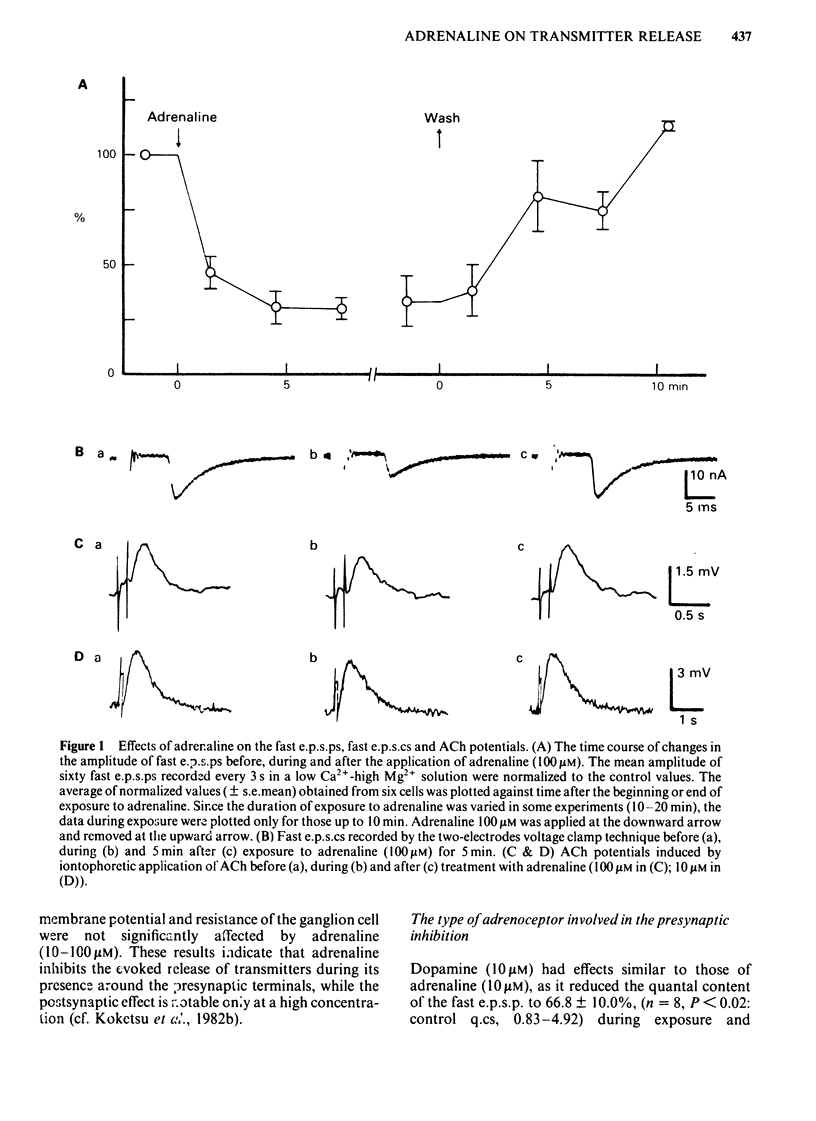
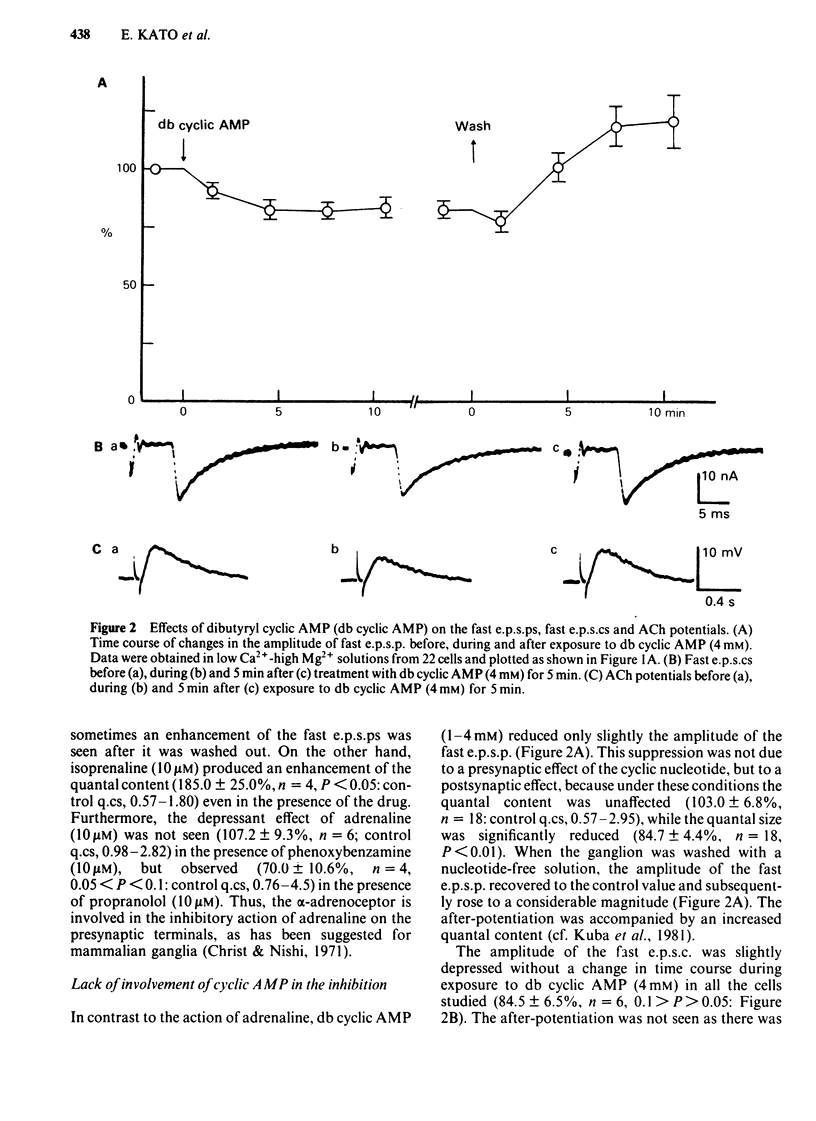
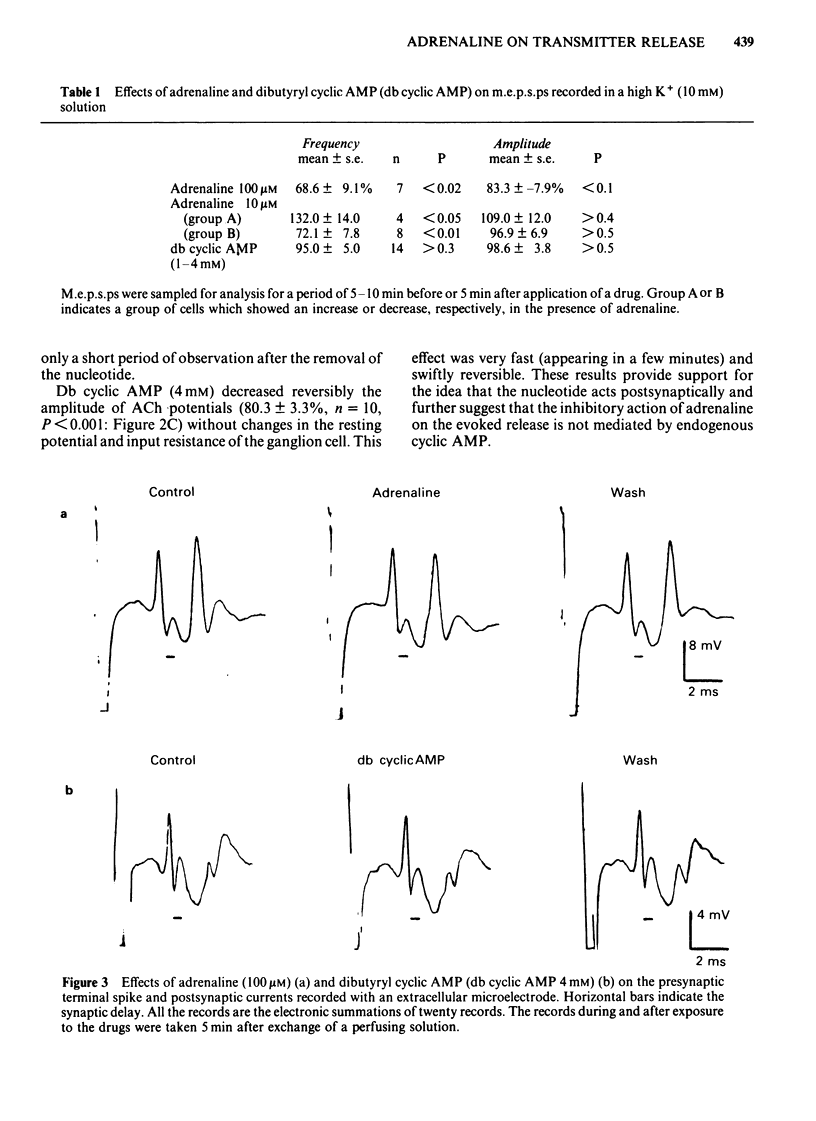
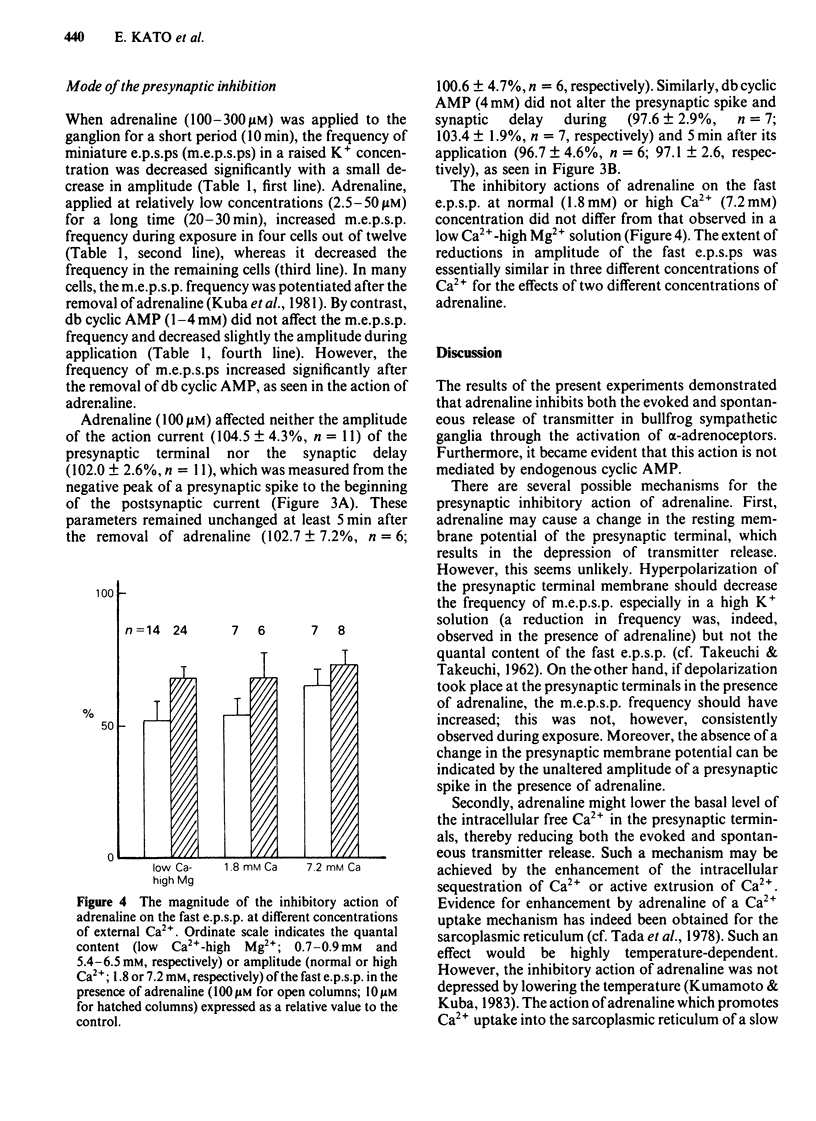
Abstract
The effects of adrenaline and dibutyryl adenosine 3':5' - cyclic monophosphate (db cyclic AMP) on nicotinic transmission in bullfrog sympathetic ganglia were compared by use of an intracellular recording technique. The evoked release of transmitter, acetylcholine (ACh), was decreased in the presence of adrenaline (10-100 microM), while the postsynaptic sensitivity to ACh was unchanged (10 microM adrenaline) or slightly reduced (100 microM). Transmitter release was similarly inhibited by dopamine (10 microM), but not by isoprenaline (10 microM). The inhibitory action of adrenaline on transmitter release was blocked by phenoxybenzamine but not by propranolol. The inhibition of transmitter release was independent of the external calcium concentration. The evoked release of transmitter and the electrical properties of the postsynaptic membrane were unchanged during exposure to db cyclic AMP (1-4 mM), while the postsynaptic sensitivity to ACh was slightly but significantly depressed. The spontaneous release of transmitter in a high K+ (10 mM) solution was decreased in the presence of adrenaline (100-300 microM), but unchanged with db cyclic AMP (4 mM). In contrast to the effects during exposure, both the evoked and spontaneous release of transmitter were enhanced after the removal of adrenaline or db cyclic AMP. Neither adrenaline (100 microM) nor db cyclic AMP (4 mM) affected the presynaptic spike and synaptic delay. It is concluded that adrenaline mainly inhibits the release of ACh from the presynaptic terminals through its alpha-action, while db cyclic AMP reduces slightly the postsynaptic sensitivity to ACh and that both agents facilitate transmitter release when they are removed from the presynaptic terminals. It is further suggested that the inhibitory action of adrenaline is independent of endogenous cyclic AMP and calcium ions.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Blaustein M. P., Ratzlaff R. W., Schweitzer E. S. Calcium buffering in presynaptic nerve terminals. II. Kinetic properties of the nonmitochondrial Ca sequestration mechanism. J Gen Physiol. 1978 Jul;72(1):43–66. doi: 10.1085/jgp.72.1.43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Christ D. D., Nishi S. Effects of adrenaline on nerve terminals in the superior cervical ganglion of the rabbit. Br J Pharmacol. 1971 Feb;41(2):331–338. doi: 10.1111/j.1476-5381.1971.tb08033.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Christ D. D., Nishi S. Site of adrenaline blockade in the superior cervical ganglion of the rabbit. J Physiol. 1971 Feb;213(1):107–117. doi: 10.1113/jphysiol.1971.sp009371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DEL CASTILLO J., KATZ B. Quantal components of the end-plate potential. J Physiol. 1954 Jun 28;124(3):560–573. doi: 10.1113/jphysiol.1954.sp005129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dun N., Nishi S. Effects of dopamine on the superior cervical ganglion of the rabbit. J Physiol. 1974 May;239(1):155–164. doi: 10.1113/jphysiol.1974.sp010560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fabiato A. Calcium release in skinned cardiac cells: variations with species, tissues, and development. Fed Proc. 1982 May;41(7):2238–2244. [PubMed] [Google Scholar]
- Ginsborg B. L. On the presynaptic acetylcholine receptors in sympathetic ganglia of the frog. J Physiol. 1971 Jul;216(1):237–246. doi: 10.1113/jphysiol.1971.sp009521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hagiwara S., Byerly L. Calcium channel. Annu Rev Neurosci. 1981;4:69–125. doi: 10.1146/annurev.ne.04.030181.000441. [DOI] [PubMed] [Google Scholar]
- Hagiwara S., Takahashi K. Surface density of calcium ions and calcium spikes in the barnacle muscle fiber membrane. J Gen Physiol. 1967 Jan;50(3):583–601. doi: 10.1085/jgp.50.3.583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horn J. P., McAfee D. A. Alpha-drenergic inhibition of calcium-dependent potentials in rat sympathetic neurones. J Physiol. 1980 Apr;301:191–204. doi: 10.1113/jphysiol.1980.sp013198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jenkinson D. H., Stamenović B. A., Whitaker B. D. The effect of noradrenaline on the end-plate potential in twitch fibres of the frog. J Physiol. 1968 Apr;195(3):743–754. doi: 10.1113/jphysiol.1968.sp008486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kato E., Kuba K. Inhibition of transmitter release in bullfrog sympathetic ganglia induced by gamma-aminobutyric acid. J Physiol. 1980 Jan;298:271–283. doi: 10.1113/jphysiol.1980.sp013080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koketsu K., Akasu T., Miyagawa M., Hirai K. Modulation of nicotinic transmission by biogenic amines in bullfrog sympathetic ganglia. J Auton Nerv Syst. 1982 Jul;6(1):47–53. doi: 10.1016/0165-1838(82)90021-2. [DOI] [PubMed] [Google Scholar]
- Koketsu K., Miyagawa M., Akasu T. Catecholamine modulates nicotinic ACh-receptor sensitivity. Brain Res. 1982 Mar 25;236(2):487–491. doi: 10.1016/0006-8993(82)90732-6. [DOI] [PubMed] [Google Scholar]
- Kuba K. Effects of catecholamines on the neuromuscular junction in the rat diaphragm. J Physiol. 1970 Dec;211(3):551–570. doi: 10.1113/jphysiol.1970.sp009293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuba K., Kato E., Kumamoto E., Koketsu K., Hirai K. Sustained potentiation of transmitter release by adrenaline and dibutyryl cyclic AMP in sympathetic ganglia. Nature. 1981 Jun 25;291(5817):654–656. doi: 10.1038/291654a0. [DOI] [PubMed] [Google Scholar]
- Kuba K., Koketsu K. Synaptic events in sympathetic ganglia. Prog Neurobiol. 1978;11(2):77–169. doi: 10.1016/0301-0082(78)90010-2. [DOI] [PubMed] [Google Scholar]
- Kuba K., Nishi S. Characteristics of fast excitatory postsynaptic current in bullfrog sympathetic ganglion cells. Effects of membrane potential, temperature and Ca ions. Pflugers Arch. 1979 Jan 31;378(3):205–212. doi: 10.1007/BF00592737. [DOI] [PubMed] [Google Scholar]
- Kuba K., Tomita T. Noradrenaline action on nerve terminal in the rat diaphragm. J Physiol. 1971 Aug;217(1):19–31. doi: 10.1113/jphysiol.1971.sp009557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumamoto E., Kuba K. Independence of presynaptic bimodal actions of adrenaline in sympathetic ganglia. Brain Res. 1983 Apr 18;265(2):344–347. doi: 10.1016/0006-8993(83)90354-2. [DOI] [PubMed] [Google Scholar]
- Minota S., Koketsu K. Effects of adrenaline on the action potential of sympathetic ganglion cells in bullfrogs. Jpn J Physiol. 1977;27(3):353–366. doi: 10.2170/jjphysiol.27.353. [DOI] [PubMed] [Google Scholar]
- Miyamota M. D., Breckenridge B. M. A cyclic adenosine monophosphate link in the catecholamine enhancement of transmitter release at the neuromuscular junction. J Gen Physiol. 1974 May;63(5):609–624. doi: 10.1085/jgp.63.5.609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- NISHI S., KOKETSU K. Electrical properties and activities of single sympathetic neurons in frogs. J Cell Comp Physiol. 1960 Feb;55:15–30. doi: 10.1002/jcp.1030550104. [DOI] [PubMed] [Google Scholar]
- Nachshen D. A., Drapeau P. A buffering model for calcium-dependent neurotransmitter release. Biophys J. 1982 May;38(2):205–208. doi: 10.1016/S0006-3495(82)84548-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- TAKEUCHI A., TAKEUCHI N. Electrical changes in pre- and postsynaptic axons of the giant synapse of Loligo. J Gen Physiol. 1962 Jul;45:1181–1193. doi: 10.1085/jgp.45.6.1181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tada M., Yamamoto T., Tonomura Y. Molecular mechanism of active calcium transport by sarcoplasmic reticulum. Physiol Rev. 1978 Jan;58(1):1–79. doi: 10.1152/physrev.1978.58.1.1. [DOI] [PubMed] [Google Scholar]


