Abstract
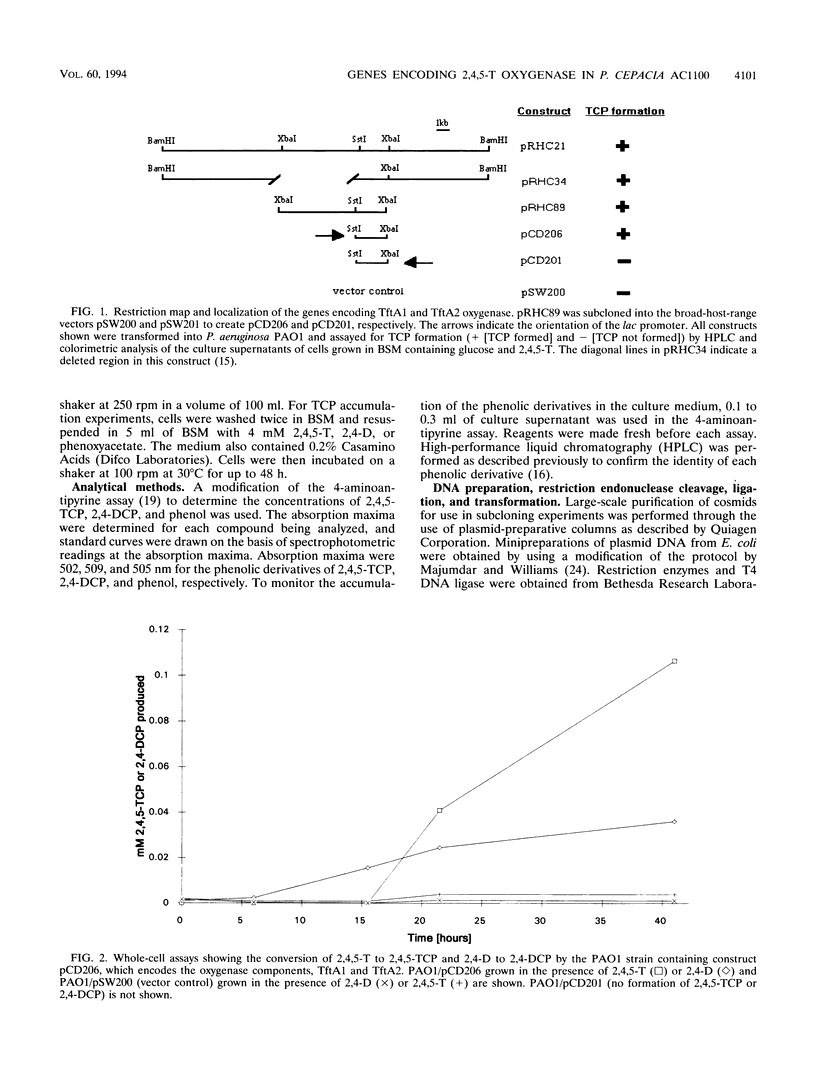
Pseudomonas cepacia AC1100 is able to use the chlorinated aromatic compound 2,4,5-trichlorophenoxyacetic acid (2,4,5-T) as the sole source of carbon and energy. One of the early steps in this pathway is the conversion of 2,4,5-T to 2,4,5-trichlorophenol (2,4,5-TCP). 2,4,5-TCP accumulates in the culture medium when AC1100 is grown in the presence of 2,4,5-T. A DNA region from the AC1100 genome has been subcloned as a 2.7-kb SstI-XbaI DNA fragment, which on transfer to Pseudomonas aeruginosa PAO1 allows the conversion of 2,4,5-T to 2,4,5-TCP. We have determined the directions of transcription of these genes as well as the complete nucleotide sequences of the genes and the number and sizes of the polypeptides synthesized by pulse-labeling experiments. This 2.7-kb DNA fragment encodes two polypeptides with calculated molecular masses of 51 and 18 kDa. Proteins of similar sizes were seen in the T7 pulse-labeling experiment in Escherichia coli. We have designated the genes for these proteins tftA1 (which encodes the 51-kDa protein) and tftA2 (which encodes the 18-kDa protein). TftA1 and TftA2 have strong amino acid sequence homology to BenA and BenB from the benzoate 1,2-dioxygenase system of Acinetobacter calcoaceticus, as well as to XylX and XylY from the toluate 1,2-dioxygenase system of Pseudomonas putida. The Pseudomonas aeruginosa PAO1 strain containing the 2.7-kb SstI-XbaI fragment was able to convert not only 2,4,5-T to 2,4,5-TCP but also 2,4-dichlorophenoxyacetic acid to 2,4-dichlorophenol and phenoxyacetate to phenol.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barkay T., Liebert C., Gillman M. Conjugal gene transfer to aquatic bacteria detected by the generation of a new phenotype. Appl Environ Microbiol. 1993 Mar;59(3):807–814. doi: 10.1128/aem.59.3.807-814.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Don R. H., Weightman A. J., Knackmuss H. J., Timmis K. N. Transposon mutagenesis and cloning analysis of the pathways for degradation of 2,4-dichlorophenoxyacetic acid and 3-chlorobenzoate in Alcaligenes eutrophus JMP134(pJP4). J Bacteriol. 1985 Jan;161(1):85–90. doi: 10.1128/jb.161.1.85-90.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Erickson B. D., Mondello F. J. Enhanced biodegradation of polychlorinated biphenyls after site-directed mutagenesis of a biphenyl dioxygenase gene. Appl Environ Microbiol. 1993 Nov;59(11):3858–3862. doi: 10.1128/aem.59.11.3858-3862.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Franklin F. C., Bagdasarian M., Bagdasarian M. M., Timmis K. N. Molecular and functional analysis of the TOL plasmid pWWO from Pseudomonas putida and cloning of genes for the entire regulated aromatic ring meta cleavage pathway. Proc Natl Acad Sci U S A. 1981 Dec;78(12):7458–7462. doi: 10.1073/pnas.78.12.7458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fukumori F., Hausinger R. P. Alcaligenes eutrophus JMP134 "2,4-dichlorophenoxyacetate monooxygenase" is an alpha-ketoglutarate-dependent dioxygenase. J Bacteriol. 1993 Apr;175(7):2083–2086. doi: 10.1128/jb.175.7.2083-2086.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gambello M. J., Kaye S., Iglewski B. H. LasR of Pseudomonas aeruginosa is a transcriptional activator of the alkaline protease gene (apr) and an enhancer of exotoxin A expression. Infect Immun. 1993 Apr;61(4):1180–1184. doi: 10.1128/iai.61.4.1180-1184.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grant W. F. The genotoxic effects of 2,4,5-T. Mutat Res. 1979;65(2):83–119. doi: 10.1016/0165-1110(79)90001-0. [DOI] [PubMed] [Google Scholar]
- Gurbiel R. J., Batie C. J., Sivaraja M., True A. E., Fee J. A., Hoffman B. M., Ballou D. P. Electron-nuclear double resonance spectroscopy of 15N-enriched phthalate dioxygenase from Pseudomonas cepacia proves that two histidines are coordinated to the [2Fe-2S] Rieske-type clusters. Biochemistry. 1989 May 30;28(11):4861–4871. doi: 10.1021/bi00437a051. [DOI] [PubMed] [Google Scholar]
- Harayama S., Rekik M., Timmis K. N. Genetic analysis of a relaxed substrate specificity aromatic ring dioxygenase, toluate 1,2-dioxygenase, encoded by TOL plasmid pWW0 of Pseudomonas putida. Mol Gen Genet. 1986 Feb;202(2):226–234. doi: 10.1007/BF00331641. [DOI] [PubMed] [Google Scholar]
- Harker A. R., Olsen R. H., Seidler R. J. Phenoxyacetic acid degradation by the 2,4-dichlorophenoxyacetic acid (TFD) pathway of plasmid pJP4: mapping and characterization of the TFD regulatory gene, tfdR. J Bacteriol. 1989 Jan;171(1):314–320. doi: 10.1128/jb.171.1.314-320.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haugland R. A., Sangodkar U. M., Chakrabarty A. M. Repeated sequences including RS1100 from Pseudomonas cepacia AC1100 function as IS elements. Mol Gen Genet. 1990 Jan;220(2):222–228. doi: 10.1007/BF00260485. [DOI] [PubMed] [Google Scholar]
- Haugland R. A., Sangodkar U. M., Sferra P. R., Chakrabarty A. M. Cloning and characterization of a chromosomal DNA region required for growth on 2,4,5-T by Pseudomonas cepacia AC1100. Gene. 1991 Apr;100:65–73. doi: 10.1016/0378-1119(91)90351-b. [DOI] [PubMed] [Google Scholar]
- Haugland R. A., Schlemm D. J., Lyons R. P., 3rd, Sferra P. R., Chakrabarty A. M. Degradation of the chlorinated phenoxyacetate herbicides 2,4-dichlorophenoxyacetic acid and 2,4,5-trichlorophenoxyacetic acid by pure and mixed bacterial cultures. Appl Environ Microbiol. 1990 May;56(5):1357–1362. doi: 10.1128/aem.56.5.1357-1362.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karns J. S., Duttagupta S., Chakrabarty A. M. Regulation of 2,4,5-trichlorophenoxyacetic acid and chlorophenol metabolism in Pseudomonas cepacia AC1100. Appl Environ Microbiol. 1983 Nov;46(5):1182–1186. doi: 10.1128/aem.46.5.1182-1186.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kilbane J. J., Chatterjee D. K., Karns J. S., Kellogg S. T., Chakrabarty A. M. Biodegradation of 2,4,5-trichlorophenoxyacetic acid by a pure culture of Pseudomonas cepacia. Appl Environ Microbiol. 1982 Jul;44(1):72–78. doi: 10.1128/aem.44.1.72-78.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- King R. J., Short K. A., Seidler R. J. Assay for detection and enumeration of genetically engineered microorganisms which is based on the activity of a deregulated 2,4-dichlorophenoxyacetate monooxygenase. Appl Environ Microbiol. 1991 Jun;57(6):1790–1792. doi: 10.1128/aem.57.6.1790-1792.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kukor J. J., Olsen R. H., Ballou D. P. Cloning and expression of the catA and catBC gene clusters from Pseudomonas aeruginosa PAO. J Bacteriol. 1988 Oct;170(10):4458–4465. doi: 10.1128/jb.170.10.4458-4465.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurkela S., Lehväslaiho H., Palva E. T., Teeri T. H. Cloning, nucleotide sequence and characterization of genes encoding naphthalene dioxygenase of Pseudomonas putida strain NCIB9816. Gene. 1988 Dec 20;73(2):355–362. doi: 10.1016/0378-1119(88)90500-8. [DOI] [PubMed] [Google Scholar]
- Majumdar M. K., Williams D. A. Minipreparations of plasmid DNA directly from cell culture by the boiling method. Biotechniques. 1992 Sep;13(3):366–366. [PubMed] [Google Scholar]
- Menn F. M., Applegate B. M., Sayler G. S. NAH plasmid-mediated catabolism of anthracene and phenanthrene to naphthoic acids. Appl Environ Microbiol. 1993 Jun;59(6):1938–1942. doi: 10.1128/aem.59.6.1938-1942.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Messing J. New M13 vectors for cloning. Methods Enzymol. 1983;101:20–78. doi: 10.1016/0076-6879(83)01005-8. [DOI] [PubMed] [Google Scholar]
- Neidle E. L., Hartnett C., Ornston L. N., Bairoch A., Rekik M., Harayama S. Nucleotide sequences of the Acinetobacter calcoaceticus benABC genes for benzoate 1,2-dioxygenase reveal evolutionary relationships among multicomponent oxygenases. J Bacteriol. 1991 Sep;173(17):5385–5395. doi: 10.1128/jb.173.17.5385-5395.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neidle E. L., Shapiro M. K., Ornston L. N. Cloning and expression in Escherichia coli of Acinetobacter calcoaceticus genes for benzoate degradation. J Bacteriol. 1987 Dec;169(12):5496–5503. doi: 10.1128/jb.169.12.5496-5503.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neidle E., Hartnett C., Ornston L. N., Bairoch A., Rekik M., Harayama S. cis-diol dehydrogenases encoded by the TOL pWW0 plasmid xylL gene and the Acinetobacter calcoaceticus chromosomal benD gene are members of the short-chain alcohol dehydrogenase superfamily. Eur J Biochem. 1992 Feb 15;204(1):113–120. doi: 10.1111/j.1432-1033.1992.tb16612.x. [DOI] [PubMed] [Google Scholar]
- Olsen R. H., DeBusscher G., McCombie W. R. Development of broad-host-range vectors and gene banks: self-cloning of the Pseudomonas aeruginosa PAO chromosome. J Bacteriol. 1982 Apr;150(1):60–69. doi: 10.1128/jb.150.1.60-69.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pearson W. R. Rapid and sensitive sequence comparison with FASTP and FASTA. Methods Enzymol. 1990;183:63–98. doi: 10.1016/0076-6879(90)83007-v. [DOI] [PubMed] [Google Scholar]
- Russel M., Model P. Replacement of the fip gene of Escherichia coli by an inactive gene cloned on a plasmid. J Bacteriol. 1984 Sep;159(3):1034–1039. doi: 10.1128/jb.159.3.1034-1039.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanseverino J., Applegate B. M., King J. M., Sayler G. S. Plasmid-mediated mineralization of naphthalene, phenanthrene, and anthracene. Appl Environ Microbiol. 1993 Jun;59(6):1931–1937. doi: 10.1128/aem.59.6.1931-1937.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tabor S., Richardson C. C. A bacteriophage T7 RNA polymerase/promoter system for controlled exclusive expression of specific genes. Proc Natl Acad Sci U S A. 1985 Feb;82(4):1074–1078. doi: 10.1073/pnas.82.4.1074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vieira J., Messing J. Production of single-stranded plasmid DNA. Methods Enzymol. 1987;153:3–11. doi: 10.1016/0076-6879(87)53044-0. [DOI] [PubMed] [Google Scholar]
- West S. E., Kaye S. A., Hamood A. N., Iglewski B. H. Characterization of Pseudomonas aeruginosa mutants that are deficient in exotoxin A synthesis and are altered in expression of regA, a positive regulator of exotoxin A. Infect Immun. 1994 Mar;62(3):897–903. doi: 10.1128/iai.62.3.897-903.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wyndham R. C., Nakatsu C., Peel M., Cashore A., Ng J., Szilagyi F. Distribution of the catabolic transposon Tn5271 in a groundwater bioremediation system. Appl Environ Microbiol. 1994 Jan;60(1):86–93. doi: 10.1128/aem.60.1.86-93.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zylstra G. J., Gibson D. T. Toluene degradation by Pseudomonas putida F1. Nucleotide sequence of the todC1C2BADE genes and their expression in Escherichia coli. J Biol Chem. 1989 Sep 5;264(25):14940–14946. [PubMed] [Google Scholar]