Abstract
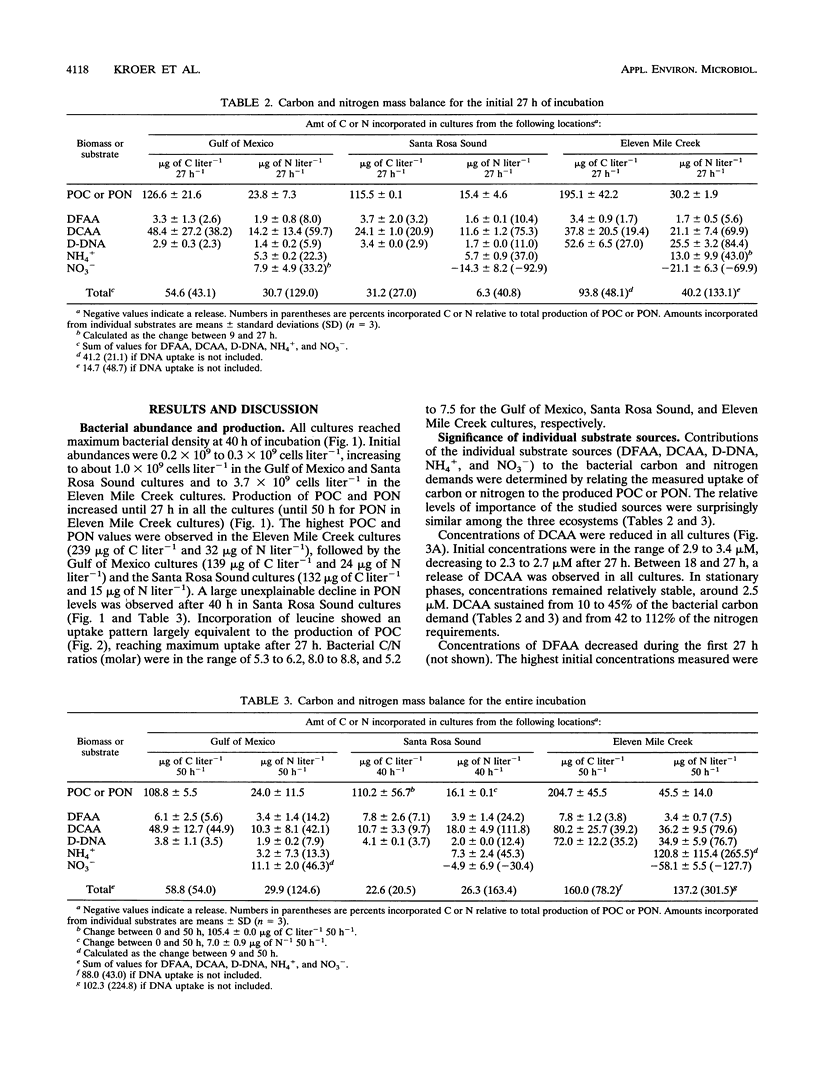
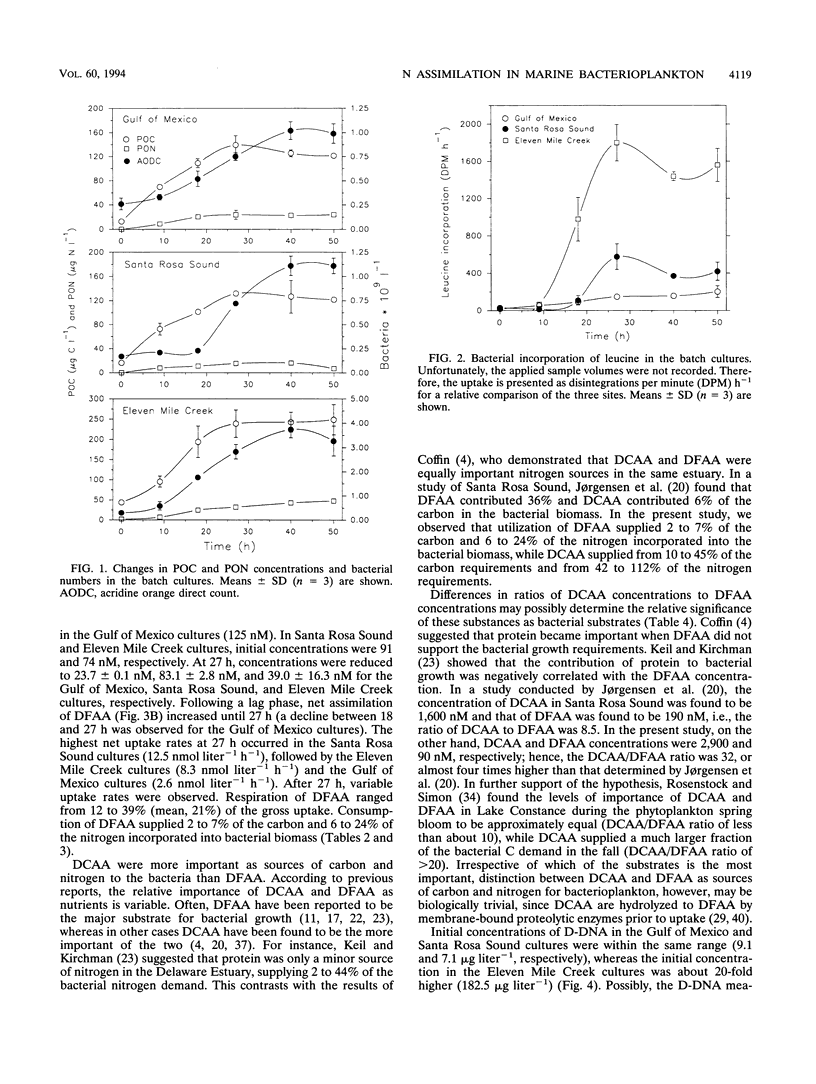
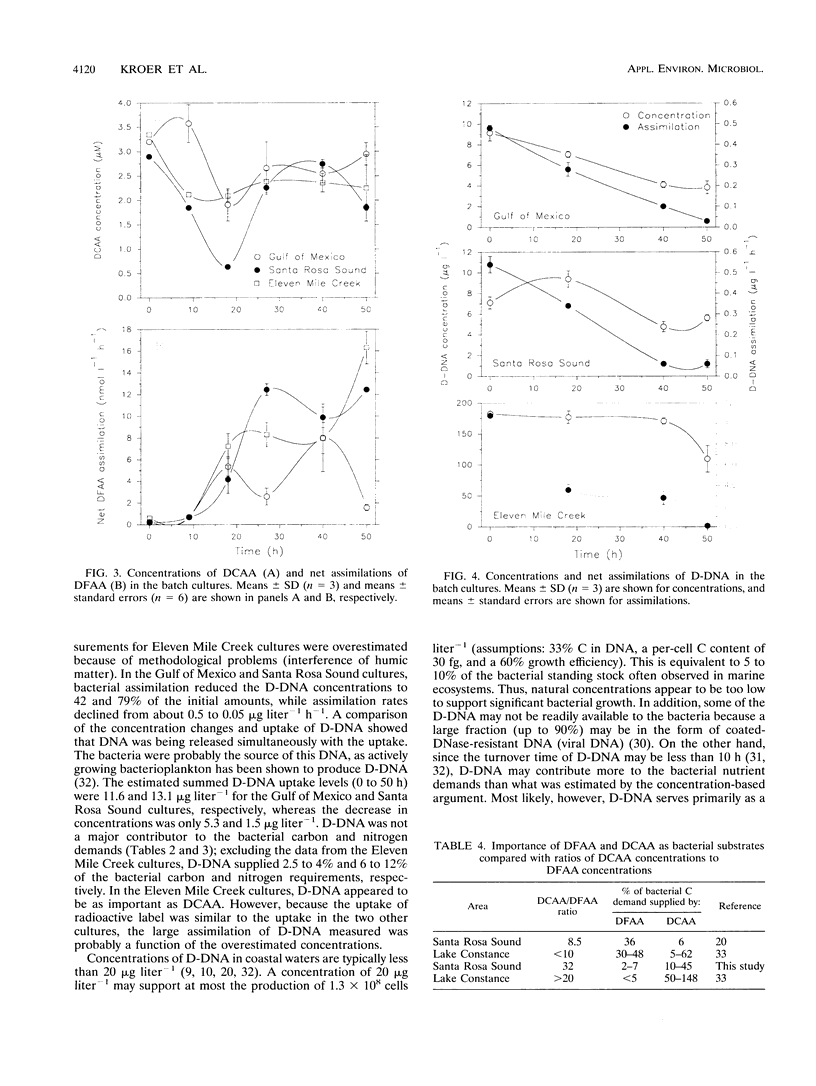
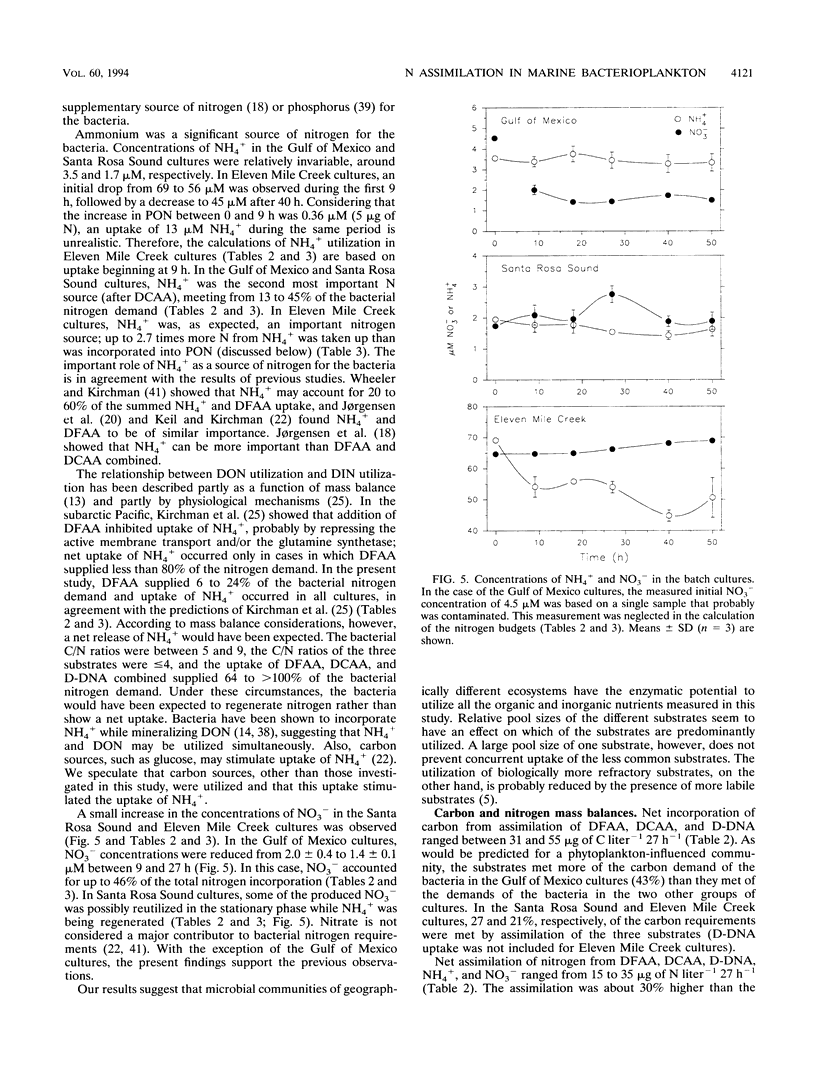
The contributions of different organic and inorganic nitrogen and organic carbon sources to heterotrophic bacterioplankton in batch cultures of oceanic, estuarine, and eutrophic riverine environments were compared. The importance of the studied compounds was surprisingly similar among the three ecosystems. Dissolved combined amino acids (DCAA) were most significant, sustaining from 10 to 45% of the bacterial carbon demands and from 42 to 112% of the bacterial nitrogen demands. Dissolved free amino acids (DFAA) supplied 2 to 7% of the carbon and 6 to 24% of the nitrogen incorporated into the bacterial biomass, while dissolved DNA (D-DNA) sustained less than 5 and 12% of the carbon and nitrogen requirements, respectively. Ammonium was the second most important source of nitrogen, meeting from 13 to 45% of the bacterial demand in the oceanic and estuarine cultures and up to 270% of the demand in riverine cultures. Nitrate was taken up in the oceanic cultures (uptake equaled up to 46% of the nitrogen demand) but was released in the two others. Assimilation of DCAA, DFAA, and D-DNA combined supplied 43% of the carbon demand of the bacteria in the oceanic cultures, while approximately 25% of the carbon requirements were met by the three substrates at the two other sites. Assimilation of nitrogen from DCAA, DFAA, D-DNA, NH4+, and NO3-, on the other hand, exceeded production of particulate organic nitrogen in one culture at 27 h and in all cultures over the entire incubation period (50 h). These results suggest that the studied nutrient sources may fully support the nitrogen needs but only partially support the carbon needs of microbial communities of geographically different ecosystems. Furthermore, a comparison of the initial concentrations of the different substrates indicated that relative pool sizes of the substrates seemed to influence which substrates were primarily being utilized by the bacteria.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Coffin R. B., Velinsky D. J., Devereux R., Price W. A., Cifuentes L. A. Stable carbon isotope analysis of nucleic acids to trace sources of dissolved substrates used by estuarine bacteria. Appl Environ Microbiol. 1990 Jul;56(7):2012–2020. doi: 10.1128/aem.56.7.2012-2020.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deflaun M. F., Paul J. H., Davis D. Simplified method for dissolved DNA determination in aquatic environments. Appl Environ Microbiol. 1986 Oct;52(4):654–659. doi: 10.1128/aem.52.4.654-659.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hobbie J. E., Daley R. J., Jasper S. Use of nuclepore filters for counting bacteria by fluorescence microscopy. Appl Environ Microbiol. 1977 May;33(5):1225–1228. doi: 10.1128/aem.33.5.1225-1228.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jørgensen N. O., Kroer N., Coffin R. B. Utilization of dissolved nitrogen by heterotrophic bacterioplankton: effect of substrate c/n ratio. Appl Environ Microbiol. 1994 Nov;60(11):4124–4133. doi: 10.1128/aem.60.11.4124-4133.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirchman D., K'nees E., Hodson R. Leucine incorporation and its potential as a measure of protein synthesis by bacteria in natural aquatic systems. Appl Environ Microbiol. 1985 Mar;49(3):599–607. doi: 10.1128/aem.49.3.599-607.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maruyama A., Oda M., Higashihara T. Abundance of Virus-Sized Non-DNase-Digestible DNA (Coated DNA) in Eutrophic Seawater. Appl Environ Microbiol. 1993 Mar;59(3):712–717. doi: 10.1128/aem.59.3.712-717.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paul J. H., Jeffrey W. H., David A. W., Deflaun M. F., Cazares L. H. Turnover of extracellular DNA in eutrophic and oligotrophic freshwater environments of southwest Florida. Appl Environ Microbiol. 1989 Jul;55(7):1823–1828. doi: 10.1128/aem.55.7.1823-1828.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paul J. H., Jeffrey W. H., DeFlaun M. F. Dynamics of extracellular DNA in the marine environment. Appl Environ Microbiol. 1987 Jan;53(1):170–179. doi: 10.1128/aem.53.1.170-179.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turk V., Rehnstam A. S., Lundberg E., Hagström A. Release of bacterial DNA by marine nanoflagellates, an intermediate step in phosphorus regeneration. Appl Environ Microbiol. 1992 Nov;58(11):3744–3750. doi: 10.1128/aem.58.11.3744-3750.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]


