998).
Figure 3.
Transcription of the rearranged IgH locus in Pax5−/− pro-B cells. (A) Schematic of transcripts of the rearranged IgH locus. Ovals represent promoters. Other symbols as described in the legend for Figure 1A. (B) RT–PCR detection of DJ (38 cycles) and VHDJ transcripts (J558, 35 cycles; 7183, 38 cycles). See the Figure 1B legend for additional details.
Sterile transcription and histone acetylation of IgH V gene segments in Pax5−/− pro-B cells
Recombination of endogenous antigen receptor loci correlates well with germ-line transcription and increased histone acetylation of the participating gene segments. For example, in B220+ CD43+ bone marrow cells from IL-7Rα-deficient mice, abundant germ-line transcripts and rearrangements are found for 7183 but not J558 V gene segments (Corcoran et al. 1998). Furthermore, IL-7R signals stimulate histone acetylation of the J558 but not the 7183 portion of the IgH locus (Chowdhury and Sen 2001). Based on this, we anticipated that gene segment accessibility would be reduced for the more 5′ V gene families in Pax5−/− pro-B cells.
RNA harvested from sorted Pax5−/− pro-B cells was examined for V7183 and VJ558 gene germ-line transcripts using RT–PCR. Germ-line V7183 transcripts were detected at approximately equal levels in the wild-type and Pax5−/− RNA samples, and surprisingly, the same was true for germ-line VJ558 transcripts, which were easily detected in both samples (Fig. 4A). Similar experiments were performed on RNA isolated from sorted CD43+ B220+ bone marrow cells from IL-7Rα−/− mice, and revealed that although germ-line V7183 transcripts were equally abundant in wild-type and IL-7Rα−/− cells, levels of germ-line VJ558 transcripts were much reduced in the IL-7Rα−/− sample (data not shown), confirming previous results (Corcoran et al. 1998).
Figure 4.
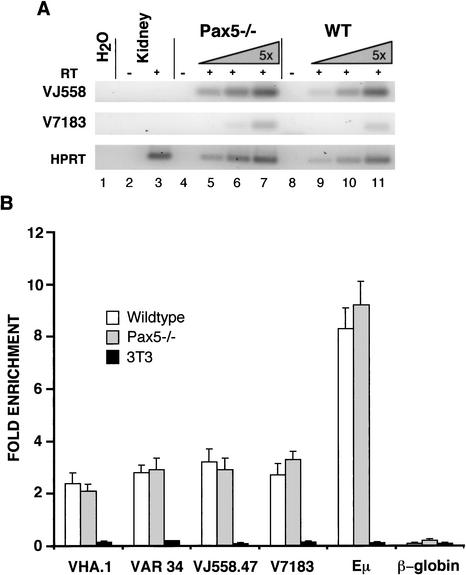
Accessibility of the IgH V gene cluster in Pax5−/− pro-B cells. (A) RT–PCR detection of germ-line transcripts of 7183 and J558 VH genes (38 cycles) in B220+ c-kit+ cells. See Figure 1B legend for additional details. (B) Chromatin immunoprecipitation assays were carried out with anti-acetylated histone H3 antibody using purified B220+ c-kit+ pro-B cells from Pax5−/− mice and wild-type littermates, and using NIH 3T3 cells. The Y-axis indicates the relative enrichment of target genes as the fold difference between the input fraction and the immunoprecipitated fraction, and the X-axis indicates the different gene segments analyzed. VHA.1 is one of the most 5′ VH genes; VAR34 is in the middle of the J558 gene cluster; and J558.47 is one of the most 3′ J558 genes. The μ enhancer region (μE) was used as a positive control for the pro-B cells and the β-globin gene as a negative control. NIH 3T3 cells served as a negative control for the VH locus. Results shown are an average of two to four independent experiments done in duplicate, with the error bars representing the standard error.
We also assessed VH gene family accessibility by measuring histone acetylation using chromatin immunoprecipitation (ChIP). Chromatin was prepared from sorted Pax5−/− and wild-type pro-B cells, and immunoprecipitated with antibodies to acetylated histone H3 as described previously (Chowdhury and Sen 2001). The resulting immunoprecipitated DNA was quantitated using real-time PCR, and the results are presented as the fold enrichment of the target sequences in immunoprecipitated as compared with input DNA (see Materials and Methods; Fig. 4B). V7183 and VJ558 gene sequences were 2.5- to 3-fold enriched in the immunoprecipitates, and no differences in levels of histone acetylation for either gene family could be detected between wild-type and Pax5−/− pro-B cells (Fig. 4B). The J558 VH gene family occupies a large portion of the VH region and of the IgH locus as a whole (Chevillard et al. 2002). We therefore examined representative V gene segments located at the 5′ (VHA.1), middle (VAR 34), and 3′ (VJ558.47) regions of the J558 V gene family cluster to show that all areas of this cluster were regulated in a similar fashion (Fig. 4B). Several controls were included to confirm the specificity of the assay (Fig. 4B). In NIH3T3 fibroblasts, in which VH gene segments are silent (and presumably not acetylated), VH sequences were dramatically depleted in the immunoprecipitated material. The same was true for the silent β-globin locus in immunoprecipitates from pro-B and fibroblast cells. Finally, sequences from the IgH intronic enhancer, which is active and acetylated in pro-B cells (Chowdhury and Sen 2001), were dramatically enriched in immunoprecipitates from pro-B cells but not fibroblasts. We conclude that both 7183 and J558 VH gene segments are comparably histone-acetylated in B220+ c-kit+ pro-B cells from Pax5−/− and wild-type bone marrow. Thus, two independent measurements of locus opening indicate that there is no reduction in accessibility accompanying the block in VJ558 gene segment recombination in Pax5−/− pro-B cells.
Models for Pax5 activity
Our results indicate that there is a discordance between germ-line transcription and histone acetylation on the one hand, and V(D)J recombination on the other hand, for the J558 V gene family (and perhaps other distal V gene families) in Pax5−/− pro-B cells. Hence, Pax5 appears to regulate IgH V gene rearrangement through a mechanism distinct from accessibility. There are some precedents for a discordance between germ-line transcription and recombination, but these studies examined either immortalized cell lines or transgenic substrates that might be developmentally disregulated and/or missing important regulatory regions (Okada et al. 1994; Alvarez et al. 1995; Angelin-Duclos and Calame 1998; Cherry and Baltimore 1999). In addition, there is an intriguing report of a Vβ gene segment that remains acetylated at a stage of T-cell development when TCR Vβ recombination is thought to be suppressed by allelic exclusion (Tripathi et al. 2002). Finally, a recent study using artificial TCRβ miniloci in a B-cell line revealed discrepancies between histone acetylation and recombination that depended on promoter placement (Sikes et al. 2002). The implications of these results for long-range recombination of endogenous antigen receptor gene loci remain unclear.
IL-7R signaling, by activating the transcription factor Stat5, has been implicated in regulating TCRγ locus accessibility (Ye et al. 1999, 2001), leading us to speculate previously that Stat5 and Pax5 play similar roles in regulating TCRγ and IgH locus recombination, respectively (Hesslein and Schatz 2001). The results presented here indicate that this idea was incorrect. Stat5 regulates recombination by controlling germ-line transcription and histone acetylation (Ye et al. 1999, 2001). In contrast, Pax5 appears to play a unique role in regulating 5′ VH gene rearrangement at a level distinct from accessibility.
Our results demonstrate that germ-line transcription and acetylation of 5′ VH genes can be activated in the absence of Pax5, whereas results from other studies strongly suggest that such activation depends on IL-7R signaling (Corcoran et al. 1998; Chowdhury and Sen 2001). These two genetic pathways could independently control distal VH gene recombination. Alternatively, IL-7R and Pax5 could be parts of the same pathway. For example, a subset of the IL-7-dependent activities could be controlled by Pax5, as V gene recombination is similar in mice deficient for either IL-7Rα or Pax5, and IL-7 signaling has been implicated in controlling Pax5 expression (Corcoran et al. 1998). However, analysis of mice deficient for the IL-7 signaling pathway has suggested a very early block in B-cell lymphopoiesis in adult mice (Carvalho et al. 2001; Miller et al. 2002). Thus, the inability to detect Pax5 expression in the bone marrow of these mice could be explained by the severe reduction in the number of pro-B cells. More study is needed to define the role of IL-7 and its receptor in B-cell lymphopoiesis, and to delineate the relationship of this signaling pathway to Pax5 expression and function.
Pax5−/− pro-B cells have the unique ability to differentiate into all other hematopoietic cell-types, excluding B cells (Nutt et al. 1999; Rolink et al. 1999). Because VH-to-DJ recombination is a hallmark of the B-cell lineage, our finding of V-to-DJ recombination in Pax5−/− pro-B cells raises the interesting question of whether pro-B cells with such rearrangements can develop into other lineages.
V(D)J recombination requires that two RSSs be brought into close proximity for both DNA cleavage and the subsequent joining reaction. We have proposed that nuclear organization and chromatin structure facilitate and/or regulate the regional localization of distant RSSs for subsequent synapsis (Hesslein and Schatz 2001). Recently, a series of provocative experiments have shown that nuclear location and a high state of compaction of the IgH locus correlate with the recombination status of the cell (Kosak et al. 2002). Importantly, in IL-7Rα−/− pro-B cells, the IgH locus remains at the nuclear periphery and fails to undergo compaction, which presumably interferes with long-range RSS interactions (Kosak et al. 2002). We speculate that Pax5 may be involved in such a mechanism and may help to bring VH and DJH gene segments into close proximity for subsequent recombination.
Materials and methods
Mice
Pax5−/−, RAG1−/−, and C57BL/6 mice were obtained from M. Busslinger, Richard Flavell, and the National Cancer Institute, respectively, and were housed in pathogen-free animal facilities at Yale University School of Medicine. At the time of harvest, mice were age matched with littermates, RAG1−/−, and/or C57BL/6 controls.
FACS sorting and analysis
Bone marrow cells were isolated and stained with the following antibodies from BD PharMingen: αc-kit-FITC (CD117), αB220-Cy-Chrome (CD45R), αMac-1-PE (CD11b), αTer-119-PE (LY-76), αGr-1-PE (LY-6G), and αThy1.2-PE (CD90). Streptavidin-FITC from Vector Laboratories was used to detect primary antibodies with a biotin conjugate. Cell sorting and analysis were performed on FACSVantage and FACSCalibur or FACScan flow cytometers, respectively, using CellQuest software.
Recombination analysis
DNA was isolated by placing sorted cells into agarose plugs followed by digestion with proteinase K as described (Papavasiliou and Schatz 2000). HEK 293T cells (a human embryonic kidney cell line) were placed in the plugs to serve as carrier. Aliquots of agarose plugs containing progenitor B-cell DNA were serially diluted into plugs containing HEK 293T DNA to maintain equal amounts of agarose and genomic DNA. All PCR assays for recombination were performed as described (Schlissel et al. 1991) using primers from Schlissel et al. (1991) and Angelin-Duclos and Calame (1998). PCR cycle numbers were adjusted to be in the linear range, based on serial dilution. PCR products were run on agarose gels, transferred, and Southern-blotted using oligonucleotide probes as described (Schlissel et al. 1991). Band intensities were quantitated with a PhosphorImager and analyzed using ImageQuant software (Molecular Dynamics). Experimental band intensities were normalized to the loading control, which was a PCR product from a nonrecombining locus (RAG2), detected by staining with SYBR green (Molecular Probes) and quantitated with a FluorImager (Molecular Dynamics).
RT–PCR
RNA was harvested from sorted cells using RNAzol B (Tel-Test) as directed by the manufacturer. RNA was incubated with DNase I at 37°C for 10 min followed by heat inactivation at 75°C for 10 min. Then 0.5 μg of RNA was subjected to reverse transcription using SuperScriptII (GIBCO-BRL) primed with random hexamers at 37°C for 2 h. The RT reaction was serially diluted and then subjected to PCR, adjusted to be in the linear range. PCR reactions for DJ, Iμ, μ0, and IgM transcripts were performed with the primers and protocol as described (Schlissel et al. 1991). The primer sequences for detecting V gene sterile transcripts are J558-5′, CGAGCTCTCCARCACAGCCTWCATGCARCTCARC; J558-3′, CTCAGTCYCAGKGCAGYTTSCTRCT; 7183-5′, CGGTACCAAGA ASAMCCTGTWCCTGCAAATGASC; and 7183-3′, GTCTCTCCGCG CCCCCTGCTGGTCC. All RT–PCR products were separated on agarose gels and stained using SYBR green.
Histone acetylation
Chromatin immunoprecipitations were performed as described (Chowdhury and Sen 2001) with the following modifications. Formaldehyde cross-linked chromatin prepared from ∼106 cells was incubated with 5 μg of anti-acetylated histone H3 antibody (against residues 1–21 of histone H3, diacetylated at K9 and K14; Upstate Biotechnology). Antibody-bound DNA was collected by adsorption to protein A-agarose, de-cross-linked, and purified.
Real-time PCR was performed using SYBR green and a Rotorgene 2000 Realtime Cycler (Corbett Research). The input DNA, which is purified from chromatin prior to immunoprecipitation, and immunoprecipitated (IP) DNA were quantified using picogreen fluorescence (Molecular Probes; excitation 480 nm, emission 520 nm). Real-time PCR was carried out using 0.5 ng of DNA at 95°C for 90 sec, followed by 45 cycles of 95°C for 15 sec, 60°C for 30 sec, and 72°C for 30 sec. The primer sequences are VHA.1-5′, CCTTCGCCCCAATCCACC; VHA.1-3′, CAAGTAACCCT CAAGAGAATGGAGACTC; VH7183-5′, CTCCAGAGACAATACCAA GAAGACCC; VH7183-3′, CCCCCTGCTGGTCCTAGATG; β-globin-5′, GCCTTGCCTGTTCCTGCTC; and β-globin-3′, CAGACCATAAA CTGTATTTTTCTTATTGAGCCC. All other VHJ588 and the Iμ intronic enhancer primers are as published (Chowdhury and Sen 2001). The endpoint used in real-time PCR quantification, Ct, is defined as the PCR cycle number that crosses an arbitrarily placed signal threshold and is a function of the amount of target DNA present in the starting material. For each experiment, the threshold was set at a point at which real-time PCR amplification was linear. The fold difference of a given target sequence was calculated by subtracting the Ct determined for the target sequence of the IP sample from the Ct determined for the target sequence of the input sample and taking the resulting power of 2, that is, 2−ΔCt (Litt et al. 2001). This gives a measure of the fold enrichment of the sequence of interest in the immunoprecipitate compared with the total sample before immunoprecipitation (input).
Acknowledgments
We thank Meinrad Busslinger and Richard Flavell for the generous gifts of Pax5−/− and RAG1−/− mice, respectively. We thank Claire Wickens and Shoaming Zhou for assistance in mantaining the mouse colonies and Tom Taylor and Gouzel Tokmoulina for assistance with flow cytometry. We are grateful to Paul Kincade, Patrick Fields, Isabelle Villey, Sebastian Fugmann, Alfred Lee, Chia-Lun Tsai, Nina Papavasiliou, and Thomas Welte for helpful suggestions and discussions. D.G.T.H. was supported by Training Grant AI07019 from the National Institutes of Health. This work was supported by grants AI32524 to D.G.S., GM40924 to A.L.M.B., and GM38925 and AI51480 to R.S. from the National Institutes of Health. D.G.S. is an investigator of the Howard Hughes Medical Institute.
The publication costs of this article were defrayed in part by payment of page charges. This article must therefore be hereby marked “advertisement” in accordance with 18 USC section 1734 solely to indicate this fact.
Footnotes
E-MAIL david.schatz@yale.edu; FAX (203) 737-1764.
Article and publication are at http://www.genesdev.org/cgi/doi/10.1101/gad.1031403.
References
- Alvarez JD, Anderson SJ, Loh DY. V(D)J recombination and allelic exclusion of a TCR β-chain minilocus occurs in the absence of a functional promoter. J Immunol. 1995;155:1191–1202. [PubMed] [Google Scholar]
- Angelin-Duclos C, Calame K. Evidence that immunoglobulin VH-DJ recombination does not require germ line transcription of the recombining variable gene segment. Mol Cell Biol. 1998;18:6253–6264. doi: 10.1128/mcb.18.11.6253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carvalho TL, Mota-Santos T, Cumano A, Demengeot J, Vieira P. Arrested B lymphopoiesis and persistence of activated B cells in adult interleukin 7−/− mice. J Exp Med. 2001;194:1141–1150. doi: 10.1084/jem.194.8.1141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cherry SR, Baltimore D. Chromatin remodeling directly activates V(D)J recombination. Proc Natl Acad Sci. 1999;96:10788–10793. doi: 10.1073/pnas.96.19.10788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chevillard C, Ozaki J, Herring CD, Riblet R. A three-megabase yeast artificial chromosome contig spanning the C57BL mouse Igh locus. J Immunol. 2002;168:5659–5666. doi: 10.4049/jimmunol.168.11.5659. [DOI] [PubMed] [Google Scholar]
- Chowdhury D, Sen R. Stepwise activation of the immunoglobulin μ heavy chain gene locus. EMBO J. 2001;20:6394–6403. doi: 10.1093/emboj/20.22.6394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corcoran AE, Riddell A, Krooshoop D, Venkitaraman AR. Impaired immunoglobulin gene rearrangement in mice lacking the IL-7 receptor. Nature. 1998;391:904–907. doi: 10.1038/36122. [DOI] [PubMed] [Google Scholar]
- Ehlich A, Martin V, Muller W, Rajewsky K. Analysis of the B-cell progenitor compartment at the level of single cells. Curr Biol. 1994;4:573–583. doi: 10.1016/s0960-9822(00)00129-9. [DOI] [PubMed] [Google Scholar]
- Fugmann SD, Lee AI, Shockett PE, Villey IJ, Schatz DG. The RAG proteins and V(D)J recombination: Complexes, ends, and transposition. Annu Rev Immunol. 2000;18:495–527. doi: 10.1146/annurev.immunol.18.1.495. [DOI] [PubMed] [Google Scholar]
- Gellert M. V(D)J recombination: Rag proteins, repair factors, and regulation. Annu Rev Biochem. 2002;71:101–132. doi: 10.1146/annurev.biochem.71.090501.150203. [DOI] [PubMed] [Google Scholar]
- Goebel P, Janney N, Valenzuela JR, Romanow WJ, Murre C, Feeney AJ. Localized gene-specific induction of accessibility to V(D)J recombination induced by E2A and early B cell factor in nonlymphoid cells. J Exp Med. 2001;194:645–656. doi: 10.1084/jem.194.5.645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hardy R, Carmack CE, Shinton SA, Kemp JD, Hayakawa K. Resolution and characterization of pro-B and pre-pro-B cell stages in normal mouse bone marrow. J Exp Med. 1991;173:1213–1225. doi: 10.1084/jem.173.5.1213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hesslein DG, Schatz DG. Factors and forces controlling V(D)J recombination. Adv Immunol. 2001;78:169–232. doi: 10.1016/s0065-2776(01)78004-2. [DOI] [PubMed] [Google Scholar]
- Kosak ST, Skok JA, Medina KL, Riblet R, Le Beau MM, Fisher AG, Singh H. Subnuclear compartmentalization of immunoglobulin loci during lymphocyte development. Science. 2002;296:158–162. doi: 10.1126/science.1068768. [DOI] [PubMed] [Google Scholar]
- Krangel MS. V(D)J recombination becomes accessible. J Exp Med. 2001;193:F27–F30. doi: 10.1084/jem.193.7.f27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Litt MD, Simpson M, Recillas-Targa F, Prioleau MN, Felsenfeld G. Transitions in histone acetylation reveal boundaries of three separately regulated neighboring loci. EMBO J. 2001;20:2224–2235. doi: 10.1093/emboj/20.9.2224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller JP, Izon D, DeMuth W, Gerstein R, Bhandoola A, Allman D. The earliest step in B lineage differentiation from common lymphoid progenitors is critically dependent upon interleukin 7. J Exp Med. 2002;196:705–711. doi: 10.1084/jem.20020784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nutt SL, Urbanek P, Rolink A, Busslinger M. Essential functions of PAX5 (BSAP) in pro-B cell development—Difference between fetal and adult B lymphopoiesis and reduced V-to-DJ recombination at the IgH locus. Genes & Dev. 1997;11:476–491. doi: 10.1101/gad.11.4.476. [DOI] [PubMed] [Google Scholar]
- Nutt SL, Heavey B, Rolink AG, Busslinger M. Commitment to the B-lymphoid lineage depends on the transcription factor Pax5. Nature. 1999;401:556–562. doi: 10.1038/44076. [DOI] [PubMed] [Google Scholar]
- Okada A, Mendelsohn M, Alt F. Differential activation of transcription versus recombination of transgenic T cell receptor β variable region gene segments in B and T lineage cells. J Exp Med. 1994;180:261–272. doi: 10.1084/jem.180.1.261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Papavasiliou FN, Schatz DG. Cell-cycle-regulated DNA double-stranded breaks in somatic hypermutation of immunoglobulin genes. Nature. 2000;408:216–221. doi: 10.1038/35041599. [DOI] [PubMed] [Google Scholar]
- Peschon JJ, Morrissey PJ, Grabstein KH, Ramsdell FJ, Maraskovsky E, Gliniak BC, Park LS, Ziegler SF, Williams DE, Ware CB, et al. Early lymphocyte expansion is severely impaired in interleukin 7 receptor-deficient mice. J Exp Med. 1994;180:1955–1960. doi: 10.1084/jem.180.5.1955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rolink A, Grawunder U, Winkler TH, Karasuyama H, Melchers F. IL-2 receptor α chain (CD25, TAC) expression defines a crucial stage in pre-B cell development. Int Immunol. 1994;6:1257–1264. doi: 10.1093/intimm/6.8.1257. [DOI] [PubMed] [Google Scholar]
- Rolink AG, Nutt SL, Melchers F, Busslinger M. Long-term in vivo reconstitution of T-cell development by Pax5-deficient B-cell progenitors. Nature. 1999;401:603–606. doi: 10.1038/44164. [DOI] [PubMed] [Google Scholar]
- Sakai E, Bottaro A, Davidson L, Sleckman BP, Alt FW. Recombination and transcription of the endogenous Ig heavy chain locus is effected by the Ig heavy chain intronic enhancer core region in the absence of the matrix attachment regions. Proc Natl Acad Sci. 1999;96:1526–1531. doi: 10.1073/pnas.96.4.1526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlissel MS, Corcoran LM, Baltimore D. Virus-transformed pre-B cells show ordered activation but not inactivation of immunoglobulin gene rearrangement and transcription. J Exp Med. 1991;173:711–720. doi: 10.1084/jem.173.3.711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Serwe M, Sablitzky F. V(D)J recombination in B-cells is impaired but not blocked by targeted deletion of the immunoglobulin heavy chain intron enhancer. EMBO J. 1993;12:2321–2327. doi: 10.1002/j.1460-2075.1993.tb05886.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sikes ML, Meade A, Tripathi R, Krangel MS, Oltz EM. Regulation of V(D)J recombination: A dominant role for promoter positioning in gene segment accessibility. Proc Natl Acad Sci. 2002;99:12309–12314. doi: 10.1073/pnas.182166699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ten Boekel E, Melchers F, Rolink A. The status of Ig loci rearrangements in single cells from different stages of B cell development. Int Immunol. 1995;7:1013–1019. doi: 10.1093/intimm/7.6.1013. [DOI] [PubMed] [Google Scholar]
- Thevenin C, Nutt SL, Busslinger M. Early function of Pax5 (BSAP) before the pre-B cell receptor stage of B lymphopoiesis. J Exp Med. 1998;188:735–744. doi: 10.1084/jem.188.4.735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tripathi R, Jackson A, Krangel MS. A change in the structure of Vβ chromatin associated with TCR β allelic exclusion. J Immunol. 2002;168:2316–2324. doi: 10.4049/jimmunol.168.5.2316. [DOI] [PubMed] [Google Scholar]
- Urbanek P, Wang ZQ, Fetka I, Wagner EF, Busslinger M. Complete block of early B cell differentiation and altered patterning of the posterior midbrain in mice lacking Pax5/BSAP. Cell. 1994;79:901–912. doi: 10.1016/0092-8674(94)90079-5. [DOI] [PubMed] [Google Scholar]
- Yancopoulos GD, Alt FW. Developmentally controlled and tissue-specific expression of unrearranged VH gene segments. Cell. 1985;40:271–281. [PubMed] [Google Scholar]
- Ye SK, Maki K, Kitamura T, Sunaga S, Akashi K, Domen J, Weissman IL, Honjo T, Ikuta K. Induction of germline transcription in the TCR γ locus by Stat5: Implications for accessibility control by the IL-7 receptor. Immunity. 1999;11:213–223. doi: 10.1016/s1074-7613(00)80096-5. [DOI] [PubMed] [Google Scholar]
- Ye SK, Agata Y, Lee HC, Kurooka H, Kitamura T, Shimizu A, Honjo T, Ikuta K. The IL-7 receptor controls the accessibility of the TCR γ locus by Stat5 and histone acetylation. Immunity. 2001;15:813–823. doi: 10.1016/s1074-7613(01)00230-8. [DOI] [PubMed] [Google Scholar]