Abstract
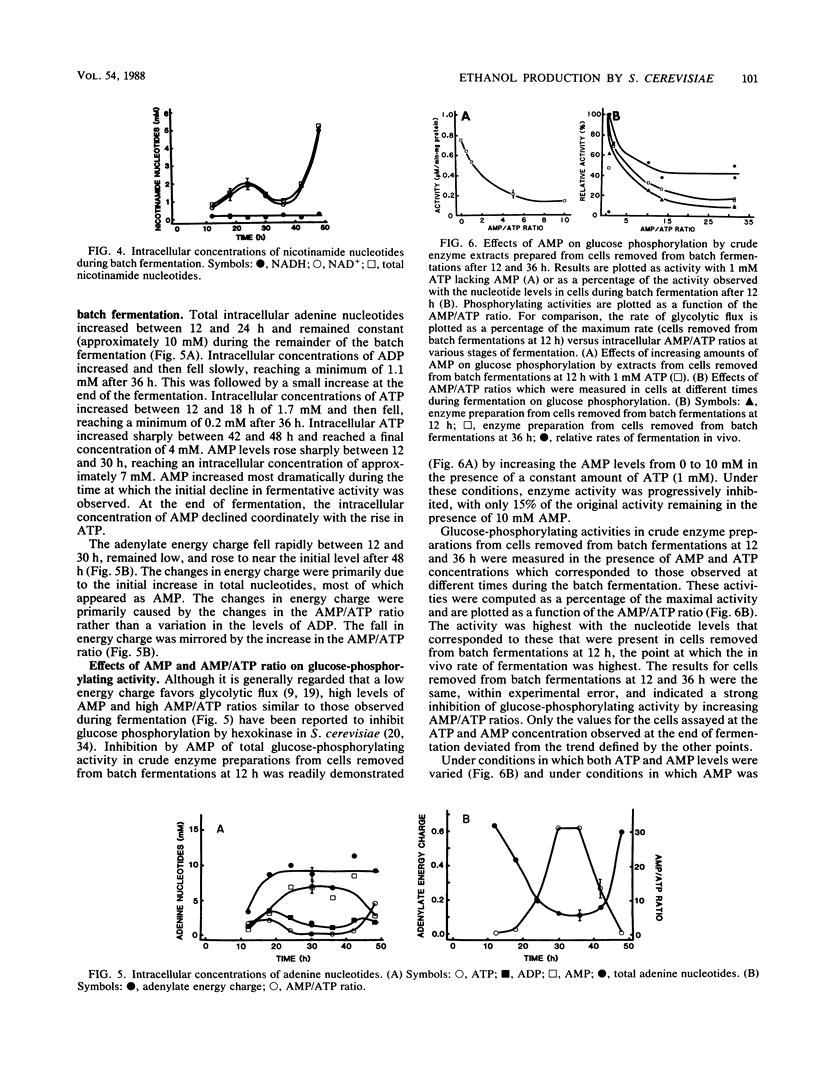
A general hypothesis is presented for the decline in the rate of ethanol production (per unit of cell protein) during batch fermentation. Inhibition of ethanol production is proposed to result from the intracellular accumulation of AMP during the transition from growth to the stationary phase. AMP acts as a competitive inhibitor of hexokinase with respect to ATP. When assayed in vitro in the presence of ATP and AMP concentrations equivalent to those within cells at different stages of fermentation, hexokinase activity declined in parallel with the in vivo decline in the rate of ethanol production. The coupling of glycolytic flux and fermentation to cell growth via degradation products of RNA may be of evolutionary advantage for Saccharomyces cerevisiae. Such a coupling would reduce the exposure of nongrowing cells to potentially harmful concentrations of waste products from metabolism and would conserve nutrients for future growth under more favorable conditions.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- ANDREASEN A. A., STIER T. J. Anaerobic nutrition of Saccharomyces cerevisiae. II. Unsaturated fatty acid requirement for growth in a defined medium. J Cell Physiol. 1954 Jun;43(3):271–281. doi: 10.1002/jcp.1030430303. [DOI] [PubMed] [Google Scholar]
- Ball W. J., Jr, Atkinson D. E. Adenylate energy charge in Saccharomyces cerevisiae during starvation. J Bacteriol. 1975 Mar;121(3):975–982. doi: 10.1128/jb.121.3.975-982.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bisson L. F., Fraenkel D. G. Expression of kinase-dependent glucose uptake in Saccharomyces cerevisiae. J Bacteriol. 1984 Sep;159(3):1013–1017. doi: 10.1128/jb.159.3.1013-1017.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bisson L. F., Neigeborn L., Carlson M., Fraenkel D. G. The SNF3 gene is required for high-affinity glucose transport in Saccharomyces cerevisiae. J Bacteriol. 1987 Apr;169(4):1656–1662. doi: 10.1128/jb.169.4.1656-1662.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boucherie H. Protein synthesis during transition and stationary phases under glucose limitation in Saccharomyces cerevisiae. J Bacteriol. 1985 Jan;161(1):385–392. doi: 10.1128/jb.161.1.385-392.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casey G. P., Ingledew W. M. Ethanol tolerance in yeasts. Crit Rev Microbiol. 1986;13(3):219–280. doi: 10.3109/10408418609108739. [DOI] [PubMed] [Google Scholar]
- Chapman A. G., Atkinson D. E. Adenine nucleotide concentrations and turnover rates. Their correlation with biological activity in bacteria and yeast. Adv Microb Physiol. 1977;15:253–306. doi: 10.1016/s0065-2911(08)60318-5. [DOI] [PubMed] [Google Scholar]
- Chapman A. G., Fall L., Atkinson D. E. Adenylate energy charge in Escherichia coli during growth and starvation. J Bacteriol. 1971 Dec;108(3):1072–1086. doi: 10.1128/jb.108.3.1072-1086.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ciriacy M., Breitenbach I. Physiological effects of seven different blocks in glycolysis in Saccharomyces cerevisiae. J Bacteriol. 1979 Jul;139(1):152–160. doi: 10.1128/jb.139.1.152-160.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clifton D., Weinstock S. B., Fraenkel D. G. Glycolysis mutants in Saccharomyces cerevisiae. Genetics. 1978 Jan;88(1):1–11. doi: 10.1093/genetics/88.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dombek K. M., Ingram L. O. Determination of the intracellular concentration of ethanol in Saccharomyces cerevisiae during fermentation. Appl Environ Microbiol. 1986 Jan;51(1):197–200. doi: 10.1128/aem.51.1.197-200.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dombek K. M., Ingram L. O. Ethanol production during batch fermentation with Saccharomyces cerevisiae: changes in glycolytic enzymes and internal pH. Appl Environ Microbiol. 1987 Jun;53(6):1286–1291. doi: 10.1128/aem.53.6.1286-1291.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dombek K. M., Ingram L. O. Magnesium limitation and its role in apparent toxicity of ethanol during yeast fermentation. Appl Environ Microbiol. 1986 Nov;52(5):975–981. doi: 10.1128/aem.52.5.975-981.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fromm H. J. Comments on the kinetics and mechanism of yeast hexokinase action. Is the binding sequence of substrates to the enzyme ordered or random? Eur J Biochem. 1969 Jan;7(3):385–392. doi: 10.1111/j.1432-1033.1969.tb19620.x. [DOI] [PubMed] [Google Scholar]
- Holmsen H., Dangelmaier C. A., Akkerman J. W. Determination of levels of glycolytic intermediates and nucleotides in platelets by pulse-labeling with [32P]orthophosphate. Anal Biochem. 1983 May;131(1):266–272. doi: 10.1016/0003-2697(83)90165-3. [DOI] [PubMed] [Google Scholar]
- Ingram L. O., Buttke T. M. Effects of alcohols on micro-organisms. Adv Microb Physiol. 1984;25:253–300. doi: 10.1016/s0065-2911(08)60294-5. [DOI] [PubMed] [Google Scholar]
- Jiménez J., Benítez T. Adaptation of yeast cell membranes to ethanol. Appl Environ Microbiol. 1987 May;53(5):1196–1198. doi: 10.1128/aem.53.5.1196-1198.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karp M. T., Raunio R. P., Lövgren T. N. Simultaneous extraction and combined bioluminescent assay of NAD+ and NADH. Anal Biochem. 1983 Jan;128(1):175–180. doi: 10.1016/0003-2697(83)90359-7. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lang J. M., Cirillo V. P. Glucose transport in a kinaseless Saccharomyces cerevisiae mutant. J Bacteriol. 1987 Jul;169(7):2932–2937. doi: 10.1128/jb.169.7.2932-2937.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leão C., Van Uden N. Effects of ethanol and other alkanols on passive proton influx in the yeast Saccharomyces cerevisiae. Biochim Biophys Acta. 1984 Jul 11;774(1):43–48. doi: 10.1016/0005-2736(84)90272-4. [DOI] [PubMed] [Google Scholar]
- Maitra P. K., Lobo Z. Yeast pyruvate kinase: a mutant from catalytically insensitive to fructose 1,6-bisphosphate. Eur J Biochem. 1977 Sep;78(2):353–360. doi: 10.1111/j.1432-1033.1977.tb11747.x. [DOI] [PubMed] [Google Scholar]
- McFarlane E. S. Ribonuclease activity during G1 arrest of the yeast Saccharomyces cerevisiae. Arch Microbiol. 1980 Feb;124(2-3):243–247. doi: 10.1007/BF00427733. [DOI] [PubMed] [Google Scholar]
- Navon G., Shulman R. G., Yamane T., Eccleshall T. R., Lam K. B., Baronofsky J. J., Marmur J. Phosphorus-31 nuclear magnetic resonance studies of wild-type and glycolytic pathway mutants of Saccharomyces cerevisiae. Biochemistry. 1979 Oct 16;18(21):4487–4499. doi: 10.1021/bi00588a006. [DOI] [PubMed] [Google Scholar]
- Polakis E. S., Bartley W. Changes in the intracellular concentrations of adenosine phosphates and nicotinamide nucleotides during the aerobic growth cycle of yeast on different carbon sources. Biochem J. 1966 Jun;99(3):521–533. doi: 10.1042/bj0990521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rudolph F. B., Fromm H. J. Computer simulation studies with yeast hexokinase and additional evidence for the random Bi Bi mechanism. J Biol Chem. 1971 Nov;246(21):6611–6619. [PubMed] [Google Scholar]
- Schuddemat J., Van den Broek P. J., Van Steveninck J. Effect of xylose incubation on the glucose transport system in Saccharomyces cerevisiae. Biochim Biophys Acta. 1986 Oct 23;861(3):489–493. doi: 10.1016/0005-2736(86)90459-1. [DOI] [PubMed] [Google Scholar]
- Swida U., Schulz-Harder B., Kücherer C., Käufer N. The occurrence of two ribosomal ribonucleases depending on growth phase in yeast. Induction of ribonuclease in glucose-starved cells. Biochim Biophys Acta. 1981 Jan 29;652(1):129–138. doi: 10.1016/0005-2787(81)90216-1. [DOI] [PubMed] [Google Scholar]
- Thomas D. S., Hossack J. A., Rose A. H. Plasma-membrane lipid composition and ethanol tolerance in Saccharomyces cerevisiae. Arch Microbiol. 1978 Jun 26;117(3):239–245. doi: 10.1007/BF00738541. [DOI] [PubMed] [Google Scholar]
- Thompson J. In vivo regulation of glycolysis and characterization of sugar: phosphotransferase systems in Streptococcus lactis. J Bacteriol. 1978 Nov;136(2):465–476. doi: 10.1128/jb.136.2.465-476.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- den Hollander J. A., Ugurbil K., Brown T. R., Bednar M., Redfield C., Shulman R. G. Studies of anaerobic and aerobic glycolysis in Saccharomyces cerevisiae. Biochemistry. 1986 Jan 14;25(1):203–211. doi: 10.1021/bi00349a029. [DOI] [PubMed] [Google Scholar]
- den Hollander J. A., Ugurbil K., Shulman R. G. 31P and 13C NMR studies of intermediates of aerobic and anaerobic glycolysis in Saccharomyces cerevisiae. Biochemistry. 1986 Jan 14;25(1):212–219. doi: 10.1021/bi00349a030. [DOI] [PubMed] [Google Scholar]