Abstract
Cells with a verapamil-sensitive ability to efflux Hoechst 33342 (termed side population [SP] cells) have been identified in adult marrow from several species including humans and in several tissues from adult mice. In mice, the SP phenotype appears to be a common feature of stem cells, but human SP cells have been less well characterized. We show here, for the first time to our knowledge, that SP cells are present in the second-trimester human fetal liver. They include all of the transplantable human hematopoietic stem cell activity detectable in NOD/SCID mice and also certain other, more differentiated hematopoietic cell types. Notably, the stem cell activity was confined to the CD34+CD38– SP+ population, and isolation of these cells gave an approximately tenfold enrichment of transplantable stem cells. This subset was not, however, coenriched in hematopoietic progenitors detectable by either short- or long-term in vitro assays, indicating most of these to be distinct from transplantable stem cells. These findings suggest that the SP phenotype is an important and distinguishing property of human hematopoietic stem cells and that early in ontogeny they express CD34.
Introduction
Identification of molecular markers that characterize transplantable human cells with stem cell potential and that allow their selective purification remains an important prerequisite for many applied as well as basic studies. This has been recently underscored by reports that hematopoietic stem cells (HSCs) can generate other types of tissue progeny to correct damage to organs including the liver, skeletal muscle, and heart (1–3). The first and still most widely used marker for purifying human HSCs is the CD34 sialomucin antigen (4). Highly enriched populations of human cells expressing CD34+ reconstitute multilineage hematopoiesis not only in sublethally irradiated immunodeficient mice (5, 6), but also in human recipients (7–10). Initially, it was assumed that in humans, only cells expressing CD34 would display HSC activity, as most types of primitive hematopoietic cells appeared to be exclusively CD34+ (11, 12), and the frequency of CD34+ cells is now commonly used to anticipate the adequacy of clinical hematopoietic cell transplants. Recently, however, several groups including ours have provided evidence of various types of primitive human hematopoietic progenitors that do not express detectable levels of CD34 (13–17). On the other hand, studies in mice indicate that CD34 expression may be subject to modulation on HSCs, as it is expressed on murine HSCs during development (18, 19) but not in the adult (20), unless homeostasis is perturbed (21).
In 1996, Goodell et al. reported a new method of obtaining enriched populations of HSCs from adult mouse bone marrow in a single step (22). This procedure exploits the ability of HSCs to efflux the fluorescent dye Hoechst 33342, which, like the activity of P-glycoprotein (encoded by the MDR gene), is verapamil-sensitive (23). The Hoechst 33342lo cells thus isolated were called side population (SP) cells (22) and, not surprisingly, were found to have the same CD34– Sca-1+ lineage marker-negative (lin–) phenotype independently established for HSCs in adult mouse bone marrow (20, 24, 25). SP cells have since been identified in adult bone marrow from several species (including humans) (26). To date, a description of the functional activities of human SP cells in normal individuals has been limited to a single in vitro study of cord blood cells that showed a partitioning of the myeloid and NK (lymphoid) progenitor activities in the CD34+ and CD34– subsets of the SP cells (27). We now demonstrate that SP cells are already present in the second-trimester human fetal liver. There they constitute a phenotypically and functionally heterogeneous population but, importantly, contain all of the cells able to repopulate nonobese diabetic/LtSz-scid/scid (NOD/SCID) mice and only a minor subset of progenitors detectable by available in vitro assays. The present studies also provide the first evidence to our knowledge that transplantable HSCs in human fetal liver, like those present in the early mouse embryo (18), are exclusively CD34+, suggesting a possible parallelism in the regulation of this surface marker on human HSCs.
Methods
Human cells.
Human fetal livers were obtained from 14- to 21-week-old aborted fetuses according to approved institutional procedures. Single-cell suspensions were prepared using dispase (Sigma Chemical Co., St. Louis, Missouri, USA) (28), and the cells were then usually cryopreserved at –135°C. For initial studies of lineage (lin) marker expression on fetal liver SP cells, low-density (<1.077 g/ml) cells were first isolated by centrifugation on Ficoll-Hypaque (Pharmacia Biotech A/B, Uppsala, Sweden). However, for later progenitor assays, this first step was omitted, which enabled higher yields of SP cells to be obtained after the removal of lin+ cells (data not shown).
Animals.
NOD/SCID mice (29) were bred and maintained under defined sterile conditions in our animal facility. At 6–8 weeks of age, they were irradiated with 350 cGy of 137Cs γ-rays and then injected intravenously the next day with human test cells (as indicated) plus 106 irradiated (1,500 cGy) human bone marrow carrier cells.
Cell purification.
Human fetal liver cells were thawed and depleted of lin+ cells using a StemSep column (StemCell Technologies, Vancouver, British Columbia, Canada) according to the supplier’s directions after staining the cells with the following cocktail of mAb’s: CD2, CD3, CD14, CD16, CD19, CD24, CD56, CD66b, and glycophorin A. The lin– cells collected after passage of the cells through the column were kept overnight at 4°C in Iscove’s medium supplemented with a serum substitute (BIT 9500; StemCell Technologies) plus 40 μg/ml of human low-density lipoprotein (Sigma Chemical Co.) and were then incubated with 5 μg/ml Hoechst 33342 (Sigma Chemical Co.) at 37°C for 90 minutes at 106 cells/ml. A parallel aliquot was stained with the Hoechst dye in the presence of 50 μM verapamil (Sigma Chemical Co.). Cells were then washed, resuspended in ice-cold HBSS containing 2% FBS (HF; StemCell Technologies) plus 5% pooled normal human serum (Sigma Chemical Co.) (HF/5% HS) at 107 cells/ml, and then stained for 30 minutes on ice with various mAb’s: FITC-conjugated CD34 (8G12; kindly provided by P.M. Lansdorp, Terry Fox Laboratory, Vancouver, British Columbia, Canada; ref. 30), CD3 (Leu-4), CD7 (Leu-9), CD14 (MφP9), CD16 (NKP15), and CD66b (G10F5), and PE-conjugated CD2 (Leu-5b), CD19 (Leu-12), CD33 (P67.6), CD34 (anti–HPCA-2), CD36 (CB38), CD38 (HB7), CD45 (HI30), CD56 (Leu-19), all from Becton Dickinson Immunocytometry Systems (San Jose, California, USA), and CD71 and glycophorin A (OKT9 and 10F7, respectively, kindly provided by P.M. Lansdorp) either for analysis or sorting. Stained cells were washed once in HF and then again in HF with 2 μg/ml propidium iodide (PI; Sigma Chemical Co.). Cells were analyzed on a FACSort using CellQuest software and were sorted on a dual laser FACStar+ (all, Becton Dickinson Immunocytometry Systems). Hoechst 33342 was excited at 360 nm, and fluorescence emission was detected using 424/BP44 and 660/BP20 optical filters (Omega Optical Inc., Brattleboro, Vermont, USA) and a 640-nm short-pass dichroic mirror (Omega Optical Inc.) to separate emission wavelengths.
Progenitor assays.
Cells were plated in methylcellulose medium (MethoCult H4230; StemCell Technologies) with 3 U/ml human erythropoietin (StemCell Technologies), 50 ng/ml of recombinant human Steel factor (SF; prepared in the Terry Fox Laboratory), and 20 ng/ml each of human IL-3 (Novartis, Basel, Switzerland), human IL-6 (Cangene, Mississauga, Ontario, Canada.), human G-CSF (StemCell Technologies), and human GM-CSF (Novartis) to assess their direct granulopoietic, erythropoietic, and multilineage colony-forming cell (CFC) content, as described elsewhere (31). For long-term culture-initiating cell (LTC-IC) assays, test cells were first cocultured for 6 weeks at 37°C with preestablished, irradiated feeder layers of mouse fibroblasts engineered to produce human IL-3, G-CSF, and SF (31). At the end of 6 weeks, a single-cell suspension was prepared from the whole culture and assayed for its CFC content.
Measurements of human cells able to repopulate NOD/SCID mice (competitive repopulating units or CRUs).
Bone marrow cells were harvested from the femurs of primary and secondary mice at the times indicated. The bone marrow cells were suspended in HF/5% HS and then first incubated for 10 minutes at 4°C with HF/5% HS supplemented with 3 mg/ml of an antimouse Fc receptor antibody (2.4G2) to block Fc receptors and prevent subsequent nonspecific antibody binding (32). The cells were then stained for 30 minutes at 4°C with antihuman CD34-FITC, CD19-PE, and CD20-PE (Becton Dickinson Immunocytometry Systems) or antihuman CD71-PE, CD45-PE, CD15-FITC, and CD66b-FITC as described previously (33, 34). Various phenotypes within the viable (PI–) fraction were then determined. A mouse was considered positive for a given population if, and only if, more than five of 20,000 events analyzed had a fluorescence greater than that exhibited by at least 99.99% of the cells stained with an irrelevant isotype-matched control antibody labeled with the corresponding fluorochrome. A mouse was considered positive for human CRU engraftment if, and only if, it was positive for both human lymphoid and human myeloid cells (33, 35). CRU frequencies were calculated with the L-calc software program (StemCell Technologies) that uses Poisson statistics and the method of maximum likelihood.
Statistical analysis.
Significant differences (P < 0.05) were established using the Student’s t test.
Results
SP cells in human fetal liver are phenotypically heterogeneous.
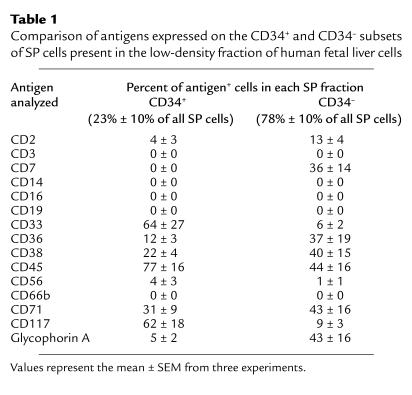
Initial Hoechst 33342 staining of low-density human fetal liver cells showed the SP fraction to be small (0.14% ± 0.07%; n = 3), but readily detectable. Further analyses revealed these fetal liver SP cells to contain a heterogeneous mixture of phenotypes (Table 1). About 80% were CD34–, most of which expressed at least one of the various lin markers evaluated. The most prominent of these appeared to be members of the erythroid and lymphoid lineages based on their expression of glycophorin A (a marker of relatively mature erythroid precursors) or CD7 (a marker typical of some lymphoid precursors). The high numbers of SP cells found to express CD36 and CD71 (markers also expressed at elevated levels on early erythroid cells) and CD38 (present on many early lymphoid cells) are consistent with the data for glycophorin A and CD7. Within the remaining approximately 20% of low-density SP fetal liver cells that were CD34+, the most prominent lin+ subset expressed the granulopoietic marker CD33. However, 5% of the CD34+ SP cells were glycophorin A+, indicating the presence of erythroid precursors within this subset also. CD45, a marker expressed on all hematopoietic cells except for terminally differentiating erythroblasts, was found on 77% and 44% of the CD34+ and CD34– subsets, respectively, of the low-density SP fetal liver cells (Table 1). The higher numbers of CD45– cells in the CD34– fraction of SP cells are consistent with these being late erythroid cells, as also indicated by removal of many of the CD45– cells in the lin+ cell depletion step (data not shown).
Table 1.
Comparison of antigens expressed on the CD34+ and CD34– subsets of SP cells present in the low-density fraction of human fetal liver cells
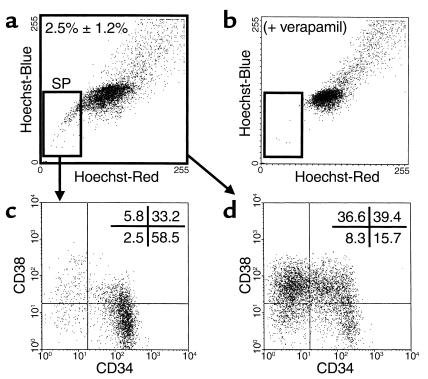
Initial studies indicated that centrifugation on Ficoll-Hypaque reduced the yield of SP cells approximately twofold (data not shown). Therefore, subsequent studies to determine which progenitors express an SP phenotype were undertaken with lin– fetal liver cells (cells not expressing CD2, 3, 14, 16, 19, 24, 56, 66b, or glycophorin A) that had not been subjected to a density centrifugation procedure. Removal of these lin+ cells increased the proportion of SP cells in the population thus obtained to 2.5% ± 1.2% (n = 9; Figure 1, a and b), as expected from the phenotyping analyses (Table 1). The proportion of lin– cells that expressed CD34 was also markedly and selectively increased to 78% ± 6% (vs. 23% ± 13%; Table 1) as was the subset of lin– SP cells that possessed the CD34+CD38– phenotype associated with transplantable stem cell activity in cord blood (5, 33) (from 18% ± 11% [Table 1] to 62% ± 6% [n = 9; see also Figure 1, c and d]).
Figure 1.
Representative FACS dot plot showing the presence and phenotypes of SP cells in the lin– fraction of human fetal liver cells. Cells were depleted of lin+ cells and stained with Hoechst 33342 and antibodies to CD34 and CD38 as described in Methods. (a) Small gated cell population identifies the SP cells (2.5% ± 1.2% of the total lin– fetal liver population; n = 9) that disappear in the presence of verapamil (b). The distribution of cells according to their expression of CD34 and CD38 in the SP (c) and total (d) fractions of the lin– fetal liver population analyzed here is also shown.
Very few progenitors in human fetal liver that are detected by in vitro assays have a lin– CD34+ CD38– SP phenotype.
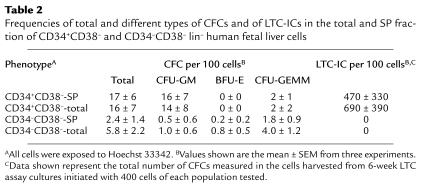
Table 2 shows the numbers of progenitors detectable by both short-term clonogenic and long-term stromal-based coculture assays detected per hundred total or SP+ CD34+CD38– lin– fetal liver cells. Interestingly, both the frequency of clonogenic progenitors (CFCs) in the SP fraction of the CD34+CD38– lin– fetal liver cells and the types of colonies they produced were indistinguishable (P > 0.05) from those generated by the total CD34+CD38– lin– cell population (> 80% granulopoietic, with the remainder being multilineage). The frequency of the more primitive LTC-ICs in the total and SP subset of CD34+CD38– lin– fetal liver cells was also similar (∼7% and 10%, respectively, assuming both will have produced, on average, 72 clonogenic progenitors per LTC-IC at the 6-week time point when their CFC output activity was assessed; ref. 36). Thus, isolation of the SP subset within the CD34+CD38– lin– population did not enrich further for LTC-ICs. Nevertheless, the frequency of LTC-ICs obtained here for the CD34+CD38– lin– cells was seven to tenfold higher than the values that we have previously reported for the total (low-density) lin– fetal liver cell population (36).
Table 2.
Frequencies of total and different types of CFCs and of LTC-ICs in the total and SP fraction of CD34+CD38– and CD34–CD38– lin– human fetal liver cells
All of the transplantable human hematopoietic stem cells present in human fetal liver have a lin– SP+ CD34+ CD38– phenotype.
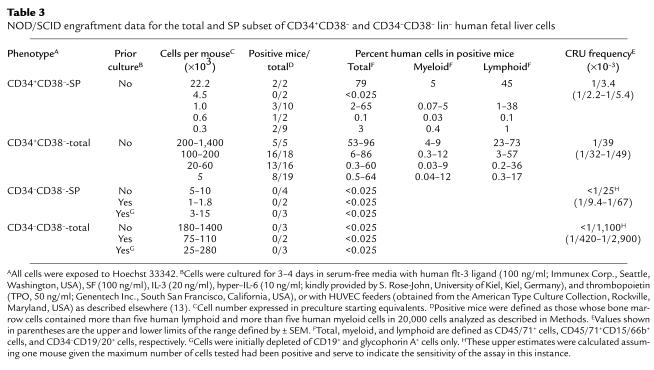
We next compared the frequency of human cells in the SP versus the total lin– CD34+CD38– fraction that were able to regenerate both lymphoid and myeloid progeny in transplanted NOD/SCID mice. Varying numbers of total CD34+CD38– lin– fetal liver cells and the SP subset of these were injected into sublethally irradiated NOD/SCID mice in a total of 13 and four experiments, respectively. Six to 24 weeks later, the mice were assessed for the presence of human lymphoid (B-lineage markers) and human myeloid (granulopoietic) cells in their bone marrow as described in Methods. The results are shown in Table 3. In contrast to the results for the progenitors measured by both short- and long-term in vitro assays, the CRU activity was exclusively associated with the SP subset of the CD34+CD38– lin– population. Most of the positive mice were highly repopulated with both lymphoid and myeloid human cells; however, some mice were not. Accordingly, Poisson statistics could be used to calculate the frequencies of transplantable human stem cells (CRUs) in the original populations transplanted, and these showed an approximately tenfold enrichment in the frequency of CRU in the SP subset of the CD34+CD38– population. However, the absolute value obtained here for the frequency of CRU in the CD34+CD38– lin– fetal liver cell population is somewhat lower than what we anticipated based on our previous data for lin– fetal liver cells (35). This was found in separate studies to be due in part to an approximately threefold loss of CRU from the toxic effect of exposing the cells to Hoechst 33342, and in part to the lack of removal in the present study of cells with a density >1.077 g/ml (which increased CRU yields but also decreased their final frequency in the lin– CD34+CD38– SP cells another twofold; data not shown).
Table 3.
NOD/SCID engraftment data for the total and SP subset of CD34+CD38– and CD34–CD38– lin– human fetal liver cells
Transplantation of NOD/SCID mice with cells from the non-SP fraction of the CD34+CD38– lin– population failed to detect CRUs in this fraction at a frequency that could not be explained by contamination by SP cells, i.e., in a total of six experiments, in which six mice were injected with 15 × 103 to 285 ×103 non-SP CD34+CD38– lin– fetal liver cells, only a single positive mouse was detected when these were analyzed 12 weeks later, and this was the mouse that received the largest number of non-SP cells.
To assess the in vivo self-renewal activity of the CRUs isolated in the SP fraction of the CD34+CD38– lin– fetal liver cells, bone marrow cells from both femurs and tibias of three highly engrafted primary recipients (containing >30% human cells) from two different experiments were harvested 7–8 weeks after transplant and injected individually into three secondary NOD/SCID mice. In each of these secondary mice, a large number of human lymphoid and myeloid progeny was detected 6–8 weeks after transplant (CD45/71+: 86%, 90%, 76%; CD45/71+CD15/66b+: 5%, 6%, 7%; and CD34–CD19/20+: 42%, 68%, 59%). Results for one of these is shown in Figure 2. These findings are similar to what we have seen previously in secondary recipients of cells from mice originally transplanted with unseparated lin– human fetal liver cells (35).
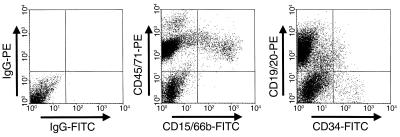
Figure 2.
Representative FACS dot plot showing human multilineage engraftment in a secondary NOD/SCID recipient of initially transplanted SP fetal liver cells. The bone marrow cells were removed from both femurs and both tibias of a first NOD/SCID mouse transplanted 7 weeks previously with 22,000 SP CD34+CD38– lin– human fetal liver cells, and all of these cells (∼25% of the total bone marrow of that mouse [ref. 48], of which 76% were human) were then transplanted into a second NOD/SCID mouse. The analysis shown is for the bone marrow cells removed from the secondary mouse another 6 weeks later.
Human fetal liver contains some primitive progenitors that do not express CD34, but these are not detectable in long-term culture assays and do not engraft NOD/SCID mice.
As illustrated in Figure 1, only a small proportion of the SP lin– cells were CD34–CD38– (6.4% ± 1.4%; n = 9), and these comprised only 0.8% ± 0.4% (n = 9) of the total CD34–CD38– lin– population. The results of assays for CFCs and LTC-ICs in the total and SP subset of the CD34–CD38– lin– population are given in Table 2, and for CRUs in Table 3. The frequency of CFCs in the total CD34–CD38– lin– population was two- to threefold lower than in the total CD34+CD38– lin– population. The CD34–CD38– CFCs were also made up of a significantly different (P < 0.05) spectrum of CFC types; i.e. they contained a much higher proportion of multilineage CFCs and many fewer granulopoietic CFCs. Moreover, although isolation of the SP subset of CD34–CD38– cells resulted in a two to threefold reduction in CFC frequency, the distribution of progenitor subtypes in the SP fraction of the CD34–CD38– population did not change. Neither LTC-ICs nor CRUs could be detected in either the total or the SP subset of CD34–CD38– lin– fetal liver cells in any of three to four experiments, even when these were assayed at the same or much larger numbers than were sufficient to detect such activities in the corresponding subsets of CD34+ cells. Failure to detect CRU activity in any of the CD34–CD38– cells also included experiments in which these were first cultured for 3–4 days with human growth factors or on top of human umbilical vein endothelial cell (HUVEC) feeders under conditions reported to stimulate the generation of NOD/SCID repopulating cells from CD34– cord blood cells (13).
Discussion
We show here that cells able to efflux Hoechst 33342 and thus designated as SP cells are readily detectable in human fetal liver and comprise a heterogeneous mixture of cell types as indicated by their surface marker profile, as previously established for cells in other hematopoietic tissues of human or nonhuman origin (26, 27). That the SP phenotype has been reported to be broadly associated with in vivo HSC activity (22, 26) and with stem cell activity for other tissues (2, 37) prompted us to investigate its association with human HSC properties. For this, we measured the ability of the various fractions of human fetal liver cells isolated to engraft sublethally irradiated NOD/SCID mice with both lymphoid and myeloid progeny when assayed at limiting doses (i.e., doses of cells that give no human cell engraftment of some mice). We also examined their in vivo self-renewal activity using secondary transplants (6, 34, 35). Our results show that all of the transplantable HSCs present in the CD34+CD38– lin– population of the second-trimester human fetal liver are contained within the SP fraction. This provides the first evidence of a human stem cell population that shares this phenotype and predicts the future extension of these observations to other human tissues both during development and in the adult.
We focused here on the CD34+CD38– lin– subset because previous studies had indicated most of the human HSCs able to engraft NOD/SCID mice have this phenotype (5, 33). We found that only about 10% of the CD34+CD38– lin– cells in human fetal liver have an SP phenotype, and isolation of these SP cells resulted in a corresponding enrichment in the transplantable HSC content of this subset. Accordingly, it would be expected that this additional purification step would allow a more highly enriched population of transplantable HSCs to be isolated than has been previously attainable, which would constitute a major advance for future biologic and molecular analyses. Unfortunately this expectation was not realized here because of the inherent toxicity of exposing the cells to Hoechst 33342 and the elimination of an initial low-density centrifugation step in order to improve the final yield of SP cells. Recently, it was reported that the SP phenotype is due to the selective expression on stem cells of ABC-G2/BCRP (38, 39), which, like the MDR/P-glycoprotein (23), is a verapamil-sensitive member of the ABC transporter family (40–42) and is likewise expressed on the cell surface (43, 44). Thus, in the future it may be possible for SP cells to be isolated using nontoxic, specific antibody-based procedures.
Our findings with CD34+CD38– lin– SP fetal liver cells provide new evidence that cells detected using in vitro stromal-based cultures, even after prolonged periods in the presence of increased level of human growth factors (31, 45), cannot necessarily be equated with cells able to serially engraft NOD/SCID mice or fetal sheep (35, 46). This was demonstrated in the present studies by the nonselective partitioning of LTC-ICs seen between the SP and non-SP subsets of the CD34+CD38– lin– cells that contrasted with the complete segregation of the transplantable HSC activity in the SP fraction. Such findings suggest that less than 10% of the LTC-ICs identified in human fetal liver with the 6-week assay used here (31) would have transplantable HSC activity. These findings underscore the need for similar comparative studies of SP and non-SP subsets from other sources of human cells with in vitro and in vivo hematopoietic activity.
In marked contrast to the results for CD34+CD38– lin– fetal liver cells, we did not detect any in vivo reconstituting activity in the corresponding CD34– population of human fetal liver cells. Although this finding appears to contrast with previously reported results for adult and neonatal sources of transplantable human hematopoietic cells (13, 14, 16, 17), this apparent discrepancy may be due to the different ontological status of the cells being compared, as CD34 expression on transplantable murine HSCs has been found to change during development (19). In addition, our findings do not exclude the possibility that CD34– progenitors restricted to the T cell and/or NK lineages may exist in human fetal liver since differentiation of human cells along these lineages has not been seen in NOD/SCID mice. The observation that all transplantable cells in human fetal liver that have B-lineage and myelopoietic potential are CD34+ is thus consistent with the concept that CD34 expression may be altered in HSCs according to their activation status, as first indicated by the finding that CD34 expression in CD34– lin– hematopoietic cells can be upregulated by exposure to certain feeders or growth factors in vitro, or after their stimulation in vivo (13, 15, 17, 21, 26). We have recently demonstrated differences in the level of expression of several other genes in freshly isolated CD34+CD38– lin– cells from human fetal liver compared with cord blood or adult bone marrow (47). Moreover, some of these changes in gene expression are also triggered by growth factor activation of adult human bone marrow CD34+CD38– lin– cells even before their exit from G0. Taken together, our present findings predict that the SP phenotype will be an important marker of human cells with stem cell potential and may be more consistent than expression of CD34.
Acknowledgments
We thank G. Thornbury and R. Zapf for assistance in cell sorting; members of the Stem Cell Assay Service for performing some of the cell separations; A. Ahamed for help in preparing the manuscript; and P. Lansdorp, S. Rose-John, Cangene, Genentech, Immunex, Novartis, and StemCell for valuable gifts of reagents. This work was supported by an NIH grant (NHLBI PO1 55435) and by the National Cancer Institute of Canada (NCIC) with funds from the Terry Fox Run and the Canadian Cancer Society. N. Uchida was supported by funds from Kirin Brewery Inc. and a Canadian Institutes of Health Research Postdoctoral Fellowship. C. Eaves was a Terry Fox Cancer Research Scientist of the NCIC.
References
- 1.Lagasse E, et al. Purified hematopoietic stem cells can differentiate into hepatocytes in vivo. Nat Med. 2000;6:1229–1234. doi: 10.1038/81326. [DOI] [PubMed] [Google Scholar]
- 2.Gussoni E, et al. Dystrophin expression in the mdx mouse restored by stem cell transplantation. Nature. 1999;401:390–394. doi: 10.1038/43919. [DOI] [PubMed] [Google Scholar]
- 3.Orlic D, et al. Bone marrow cells regenerate infarcted myocardium. Nature. 2001;410:701–705. doi: 10.1038/35070587. [DOI] [PubMed] [Google Scholar]
- 4.Krause DS, et al. Characterization of murine CD34, a marker for hematopoietic progenitor and stem cells. Blood. 1994;84:691–701. [PubMed] [Google Scholar]
- 5.Bhatia M, Wang JCY, Kapp U, Bonnet D, Dick JE. Purification of primitive human hematopoietic cells capable of repopulating immune-deficient mice. Proc Natl Acad Sci USA. 1997;94:5320–5325. doi: 10.1073/pnas.94.10.5320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hogan CJ, et al. Engraftment and development of human CD34+-enriched cells from umbilical cord blood in NOD/LtSz-scid/scid mice. Blood. 1997;90:85–96. [PubMed] [Google Scholar]
- 7.Bensinger WI, et al. Transplantation of allogeneic CD34+ peripheral blood stem cells in patients with advanced hematologic malignancy. Blood. 1996;88:4132–4138. [PubMed] [Google Scholar]
- 8.Dunbar CE, et al. Retrovirally marked CD34-enriched peripheral blood and bone marrow cells contribute to long-term engraftment after autologous transplantation. Blood. 1995;85:3048–3057. [PubMed] [Google Scholar]
- 9.Negrin RS, et al. Transplantation of highly purified CD34+ Thy-1+ hematopoietic stem cells in patients with metastatic breast cancer. Biol Blood Marrow Transplant. 2000;6:262–271. doi: 10.1016/s1083-8791(00)70008-5. [DOI] [PubMed] [Google Scholar]
- 10.Michallet M, et al. Transplantation with selected autologous peripheral blood CD34+ Thy1+ hematopoietic stem cells (HSCs) in multiple myeloma: impact of HSC dose on engraftment, safety, and immune reconstitution. Exp Hematol. 2000;28:858–870. doi: 10.1016/s0301-472x(00)00169-7. [DOI] [PubMed] [Google Scholar]
- 11.Terstappen LWMM, Huang S, Safford M, Lansdorp PM, Loken MR. Sequential generations of hematopoietic colonies derived from single nonlineage-committed CD34+CD38- progenitor cells. Blood. 1991;77:1218–1227. [PubMed] [Google Scholar]
- 12.Sutherland HJ, Lansdorp PM, Henkelman DH, Eaves AC, Eaves CJ. Functional characterization of individual human hematopoietic stem cells cultured at limiting dilution on supportive marrow stromal layers. Proc Natl Acad Sci USA. 1990;87:3584–3588. doi: 10.1073/pnas.87.9.3584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bhatia M, Bonnet D, Murdoch B, Gan OI, Dick J. A newly discovered class of human hematopoietic cells with SCID-repopulating activity. Nat Med. 1998;4:1038–1045. doi: 10.1038/2023. [DOI] [PubMed] [Google Scholar]
- 14.Zanjani ED, Almeida-Porada G, Livingston AG, Flake AW, Ogawa M. Human bone marrow CD34- cells engraft in vivo and undergo multilineage expression that includes giving rise to CD34+ cells. Exp Hematol. 1998;26:353–360. [PubMed] [Google Scholar]
- 15.Fujisaki T, Berger MG, Rose-John S, Eaves CJ. Rapid differentiation of a rare subset of adult human lin-CD34-CD38- cells stimulated by multiple growth factors in vitro. Blood. 1999;94:1926–1932. [PubMed] [Google Scholar]
- 16.Gallacher L, et al. Isolation and characterization of human CD34-Lin- and CD34 +Lin- hematopoietic stem cells using cell surface markers AC133 and CD7. Blood. 2000;95:2813–2820. [PubMed] [Google Scholar]
- 17.Nakamura Y, et al. Ex vivo generation of CD34+ cells from CD34- hematopoietic cells. Blood. 1999;94:4053–4059. [PubMed] [Google Scholar]
- 18.Yoder MC, et al. Characterization of definitive lymphohematopoietic stem cells in the day 9 murine yolk sac. Immunity. 1997;7:335–344. doi: 10.1016/s1074-7613(00)80355-6. [DOI] [PubMed] [Google Scholar]
- 19.Ito T, Tajima F, Ogawa M. Developmental changes of CD34 expression by murine hematopoietic stem cells. Exp Hematol. 2000;28:1269–1273. doi: 10.1016/s0301-472x(00)00535-x. [DOI] [PubMed] [Google Scholar]
- 20.Osawa M, Hanada K, Hamada H, Nakauchi H. Long-term lymphohematopoietic reconstitution by a single CD34-low/negative hematopoietic stem cell. Science. 1996;273:242–245. doi: 10.1126/science.273.5272.242. [DOI] [PubMed] [Google Scholar]
- 21.Sato T, Laver JH, Ogawa M. Reversible expression of CD34 by murine hematopoietic stem cells. Blood. 1999;94:2548–2554. [PubMed] [Google Scholar]
- 22.Goodell MA, Brose K, Paradis G, Conner AS, Mulligan RC. Isolation and functional properties of murine hematopoietic stem cells that are replicating in vivo. J Exp Med. 1996;183:1797–1806. doi: 10.1084/jem.183.4.1797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Yusa K, Tsuruo T. Reversal mechanism of multidrug resistance by verapamil: direct binding of verapamil to P-glycoprotein on specific sites and transport of verapamil outward across the plasma membrane of K562/ADM cells. Cancer Res. 1989;49:5002–5006. [PubMed] [Google Scholar]
- 24.Spangrude GJ, et al. Mouse hematopoietic stem cells. Blood. 1991;78:1395–1402. [PubMed] [Google Scholar]
- 25.Miller CL, Eaves CJ. Expansion in vitro of adult murine hematopoietic stem cells with transplantable lympho-myeloid reconstituting ability. Proc Natl Acad Sci USA. 1997;94:13648–13653. doi: 10.1073/pnas.94.25.13648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Goodell MA, et al. Dye efflux studies suggest that hematopoietic stem cells expressing low or undetectable levels of CD34 antigen exist in multiple species. Nat Med. 1997;3:1337–1345. doi: 10.1038/nm1297-1337. [DOI] [PubMed] [Google Scholar]
- 27.Storms RW, Goodell MA, Fisher A, Mulligan RC, Smith C. Hoechst dye efflux reveals a novel CD7+CD34- lymphoid progenitor in human unbilical cord blood. Blood. 2000;96:2125–2133. [PubMed] [Google Scholar]
- 28.Nishikawa S-I, et al. In vitro generation of lymphohematopoietic cells from endothelial cells purified from murine embryos. Immunity. 1998;8:761–769. doi: 10.1016/s1074-7613(00)80581-6. [DOI] [PubMed] [Google Scholar]
- 29.Shultz LD, et al. Multiple defects in innate and adaptive immunologic function in NOD/LtSz-scid mice. J Immunol. 1995;154:180–191. [PubMed] [Google Scholar]
- 30.Schmitt C, Eaves CJ, Lansdorp PM. Expression of CD34 on human B cell precursors. Clin Exp Immunol. 1991;85:168–173. doi: 10.1111/j.1365-2249.1991.tb05699.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hogge DE, Lansdorp PM, Reid D, Gerhard B, Eaves CJ. Enhanced detection, maintenance and differentiation of primitive human hematopoietic cells in cultures containing murine fibroblasts engineered to produce human Steel factor, interleukin-3 and granulocyte colony-stimulating factor. Blood. 1996;88:3765–3773. [PubMed] [Google Scholar]
- 32.Unkeless JC. Characterization of a monoclonal antibody directed against mouse macrophage and lymphocyte Fc receptors. J Exp Med. 1979;150:580–596. doi: 10.1084/jem.150.3.580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Conneally E, Cashman J, Petzer A, Eaves C. Expansion in vitro of transplantable human cord blood stem cells demonstrated using a quantitative assay of their lympho-myeloid repopulating activity in nonobese diabetic-scid/scid mice. Proc Natl Acad Sci USA. 1997;94:9836–9841. doi: 10.1073/pnas.94.18.9836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Cashman J, Bockhold K, Hogge DE, Eaves AC, Eaves CJ. Sustained proliferation, multi-lineage differentiation and maintenance of primitive human haematopoietic cells in NOD/SCID mice transplanted with human cord blood. Br J Haematol. 1997;97:1026–1036. doi: 10.1046/j.1365-2141.1997.3233140.x. [DOI] [PubMed] [Google Scholar]
- 35.Holyoake TL, Nicolini FE, Eaves CJ. Functional differences between transplantable human hematopoietic stem cells from fetal liver, cord blood, and adult marrow. Exp Hematol. 1999;27:1418–1427. doi: 10.1016/s0301-472x(99)00078-8. [DOI] [PubMed] [Google Scholar]
- 36.Nicolini FE, et al. Unique differentiation programs of human fetal liver stem cells revealed both in vitro and in vivo in NOD/SCID mice. Blood. 1999;94:2686–2695. [PubMed] [Google Scholar]
- 37.Jackson KA, Mi T, Goodell MA. Hematopoietic potential of stem cells isolated from murine skeletal muscle. Proc Natl Acad Sci USA. 1999;96:14482–14486. doi: 10.1073/pnas.96.25.14482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Zhou S, et al. Expression of the breast cancer resistance protein (BCRP) in side population (SP) stem cells, natural killer (NK) lymphocytes, and erythroid progenitor cells. Blood. 2000;96:820a. (Abstr.) [Google Scholar]
- 39.Scharenberg CW, Harkey MA, Torok-Storb BJ. The transporter ABCG2 effluxes hoechst and its expression is restricted to the most primitive human hematopoietic cells. Blood. 2000;96:666a. (Abstr.) [Google Scholar]
- 40.Allikmets R, Schriml LM, Hutchinson A, Romano-Spica V, Dean M. A human placenta-specific ATP-binding cassette gene (ABCP) on chromosome 4q22 that is involved in multidrug resistance. Cancer Res. 1998;58:5337–5339. [PubMed] [Google Scholar]
- 41.Doyle LA, et al. A multidrug resistance transporter from human MCF-7 breast cancer cells. Proc Natl Acad Sci USA. 1998;95:15665–15670. doi: 10.1073/pnas.95.26.15665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Miyake K, et al. Molecular cloning of cDNAs which are highly overexpressed in mitoxantrone-resistant cells: demonstration of homology to ABC transport genes. Cancer Res. 1999;59:8–13. [PubMed] [Google Scholar]
- 43.Scheffer GL, et al. Breast cancer resistance protein is localized at the plasma membrane in mitoxantrone- and topotecan-resistant cell lines. Cancer Res. 2000;60:2589–2593. [PubMed] [Google Scholar]
- 44.Rocchi E, et al. The product of the ABC half-transporter gene ABCG2 (BCRP/MXR/ABCP) is expressed in the plasma membrane. Biochem Biophys Res Commun. 2000;271:42–46. doi: 10.1006/bbrc.2000.2590. [DOI] [PubMed] [Google Scholar]
- 45.Hao QL, Thiemann FT, Petersen D, Smogorzewska EM, Crooks GM. Extended long-term culture reveals a highly quiescent and primitive human hematopoietic progenitor population. Blood. 1996;88:3306–3313. [PubMed] [Google Scholar]
- 46.Civin CI, et al. Sustained, retransplantable, multilineage engraftment of highly purified adult human bone marrow stem cells in vivo. Blood. 1996;88:4102–4109. [PubMed] [Google Scholar]
- 47.Oh I-H, Lau A, Eaves CJ. During ontogeny primitive (CD34+CD38-) hematopoietic cells show altered expression of a subset of genes associated with early cytokine and differentiation responses of their adult counterparts. Blood. 2000;96:4160–4168. [PubMed] [Google Scholar]
- 48.Boggs DR. The total marrow mass of the mouse: a simplified method of measurement. Am J Hematol. 1984;16:277–286. doi: 10.1002/ajh.2830160309. [DOI] [PubMed] [Google Scholar]