Abstract
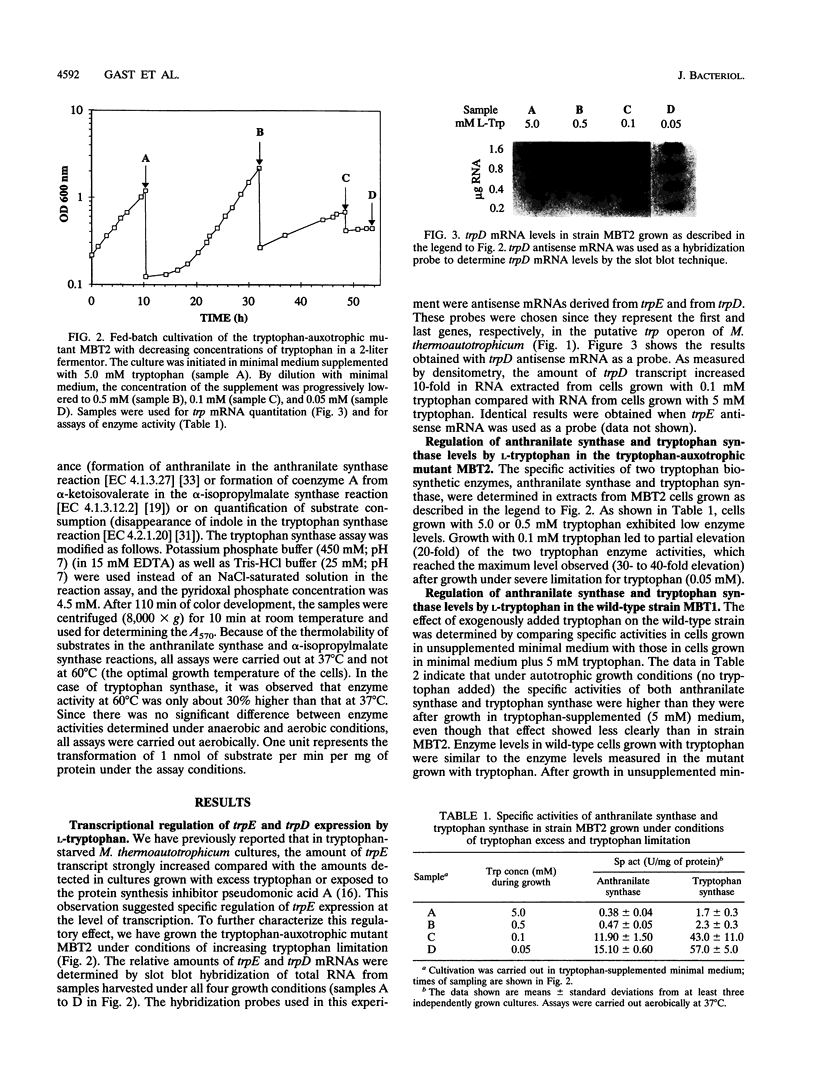
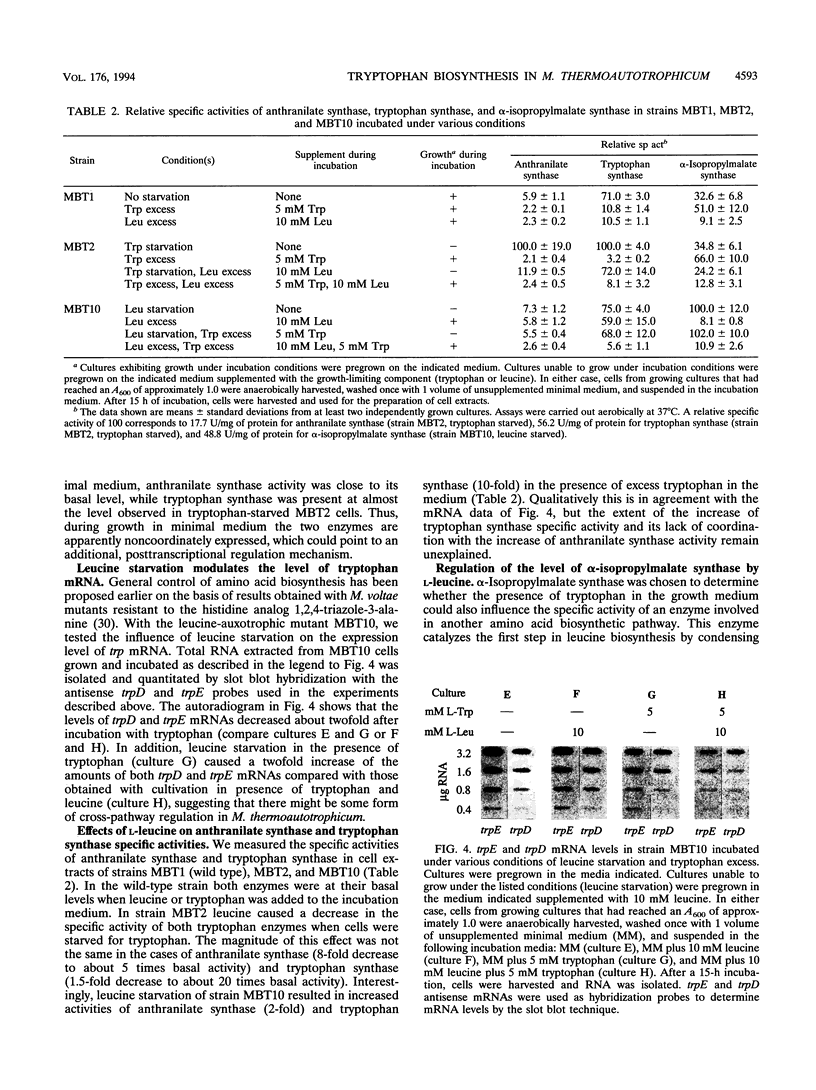
A tryptophan-auxotrophic mutant of the archaeon Methanobacterium thermoautotrophicum Marburg was grown with growth-promoting and growth-limiting concentrations of tryptophan. The specific activities of anthranilate synthase (TrpEG) and tryptophan synthase (TrpB) increased 30- to 40-fold in tryptophan-starved cells. Levels of trpE-specific and trpD-specific mRNAs (transcripts of the first and the last genes, respectively, of the M. thermoautotrophicum Marburg trp gene cluster) increased about 10-fold upon starvation for tryptophan. Thus, the expression of the trp genes appears to be regulated primarily at the level of transcription. These data support transcription of trp genes as an operon and support a regulatory model involving a repressor. Anthranilate synthase was feedback inhibited by L-tryptophan, with a Ki of 3.0 microM. In a leucine-auxotrophic mutant starved for L-leucine, the level of alpha-isopropylmalate synthase (LeuA) was 10-fold higher than in cells grown with L-leucine. In addition to the finding of specific regulation of gene expression by the end products of their respective pathways, it was found that the levels of anthranilate synthase and alpha-isopropylmalate synthase were reduced upon growth in the presence of amino acids of other families, such as L-alanine, L-proline, or L-arginine. Conversely, starvation for tryptophan caused a slight elevation of alpha-isopropylmalate synthase and starvation for leucine caused a significant increase of anthranilate synthase and tryptophan synthase specific activities. The latter effect was also observed at the level of trp-specific mRNA and is reminiscent of general amino acid control.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Beauclerk A. A., Hummel H., Holmes D. J., Böck A., Cundliffe E. Studies of the GTPase domain of archaebacterial ribosomes. Eur J Biochem. 1985 Sep 2;151(2):245–255. doi: 10.1111/j.1432-1033.1985.tb09095.x. [DOI] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1006/abio.1976.9999. [DOI] [PubMed] [Google Scholar]
- Braus G. H. Aromatic amino acid biosynthesis in the yeast Saccharomyces cerevisiae: a model system for the regulation of a eukaryotic biosynthetic pathway. Microbiol Rev. 1991 Sep;55(3):349–370. doi: 10.1128/mr.55.3.349-370.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caligiuri M. G., Bauerle R. Identification of amino acid residues involved in feedback regulation of the anthranilate synthase complex from Salmonella typhimurium. Evidence for an amino-terminal regulatory site. J Biol Chem. 1991 May 5;266(13):8328–8335. [PubMed] [Google Scholar]
- Chang M., Hadero A., Crawford I. P. Sequence of the Pseudomonas aeruginosa trpI activator gene and relatedness of trpI to other procaryotic regulatory genes. J Bacteriol. 1989 Jan;171(1):172–183. doi: 10.1128/jb.171.1.172-183.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cimmino C., Scoarughi G. L., Donini P. Stringency and relaxation among the halobacteria. J Bacteriol. 1993 Oct;175(20):6659–6662. doi: 10.1128/jb.175.20.6659-6662.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crawford I. P. Evolution of a biosynthetic pathway: the tryptophan paradigm. Annu Rev Microbiol. 1989;43:567–600. doi: 10.1146/annurev.mi.43.100189.003031. [DOI] [PubMed] [Google Scholar]
- Fuchs G., Stupperich E., Thauer R. K. Acetate assimilation and the synthesis of alanine, aspartate and glutamate in Methanobacterium thermoautotrophicum. Arch Microbiol. 1978 Apr 27;117(1):61–66. doi: 10.1007/BF00689352. [DOI] [PubMed] [Google Scholar]
- Graf R., Mehmann B., Braus G. H. Analysis of feedback-resistant anthranilate synthases from Saccharomyces cerevisiae. J Bacteriol. 1993 Feb;175(4):1061–1068. doi: 10.1128/jb.175.4.1061-1068.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanahan D. Studies on transformation of Escherichia coli with plasmids. J Mol Biol. 1983 Jun 5;166(4):557–580. doi: 10.1016/s0022-2836(83)80284-8. [DOI] [PubMed] [Google Scholar]
- Hinnebusch A. G. Mechanisms of gene regulation in the general control of amino acid biosynthesis in Saccharomyces cerevisiae. Microbiol Rev. 1988 Jun;52(2):248–273. doi: 10.1128/mr.52.2.248-273.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jarrell K. F., Faguy D., Hebert A. M., Kalmokoff M. L. A general method of isolating high molecular weight DNA from methanogenic archaea (archaebacteria). Can J Microbiol. 1992 Jan;38(1):65–68. doi: 10.1139/m92-010. [DOI] [PubMed] [Google Scholar]
- Jenal U., Rechsteiner T., Tan P. Y., Bühlmann E., Meile L., Leisinger T. Isoleucyl-tRNA synthetase of Methanobacterium thermoautotrophicum Marburg. Cloning of the gene, nucleotide sequence, and localization of a base change conferring resistance to pseudomonic acid. J Biol Chem. 1991 Jun 5;266(16):10570–10577. [PubMed] [Google Scholar]
- Jenal U., Thurner C., Leisinger T. Transcription of the ileS operon in the archaeon Methanobacterium thermoautotrophicum Marburg. J Bacteriol. 1993 Sep;175(18):5945–5952. doi: 10.1128/jb.175.18.5945-5952.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones W. J., Nagle D. P., Jr, Whitman W. B. Methanogens and the diversity of archaebacteria. Microbiol Rev. 1987 Mar;51(1):135–177. doi: 10.1128/mr.51.1.135-177.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kohlhaw G., Leary T. R., Umbarger H. E. Alpha-isopropylmalate synthase from Salmonella typhimurium. Purification and properties. J Biol Chem. 1969 Apr 25;244(8):2218–2225. [PubMed] [Google Scholar]
- Lam W. L., Logan S. M., Doolittle W. F. Genes for tryptophan biosynthesis in the halophilic archaebacterium Haloferax volcanii: the trpDFEG cluster. J Bacteriol. 1992 Mar;174(5):1694–1697. doi: 10.1128/jb.174.5.1694-1697.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meile L., Abendschein P., Leisinger T. Transduction in the archaebacterium Methanobacterium thermoautotrophicum Marburg. J Bacteriol. 1990 Jun;172(6):3507–3508. doi: 10.1128/jb.172.6.3507-3508.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meile L., Stettler R., Banholzer R., Kotik M., Leisinger T. Tryptophan gene cluster of Methanobacterium thermoautotrophicum Marburg: molecular cloning and nucleotide sequence of a putative trpEGCFBAD operon. J Bacteriol. 1991 Aug;173(16):5017–5023. doi: 10.1128/jb.173.16.5017-5023.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melton D. A., Krieg P. A., Rebagliati M. R., Maniatis T., Zinn K., Green M. R. Efficient in vitro synthesis of biologically active RNA and RNA hybridization probes from plasmids containing a bacteriophage SP6 promoter. Nucleic Acids Res. 1984 Sep 25;12(18):7035–7056. doi: 10.1093/nar/12.18.7035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sibold L., Henriquet M. Cloning of the trp genes from the archaebacterium Methanococcus voltae: nucleotide sequence of the trpBA genes. Mol Gen Genet. 1988 Nov;214(3):439–450. doi: 10.1007/BF00330478. [DOI] [PubMed] [Google Scholar]
- Sment K. A., Konisky J. Excretion of amino acids by 1,2,4-triazole-3-alanine-resistant mutants of Methanococcus voltae. Appl Environ Microbiol. 1989 May;55(5):1295–1297. doi: 10.1128/aem.55.5.1295-1297.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stettler R., Leisinger T. Physical map of the Methanobacterium thermoautotrophicum Marburg chromosome. J Bacteriol. 1992 Nov;174(22):7227–7234. doi: 10.1128/jb.174.22.7227-7234.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tutino M. L., Scarano G., Marino G., Sannia G., Cubellis M. V. Tryptophan biosynthesis genes trpEGC in the thermoacidophilic archaebacterium Sulfolobus solfataricus. J Bacteriol. 1993 Jan;175(1):299–302. doi: 10.1128/jb.175.1.299-302.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]




