Abstract
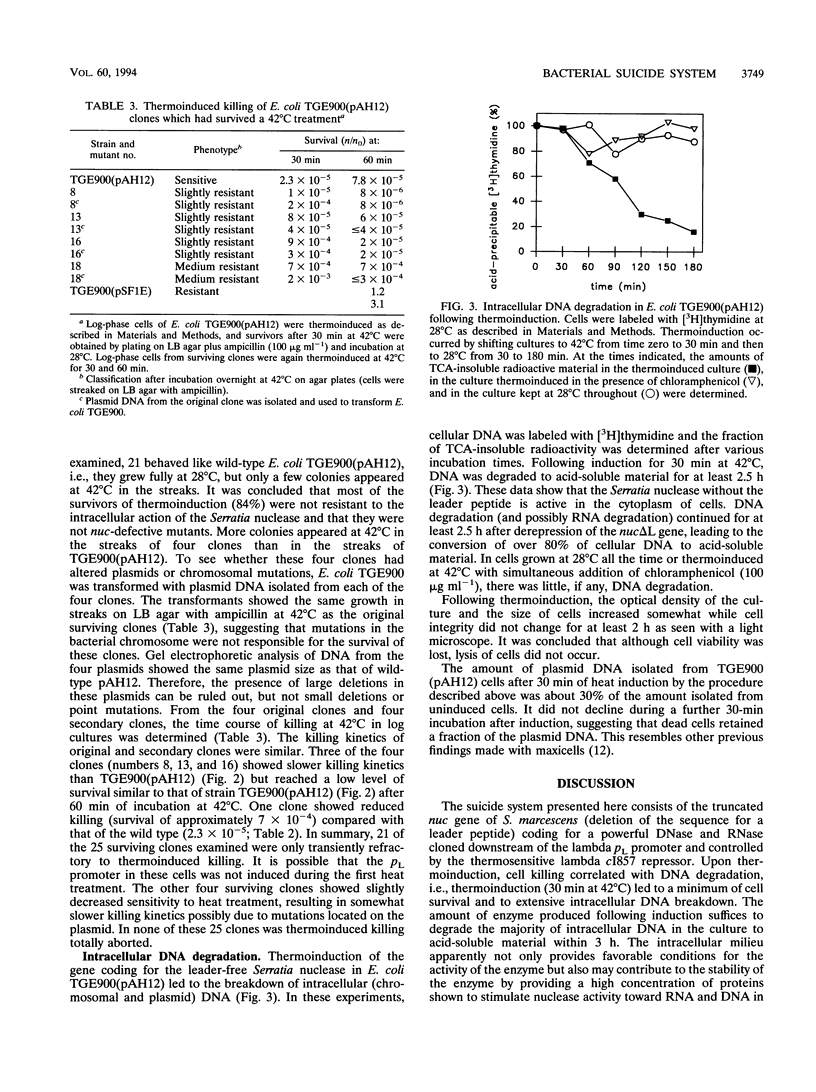
The potential risks associated with the intentional or unintentional release of genetically engineered microorganisms led to the construction of biological containment systems by which bacteria are killed in a controlled suicide process. In previously published suicide systems, cell killing was caused by proteins destroying the cell membrane or cell wall. Here a conditional cell killing system based on the intracellular degradation of cellular DNA is presented. The nuclease gene used was that of the extracellular nuclease of Serratia marcescens. The nuclease gene was deleted for the leader-coding sequence, and the truncated gene was put under the control of the lambda pL promoter. Following thermoinduction of the nuclease gene cassette in Escherichia coli, cell survival dropped to 2 x 10(-5), and more than 80% of the radioactively labeled DNA was converted to acid-soluble material within 2.5 h in the absence of cell lysis. The majority (84%) of clones which survived thermoinduced killing turned out to be as sensitive to a second thermoinduction as the original strain. The other clones showed somewhat slower killing kinetics or slightly higher final levels of survivors. The suicide system described combines the regulated killing of cells with the destruction of intracellular DNA otherwise potentially available for horizontal gene transfer processes.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ahrenholtz I., Lorenz M. G., Wackernagel W. The extracellular nuclease of Serratia marcescens: studies on the activity in vitro and effect on transforming DNA in a groundwater aquifer microcosm. Arch Microbiol. 1994;161(2):176–183. doi: 10.1007/BF00276480. [DOI] [PubMed] [Google Scholar]
- Ball T. K., Saurugger P. N., Benedik M. J. The extracellular nuclease gene of Serratia marcescens and its secretion from Escherichia coli. Gene. 1987;57(2-3):183–192. doi: 10.1016/0378-1119(87)90121-1. [DOI] [PubMed] [Google Scholar]
- Ball T. K., Suh Y., Benedik M. J. Disulfide bonds are required for Serratia marcescens nuclease activity. Nucleic Acids Res. 1992 Oct 11;20(19):4971–4974. doi: 10.1093/nar/20.19.4971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bayer I., Fliess A., Greipel J., Urbanke C., Maass G. Modulation of the affinity of the single-stranded DNA-binding protein of Escherichia coli (E. coli SSB) to poly(dT) by site-directed mutagenesis. Eur J Biochem. 1989 Feb 1;179(2):399–404. doi: 10.1111/j.1432-1033.1989.tb14567.x. [DOI] [PubMed] [Google Scholar]
- Biedermann K., Jepsen P. K., Riise E., Svendsen I. Purification and characterization of a Serratia marcescens nuclease produced by Escherichia coli. Carlsberg Res Commun. 1989;54(1):17–27. doi: 10.1007/BF02910469. [DOI] [PubMed] [Google Scholar]
- Birnboim H. C., Doly J. A rapid alkaline extraction procedure for screening recombinant plasmid DNA. Nucleic Acids Res. 1979 Nov 24;7(6):1513–1523. doi: 10.1093/nar/7.6.1513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Contreras A., Molin S., Ramos J. L. Conditional-suicide containment system for bacteria which mineralize aromatics. Appl Environ Microbiol. 1991 May;57(5):1504–1508. doi: 10.1128/aem.57.5.1504-1508.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Courtney M., Buchwalder A., Tessier L. H., Jaye M., Benavente A., Balland A., Kohli V., Lathe R., Tolstoshev P., Lecocq J. P. High-level production of biologically active human alpha 1-antitrypsin in Escherichia coli. Proc Natl Acad Sci U S A. 1984 Feb;81(3):669–673. doi: 10.1073/pnas.81.3.669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friedhoff P., Gimadutdinow O., Rüter T., Wende W., Urbanke C., Thole H., Pingoud A. A procedure for renaturation and purification of the extracellular Serratia marcescens nuclease from genetically engineered Escherichia coli. Protein Expr Purif. 1994 Feb;5(1):37–43. doi: 10.1006/prep.1994.1005. [DOI] [PubMed] [Google Scholar]
- Gerdes K., Rasmussen P. B., Molin S. Unique type of plasmid maintenance function: postsegregational killing of plasmid-free cells. Proc Natl Acad Sci U S A. 1986 May;83(10):3116–3120. doi: 10.1073/pnas.83.10.3116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heinemann J. A., Ankenbauer R. G. Retrotransfer of IncP plasmid R751 from Escherichia coli maxicells: evidence for the genetic sufficiency of self-transferable plasmids for bacterial conjugation. Mol Microbiol. 1993 Oct;10(1):57–62. doi: 10.1111/j.1365-2958.1993.tb00903.x. [DOI] [PubMed] [Google Scholar]
- Jensen L. B., Ramos J. L., Kaneva Z., Molin S. A substrate-dependent biological containment system for Pseudomonas putida based on the Escherichia coli gef gene. Appl Environ Microbiol. 1993 Nov;59(11):3713–3717. doi: 10.1128/aem.59.11.3713-3717.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knudsen S. M., Karlström O. H. Development of efficient suicide mechanisms for biological containment of bacteria. Appl Environ Microbiol. 1991 Jan;57(1):85–92. doi: 10.1128/aem.57.1.85-92.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lorenz M. G., Wackernagel W. Bacterial gene transfer by natural genetic transformation in the environment. Microbiol Rev. 1994 Sep;58(3):563–602. doi: 10.1128/mr.58.3.563-602.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Molin S., Boe L., Jensen L. B., Kristensen C. S., Givskov M., Ramos J. L., Bej A. K. Suicidal genetic elements and their use in biological containment of bacteria. Annu Rev Microbiol. 1993;47:139–166. doi: 10.1146/annurev.mi.47.100193.001035. [DOI] [PubMed] [Google Scholar]
- Nestle M., Roberts W. K. An extracellular nuclease from Serratia marcescens. II. Specificity of the enzyme. J Biol Chem. 1969 Oct 10;244(19):5219–5225. [PubMed] [Google Scholar]
- Oelgeschläger T., Geiger R., Rüter T., Alves J., Fliess A., Pingoud A. Probing the function of individual amino acid residues in the DNA binding site of the EcoRI restriction endonuclease by analysing the toxicity of genetically engineered mutants. Gene. 1990 Apr 30;89(1):19–27. doi: 10.1016/0378-1119(90)90201-2. [DOI] [PubMed] [Google Scholar]
- Recorbet G., Robert C., Givaudan A., Kudla B., Normand P., Faurie G. Conditional suicide system of Escherichia coli released into soil that uses the Bacillus subtilis sacB gene. Appl Environ Microbiol. 1993 May;59(5):1361–1366. doi: 10.1128/aem.59.5.1361-1366.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Romanowski G., Lorenz M. G., Sayler G., Wackernagel W. Persistence of free plasmid DNA in soil monitored by various methods, including a transformation assay. Appl Environ Microbiol. 1992 Sep;58(9):3012–3019. doi: 10.1128/aem.58.9.3012-3019.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Romanowski G., Lorenz M. G., Wackernagel W. Use of polymerase chain reaction and electroporation of Escherichia coli to monitor the persistence of extracellular plasmid DNA introduced into natural soils. Appl Environ Microbiol. 1993 Oct;59(10):3438–3446. doi: 10.1128/aem.59.10.3438-3446.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sargentini N. J., Smith K. C. Quantitation of the involvement of the recA, recB, recC, recF, recJ, recN, lexA, radA, radB, uvrD, and umuC genes in the repair of X-ray-induced DNA double-strand breaks in Escherichia coli. Radiat Res. 1986 Jul;107(1):58–72. [PubMed] [Google Scholar]
- Schweder T., Schmidt I., Herrmann H., Neubauer P., Hecker M., Hofmann K. An expression vector system providing plasmid stability and conditional suicide of plasmid-containing cells. Appl Microbiol Biotechnol. 1992 Oct;38(1):91–93. doi: 10.1007/BF00169425. [DOI] [PubMed] [Google Scholar]
- Tanhauser S. M., Jewell D. A., Tu C. K., Silverman D. N., Laipis P. J. A T7 expression vector optimized for site-directed mutagenesis using oligodeoxyribonucleotide cassettes. Gene. 1992 Aug 1;117(1):113–117. doi: 10.1016/0378-1119(92)90498-e. [DOI] [PubMed] [Google Scholar]


