Abstract
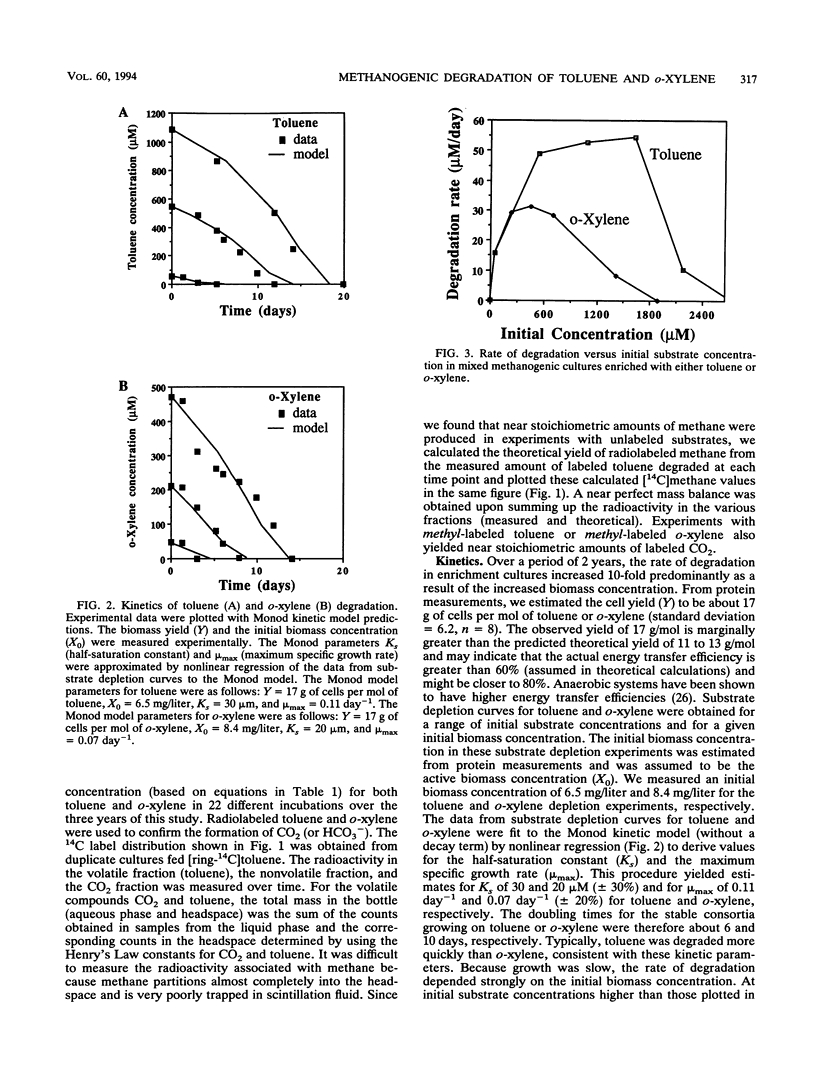
Toluene and o-xylene were completely mineralized to stoichiometric amounts of carbon dioxide, methane, and biomass by aquifer-derived microorganisms under strictly anaerobic conditions. The source of the inoculum was creosote-contaminated sediment from Pensacola, Fla. The adaptation periods before the onset of degradation were long (100 to 120 days for toluene degradation and 200 to 255 days for o-xylene). Successive transfers of the toluene- and o-xylene-degrading cultures remained active. Cell density in the cultures progressively increased over 2 to 3 years to stabilize at approximately 10(9) cells per ml. Degradation of toluene and o-xylene in stable mixed methanogenic cultures followed Monod kinetics, with inhibition noted at substrate concentrations above about 700 microM for o-xylene and 1,800 microM for toluene. The cultures degraded toluene or o-xylene but did not degrade m-xylene, p-xylene, benzene, ethylbenzene, or naphthalene. The degradative activity was retained after pasteurization or after starvation for 1 year. Degradation of toluene and o-xylene was inhibited by the alternate electron acceptors oxygen, nitrate, and sulfate. Degradation was also inhibited by the addition of preferred substrates such as acetate, H2, propionate, methanol, acetone, glucose, amino acids, fatty acids, peptone, and yeast extract. These data suggest that the presence of natural organic substrates or contaminants may inhibit anaerobic degradation of pollutants such as toluene and o-xylene at contaminated sites.
Full text
PDF








Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Beller H. R., Grbić-Galić D., Reinhard M. Microbial degradation of toluene under sulfate-reducing conditions and the influence of iron on the process. Appl Environ Microbiol. 1992 Mar;58(3):786–793. doi: 10.1128/aem.58.3.786-793.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1006/abio.1976.9999. [DOI] [PubMed] [Google Scholar]
- Brock T. D., Od'ea K. Amorphous ferrous sulfide as a reducing agent for culture of anaerobes. Appl Environ Microbiol. 1977 Feb;33(2):254–256. doi: 10.1128/aem.33.2.254-256.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dean B. J. Recent findings on the genetic toxicology of benzene, toluene, xylenes and phenols. Mutat Res. 1985 Nov;154(3):153–181. doi: 10.1016/0165-1110(85)90016-8. [DOI] [PubMed] [Google Scholar]
- Dolfing J., Zeyer J., Binder-Eicher P., Schwarzenbach R. P. Isolation and characterization of a bacterium that mineralizes toluene in the absence of molecular oxygen. Arch Microbiol. 1990;154(4):336–341. doi: 10.1007/BF00276528. [DOI] [PubMed] [Google Scholar]
- Edwards E. A., Edwards A. M., Grbić-Galić D. A method for detection of aromatic metabolites at very low concentrations: application to detection of metabolites of anaerobic toluene degradation. Appl Environ Microbiol. 1994 Jan;60(1):323–327. doi: 10.1128/aem.60.1.323-327.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edwards E. A., Wills L. E., Reinhard M., Grbić-Galić D. Anaerobic degradation of toluene and xylene by aquifer microorganisms under sulfate-reducing conditions. Appl Environ Microbiol. 1992 Mar;58(3):794–800. doi: 10.1128/aem.58.3.794-800.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans P. J., Mang D. T., Young L. Y. Degradation of toluene and m-xylene and transformation of o-xylene by denitrifying enrichment cultures. Appl Environ Microbiol. 1991 Feb;57(2):450–454. doi: 10.1128/aem.57.2.450-454.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grbić-Galić D., Vogel T. M. Transformation of toluene and benzene by mixed methanogenic cultures. Appl Environ Microbiol. 1987 Feb;53(2):254–260. doi: 10.1128/aem.53.2.254-260.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuhn E. P., Zeyer J., Eicher P., Schwarzenbach R. P. Anaerobic degradation of alkylated benzenes in denitrifying laboratory aquifer columns. Appl Environ Microbiol. 1988 Feb;54(2):490–496. doi: 10.1128/aem.54.2.490-496.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lovley D. R., Lonergan D. J. Anaerobic Oxidation of Toluene, Phenol, and p-Cresol by the Dissimilatory Iron-Reducing Organism, GS-15. Appl Environ Microbiol. 1990 Jun;56(6):1858–1864. doi: 10.1128/aem.56.6.1858-1864.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith M. R. The biodegradation of aromatic hydrocarbons by bacteria. Biodegradation. 1990;1(2-3):191–206. doi: 10.1007/BF00058836. [DOI] [PubMed] [Google Scholar]
- Thauer R. K., Jungermann K., Decker K. Energy conservation in chemotrophic anaerobic bacteria. Bacteriol Rev. 1977 Mar;41(1):100–180. doi: 10.1128/br.41.1.100-180.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vogel T. M., Grbìc-Galìc D. Incorporation of Oxygen from Water into Toluene and Benzene during Anaerobic Fermentative Transformation. Appl Environ Microbiol. 1986 Jul;52(1):200–202. doi: 10.1128/aem.52.1.200-202.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zehnder A. J., Huser B. A., Brock T. D., Wuhrmann K. Characterization of an acetate-decarboxylating, non-hydrogen-oxidizing methane bacterium. Arch Microbiol. 1980 Jan;124(1):1–11. doi: 10.1007/BF00407022. [DOI] [PubMed] [Google Scholar]
- Zeyer J., Kuhn E. P., Schwarzenbach R. P. Rapid microbial mineralization of toluene and 1,3-dimethylbenzene in the absence of molecular oxygen. Appl Environ Microbiol. 1986 Oct;52(4):944–947. doi: 10.1128/aem.52.4.944-947.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]